Antimicrobial, Antibiofilm, and Antioxidant Properties of Essential Oil of Foeniculum vulgare Mill. Leaves
Abstract
:1. Introduction
2. Results and Discussion
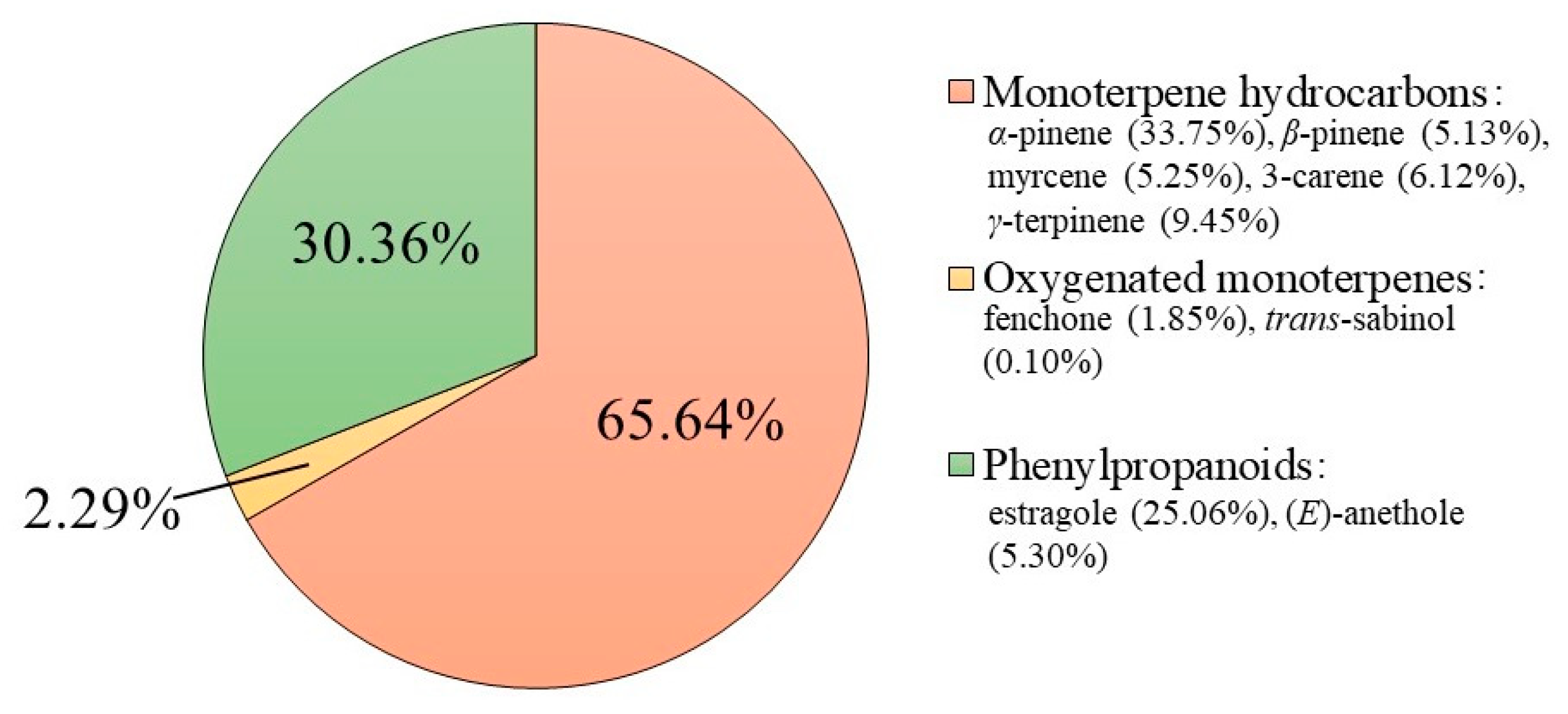
2.1. Chemical Profiling of F. vulgare subsp. vulgare var. vulgare EO
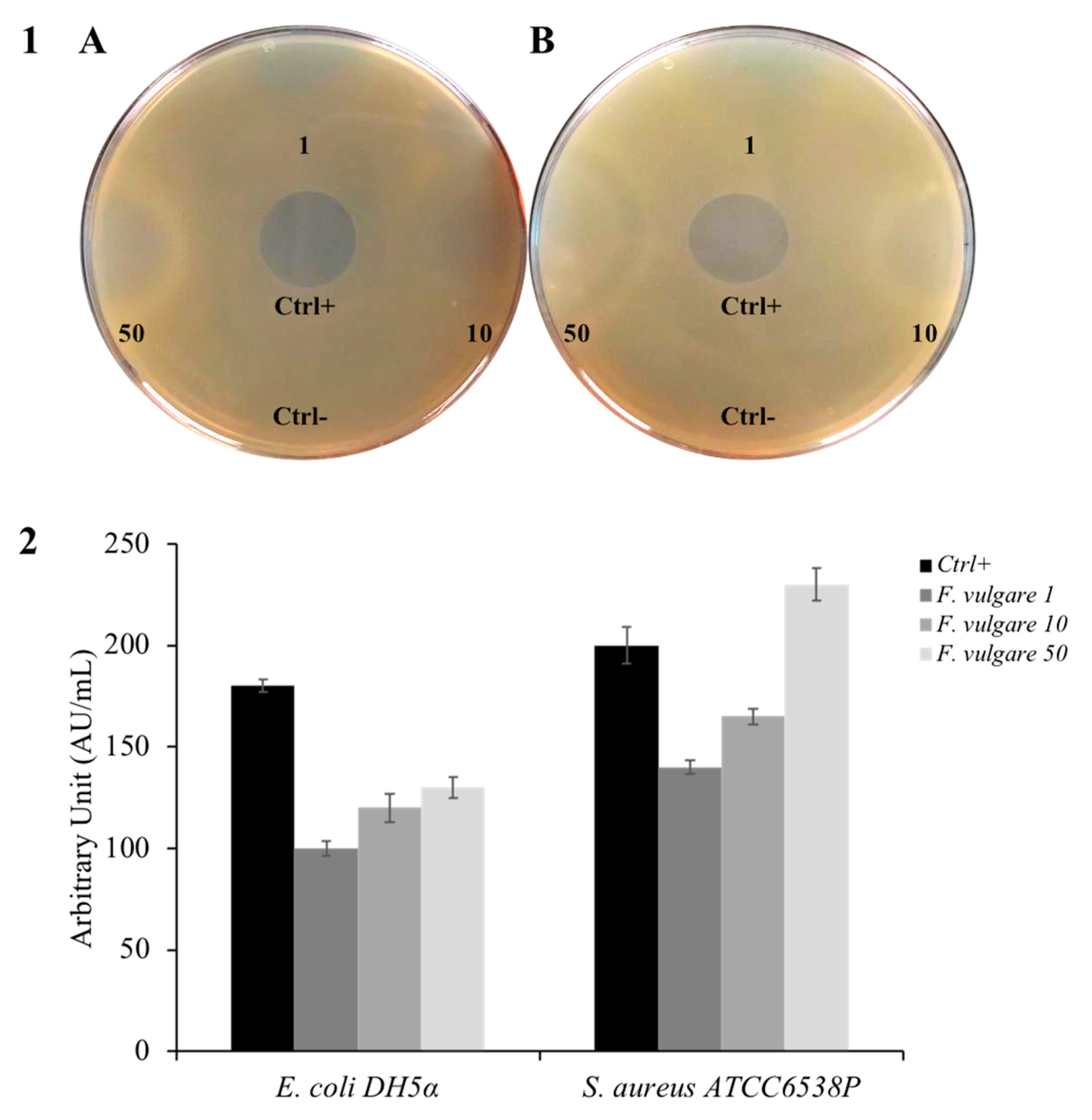
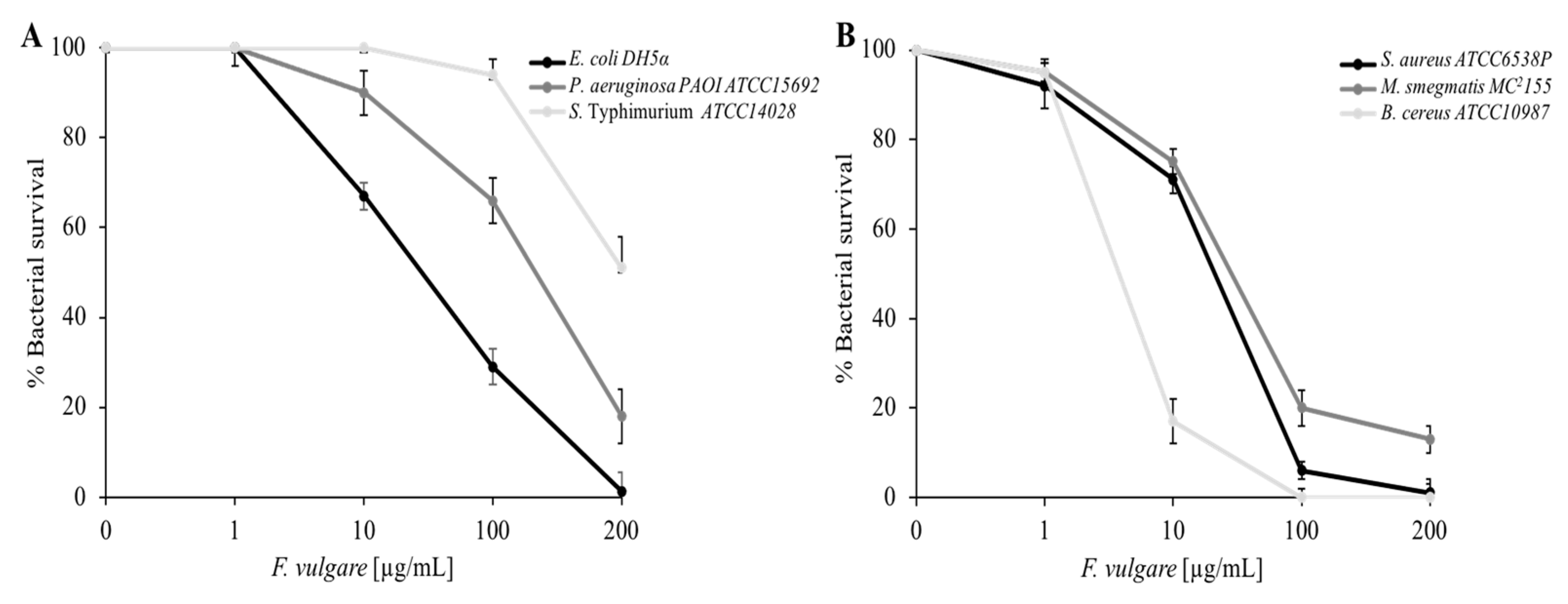
2.2. Antimicrobial Activity of F. vulgare subsp. vulgare var. vulgare EO
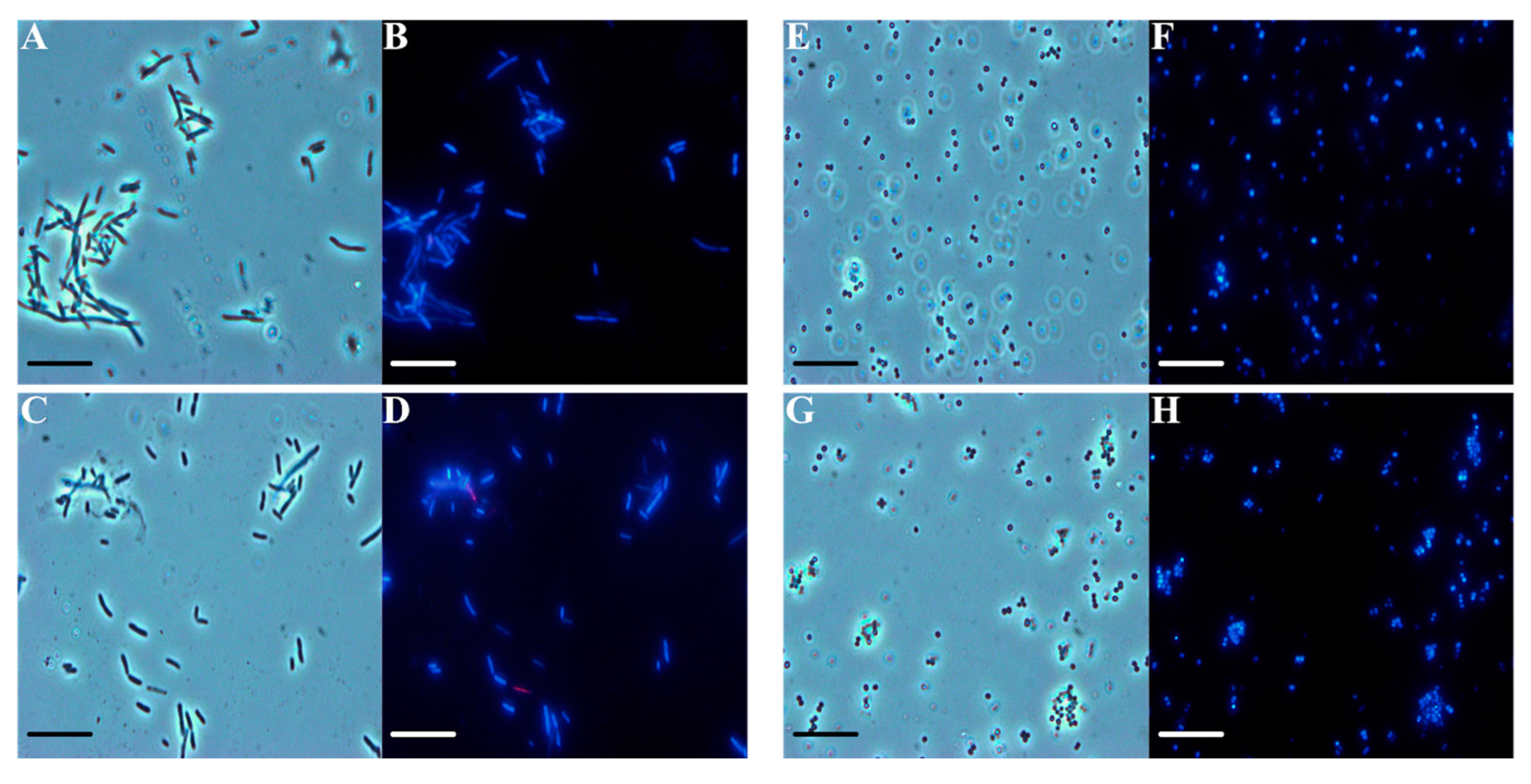
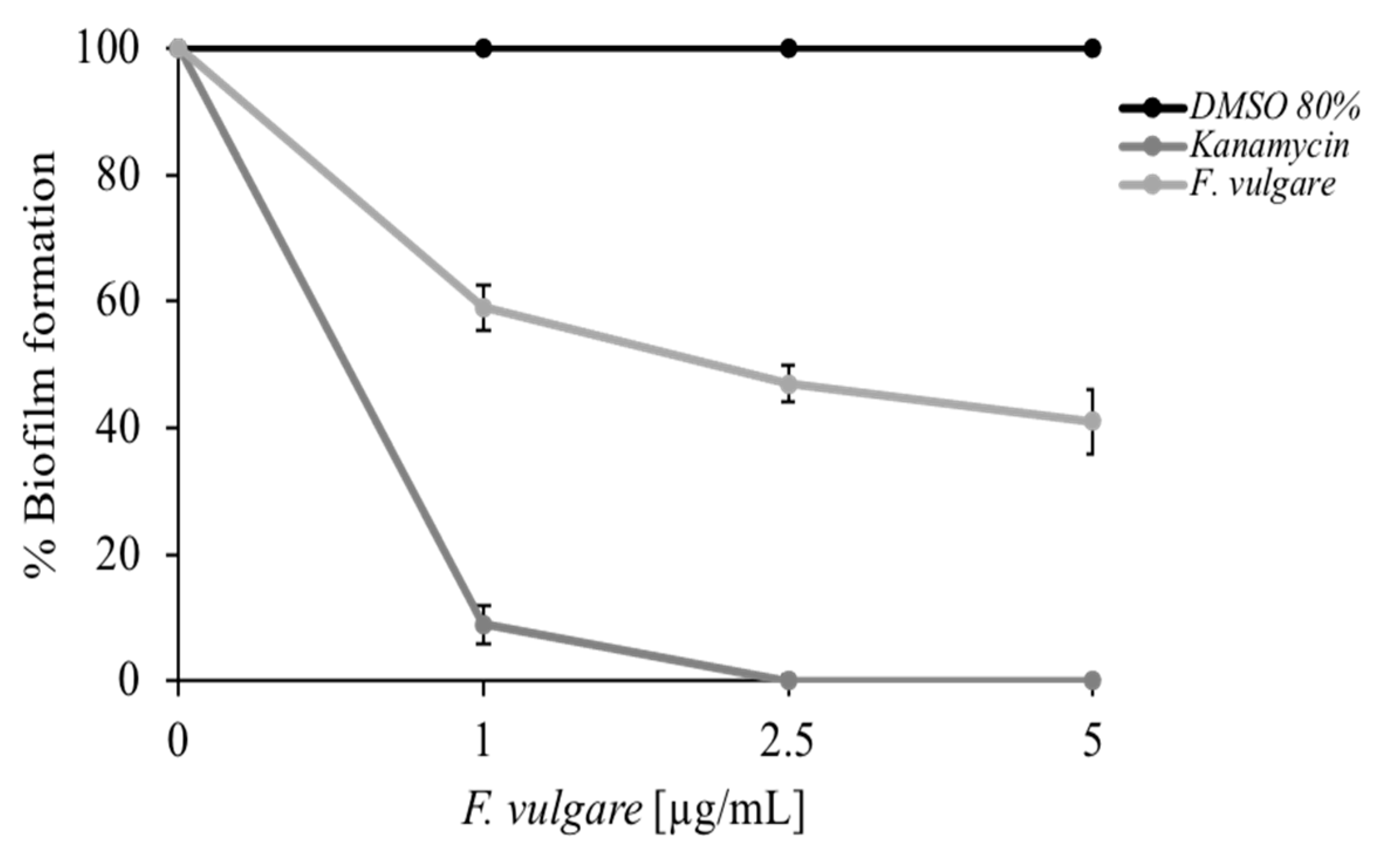
2.3. Antibiofilm Activity of F. vulgare subsp. vulgare var. vulgare EO
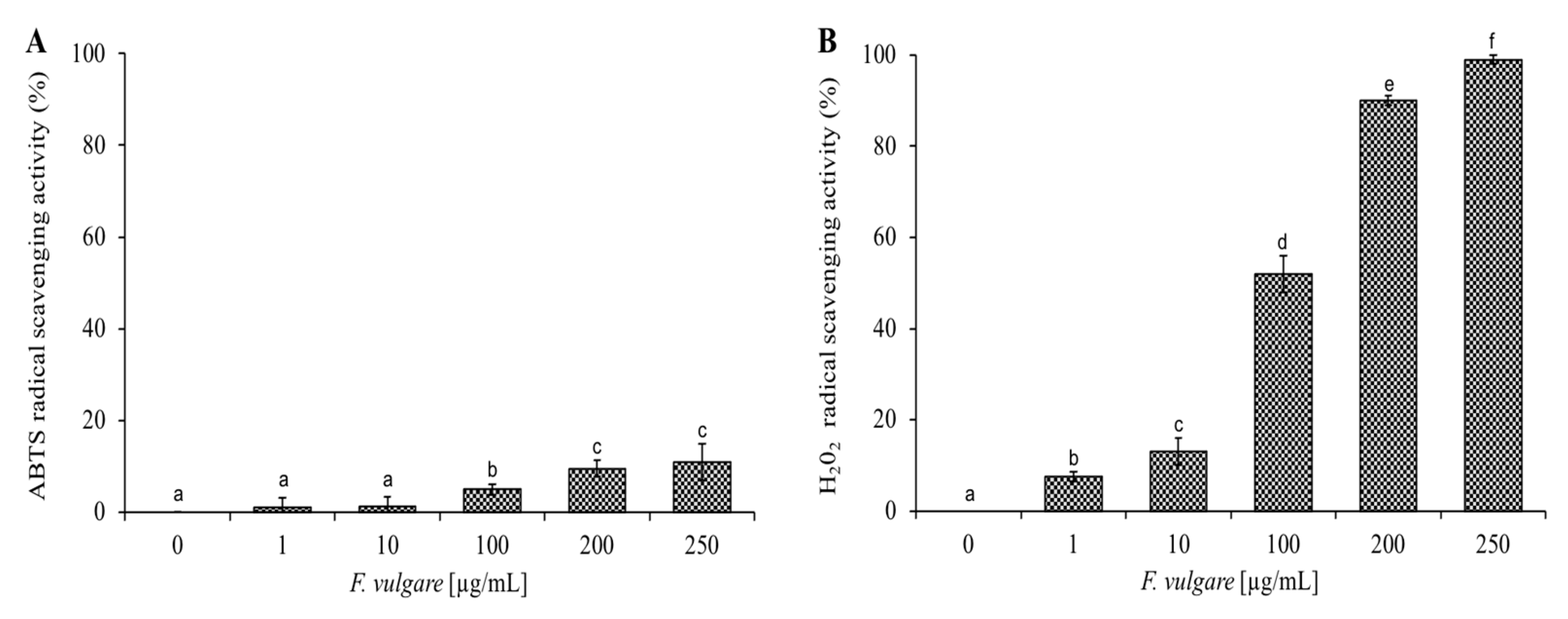
2.4. Antioxidant Activity of F. vulgare subsp. vulgare var. vulgare EO
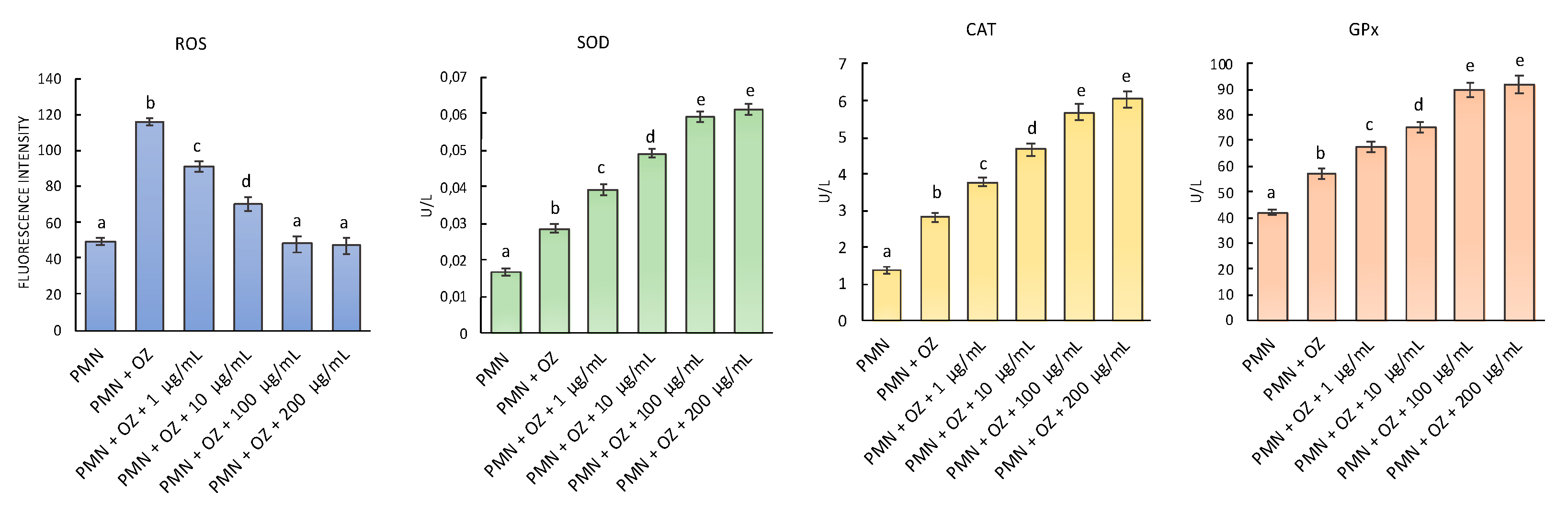
2.5. ROS Generation and Antioxidant Enzymes Activity on Polymorphonuclear Leukocytes (PMN)
3. Materials and Methods
3.1. Plant Material and Isolation of Essential Oil
3.2. GC-MS Analysis
3.3. Bacterial Strains
3.4. Antimicrobial Activity Assay
3.5. Determination of Minimal Inhibitory Concentration
3.6. Antibiofilm Activity Assay
3.7. DAPI/PI Dual Staining and Fluorescence Microscopy Image Acquisition
3.8. ABTS Scavenging Capacity Assay
3.9. Hydrogen Peroxide Scavenging Assay
3.10. Eukaryotic Cell Culture
3.11. ROS Generation and Antioxidant Enzymes Activity on Polymorphonuclear Leukocytes (PMN)
3.12. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Plants of World Online (POWO). Available online: https://powo.science.kew.org/ (accessed on 5 November 2022).
- Sayed-Ahmad, B.; Talou, T.; Saad, Z.; Hijazi, A.; Merah, O. The Apiaceae: Ethnomedicinal family as source for industrial uses. Ind. Crop. Prod. 2017, 109, 661–671. [Google Scholar] [CrossRef] [Green Version]
- Berenbaum, M.R. Evolution of specialization in insect-umbellifer associations. Annu. Rev. Entomol. 1990, 35, 319–343. [Google Scholar] [CrossRef]
- Acìmovìc, M.G.; Kostadinovìc, L.M.; Popovìc, S.J.; Dojcinovìc, N.S. Apiaceae seeds as functional food. J. Agric. Sci. 2015, 60, 237–246. [Google Scholar]
- Badalamenti, N.; Vaglica, A.; Maggio, A.; Bruno, M. A new ferulol derivative isolated from the aerial parts of Ferulago nodosa (L.) Boiss. growing in Sicily (Italy). Nat. Prod. Res. 2022; in press. [Google Scholar] [CrossRef]
- Badalamenti, N.; Vaglica, A.; Ilardi, V.; Bruno, M. The chemical composition of essential oil from Seseli tortuosum subsp. tortuosum and S. tortuosum subsp. maritimum (Apiaceae) aerial parts growing in Sicily (Italy). Nat. Prod. Res. 2022; in press. [Google Scholar] [CrossRef]
- Badalamenti, N.; Vaglica, A.; Ilardi, V.; Bruno, M. The chemical composition of essential oil from Seseli bocconei (Apiaceae) aerial parts growing in Sicily (Italy). Nat. Prod. Res. 2022; in press. [Google Scholar] [CrossRef]
- Badalamenti, N.; Modica, A.; Ilardi, V.; Bruno, M.; Maresca, V.; Zanfardino, A.; Di Napoli, M.; Castagliuolo, G.; Varcamonti, M.; Basile, A. Daucus carota subsp. maximus (Desf.) Ball from Pantelleria, Sicily (Italy): Isolation of essential oils and evaluation of their bioactivity. Nat. Prod. Res. 2022, 36, 5842–5847. [Google Scholar] [CrossRef]
- D’Agostino, G.; Giambra, B.; Palla, F.; Bruno, M.; Badalamenti, N. The application of the essential oils of Thymus vulgaris L. and Crithmum maritimum L. as biocidal on two Tholu Bommalu Indian leather puppets. Plants 2021, 10, 1508. [Google Scholar] [CrossRef]
- Badalamenti, N.; Bruno, M.; Gagliano Candela, R.; Maggi, F. Chemical composition of the essential oil of Elaeoselinum asclepium (L.) Bertol subsp. meoides (Desf.) Fiori (Umbelliferae) collected wild in Central Sicily and its antimicrobial activity. Nat. Prod. Res. 2022, 36, 789–797. [Google Scholar] [CrossRef]
- Badalamenti, N.; Ilardi, V.; Rosselli, S.; Bruno, M.; Maggi, F.; Leporini, M.; Falco, T.; Loizzo, M.R.; Tundis, R. Ferulago nodosa subsp. geniculata (Guss.) Troia & Raimondo from Sicily (Italy): Isolation of essential oil and evaluation of its bioactivity. Molecules 2020, 25, 3249. [Google Scholar]
- Badalamenti, N.; Ilardi, V.; Rosselli, S.; Bruno, M. The ethnobotany, phytochemistry and biological properties of genus Ferulago—A review. J. Ethnopharmacol. 2021, 27, 114050. [Google Scholar] [CrossRef] [PubMed]
- Lauricella, M.; Maggio, A.; Badalamenti, N.; Bruno, M.; D’Angelo, G.D.; D’Anneo, A. Essential oil of Foeniculum vulgare subsp. piperitum fruits exerts an anti-tumor effect in triple-negative breast cancer cells. Mol. Med. Rep. 2022, 26, 243. [Google Scholar] [CrossRef] [PubMed]
- Badalamenti, N.; Ilardi, V.; Bruno, M.; Pavela, R.; Boukouvala, M.C.; Kavallieratos, N.G.; Maggi, F.; Canale, A.; Benelli, G. Chemical composition and broad-spectrum insecticidal activity of the flower essential oil from an ancient Sicilian food plant, Ridolfia segetum. Agriculture 2021, 11, 304. [Google Scholar] [CrossRef]
- Basile, S.; Badalamenti, N.; Riccobono, O.; Guarino, S.; Ilardi, V.; Bruno, M.; Peri, E. Chemical composition and evaluation of insecticidal activity of Calendula incana subsp. maritima and Laserpitium siler subsp. siculum essential oils against stored products pests. Molecules 2022, 27, 588. [Google Scholar] [CrossRef]
- Plants of the World Online (POWO). Available online: https://powo.science.kew.org/taxon/urn:lsid:ipni.org:names:842680-1 (accessed on 10 December 2022).
- Ilardi, V.; Badalamenti, N.; Bruno, M. Chemical composition of the essential oil from different vegetative parts of Foeniculum vulgare subsp. piperitum (Ucria) Coutinho (Umbelliferae) growing wild in Sicily. Nat. Prod. Res. 2022, 36, 3587–3597. [Google Scholar] [CrossRef] [PubMed]
- Diaaz-Maroto, M.C.; Pea rez-Coello, M.S.; Esteban, J.; Sanz, J. Comparison of the volatile composition of wild fennel samples (Foeniculum vulgare Mill.) from Central Spain. J. Agric. Food Chem. 2006, 54, 6814–6818. [Google Scholar] [CrossRef] [PubMed]
- Rather, M.A.; Dar, B.A.; Sofi, S.N.; Bhat, B.A.; Qurishi, M.A. Foeniculum vulgare: A comprehensive review of its traditional use, phytochemistry, pharmacology, and safety. Arab. J. Chem. 2012, 9, S1574–S1583. [Google Scholar] [CrossRef] [Green Version]
- Diaaz-Maroto, M.C.; Hidalgo, I.J.D.; Saanchez-Palomo, E.; Pearez-Coello, M.S. Volatile components and key odorants offennel (Foeniculum vulgare Mill.) and thyme (Thymus vulgaris L.) oil extracts obtained by simultaneous distillation–extraction and supercritical fluid extraction. J. Agric. Food Chem. 2005, 53, 5385–5389. [Google Scholar] [CrossRef]
- Mohsenzadeh, M. Evaluation of antibacterial activity of selected Iranian essential oils against Staphylococcus aureus and Escherichia coli in nutrient broth medium. Pak. J. Biol. Sci. 2007, 10, 3693–3697. [Google Scholar]
- Dadalioglu, I.; Evrendilek, G.A. Chemical compositions and antibacterial effects of essential oils of Turkish oregano (Origanum minutiflorum), bay laurel (Laurus nobilis), Spanish lavender (Lavandula stoechas L.), and fennel (Foeniculum vulgare) on common foodborne pathogens. J. Agric. Food Chem. 2004, 52, 8255–8260. [Google Scholar] [CrossRef]
- Cantore, P.L.; Iacobelli, N.S.; Marco, A.D.; Capasso, F.; Senatore, F. Antibacterial activity of Coriandrum sativum L. and Foeniculum vulgare Miller var. vulgare (Miller) essential oils. J. Agric. Food Chem. 2004, 52, 7862–7866. [Google Scholar] [CrossRef] [PubMed]
- Abed, K.F. Antimicrobial activity of essential oils of some medicinal plants from Saudi Arabia. Saudi J. Biol. Sci. 2007, 14, 53–60. [Google Scholar]
- Shahat, A.A.; Ibrahim, A.Y.; Hendawy, S.F.; Omer, E.A.; Hammouda, F.M.; Rahman, F.H.A.; Saleh, M.A. Chemical composition, antimicrobial and antioxidant activities of essential oils from organically cultivated fennel cultivars. Molecules 2011, 16, 1366–1377. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El-Soud, N.A.; El-Laithy, N.; El-Saeed, G.; Wahby, M.S.; Khalil, M.; Morsy, F.; Shaffie, N. Antidiabetic activities of Foeniculum vulgare Mill. Essential oil in Streptozotocin induced diabetic rats. Maced. J. Med. Sci. 2011, 173, 1857–5773. [Google Scholar]
- Lee, S.H. Acaricidal activity of constituents identified in Foeniculum vulgare fruit oil against Dermatophagoides spp. (Acari: Pyroglyphidae). J. Agric. Food Chem. 2004, 52, 2887–2889. [Google Scholar] [CrossRef]
- Araque, M.; Rojas, L.B.; Usubillaga, A. Antimicrobial activities of essential oil of Foeniculum vulgare Miller agains multiresistant gram negative Bacillus from nosocomial infections. Ciencia 2007, 15, 366–370. [Google Scholar]
- Serviï, H.; Şen, A.; Serviï, E.Y.; Doğan, A. Chemical Composition and Biological Activities of Essential Oils of Foeniculum vulgare Mill. and Daucus carota, L. Growing Wild in Turkey. J. Res. Pharm. 2021, 25, 142–152. [Google Scholar] [CrossRef]
- Esquivel-Ferriño, P.C.; Favela-Hernández, J.M.J.; Garza-González, E.; Waksman, N.; Ríos, M.Y.; Camacho-Corona, M. del R. Antimycobacterial Activity of Constituents from Foeniculum vulgare Var. Dulce Grown in Mexico. Molecules 2012, 17, 8471–8482. [Google Scholar] [CrossRef]
- Martins, M.R.; Tinoco, M.T.; Almeida, A.S.; Cruz-Morais, J. Chemical composition, antioxidant and antimicrobial properties of three es- sential oils from portuguese flora. J. Pharmacogn. 2012, 3, 39–44. [Google Scholar]
- Ruberto, G.; Baratta, M.T.; Deans, S.G.; Dorman, H.J.D. Antioxidant and Antimicrobial Activity of Foeniculum vulgare and Crithmum maritimum Essential Oils. Planta Med. 2000, 66, 687–693. [Google Scholar] [CrossRef]
- Conner, D.E. Naturally Occurring Compounds. In Antimicrobials in Foods; Davidson, P., Branen, A.L., Eds.; Marcel Dekker Publishing Company: New York, NY, USA, 1991. [Google Scholar]
- Gulfraz, M.; Mehmood, S.; Minhas, N.; Jabeen, N.; Kausar, R.; Jabeen, K.; Arshad, G. Composition and antimicrobial properties of essential oil of Foeniculum vulgare. Afr. J. Biotechnol. 2008, 7, 4364–4368. [Google Scholar]
- Sochorová, Z.; Petráčková, D.; Sitařová, B.; Buriánková, K.; Bezoušková, S.; Benada, O.; Kofroňová, O.; Janeček, J.; Halada, P.; Weiser, J. Morphological and proteomic analysis of early stage air-liquid interface biofilm formation in Mycobacterium smegmatis. Microbiology 2014, 160, 1346–1356. [Google Scholar] [CrossRef] [Green Version]
- Mishra, R.; Panda, A.K.; De Mandal, S.; Shakeel, M.; Bisht, S.S.; Khan, J. Natural anti-biofilm agents: Strategies to control biofilm-forming pathogens. Front. Microbiol. 2020, 11, 566325. [Google Scholar] [CrossRef] [PubMed]
- Badalamenti, N.; Maresca, V.; Di Napoli, M.; Bruno, M.; Basile, A.; Zanfardino, A. Chemical composition and biological activities of Prangos ferulacea essential oils. Molecules 2022, 27, 7430. [Google Scholar] [CrossRef] [PubMed]
- Ruberto, G.; Baratta, M.T. Antioxidant activity of selected components in two lipid model systems. Food Chem. 2000, 169, 167–174. [Google Scholar] [CrossRef]
- Determination of Essential Oils in Herbal Drugs, 2.8.12. In European Pharmacopoeia 6.0; The Stationery Office/Tso: London, UK, 2008; pp. 251–252. Available online: http://www.sifit.org/r/pdf/english_index_84.pdf (accessed on 14 November 2022).
- Rigano, D.; Formisano, C.; Rosselli, S.; Badalamenti, N.; Bruno, M. GC and GC-MS analysis of volatile compounds from Ballota nigra subsp. uncinata collected in Aeolian Islands, Sicily (Southern Italy). Nat. Prod. Commun. 2020, 15, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Bauer, A.W.; Kirby, W.M.; Sherris, J.C.; Turck, M. Antibiotic susceptibility testing by a standardized single disk method. Am. J. Clin. Pathol. 1966, 45, 493–496. [Google Scholar] [CrossRef]
- Di Napoli, M.; Varcamonti, M.; Basile, A.; Bruno, M.; Maggi, F.; Zanfardino, A. Anti-Pseudomonas aeruginosa activity of hemlock (Conium maculatum, Apiaceae) essential oil. Nat. Prod. Res. 2019, 33, 3436–3440. [Google Scholar] [CrossRef]
- Badalamenti, N.; Russi, S.; Bruno, M.; Maresca, V.; Vaglica, A.; Ilardi, V.; Zanfardino, A.; Di Napoli, M.; Varcamonti, M.; Cianciullo, P.; et al. Dihydrophenanthrenes from a Sicilian accession of Himantoglossum robertianum (Loisel.) P. delforge showed antioxidant, antimicrobial, and antiproliferative pctivities. Plants 2021, 10, 2776. [Google Scholar] [CrossRef]
- Prencipe, F.; Zanfardino, A.; Di Napoli, M.; Rossi, F.; D’Errico, S.; Piccialli, G.; Mangiatordi, G.F.; Saviano, M.; Ronga, L.; Varcamonti, M.; et al. Silver (I) n-heterocyclic carbene complexes: A winning and broad spectrum of antimicrobial properties. Int. J. Mol. Sci. 2021, 22, 2497. [Google Scholar] [CrossRef]
- Di Napoli, M.; Maresca, V.; Varcamonti, M.; Bruno, M.; Badalamenti, N.; Basile, A.; Zanfardino, A. (+)-(E)-Chrysanthenyl acetate: A molecule with interesting biological properties contained in the Anthemis secundiramea (Asteraceae) Flowers. Appl. Sci. 2020, 10, 6808. [Google Scholar] [CrossRef]
- Di Napoli, M.; Maresca, V.; Sorbo, S.; Varcamonti, M.; Basile, A.; Zanfardino, A. Proteins of the fruit pulp of Acca sellowiana have antimicrobial activity directed against the bacterial membranes. Nat. Prod. Res. 2021, 35, 2942–2946. [Google Scholar] [CrossRef] [PubMed]
- Napolitano, A.; DiNapoli, M.; Castagliuolo, G.; Badalamenti, N.; Cicio, A.; Bruno, M.; Piacente, S.; Maresca, V.; Cianciullo, P.; Capasso, L.; et al. The chemical composition of the aerial parts of Stachys spreitzenhoferi (Lamiaceae) growing in Kythira Island (Greece), and their antioxidant, antimicrobial, and antiproliferative properties. Phytochemistry 2022, 203, 113373. [Google Scholar] [CrossRef]
- Beers, R.; Sizer, I. A spectrophotometric method for measuring the breakdown of hydrogen peroxide by catalase. J. Biol. Chem. 1952, 195, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Vitiello, G.; Zanfardino, A.; Tammaro, O.; Di Napoli, M.; Caso, M.F.; Pezzella, A.; Varcamonti, M.; Silvestri, B.; D’Errico, G.; Costantini, A.; et al. Bioinspired hybrid eumelanin-TiO2 antimicrobial nanostructures: The key role of organo-inorganic frameworks in tuning eumelanin’s biocide action mechanism through membrane interaction. RSC Adv. 2018, 8, 28275–28283. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zanfardino, A.; Bosso, A.; Gallo, G.; Pistorio, V.; Di Napoli, M.; Gaglione, R.; Dell’Olmo, E.; Varcamonti, M.; Notomista, E.; Arciello, A.; et al. Human apolipoprotein E as a reservoir of cryptic bioactive peptides: The case of ApoE 133-167. J. Pept. Sci. 2018, 24, e3095. [Google Scholar] [CrossRef]
- Harbeck, R.J.; Hoffman, A.A.; Redecker, S.; Biundo, T.; Kurnick, J. The isolation and functional activity of polymorphonuclear leukocytes and lymphocytes separated from whole blood on a single Percoll density gradient. Clin. Immunol. Immunolpathol. 1982, 23, 682–690. [Google Scholar] [CrossRef]
- Manna, A.; Saha, P.; Sarkar, A.; Mukhopadhyay, D.; Bauri, A.K.; Kumar, D.; Das, P.; Chattopadhyay, S.; Chatterjee, M.; Gartel, A.L. Malabaricone-A induces a redox imbalance that mediates apoptosis in u937 cell line. PLoS ONE 2012, 7, e36938. [Google Scholar] [CrossRef]
- Barbosa, P.O.; Pala, D.; Silva, C.T.; Oliveira de Souza, M.; Ferreira do Amaral, J.; Lima Vieira, R.A.; Andrezza de Freitas Folly, G.; Pinheiro Volp, A.C.; Nascimento de Freitas, R. Açai (Euterpe oleracea Mart.) pulp dietary intake improves cellular antioxidant enzymes and biomarkers of serum in healthy women. Nutrition 2016, 32, 674–680. [Google Scholar] [CrossRef]
| Strains | MIC100 [µg/mL] |
|---|---|
| E. coli DH5α | 250 |
| P. aeruginosa PAO1 ATCC15692 | >250 |
| S. Typhimurium ATCC14028 | >250 |
| S. aureus ATCC6538P | 250 |
| M. smegmatis MC2155 | >250 |
| B. cereus ATCC10987 | 250 |
| Sample | IC50 of ABTS (µg/mL) | Sample | IC50 of H2O2 (µg/mL) |
|---|---|---|---|
| F. vulgare subsp. vulgare var. vulgare | >1000 | F. vulgare subsp. vulgare var. vulgare | 100 |
| Ascorbic acid | 0.03 | Resveratrol | 0.05 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Napoli, M.; Castagliuolo, G.; Badalamenti, N.; Maresca, V.; Basile, A.; Bruno, M.; Varcamonti, M.; Zanfardino, A. Antimicrobial, Antibiofilm, and Antioxidant Properties of Essential Oil of Foeniculum vulgare Mill. Leaves. Plants 2022, 11, 3573. https://doi.org/10.3390/plants11243573
Di Napoli M, Castagliuolo G, Badalamenti N, Maresca V, Basile A, Bruno M, Varcamonti M, Zanfardino A. Antimicrobial, Antibiofilm, and Antioxidant Properties of Essential Oil of Foeniculum vulgare Mill. Leaves. Plants. 2022; 11(24):3573. https://doi.org/10.3390/plants11243573
Chicago/Turabian StyleDi Napoli, Michela, Giusy Castagliuolo, Natale Badalamenti, Viviana Maresca, Adriana Basile, Maurizio Bruno, Mario Varcamonti, and Anna Zanfardino. 2022. "Antimicrobial, Antibiofilm, and Antioxidant Properties of Essential Oil of Foeniculum vulgare Mill. Leaves" Plants 11, no. 24: 3573. https://doi.org/10.3390/plants11243573