Key Challenges in Diamond Coating of Titanium Implants: Current Status and Future Prospects
Abstract
:1. Introduction
2. Diamonds for Implant Technology
3. Strategies for Diamond Coating
3.1. Coating by CVD-Grown Diamond
3.2. Coating by Detonation Nanodiamond
4. Post-Processing and Surface Modifications
5. Addressing the Topic of Conductivity
6. Summary and Perspectives
Funding
Conflicts of Interest
References
- Wang, L.; Wang, C.; Wu, S.; Fan, Y.; Li, X. Influence of the mechanical properties of biomaterials on degradability, cell behaviors and signaling pathways: Current progress and challenges. Biomater. Sci. 2020, 8, 2714–2733. [Google Scholar] [CrossRef]
- Hasan, A.; Byambaa, B.; Morshed, M.; Cheikh, M.I.; Shakoor, R.A.; Mustafy, T.; Marei, H.E. Advances in osteobiologic materials for bone substitutes. J. Tissue Eng. Regen. Med. 2018, 12, 1448–1468. [Google Scholar] [CrossRef]
- American Joint Replacement Registry Releases 2021, Annual Report of American Academy of Orthopaedic Surgeons. Newsroom 11/12/2021. Available online: https://www.aaos.org/aaos-home/newsroom/press-releases/american-joint-replacement-registry-releases-2021-annual-report (accessed on 25 November 2022).
- Orthopedic Implants Market Analysis, by Application (Spinal fusion, Long bone, Foot & Ankle, Craniomaxillofacial, Joint replacement, Dental), and Segment Forecasts to 2024. Medical Devices, 80 Report ID: GVR-1-68038-020-0, Grand View Research, San Francisco (CA, U.S). Available online: https://www.grandviewresearch.com/industry-analysis/orthopedic-implants-market (accessed on 25 November 2022).
- Quinn, J.; McFadden, R.; Chan, C.-W.; Carson, L. Titanium for Orthopedic Applications: An Overview of Surface Modification to Improve Biocompatibility and Prevent Bacterial Biofilm Formation. iScience 2020, 23, 101745. [Google Scholar] [CrossRef]
- Langer, R.; Tirrell, D.A. Designing materials for biology and medicine. Nature 2004, 428, 487–492. [Google Scholar] [CrossRef]
- Sadtler, K.; Singh, A.; Wolf, M.T.; Wang, X.; Pardoll, D.M.; Elisseeff, J.H. Design, clinical translation and immunological response of biomaterials in regenerative medicine. Nat. Rev. Mater. 2016, 1, 1–17. [Google Scholar] [CrossRef]
- Dzobo, K.; Thomford, N.E.; Senthebane, D.A.; Shipanga, H.; Rowe, A.; Dandara, C.; Pillay, M.; Motaung, K.S.C.M. Advances in regenerative medicine and tissue engineering: Innovation and transformation of medicine. Stem. Cells Int. 2018, 2018, 2495848. [Google Scholar] [CrossRef] [Green Version]
- Kim, H.; Murakami, H.; Chehroudi, B.; Textor, M.; Brunette, D.M. Effects of surface topography on the connective tissue attachment to subcutaneous implants. Int. J. Oral Maxillofac. Implant. 2006, 21, 354–365. [Google Scholar]
- Geetha, M.; Singh, A.K.; Asokamani, R.; Gogia, A.K. Ti based biomaterials, the ultimate choice for orthopaedic implants—A review. Pro. Mater. Sci. 2009, 54, 397–425. [Google Scholar] [CrossRef]
- Schwarz, F.; Sager, M.; Kadelka, I.; Ferrari, D.; Becker, J. Influence of titanium implant surface characteristics on bone regeneration in dehiscence-type defects: An experimental study in dogs. J. Clin. Periodontol. 2010, 37, 466–472. [Google Scholar] [CrossRef]
- Sjöström, T.; McNamara, L.E.; Meek, R.M.D.; Dalby, M.J.; Su, B. 2D and 3D nanopatterning of titanium for enhancing osteoinduction of stem cells at implant surfaces. Adv. Healthc. Mater. 2013, 2, 1285–1293. [Google Scholar] [CrossRef]
- Jiang, G.; He, G. Enhancement of the porous titanium with entangled wire structure for load-bearing biomedical applications. Mater. Des. 2014, 56, 241–244. [Google Scholar] [CrossRef]
- Sidambe, A.T. Biocompatibility of advanced manufactured titanium implants—A review. Materials 2014, 7, 8168–8188. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jemat, A.; Ghazali, M.J.; Razali, M.; Otsuka, Y. Surface modifications and their effects on titanium dental implants. BioMed Res. Int. 2015, 2015, 791725. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salou, L.; Hoornaert, A.; Louarn, G.; Layrolle, P. Enhanced osseointegration of titanium implants with nanostructured surfaces: An experimental study in rabbits. Acta Biomater. 2015, 11, 494–502. [Google Scholar] [CrossRef] [PubMed]
- Staruch, R.; Griffin, M.; Butler, P. Nanoscale surface modifications of orthopaedic implants: State of the art and perspectives. Open Orthop. J. 2016, 10, 920–938. [Google Scholar] [CrossRef]
- Medvedev, A.E.; Ng, H.P.; Lapovok, R.; Estrin, Y.; Lowe, T.C.; Anumalasetty, V.N. Effect of bulk microstructure of commercially pure titanium on surface characteristics and fatigue properties after surface modification by sand blasting and acid-etching. J. Mech. Behav. Biomed. Mater. 2016, 57, 55–68. [Google Scholar] [CrossRef]
- Ng, J.; Spiller, K.; Bernhard, J.; Vunjak-Novakovic, G. Biomimetic Approaches for Bone Tissue Engineering. Tissue Eng. Part B Rev. 2017, 23, 480–493. [Google Scholar] [CrossRef]
- Cao, Y.; Su, B.; Chinnaraj, S.; Jana, S.; Bowen, L.; Charlton, S.; Duan, P.; Jakubovics, N.S.; Chen, J. Nanostructured titanium surfaces exhibit recalcitrance towards Staphylococcus epidermidis biofilm formation. Sci. Rep. 2018, 8, 1071. [Google Scholar] [CrossRef] [Green Version]
- Esteban, J.; Vallet-Regí, M.; Aguilera-Correa, J.J. Antibiotics- and Heavy Metals-Based Titanium Alloy Surface Modifications for Local Prosthetic Joint Infections. Antibiotics 2021, 10, 1270. [Google Scholar] [CrossRef]
- Rodriguez, D.C.; Valderrama, P.; Wilson, T.G., Jr.; Palmer, K.; Thomas, A.; Sridhar, S.; Adaalli, A.; Burbano, M.; Wadhani, C. Titanium corrosion mechanism in the oral environment: A retrieval study. Materials 2013, 6, 5258. [Google Scholar] [CrossRef] [Green Version]
- Rutkunas, V.; Bukelskiene, V.; Sabaliauskas, V.; Balciunas, E.; Malinauskas, M.; Baltriukiene, D. Assessment of human gingival fibroblast interaction with dental implant abutment materials. J. Mater. Sci. Mater. Med. 2015, 26, 169. [Google Scholar] [CrossRef]
- Lozano, P.; Peña, M.; Herrero-Climent, M.; Rios-Santos, J.V.; Rios-Carrasco, B.; Brizuela, A.; Gil, J. Corrosion Behavior of Titanium Dental Implants with Implantoplasty. Materials 2022, 15, 1563. [Google Scholar] [CrossRef]
- Herrero-Climent, M.; Lázaro, P.; Vicente Rios, J.; Lluch, S.; Marqués-Calvo, M.S.; Guillem-Martí, J.; Gil, F.J. Influence of acid-etching after grit-blasted on osseointegration of titanium dental implants: In vitro and in vivo studies. J. Mater. Sci. Mater. Med. 2013, 24, 2047–2055. [Google Scholar] [CrossRef]
- El-wassefy, N.A.; Hammouda, I.M.; Habib, A.N.E.-d.A.; El-awady, G.Y.; Marzook, H.A. Assessment of anodized titanium implants bioactivity. Clin. Oral Implant. Res. 2014, 25, e1–e9. [Google Scholar] [CrossRef]
- Saulacic, N.; Erdösi, R.; Bosshardt, D.D.; Gruber, R.; Buser, D. Acid and Alkaline Etching of Sandblasted Zirconia Implants: A Histomorphometric Study in Miniature Pigs. Clin. Implant. Dent. Relat. Res. 2014, 16, 313–322. [Google Scholar] [CrossRef]
- Ferraris, S.; Venturello, A.; Miola, M.; Cochis, A.; Rimondini, L.; Spriano, S. Antibacterial and bioactive nanostructured titanium surfaces for bone integration. Appl. Surf. Sci. 2014, 311, 279–291. [Google Scholar] [CrossRef]
- Ozdemir, Z.; Ozdemir, A.; Basim, G.B. Application of chemical mechanical polishing process on titanium based implants. Mater. Sci. Eng. C 2016, 68, 383–396. [Google Scholar] [CrossRef] [PubMed]
- Bürgers, R.; Gerlach, T.; Hahnel, S.; Schwarz, F.; Handel, G.; Gosau, M. In vivo and in vitro biofilm formation ontwo different titanium implant surfaces. Clin. Oral Implant. Res. 2010, 21, 156–164. [Google Scholar] [CrossRef] [PubMed]
- Lorenzetti, M.; Dogša, I.; Stošicki, T.; Stopar, D.; Kalin, M.; Kobe, S.; Novak, S. The influence of surface modification on bacterial adhesion to titanium-based substrates. ACS Appl. Mater. Interfaces 2015, 7, 1644–1651. [Google Scholar] [CrossRef]
- Khatoon, Z.; McTiernan, C.D.; Suuronen, E.J.; Mah, T.-F.; Alarcon, E.I. Bacterial biofilm formation on implantable devices and approaches to its treatment and prevention. Heliyon 2018, 4, e01067. [Google Scholar] [CrossRef] [Green Version]
- Souza, J.G.S.; Bertolini, M.M.; Costa, R.C.; Nagay, B.E.; Dongari-Bagtzoglou, A.; Barão, V.A.R. Targeting implant-associated infections: Titanium surface loaded with antimicrobial. iScience 2021, 24, 102008. [Google Scholar] [CrossRef] [PubMed]
- Hao, Y.; Huang, X.; Zhou, X.; Li, M.; Ren, B.; Peng, X.; Cheng, L. Influence of Dental Prosthesis and Restorative Materials Interface on Oral Biofilms. Int. J. Mol. Sci. 2018, 14, 3157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chouirfa, H.; Bouloussa, H.; Migonney, V.; Falentin-Daudré, C. Review of titanium surface modification techniques and coatings for antibacterial applications. Acta Biomater. 2019, 83, 37–54. [Google Scholar] [CrossRef] [PubMed]
- Browne, K.; Chakraborty, S.; Chen, R.; Willcox, M.D.; Black, D.S.; Walsh, W.R.; Kumar, N. A New Era of Antibiotics: The Clinical Potential of Antimicrobial Peptides. Int. J. Mol. Sci. 2020, 21, 7047. [Google Scholar] [CrossRef]
- Subramani, K.; Jung, R.E.; Molenberg, A.; Hammerle, C.H.F. Biofilm on dental implants: A review of the literature. Int. J. Oral. Maxillofac. Implants 2009, 24, 616–662. [Google Scholar]
- Simchi, A.; Tamjid, E.; Pishbin, F.; Boccaccini, A.R. Recent progress in inorganic and composite coatings with bactericidal capability for orthopaedic applications. Nanomedicine 2011, 7, 22. [Google Scholar] [CrossRef]
- Ferraris, S.; Spriano, S. Antibacterial titanium surfaces for medical implants. Mater. Sci. Eng. C 2015, 61, 965–968. [Google Scholar] [CrossRef]
- Zhao, L.; Chu, P.K.; Zhang, Y.; Wu, Z. Antibacterial coatings on titanium implants. J. Biomed. Mater. Res. Part B Appl. Biomater. 2019, 91, 470–480. [Google Scholar] [CrossRef]
- Rahimi, E.; Rafsanjani-Abbasi, A.; Imani, A.; Hosseinpour, S.; Davoodi, A. Insights into Galvanic Corrosion Behavior of Ti-Cu Dissimilar Joint: Effect of Microstructure and Volta Potential. Materials 2018, 11, 1820. [Google Scholar] [CrossRef] [Green Version]
- Wan, Y.Z.; Raman, S.; He, F.; Huang, Y. Surface modification of medical metals by ion implantation of silver and copper. Vacuum 2007, 81, 1114–1118. [Google Scholar] [CrossRef]
- Xavier, J.R. Galvanic corrosion of copper/titanium in aircraft structures using a cyclic wet/dry corrosion test in marine environment by EIS and SECM techniques. SN Appl. Sci. 2020, 2, 1341. [Google Scholar] [CrossRef]
- Hasan, J.; Russell, J.C.; Ivanova, E.P. Antibacterial Surfaces: The Quest for a New Generation of Biomaterials. Trends Biotechnol. 2013, 31, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Raphel, J.; Holodniy, M.; Goodman, S.B.; Heilshorn, S.C. Multifunctional coatings to simultaneously promote osseointegration and prevent infection of orthopaaedic implants. Biomaterials 2016, 84, 301–314. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spriano, S.; Yamaguchi, S.; Baino, F.; Ferraris, S. A critical review of multifunctional titanium surfaces: New frontiers for improving osseointegration and host response, avoiding bacteria contamination. Acta Biomater. 2018, 79, 1–22. [Google Scholar] [CrossRef]
- Rupp, F.; Liang, L.; Geis-Gerstorfer, J.L.; Scheideler, L.; Hüttig, F. Surface characteristics of dental implants: A review. Dent. Mater. 2018, 34, 40–57. [Google Scholar] [CrossRef] [PubMed]
- Damiati, L.; Eales, M.G.; Nobbs, A.H.; Su, B.; Tsimbouri, P.M.; Salmeron-Sanchez, M.; Dalby, M.J. Impact of surface topography and coating on osteogenesis and bacterial attachment on titanium implants. J. Tissue Eng. 2018, 9, 2041731418790694. [Google Scholar] [CrossRef]
- Nazarè, M.H.; Neves, A.J. (Eds.) Properties, Growth and Applications of Diamond; ET—Technology & Engineering, INSPEC: London, UK, 2001. [Google Scholar]
- Broz, A.; Ukraintsev, E.; Kromka, A.; Rezek, B.; Hubalek Kalbacova, M. Osteoblast adhesion, migration, and proliferation variations on chemically patterned nanocrystalline diamond films evaluated by live-cell imaging. J. Biomed. Mater. Res. Part A 2017, 105A, 1469–1478. [Google Scholar] [CrossRef]
- Mani, N.; Rifai, A.; Houshyar, S.; Booth, M.A.; Fox, K. Diamond in medical devices and sensors: An overview of diamond surfaces. Med. Devices Sens. 2020, 3, 1–13. [Google Scholar] [CrossRef]
- Yang, K.H.; Narayan, R.J. Biocompatibility and functionalization of diamond for neural applications. Curr. Opin. Biomed. Eng. 2019, 10, 60–68. [Google Scholar] [CrossRef]
- Grausova, L.; Kromka, A.; Bacakova, L.; Potocky, S.; Vanecek, M.; Lisa, V. Bone and vascular endothelial cells in cultures on nanocrystalline diamond films. Diam. Relat. Mater. 2008, 17, 1405–1409. [Google Scholar] [CrossRef]
- Grausova, L.; Bacakova, L.; Kromka, A.; Potocky, S.; Vanecek, M.; Nesladek, M.; Lisa, V. Nanodiamond as promising material for bone tissue engineering. Proc. J. Nanosci. Nanotechnol. 2009, 9, 3524–3534. [Google Scholar] [CrossRef] [PubMed]
- Fox, K.; Palamara, J.; Judge, R.; Greentree, A. Diamond as a scaffold for bone growth. J. Mater. Sci. Mater. Med. 2013, 24, 849–861. [Google Scholar] [CrossRef]
- Bacakova, L.; Kopova, I.; Stankova, L.; Liskova, J.; Vacik, J.; Lavrentiev, V.; Kromka, A.; Potocky, S.; Stranska, D. Bone cells in cultures on nanocarbon based materials for potential bone tissue engineering: A review. Phys. Status Solidi Appl. Mater. 2014, 211, 2688–2702. [Google Scholar] [CrossRef]
- Pareta, R.; Yang, L.; Kothari, A.; Sirinrath, S.; Xiao, X.; Sheldon, B.W.; Webster, T.J. Tailoring nanocrystalline diamond coated on titanium for osteoblast adhesion. J. Biomed. Mater. Res. Part A 2010, 95, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Metzler, P.; von Wilmowsky, C.; Stadlinger, B.; Zemann, W.; Schlegel, K.A.; Rosiwal, S.; Rupprecht, S. Nano-crystalline diamond-coated titanium dental implants, A histomorphometric study in adult domestic pigs. J. Cranio-Maxillofac. Surg. 2013, 41, 532–538. [Google Scholar] [CrossRef]
- Ma, S.; Liu, J.; Luo, W.; Zhang, H.; Meng, X.; Guo, Y.; Cheng, S.; Wang, Q.; Zhou, Y.; Li, H. Characteristics of nanocrystalline diamond films on titanium surface and its impact on the proliferation and adhesion of cells. J. Hard Tissue Biol. 2016, 25, 6–14. [Google Scholar] [CrossRef] [Green Version]
- Skoog, S.A.; Kumar, G.; Zheng, J.; Sumant, A.V.; Goering, P.L.; Narayan, R.J. Biological evaluation of ultrananocrystalline and nanocrystalline diamond coatings. J. Mater. Sci. Mater. Med. 2016, 27, 187. [Google Scholar] [CrossRef]
- Rifai, A.; Tran, N.; Lau, D.W.; Elbourne, A.; Zhan, H.; Stacey, A.D.; Mayes, E.L.H.; Sarker, A.; Ivanova, E.P.; Crawford, R.J.; et al. Polycrystalline Diamond Coating of Additively Manufactured Titanium for Biomedical Applications. ACS Appl. Mater. Interfaces 2018, 10, 8474–8484. [Google Scholar] [CrossRef]
- Paxton, W.F.; Rozsa, J.L.; Brooks, M.M.; Running, M.P.; David, J.; Schultz, D.J.; Jasinski, J.B.; Jung, H.J.; Akram, M.Z. A scalable approach to topographically mediated antimicrobial surfaces based on diamond. J. Nanobiotechnol. 2021, 19, 458. [Google Scholar] [CrossRef]
- Fong, J.S.L.; Booth, M.A.; Rifai, A.; Fox, K.; Gelmi, A. Diamond in the Rough: Toward Improved Materials for the Bone−Implant Interface. Adv. Healthc. Mater. 2021, 10, 2100007. [Google Scholar] [CrossRef]
- Engler, A.J.; Sen, S.; Sweeney, H.L.; Discher, D.E. Matrix Elasticity Directs Stem Cell Lineage Specification. Cell 2006, 126, 677–689. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tzvetkova-Chevolleau, T.; Stéphanou, A.; Fuard, D.; Ohayon, J.; Schiavone, P.; Tracqui, P. The motility of normal and cancer cells in response to the combined influence of the substrate rigidity and anisotropic microstructure. Biomaterials 2008, 29, 1541–1551. [Google Scholar] [CrossRef] [PubMed]
- Forte, G.; Carotenuto, F.; Pagliari, F.; Pagliari, S.; Cossa, P.; Fiaccavento, R.; Ahluwalia, A.; Vozzi, G.; Vinci, B.; Serafino, A.; et al. Criticality of the Biological and Physical Stimuli Array Inducing Resident Cardiac Stem Cell Determination. Stem Cells 2008, 26, 2093–2103. [Google Scholar] [CrossRef] [PubMed]
- Handorf, A.M.; Zhou, Y.; Halanski, M.A.; Li, W.J. Tissue stiffness dictates development, homeostasis, and disease progression. Organogenesis 2015, 11, 1–15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klein, C.A.; Cardinale, G. Young’s modulus and Poisson′s ratio of CVD diamond. Diam. Relat. Mater. 1993, 2, 918–923. [Google Scholar] [CrossRef]
- Telling, R.H.; Pickard, C.J.; Payne, M.C.; Field, J.E. Theoretical Strength and Cleavage of Diamond, Phys. Rev. Lett. 2020, 84, 5160–5163. [Google Scholar] [CrossRef] [Green Version]
- Currey, J.D. Effects of differences in mineralization on the mechanical properties of bone. Phil. Trans. R. Soc. Lond. B 1984, 304, 509–518. [Google Scholar] [CrossRef]
- Ahnood, A.; Meffin, H.; Garrett, D.J.; Fox, K.; Ganesan, K.; Stacey, A.; Apollo, N.V.; Wong, Y.T.; Lichter, S.G.; Kentler, W.; et al. Diamond Devices for High Acuity Prosthetic Vision. Adv. Biosyst. 2017, 1, 1600003. [Google Scholar] [CrossRef]
- Ebisawa, K.; Kato, R.; Okada, M.; Sugimura, T.; Latif, M.A.; Hori, Y.; Narita, Y.; Ueda, M.; Honda, H.; Kagami, H. Gingival and dermal fibroblasts: Their similarities and differences revealed from gene expression. J. Biosci. Bioeng. 2011, 111, 255–258. [Google Scholar] [CrossRef]
- Nistor, P.A.; May, P.W. Diamond thin films: Giving Biomedical applications a new shine. J. R. Soc. Interface 2017, 14, 20170382. [Google Scholar] [CrossRef] [Green Version]
- Booth, L.; Catledge, S.; Nolen, D.; Thompson, R.; Vohra, Y. Synthesis and characterization of multilayered diamond coatings for biomedical implants. Materials 2011, 4, 857–867. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chu, Y.-C.; Tzeng, Y.; Auciello, O. Microwave plasma enhanced chemical vapor deposition of nanoacrystalline diamond films by bias-enhanced nucleation and bias-enhanced grow. J. Appl. Phys. 2014, 115, 024308. [Google Scholar] [CrossRef]
- Fuentes-Fernandez, E.M.A.; Alcantar Peña, J.J.; Lee, G.; Boulom, A.; Phan, H.; Smith, B.; Nguyen, T.; Sahoo, S.; Ruiz-Zepeda, F.; Arellano-Jimenez, M.J.; et al. Synthesis and characterization of microcrystalline diamond to ultrananocrystalline diamond films via Hot Filament Chemical Vapor Deposition for scaling to large area applications. Thin Solid Films 2016, 603, 62–68. [Google Scholar] [CrossRef] [Green Version]
- Baranauskas, V.; Ceragioli, H.J.; Peterlevitz, A.C.; Tosin, M.C.; Durrant, S.F. Effects of the addition of helium on the synthesis of diamond films. Thin Solid Films 2000, 377, 182–187. [Google Scholar] [CrossRef]
- Sur, D.; Tirado, P.; Alcantar, J.; Auciello, O.; Basim, G.B. Integration of Ultrananocrystalline Diamond (UNCD)-Coatings on Chemical-Mechanical Surface Nano-structured (CMNS) Titanium-Based Dental implants. MRS Adv. 2020, 5, 2261–2271. [Google Scholar] [CrossRef]
- Fries, M.; Vohra, Y. Nanostructured diamond film deposition on curved surfaces of metallic temporomandibular joint implant. J. Phys. D Appl. Phys. 2002, 35, L105. [Google Scholar] [CrossRef]
- Papo, M.J.; Catledge, S.A.; Vohra, Y.K. Mechanical wear behavior of nanocrystalline and multilayered diamond coatings on temporomandibular joint implants. J. Mater. Sci.—Mater. Med. 2004, 15, 773–777. [Google Scholar] [CrossRef]
- Steinerova, M.; Matejka, R.; Stepanovska, J.; Filova, E.; Stankova, L.; Rysova, M.; Martinova, L.; Dragounova, H.; Domonkos, M.; Artemenko, A.; et al. Human osteoblast-like SAOS-2 cells on submicron-scale fibers coated with nanocrystalline diamond films. Mater. Sci. Eng. C 2021, 121, 111792. [Google Scholar] [CrossRef] [PubMed]
- Haubner, R. Low-pressure diamond: From the unbelievable to technical products. ChemTexts 2021, 7, 10. [Google Scholar] [CrossRef]
- Greiner, N.R.; Phillips, D.S.; Johnson, J.D.; Volk, F. Diamonds in detonation soot. Nature 1988, 333, 440–442. [Google Scholar] [CrossRef]
- Mochalin, V.N.; Shenderova, O.; Ho, D.; Gogotsi, Y. The properties and applications of nanodiamonds. Nat. Nanotechnol. 2012, 7, 11–23. [Google Scholar] [CrossRef] [PubMed]
- Reina, G.; Orlanducci, S.; Gay, S.; Gismondi, A.; Lavecchia, T.; Terranova, M.L.; Tamburri, E. Routes to Control the Chemical Potential and to Modulate the Reactivity of Nanodiamond Surfaces. MRS Symp. Proc. 2015, 1734, 32–39. [Google Scholar] [CrossRef]
- Taylor, A.C.; Gonzalez, C.H.; Miller, B.S.; Edgington, R.J.; Ferretti, P.; Jackman, R.B. Surface functionalisation of nanodiamonds for human neural stem cell adhesion and proliferation. Sci. Rep. 2017, 7, 7307. [Google Scholar] [CrossRef]
- Rifai, A.; Pirogova, E.; Fox, K. Diamond, Carbon Nanotubes and Graphene for Biomedical Applications. Ref. Modul. Biomed. Sci. 2018, 2018, 2–11. [Google Scholar] [CrossRef]
- Amaral, M.; Dias, A.; Gomes, P.; Lopes, M.; Silva, R.; Santos, J.; Fernandes, M. Nanocrystalline diamond: In vitro biocompatibility assessment by MG63 and human bone marrow cells cultures. J. Biomed. Mater. Res. Part A 2008, 87, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Roseti, L.; Parisi, V.; Petretta, M.; Cavallo Desando, C.G.; Bartolotti, I.; Grigolo, B. Scaffolds for Bone Tissue Engineering: State of the art and new perspectives. Mater. Sci. Eng. C 2017, 78, 1246–1262. [Google Scholar] [CrossRef]
- Popov, V.V., Jr.; Muller-Kamskii, G.; Kovalevsky, A.; Dzhenzhera, G.; Strokin, E.; Kolomiets, A.; Ramon, J. Design and D printing of titanium bone implants: Brief review of approach and clinical cases. Biomed. Eng. Lett. 2018, 8, 337–344. [Google Scholar] [CrossRef]
- Li, X.C.; He, L.; Zhang, J.W.; Misra, R.D.K.; Liu, S.L.; Xiong, Z.H.; Tang, Z.H. Additive manufacturing of dental root-analogue implant with desired properties. Mater. Technol. 2021, 36, 894906. [Google Scholar] [CrossRef]
- Rifai, A.; Tran, N.; Reineck, P.; Elbourne, A.; Mayes, E.; Sarker, A.; Dekiwadia, C.; Ivanova, E.P.; Crawford, R.J.; Ohshima, T.; et al. Engineering the Interface: Nanodiamond Coating on.3D-Printed Titanium Promotes Mammalian Cell Growth and Inhibits Staphylococcus aureus. ACS Appl. Mater. Interfaces 2019, 11, 24588–24597. [Google Scholar] [CrossRef]
- Rifai, A.; Tran, N.; Leitch, V.; Booth, M.A.; Williams, R.; Fox, K.E. Osteoblast Cell Response on Polycrystalline Diamond-Coated Additively Manufactured Scaffolds. ACS Appl. Bio Mater. 2021, 4, 7509–7516. [Google Scholar] [CrossRef]
- Booth, M.A.; Pope, L.; Sherrell, P.C.; Stacey, A.; Tran, P.; Fox, K.E. Polycrystalline diamond coating on 3D printed titanium scaffolds: Surface characterisation and foreign body response. Mater. Sci. Eng. C 2021, 130, 112467. [Google Scholar] [CrossRef] [PubMed]
- Norouzia, N.; Onga, Y.; Damlea, V.G.; Najafib, M.B.H.; Schirhagla, R. Effect of medium and aggregation on antibacterial activity of nanodiamonds. Mater. Sci. Eng. C 2020, 112, 110930. [Google Scholar] [CrossRef] [PubMed]
- Cumont, A.; Pitt, A.R.; Lambert, P.A.; Oggioni, M.R.; Ye, H. Properties, mechanism and applications of diamond as an antibacterial material. Funct. Diam. 2021, 1, 1–28. [Google Scholar] [CrossRef]
- Li, L.; Chen, X.; Zhang, W.; Peng, K. Characterization and formation mechanism of pits on diamond {100} face etched by molten potassium nitrite. Int. J. Refract. Met. Hard Mater. 2018, 71, 129–134. [Google Scholar] [CrossRef]
- Li, J.-W.; Liu, X.-P.; Wan, L.; Fang, W.-J.; Yan, Y.-G.; Li, Y.-Y.; Gao, P.-Z. Using the thermochemical corrosion method to prepare porous diamonds. Ceram. Int. 2021, 47, 35002–35012. [Google Scholar] [CrossRef]
- Liu, D.; Zhao, J.; Lei, Y.; Wang, X.; Fu, W.; Song, X.; Long, W. Micropatterning of synthetic diamond by metal contact etching with Ti powder. Diam. Rel. Mater. 2022, 129, 109299. [Google Scholar] [CrossRef]
- Golovanov, A.V.; Bormashov, V.S.; Luparev, N.V.; Tarelkin, S.A.; Troschiev, S.Y.; Buga, S.G.; Blank, V.D. Diamond microstructuring by deep anisotropic reactive ion etching. Phys. Stat. Solidi A 2018, 215, 1800273. [Google Scholar] [CrossRef]
- Orlanducci, S.; Guglielmotti, V.; Cianchetta, I.; Sessa, V.; Tamburri, E.; Toschi, F.; Terranova, M.L.; Rossi, M. Detonation Nanodiamond as Building Blocks for Fabrication of Densely Packed Arrays of Diamond Nanowhiskers. Nanosc. Nanotechnol. Lett. 2012, 4, 338–343. [Google Scholar] [CrossRef] [Green Version]
- Reina, G.; Tamburri, E.; Orlanducci, S.; Gay, S.; Matassa, R.; Guglielmotti, V.; Lavecchia, T.; Terranova, M.L.; Rossi, M. Nanocarbon surfaces for biomedicine. Biomatter 2014, 4, e28537. [Google Scholar] [CrossRef] [Green Version]
- Merker, D.; Handzhiyski, Y.; Merz, R.; Kopnarski, M.; Reithmaier, J.P.; Popov, C.; Apostolova, M.D. Influence of surface termination of ultrananocrystalline diamond films coated on titanium on response of human osteoblast cells: A proteome study. Mater. Sci. Eng. C 2021, 128, 112289. [Google Scholar] [CrossRef]
- Yang, L.; Li, Y.; Sheldon, B.W.; Webster, T.J. Altering surface energy of nanocrystalline diamond to control osteoblast responses. J. Mater. Chem. 2012, 22, 205–214. [Google Scholar] [CrossRef]
- Liskova, J.; Babchenko, O.; Varga, M.; Kromka, A.; Hadraba, D.; Svindrych, Z.; Burdikova, Z.; Bacakova, L. Osteogenic cell differentiation on H-terminated and O-terminated nanocrystalline diamond films. Int. J. Nanomed. 2015, 10, 869–884. [Google Scholar] [CrossRef]
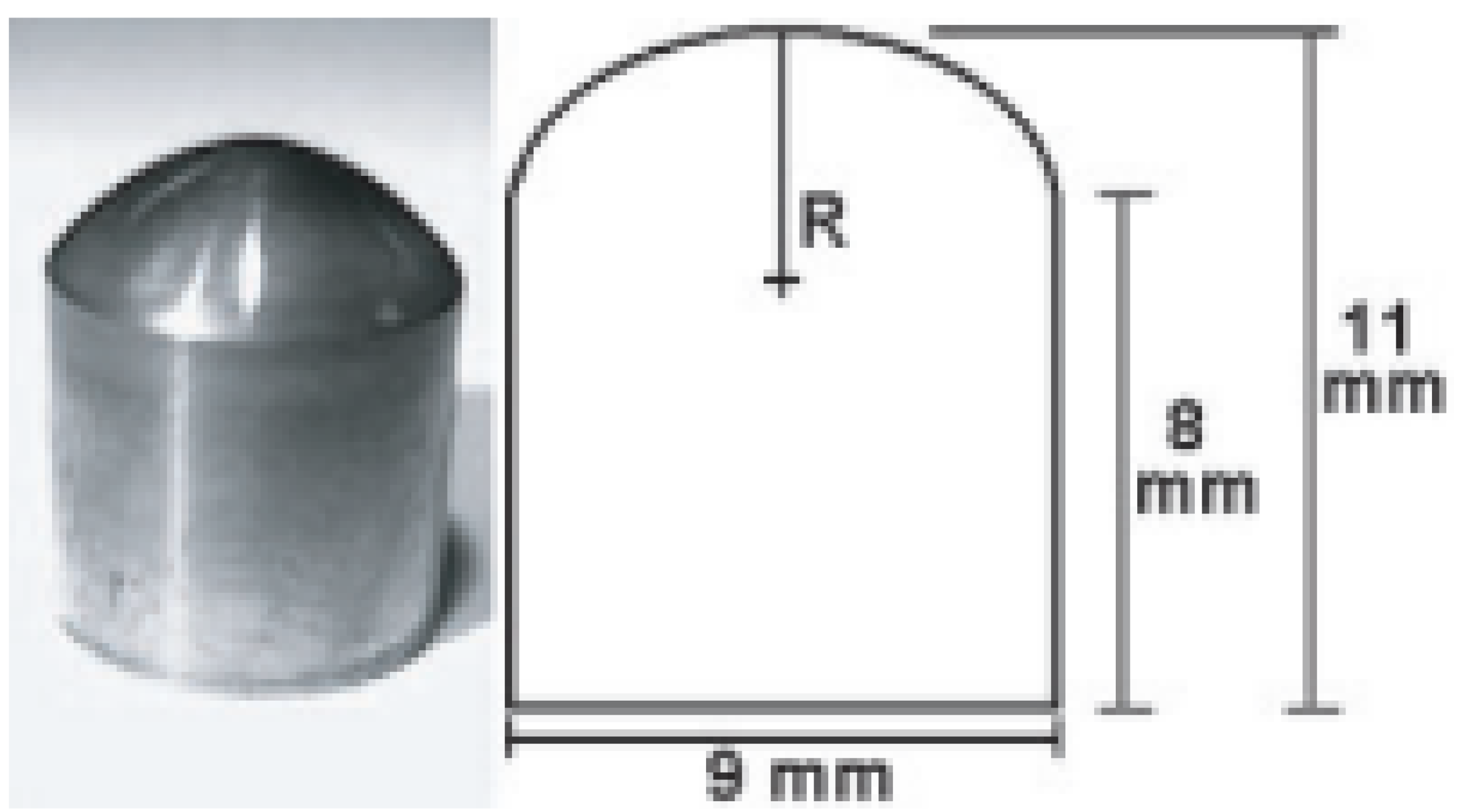
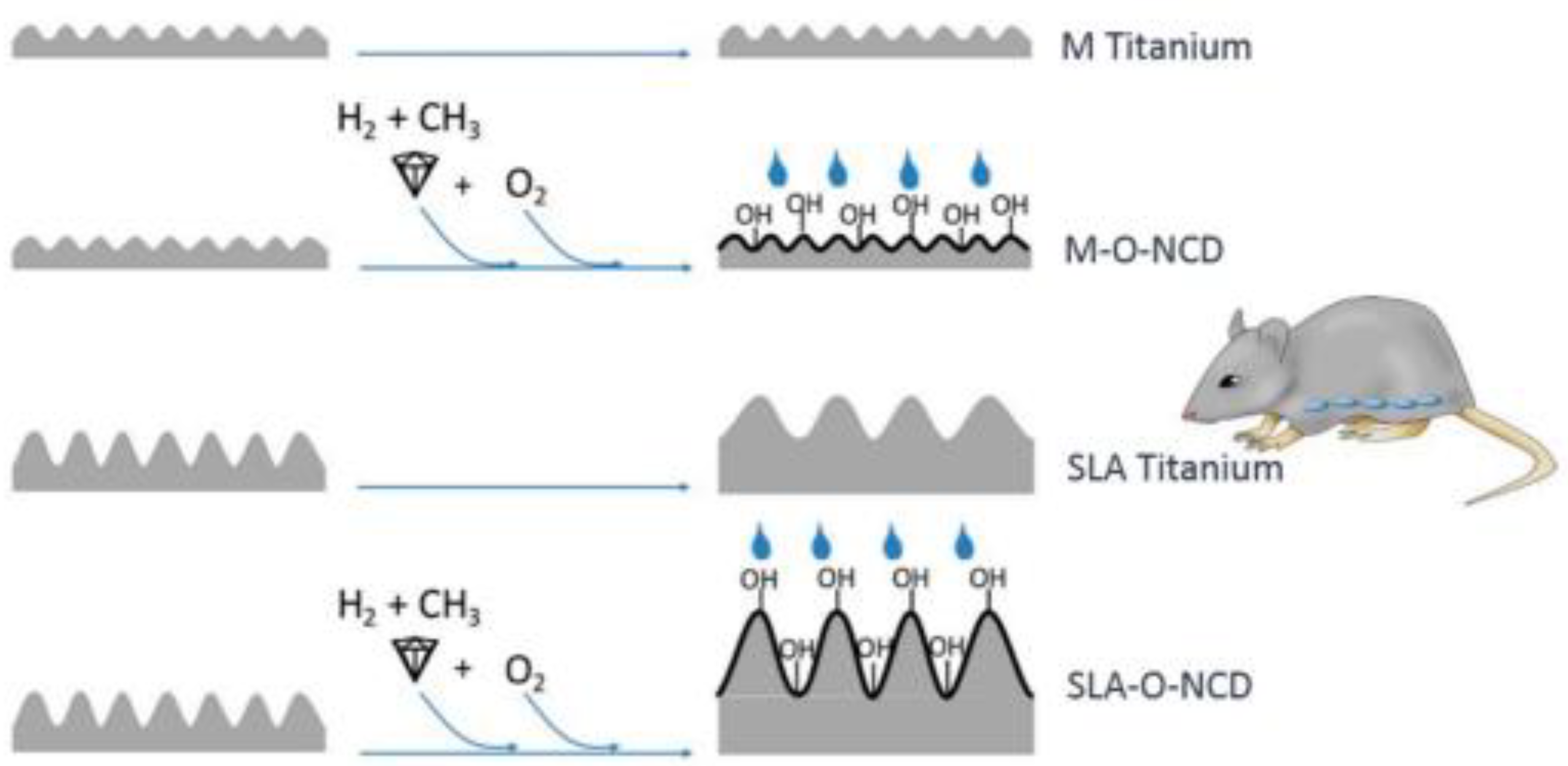
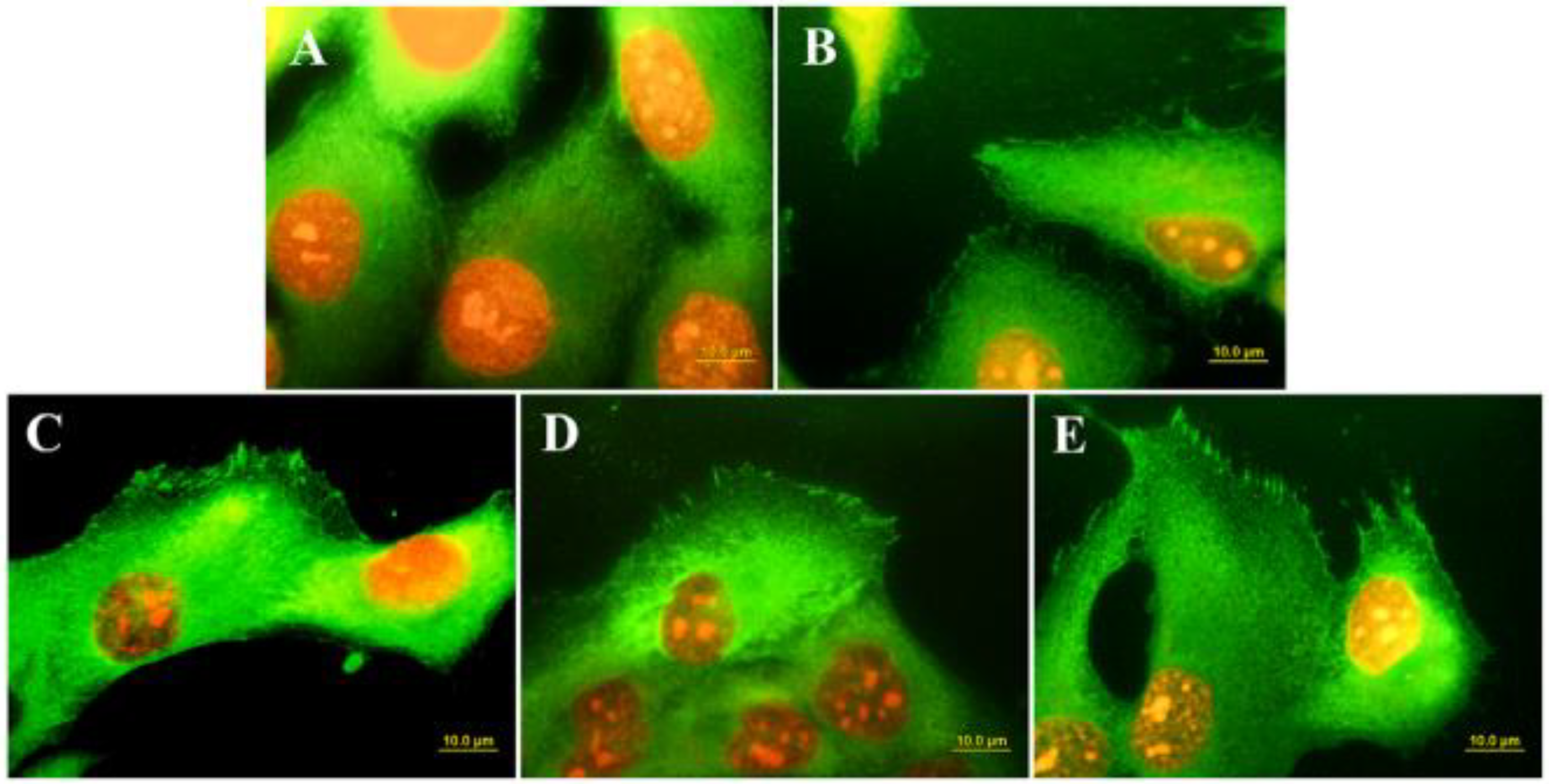
- Stigler, R.G.; Becker, K.; Bruschi, M.; Steinmüller-Nethl, D.; Gassner, R. Impact of Nano-Crystalline Diamond Enhanced Hydrophilicity on Cell Proliferation on Machined and SLA Titanium Surfaces: An In-Vivo Study in Rodents. Nanomaterials 2018, 8, 524. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martinez, M.A.F.; Balderrama, Í.; Karam, P.S.B.H.; de Oliveira, R.C.; de Oliveira, F.A.; Grandini, C.R.; Vicente, F.B.; Stavropoulos, A.; Zangrando, M.S.R.; Sant′Ana, A.C.P. Surface roughness of titanium disks influences the adhesion, proliferation and differentiation of osteogenic properties derived from human. Int. J. Implant. Dent. 2020, 6, 46. [Google Scholar] [CrossRef]
- Sikorvsky, P. Electroconductive scaffolds for tissue engineering applications. Biomater. Sci. 2020, 8, 5583–5588. [Google Scholar]
- Grausova, L.; Kromka, A.; Burdikova, Z.; Eckhardt, A.; Rezek, B.; Vacik, J.; Haenen, K.; Lisa, V.; Bacakova, L. Enhanced growth and osteogenic differentiation of human osteoblast-like cells on boron-doped nanocrystalline diamond thin films. PLoS ONE 2011, 6, 20943. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koerner, R.J.; Butterworth, L.A.; Mayer, I.V.; Dasbach, R.; Bussche, H.J. Bacterial adhesion to titanium-oxy-nitride (TiNOX) coatings with different resistivities: A novel approach for the development of biomaterials. Biomaterials 2002, 23, 2835–2840. [Google Scholar] [CrossRef] [PubMed]
- Meijs, S.; Alcaide, M.; Sørensen, C.; McDonald, M.; Sørensen, S.; Rechendorff, K.; Gerhardt, A.; Nesladek, M.; Rijkhoff, N.J.M.; Pennisi, C.P. Biofouling resistance of boron-doped diamond neural stimulation electrodes is superior to titanium nitride electrodes in vivo. J. Neural Eng. 2016, 13, 056011. [Google Scholar] [CrossRef]
- Garrett, D.J.; Saunders, A.L.; McGowan, C.; Specks, J.; Ganesan, K.; Meffin, H.; Williams, R.A.; Nayagam, D.A.X. In vivo biocompatibility of boron doped and nitrogen included conductive-diamond for use in medical implants. J. Biomed. Mater. Res. Part B 2016, 104B, 19–26. [Google Scholar] [CrossRef]
- Tong, W.; Tran, P.A.; Turnley, A.M.; Aramesh, M.; Prawer, S.; Brandt, M.; Fox, K. The influence of sterilization on nitrogen-included ultrananocrystalline diamond for biomedical applications. Mater. Sci. Eng. C 2016, 61, 324–332. [Google Scholar] [CrossRef]
- Simcox, L.J.; Pereira, R.P.A.; Wellington, E.M.H.; Macpherson, J.V. Boron Doped Diamond as a Low Biofouling Material in Aquatic Environments: Assessment of Pseudomonas aeruginosa Biofilm Formation. ACS Appl. Mater. Interfaces 2019, 11, 25024–25033. [Google Scholar] [CrossRef] [PubMed]
- Terranova, M.L.; Piccirillo, S.; Sessa, V.; Rossi, M.; Cappuccio, G. A New CVD Route for the Synthesis of Composite Diamond-Based Materials. Chem. Vap. Depos. 1999, 5, 101–105. [Google Scholar] [CrossRef]
- Tamburri, E.; Carcione, R.; Vitale, F.; Valguarnera, A.; Macis, S.; Lucci, M.; Terranova, M.L. Exploiting the Properties of Ti-Doped CVD-Grown Diamonds for the Assembling of Electrodes. Adv. Mater. Interfaces 2017, 4, 1700222. [Google Scholar] [CrossRef]
- Carcione, R.; Politi, S.; Iacob, E.; Potrich, C.; Lunelli, L.; Vanzetti, L.E.; Bartali, R.; Micheli, V.; Pepponi, G.; Terranova, M.L.; et al. Exploring a new approach for regenerative medicine: Ti-doped polycrystalline diamond layers as bioactive platforms for osteoblast-like cells growth. Appl. Surf. Sci. 2021, 540, 148334. [Google Scholar] [CrossRef]
- Fox, K.; Mani, N.; Rifai, A.; Reineck, P.; Jones, A.; Tran, P.A.; Nejad, A.; Brandt, M.; Gibson, B.C.; Greentree, A.D.; et al. 3D-Printed Diamond–Titanium Composite: A Hybrid Material for Implant Engineering. ACS Appl. Bio Mater. 2020, 3, 29–36. [Google Scholar] [CrossRef] [PubMed]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Terranova, M.L. Key Challenges in Diamond Coating of Titanium Implants: Current Status and Future Prospects. Biomedicines 2022, 10, 3149. https://doi.org/10.3390/biomedicines10123149
Terranova ML. Key Challenges in Diamond Coating of Titanium Implants: Current Status and Future Prospects. Biomedicines. 2022; 10(12):3149. https://doi.org/10.3390/biomedicines10123149
Chicago/Turabian StyleTerranova, Maria Letizia. 2022. "Key Challenges in Diamond Coating of Titanium Implants: Current Status and Future Prospects" Biomedicines 10, no. 12: 3149. https://doi.org/10.3390/biomedicines10123149



