A Comprehensive Evaluation of Studies on the Adverse Effects of Medications in Australian Aged care Facilities: A Scoping Review
Abstract
1. Introduction
- What are the types of adverse effects identification tools currently used in Australian health care settings (aged care facilities and hospitals)?
- What evaluation outcome measures have been reported for the tools in primary care settings?
- Does the tool or protocol minimise the adverse effects of medications without compromising the benefits of medications?
- Do the tools improve patients’ clinical outcomes by identifying inappropriate medication prescribed or medication errors?
- Do the tools or protocols support multi-disciplinary interventions through optimising day-to-day patient care?
- The study was intended for patients aged 65 years or older;
- The study included older patients who experienced adverse effects of medications;
- The study included older patients suffering from the adverse effects of polypharmacy and living in aged care facilities or admitted in hospitals;
- The study investigated tools that were/are currently being used in Australia.
- No data on the adverse effects;
- Study included paediatrics and we were unable to separately extract paediatric data;
- Duplicated studies;
- Study included only a single medication;
- Study population only included adults that were younger than 65 years;
- Primary objective was not the adverse effects of medications;
- Studies not in English;
- Studies focused on experimental medicines;
- Studies in phase III of a clinical trial.
- Model designs of the study were insufficiently described;
- Validation of tests were ambiguous;
- Designs and measures were not detailed;
- It was indeterminate as to whether the measure has been accepted in practice.
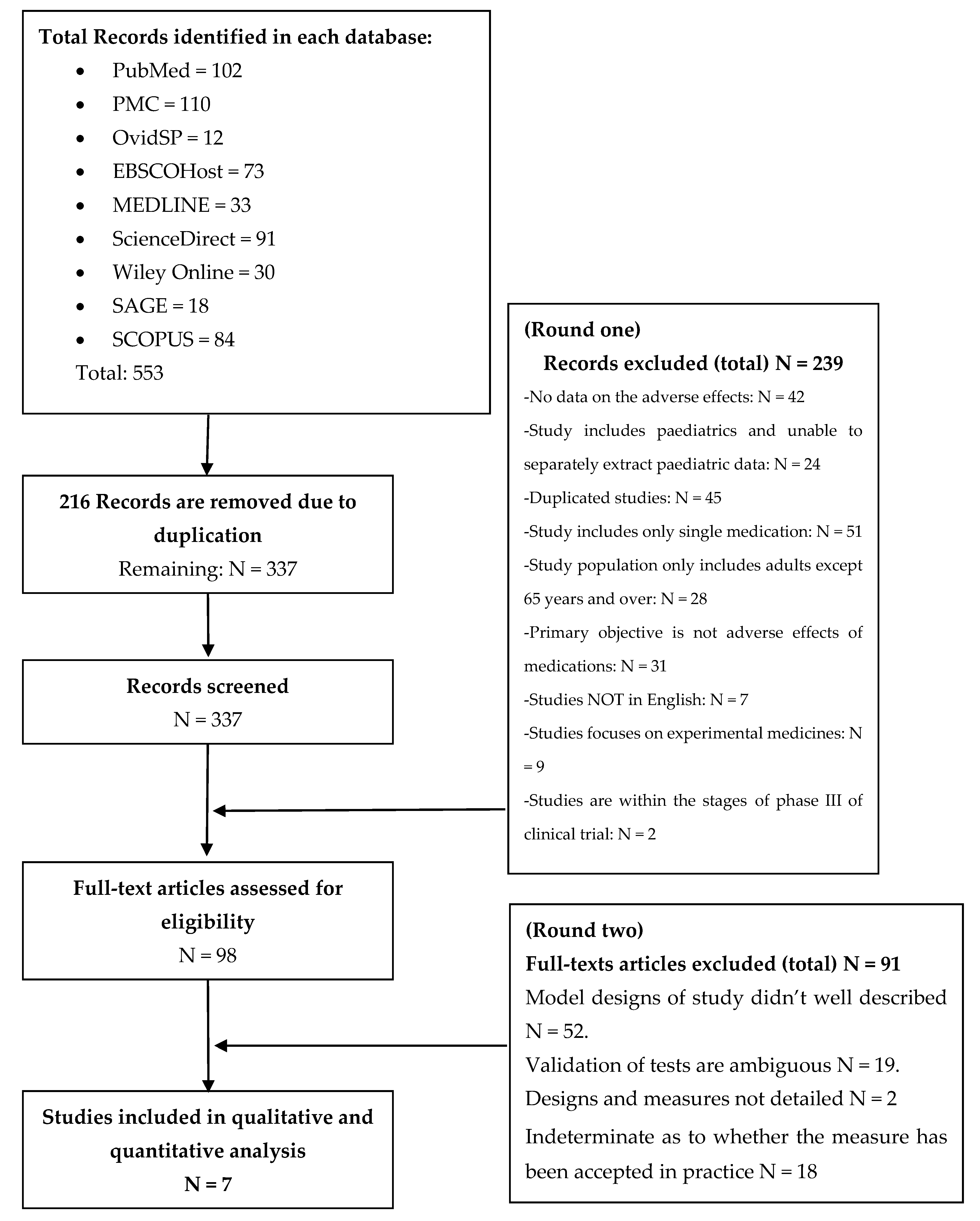
2. Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A. Search Strategies
- Medication adverse effects;
- Elderly [all] OR Senior OR Older people [all] OR People aged over 65 OR [all] Geriatric OR Old;
- Aging OR [all] Ageing OR Veteran [all] OR Older age;
- Older people OR [all] older people OR [all] oldest people Older population;
- Aged Care Facilities OR [all] Aged Care OR aged care OR [all] Nursing Home OR nursing facilities for aged care OR [all] nursing home OR Senior care [all] OR Care for older people OR [all] Care for advanced age;
- Medication monitoring OR [all] Drugs monitor OR [all] Medicine monitor OR [all] medicine monitoring OR [all] therapy monitor OR curative monitoring OR [all] treatment assessment OR [all] therapy assessment OR remedy monitor [all] OR medication observation OR medication tracking OR [all] medicine records;
- Drugs review OR Medication review OR [all] therapy regimen review OR [all] Medication check OR drugs check OR [all] medication rehearsal OR [all] medication revision OR [all] drugs revision OR [all] medication reassess OR [all] drugs reassessment OR [all] medication regimen appraisal OR [all] medication evaluation OR [all] drugs evaluation;
- Adverse effects assessment OR [all] medication harm assessment OR [all] adverse effects revision OR [all] adverse effects evaluation OR [all] adverse effects evaluation OR [all] adverse effects estimation OR [all] adverse effects judgment OR [all] unwanted medication effects OR undesirable adverse effects OR [all] harmful medication effects OR [all] unfavourable drugs effects OR [all] pernicious drugs effects;
- In Australia OR [all] In Australian heath care system OR [all] in Australian health setting;
- #1 OR #3 and #5 and #6 and #7 and #8 OR 9# [all];
- #2 OR #3 and #5 and #6 and #7 and #8 OR 10# [all];
- #3 OR #4 and #5 and #6 and #7 and #8 OR 11# OR 12# [all].
| Included Study | Setting Research |
|---|---|
| Harrison et al., 2018—EuroQol questionnaire and DQL questionnaire study [8] | Australia aged care facilities and nursing home |
| Turner et al., 2016—FRIDs study [9] | Australian public hospitals and multi-disciplinary clinics |
| Basger et al., 2008—IMU-PI tool [10] | Australian health database |
| Ashoorian et al., 2015—M3Q Tool [11] | Australian mental health clinics and public hospitals |
| Lee et al., 2017—M-DRAW tool [12] | American-based study and used by Australian HMR †/RMMR ‡ Pharmacists |
| McLeod et al., 1997—McLeod Tool [13] | Canadian-based study and used by Australian HMR †/RMMR ‡ Pharmacists |
| Nishtala et al., 2009—DBI study [14] | Australian aged care facilities and nursing home |
| Tool | Current Use in Practice | Used by | Evaluation | When not Used and Why? | Limitations in Practice |
|---|---|---|---|---|---|
| DBI study (Nishtala et al. 2009) | 62 aged care facilities in NSW. Determine DBI scores in older people in aged care homes; and evaluate the impact of RMMR on DBI score after uptake of pharmacist recommendations by GPs | Consultant Pharmacists in community and hospital settings, HMR† and RMMR‡ accredited pharmacists | N = 500 residents, SD of age = 84.0 years, 25% male. SD for medications per resident = 7.4, SD for anticholinergic and sedative = 0.9 & 0.2 respectively. Reduction in prescribed anticholinergic and sedative medications can be achieved in older people through using DBI. | DBI is a formula designed to measure the adverse effects of anticholinergic and sedative medication on the quality of life. A higher DBI score represents a lower quality of life. DBI is not a tool for frequent use. It provides a reference for developing a RMMR report and subsequent pharmacist recommendations to GPs and nursing staff | This tool did not take into account differential pharmacokinetic properties of medications. No indication for drug–drug interactions provided and no pharmacodynamic profiles among aged care home residents are developed. No questionnaire; DBI calculations estimated as a liner dose-response relationship between drug classes. Predictive capacity of DBI not established. In this study the residents were not randomised into the intervention and control groups. This tool was applied retrospectively limiting any establishment of causality. No information about their health status or their disease severity was included. |
| INSPIRED study (Harrison et al. 2018) | Cross-sectional study: analysis of 541 individuals recruited from 17 aged care facilities in Australia (from NSW, QLD, SA, WA) | Nurses and carers in aged care facilities. This study was specific to older people living with cognitive impairment and dementia. | With respect to anticholinergic and sedative medications adverse effects, the PIM (Beers)§ criteria and DBI were highly prevalent in residential aged care at 73% and 83.1% respectively. Study confirmed higher exposure to these medications in inappropriate prescriptions were associated with a lower quality of life. | This study does not present a new tool. It is a comparison between DBI and PIM (Beer’s criteria) to determine whether these tools are associated with quality of life in older adults living in aged care facilities. It was only used in those with cognitive impairment and not for other medical conditions. | This study was unable to assess causality or the direction of any observed associated issues. In addition, these is no certainty of compatibility between the proxy measures that were used and what the individual would self-report if they able to do so. |
| FRIDs study Turner et al. 2016 | Tertiary referral hospital in geriatric oncology outpatient multidisciplinary clinic. | Administrated by nurses, geriatricians, medical oncologists, geriatric oncology nurse, social workers, dietician, pharmacists, occupational therapists, and palliative care nurses. | Cohort study of older people with cancer. All data in this study verified by nurses with full access to patients’ medical records. Enabled inclusion of any omitted data to be collected. There was 79% concordance for self-reported prescribed medications compared with those obtained in an interview with clinical pharmacists in hospital wards. | Study limited to older people newly diagnosed with cancer, and previous history of falls / or orthostatic hypotension, and administrating psychotropic medications. Not applicable to older people administered psychotropic medications. | Single site data collection and not generalisable to other settings. Some patients did not know what fall was, others did not remember having fallen or they underreported the number of falls (if they fell several times). Not possible to determine if FRIDs study used at the time of fall or initiated after fall. In addition, the number of older people who received more than 3 prescribed medications of antipsychotic was small. These factors impacted the results of the adjusted multi-variate regression analysis giving wider confidence intervals. |
| IMU-PI tool Basger et al. 2008 | Study tool design informed by expert’s review, international literatures, and clinical practice guidelines for medication use in elderly. The tool used with Australian heathcare system data and cross-referenced with treatment of common medical conditions for those with the highest volume of Australian Pharmaceutical Benefits Scheme usage in 2006 and 2007. | Experts from University of Sydney, NSW | Tool design is similar to Beers and McLeod tool. This tool had set out to develop an indicator list relevant to Australia the design did not involve an expert consensus process. Instead the tool was based on Australian healthcare data. Indicators had been selected from analysis of the most commonly dispensed PBS medications and based on the most common conditions for older people receiving medical care. | This study is NOT a specific tool or questionnaire used in age care facilities. This study performed only by collection of PBS data within only a two-year window. As a result, this tool has no ability to determine or detect the adverse effects of medications nor be used in any aged care facility. | The tool was not validated yet. This tool was not designed to act as a preventative health tool to avoid adverse events. It indicates that either appropriate or inappropriate medication has been prescribed. |
| M3Q Tool Ashoorian et al. 2015 | Six public mental health clinics and one hospital in WA; 205 participants divided into intervention and control groups. | Nurses in mental health clinics | M3Q was designed specifically to assess the effects of antidepressants, antipsychotics, anxiolytic and mood stabilizers. This tool was developed to fill the gaps of lack communication between clinicians and patients. It contains closed and open response questions. It has been through rigorous validation processes; expert focus groups developed the design and psychometric testing. Focuses on patient’s list of self-reported medications and dose and they rank three bothersome side effects. Checklist of 32 possible side effects under 11 domains. | M3Q tool not applicable for older people suffering from other co-morbidities. The assessment of the psychotropic medication side effects does not reflect the reality of comorbidities and increases risk of inaccuracy. | This tool was not designed to objectively record the accurate number of psychotropic medications and their side effects. No statistically significant change was demonstrated within each group. M3Q tool was used a non-randomized convenience sample of patients. Many patients suffered other co-morbidities and were taking a number of medications not related to psychotropic medications or mental illness which may confound the assessment of side effects by clinician. A wider cross-section of patients attending GPs, pharmacies and wider representations would be worthwhile. |
| M-DRAW Tool Lee at al. 2017 | Academic medical centre pharmacy in California-USA. | Pharmacists, nurses, social workers, and patient’s carers. | M-DRAW uses a motivational interview-based intervention strategy for each identified barrier. M-DRAW provides recommendations to clinicians on how to systematically approach follow-up for each identified barrier, and also identify the root cause of non-adherence. | This tool has been designed only for identifying barriers of medication adherence. It consists of a 13-item checklist questionnaire, and the results of this tool is scaled from 1 = never to 4 = often. | The limitation of this study was small sample size which limits generalisation. Test and re-test reliability were NOT performed, short duration study, follow-up items were not well defined. No specific illness dealt with; any chronic conditions. This tool assesses only non-intentional and intentional non-adherence of medications. This tool is applicable for a pharmacist conducting RMMR for medication-adherence assessment only. |
| McLeod Tool McLeod et al. 1997 | Academic medical centres across Canada. | Pharmacists, doctors’ specialists, nurses, GPs, geriatricians. | New approach to identify inappropriate practice in prescribing medication for older people. This study has a list of 71 inappropriate practices in prescription for older people, and each practice rated from 1—not significant to 4 high significance. 3 major categories: drug contraindicated, drug-disease interactions and drug–drug interactions. The recommendation for each item could be generalizable. | This tool developed by Beers and collaborators resulting in considerable similarity between this tool and Beers criteria. This tool will be helpful for medication reviewing and preparing recommendations to GPs for consideration. | This study has no specific questionnaire and requires no interview with patients. It was designed only for detecting frequent inappropriate prescriptions for older people. The recommendation for each item was general with no further details or explanation. |
| Author | Location | No. of Patients | No. of Drugs | Drugs Most Frequently Associated with Outcomes | Validation | Most Frequent Body System Affected by ADRs | Selection was not Biased | Acceptability Low Rates of Loss to Follow-up | Blinding Outcome |
|---|---|---|---|---|---|---|---|---|---|
| Nishtala et al, 2009 | Australia—database reported ADRs of psychiatric medications collected from TGA, PBS, health care professional (including hospitals and aged care facilities), consumers. | 150475 (6751 cases and 123334 non-cases) Case is a report that include 1 or more neuropsychiatric ADRs. The non-case is a report that does not include any neuropsychiatric medication | Benzodiazepines, Anticholinergics, TCAs, and other 24 medications (CVDs, neurological, and pain management). | The following medications are results of 95% Cl for older + drug/older-drug. These medications producing more ADRs effects with older people than younger people: Cimetidine 2.24(1.7–3.0); Anticholinergic drugs 3.12(2.53-3.85); Antipsychotics 2.73(2.21–3.37); TCAs 2.31(1.93–2.77). | ADRs reports were validated by inclusion and exclusion criteria. Reports were excluded from the analysis if data for age or DOB were absent. Also, a combination of drugs including drug of interest had excluded as well from analysis. The 25 drugs of interest identified and assessed by Tune and coworkers. | CNS with major reports of agitation, anxiety, cognitive impairment, confusion, delirium, hallucinations, psychosis | The selection of report based on CNS signs such as history of hallucination, anxiety, agitation, depression, delirium and cognitive impairment. The association observed between drug exposure to the observed outcome may have been biased or distorted. Also, confounding by concomitant drug use gives concern of bias. In addition, the ADRs database are consist of reported adverse events information, thus subject to differential reporting are clearly biases. However, the authors minimised bias in this study by applying the drug of interest were not typically viewed as possessing anticholinergic characters. Furthermore, all reporting and coding had included in the analysis (not just those drugs that were coded as suspect drug for the reactions). | Not mentioned | Blinding was not reported in any stage. |
| Harrison et al, 2018 | Australia—cross-sectional analysis of 541 older people recruited from 17 residential aged care facilities around Australia. | 541 | The criteria regarding drugs of interest based on Beers criteria and PIMs for all older people exposed for more than 8 weeks: PPIs 41.5%, Benzodiazepine 30.5%, Antipsychotics 24.8%, Antidepressants (mirtazapine 17.1%, sertraline 9.5%, escitalopram 8.6%, citalopram 7.1%), and Opioids (buprenorphine 14.3%, fentanyl 9.7%, oxycodone 8.2%). Benzodiazepine 9.9%. Antipsychotics (risperidone 12.7%). | Benzodiazepine, antipsychotics, antidepressants, and opioids | PIMs identified in this study by using validated measures of Beers criteria for older people. The facilities candidates have characteristic-levels were determined from information collected in a standardised questionnaire that was validated in older residential care population. This questionnaire includes 33 questions (asked about facility-level, location, No. of direct care hours per resident, size of facility, age, sex, marital status). The measures of EQ-5D-5L which completed by proxy has been validated in residential living in aged care facilities with dementia. | CNS and musculoskeletal system | Not mentioned | Not mentioned | Not mentioned |
| Turner at al, 2016 | Australia, cross-sectional study at referral hospitals in Adelaide, geriatric oncology outpatients clinics. | 383 | Psychotropics, opioids, anxiolytics, hypnotics, sedatives, antidepressant, vasodilators in cardiac diseases, antihypertensives, diuretics, B-blockers, CCB, Renin-angiotensin system inhibitors, Alpha-antagonists, Dopaminergic agents. | Psychotropics, opioids, anxiolytics, hypnotics, sedatives, antidepressant, vasodilators in cardiac diseases, antihypertensives, diuretics, B-blockers, CCB, Renin-angiotensin system inhibitors, Alpha-antagonists, Dopaminergic agents. | This study is well-characterized cohort for older people with cancer. The validation stated from the initial appointment, and all data contained within the structured collection sheet which verified by nurses have access to participant’s medical records to allow any omitted data to be collected. Also, the validation of this study found that 77% concordance for self-reported prescription medication use when compared with participants obtained in an interview conducted by clinical pharmacist, which also comparable with medication has been taken routinely in hospital wards. | CNS and CVD | Not mentioned | Not mentioned | Not mentioned |
| Basger et al, 2008 | Australia—cross-referenced treatment of the common medical conditions with the highest 50 PBS-medications prescribed to Australians in 2006. | 50 highest used PBS-prescribed medications / documentations. | Top 50 prescribed medications in 2006 for Australian older people (>65 years old). | ACEI, ARB, Aspirin, B-adrenoceptor antagonists, Biphosphonates, Bupropion, Calcitriol, Calcium, Clopidogrel, Dipyridamole, inhaled corticosteroids, Intravaginal estrogen, Nicotine replacement medications, Paracetamol, Raloxifene, HMG-CoA (statins), Strontium, Teriparatide, Varenicline, Vitamin D, Warfarin. | The indicators of this study need to be tested and validated for relevance. However, the common anticipation is an identification of inappropriate medication use for commonly used medications in elderly Australians. | Heart failure, URI, depression, anxiety, arthritis, back pain, osteoporosis, falls, CVDs, renal impairment, GIT diseases (including GORD and ulcers), Type 2 diabetes mellitus, thyroid and parathyroid disorders, hepatic impairments, asthma and COPD, coagulation disorders. | Not mentioned | Not mentioned | Not mentioned |
| Ashoorian et al, 2015 (M3Q Tool) | Australia–adult people diagnosed with mental health conditions and they taking at least one or more psychotropic medications. Participants data collected from community and clinic public mental health services in west Australia. | 205 patients: >50% male, Mean = 43 years, SD = 13. 73% reported taking multiple psychotropic medications. | All psychotropic PBS approvals | All psychotropic PBS approvals | M3Q tool was validated by provided participants an opportunity to express the impact of psychotropic medications side effects on their lives. Furthermore, the validation pf this tool passed through rigorous process: including eight focus groups with experts’ stakeholders to develop items followed by psychometric testing assessing the validity and reliability of the M3Q questionnaire. | Schizophrenia, bipolar disorder, depression, anxiety. These diseases usually associated with more or more comorbidities | Not mentioned | Follow-up after 3 months from the date of collection. Loss to follow-up had reported as 3 interviews abandoned to answer questions, 2 patients deceased and 14 decline to participate after 3 months from the first interview. | Not mentioned |
| (Lee at al. 2017) M-DRAW Tool | This study designed in USA-California and used in Australia. The study conducted in academic medical centres pharmacy in south California. | 26 | This study is non-drug focused. This study assesses factors contributing to medication non-adherence. | PBS-approved medication prescribed in chronic condition in adult and older people. | The validity been examined by applied pilot study of the psychometric properties of the M-DRAW tool to check the tool’s reliability. The validity of the tool was examined by priming question in 4-fold number of barriers to adherence within the self-selected intervention group and control group. However, confirmed validity not clearly stated because of small sample size and lost follow-up. | CVDs (hypertension and dyslipidaemia), type 2 diabetes, and chronic pain conditions. | Not mentioned | Loss follow-up reported in this study. For this reason, the validity of this study not been completely confirmed. The follow-up assessments were not collected as planned at the initial stage of the study protocol development because of short duration of this study time. | Not mentioned |
| McLeod et al. 1997 (McLeod Tool) | This study designed in Canada and using as a tool of older people I Australia. The participants from health professional of 32 specialities (7 clinical pharmacists, 9 geriatrics, 8 family GPs, and 8 community pharmacists). | 32 health specialties recruited in academic medical centres across Canada. | CVDs drugs, psychotropic drugs, pain management drugs, and other miscellaneous drugs in older people. | B-blockers, AECIs, diuretics, CCB, benzodiazepine, TCA, barbiturate, antipsychotics, NSAIDs, phenylbutazone, warfarin, pentazocine, cimetidine, anticholinergics, antispasmodics, dipyridamole, diphenoxylate, cyclobenzaprine, methocarbamol. | Not mentioned | CVDs (including heart failure), asthma, COPD, mental issues (including dementia and insomnia), back pain, and osteoarthritis. | Not mentioned | Not mentioned | The collected list of inappropriate practice in prescribed medication underwent modifications before it is used in double-blinded controlled trial of a computer-based intervention for improving prescribing for older people. |
References
- Hillen, J.B.; Vitry, A.; Caughey, G.E. Medication-related quality of care in residential aged care: An Australian experience. Int. J. Qual. Health Care 2019, 31, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Holmes, H.M.; Min, L.C.; Yee, M.; Varadhan, R.; Basran, J.; Dale, W.; Boyd, C.M. Rationalizing prescribing for older patients with multimorbidity: Considering time to benefit. Drugs Aging 2013, 9, 655–666. [Google Scholar] [CrossRef]
- Tangiisuran, B.; Wright, J.; Van der Cammen, T.; Rajkumar, C.H. Adverse drug reactions in elderly: Challenges in identification and improving preventative strategies. Age Ageing 2009, 4, 358–359. [Google Scholar] [CrossRef]
- Fick, D.M.; Semla, T.P.; Beizer, J.; Brandt, N.; Dombrowski, R.; DuBeau, C.E. American Geriatrics Society 2015 Updated Beers Criteria for Potentially Inappropriate Medication Use in Older Adults. J. Am. Geriatr. Soc. 2015, 11, 2227–22246. Available online: https://www.ncbi.nlm.nih.gov/pubmed/26446832 (accessed on 1 November 2019).
- Kouladjian, L.; Gnjidic, D.; Chen, T.F.; Hilmer, S.N. Development, validation and evaluation of an electronic pharmacological tool: The Drug Burden Index Calculator. Res. Soc. Adm. Pharm. 2016, 6, 865–875. Available online: https://www.ncbi.nlm.nih.gov/pubmed/26655397 (accessed on 1 November 2019). [CrossRef] [PubMed]
- Castelino, R.L.; Hilmer, S.N.; Bajorek, B.V.; Nishtala, P.; Chen, T.F. Drug Burden Index and potentially inappropriate medications in community-dwelling older people: The impact of Home Medication Review. Drugs Aging 2010, 27, 135–148. Available online: https://www.ncbi.nlm.nih.gov/pubmed/20104939 (accessed on 1 November 2019). [CrossRef] [PubMed]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef] [PubMed]
- Harrison, S.L.; Kouladjian, O.L.; Bradley, C.E.; Milte, R.; Dyer, S.M.; Gnanamanickam, E.S. Associations between the Drug Burden Index, Potentially Inappropriate Medications and Quality of Life in Residential Aged Care. Drug Aging 2018, 1, 83–91. Available online: https://www.ncbi.nlm.nih.gov/pubmed/29322470 (accessed on 1 November 2019). [CrossRef] [PubMed]
- Turner, J.P.; Tervonen, H.E.; Shakib, S.; Singhal, N.; Prowse, R.; Bell, J.S. Factors associated with use of falls risk-increasing drugs among patients of a geriatric oncology outpatient clinic in Australia: A cross-sectional study. J. Eval. Clin. Pract. 2017, 2, 361–368. [Google Scholar] [CrossRef] [PubMed]
- Basger, B.J.; Chen, T.F.; Moles, R.J. Inappropriate Medication Use and Prescribing Indicators in Elderly Australians. Development of a Prescribing Indicators Tool. Drugs Aging 2008, 9, 777–793. [Google Scholar] [CrossRef] [PubMed]
- Ashoorian, D.; Davidson, R.; Rock, D.; Dragovic, M.; Clifford, R. A clinical communication tool for the assessment of psychotropic medication side effects. Psychiatry Res. 2015, 2, 643–657. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Bae, Y.H.; Worley, M.; Law, A. Validating the Modified Drug Adherence Work-Up (M-DRAW) Tool to Identify and Address Barriers to Medication Adherence. Pharmacy 2017, 3, 52. [Google Scholar] [CrossRef] [PubMed]
- McLeod, P.J.; Huang, A.R.; Tamblyn, R.M.; Gayton, D.C. Defining inappropriate practices in prescribing for elderly people: A national consensus panel. CMAJ 1997, 3, 385–391. Available online: https://www.ncbi.nlm.nih.gov/pubmed/9033421 (accessed on 1 November 2019).
- Nishtala, P.S.; Fois, R.A.; McLachlan, A.J.; Bell, J.S.; Kelly, P.J.; Chen, T.F. Anticholinergic activity of commonly prescribed medications and neuropsychiatric adverse events in older people. J. Clin. Pharmacol. 2009, 10, 1176–1184. [Google Scholar] [CrossRef] [PubMed]
- Kouladjian, L.; Gnjidic, D.; Chen, T.F.; Mangoni, A.A.; Hilmer, S.N. Drug Burden Index in older adults: Theoretical and practical issues. Clin. Interv. Aging 2014, 9, 1503–1515. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qasim, H.; Simpson, M.; Guisard, Y.; de Courten, B. A Comprehensive Evaluation of Studies on the Adverse Effects of Medications in Australian Aged care Facilities: A Scoping Review. Pharmacy 2020, 8, 56. https://doi.org/10.3390/pharmacy8020056
Qasim H, Simpson M, Guisard Y, de Courten B. A Comprehensive Evaluation of Studies on the Adverse Effects of Medications in Australian Aged care Facilities: A Scoping Review. Pharmacy. 2020; 8(2):56. https://doi.org/10.3390/pharmacy8020056
Chicago/Turabian StyleQasim, Haider, Maree Simpson, Yann Guisard, and Barbora de Courten. 2020. "A Comprehensive Evaluation of Studies on the Adverse Effects of Medications in Australian Aged care Facilities: A Scoping Review" Pharmacy 8, no. 2: 56. https://doi.org/10.3390/pharmacy8020056
APA StyleQasim, H., Simpson, M., Guisard, Y., & de Courten, B. (2020). A Comprehensive Evaluation of Studies on the Adverse Effects of Medications in Australian Aged care Facilities: A Scoping Review. Pharmacy, 8(2), 56. https://doi.org/10.3390/pharmacy8020056







