Type 2C Protein Phosphatases MoPtc5 and MoPtc7 Are Crucial for Multiple Stress Tolerance, Conidiogenesis and Pathogenesis of Magnaporthe oryzae
Abstract
:1. Introduction
2. Materials and Methods
2.1. Strains and Culture Conditions
2.2. Targeted Gene Deletion and Complementation
2.3. Infection Assays, Cuticle Penetration and Incipient Cytorrhysis Assay
2.4. Microscopy
2.5. Southern Blot Analysis, Extraction of Total RNA, qRT-PCR and Gene Expressions
2.6. Bioinformatics Analysis
2.7. Cell Wall Integrity, Osmotic and Oxidative Stresses and Cell Wall Thickness Assays
2.8. Western Blot Assay
2.9. Subcellular Localization Assays
3. Results
3.1. Phylogenetic Analysis of MoPtc5 and MoPtc7
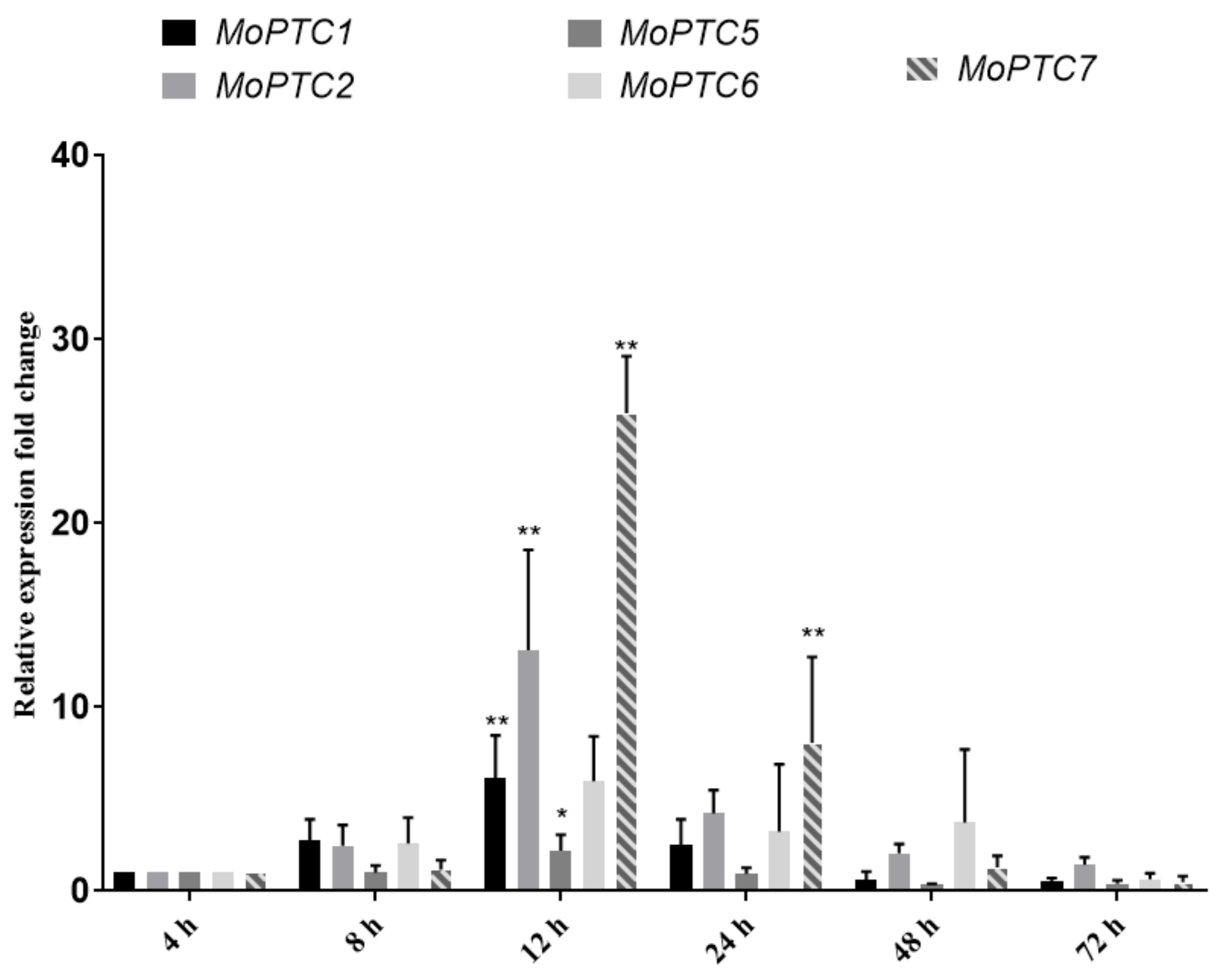
3.2. MoPtc5 and MoPtc7 Are Differentially Expressed during Pathogen-Host Interaction
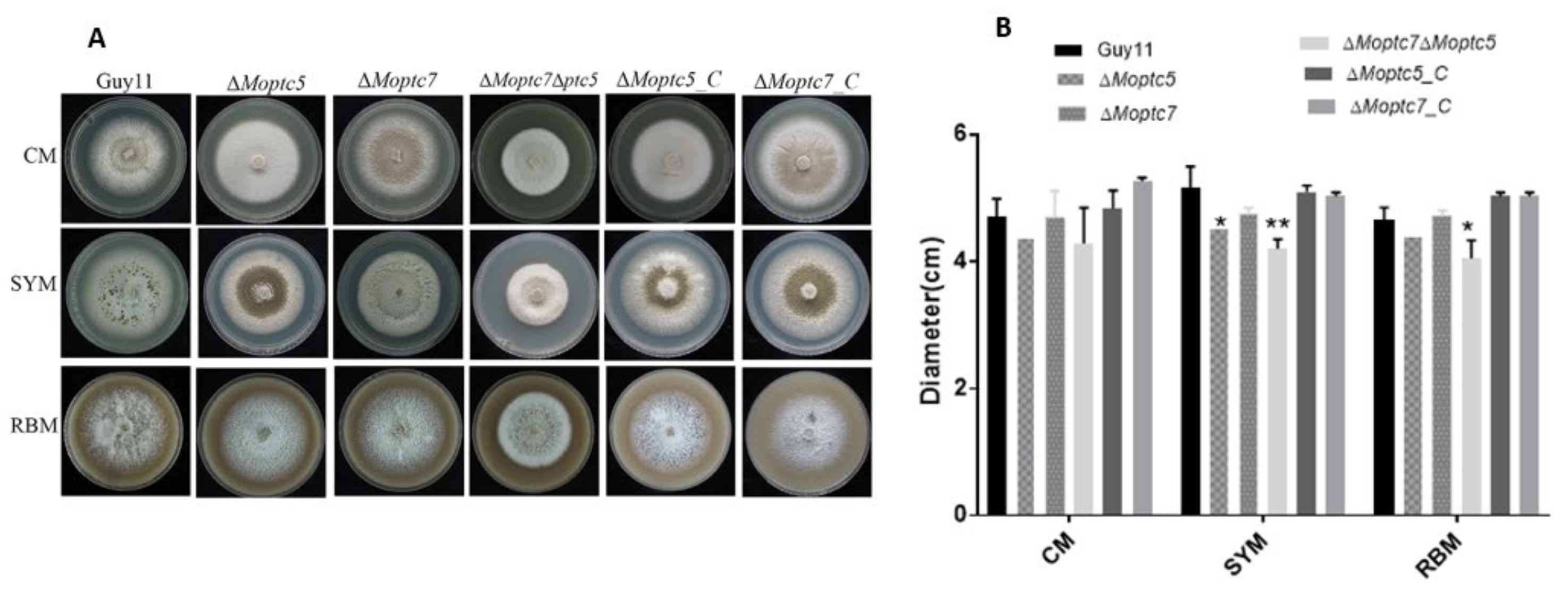
3.3. MoPtc5 and MoPtc7 Redundantly Contribute to Vegetative Growth of M. oryzae
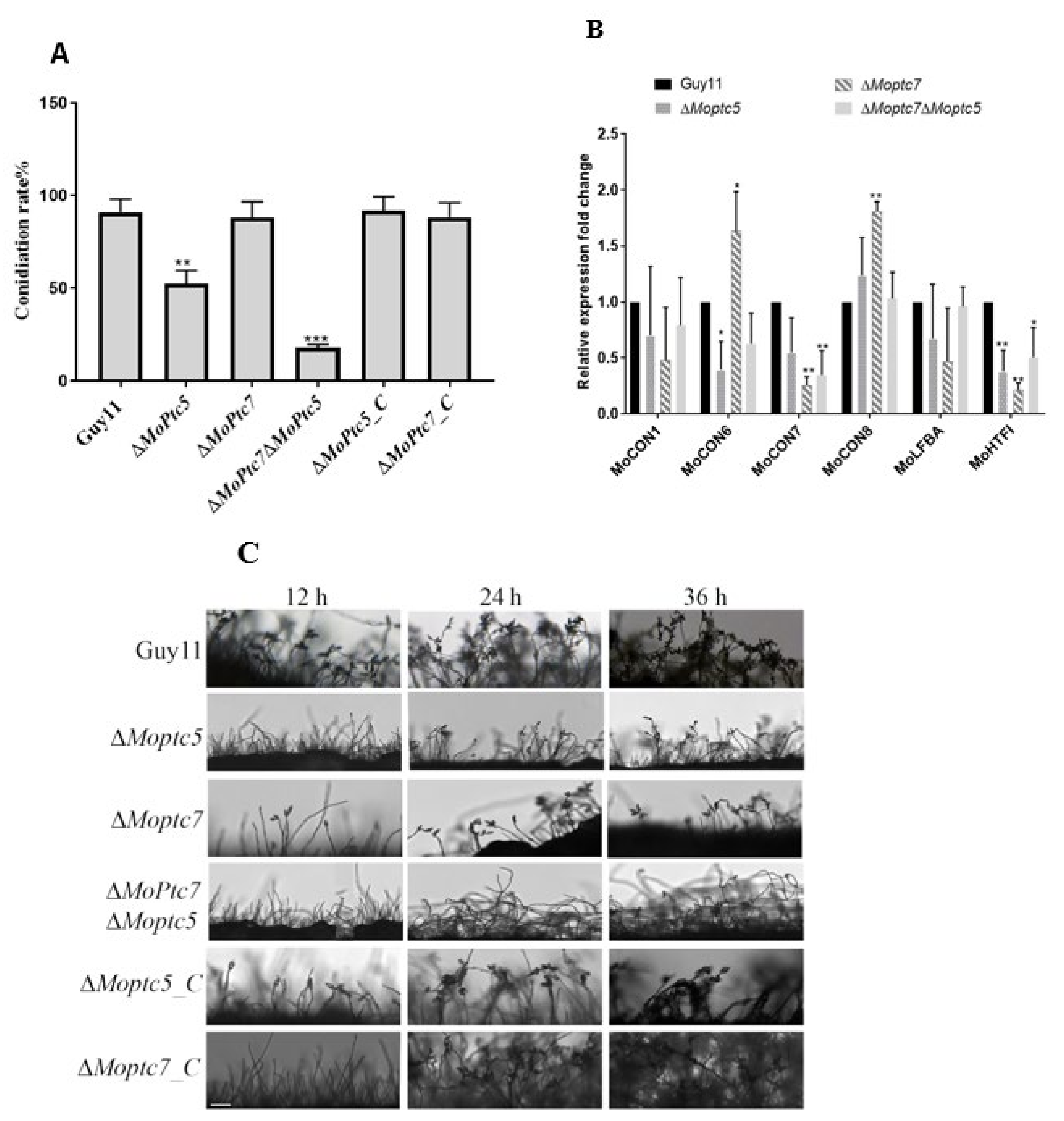
3.4. MoPtc5 Is Involved in Asexual Reproduction of the Rice Blast Fungus
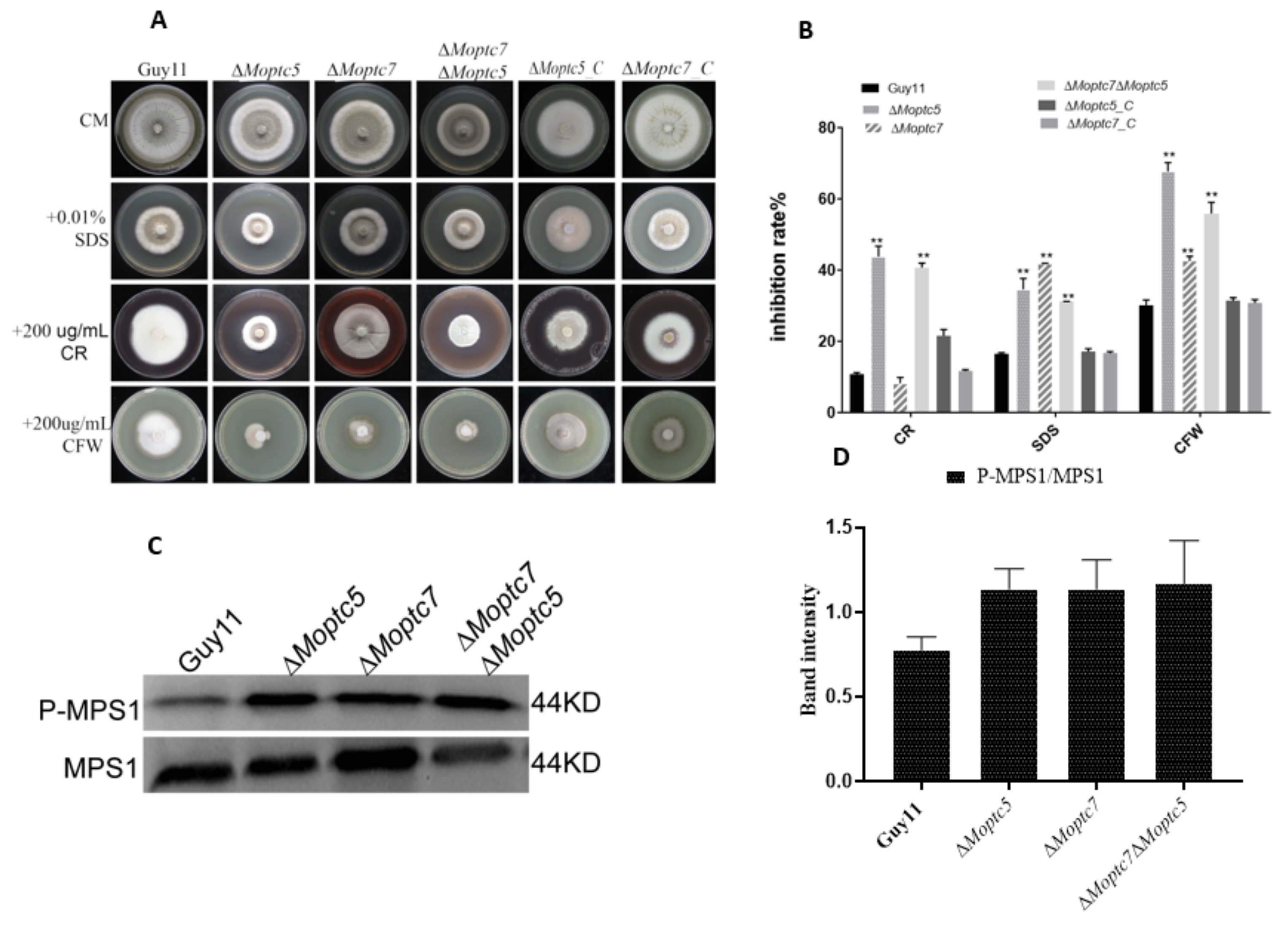
3.5. MoPtc5 and MoPtc7 Are Involved in Cell Wall Stress Resistance and Dephosphorylation of MoMps1
3.6. MoPtc5 and MoPtc7 May Mediate Chitin Synthesis to Confer Resistance to Cell Wall Stress in the Blast Fungus
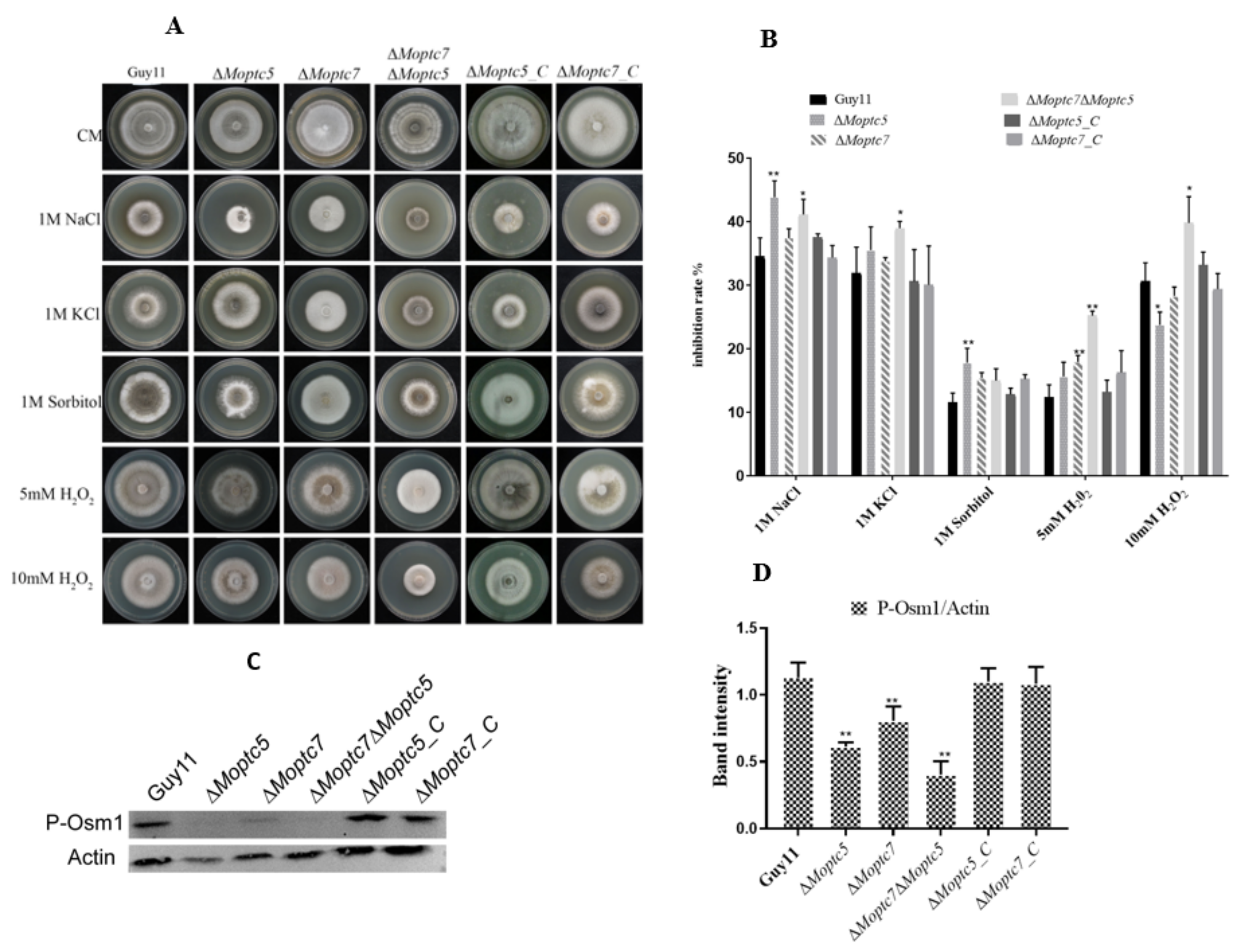
3.7. MoPtc5 and MoPtc7 Are Required for Osmotic and Oxidative Stress Tolerance
3.8. MoPtc5 and MoPtc7 Are Responsible for Appressorium Turgor Generation and Degradation of Glycogen in M. oryzae
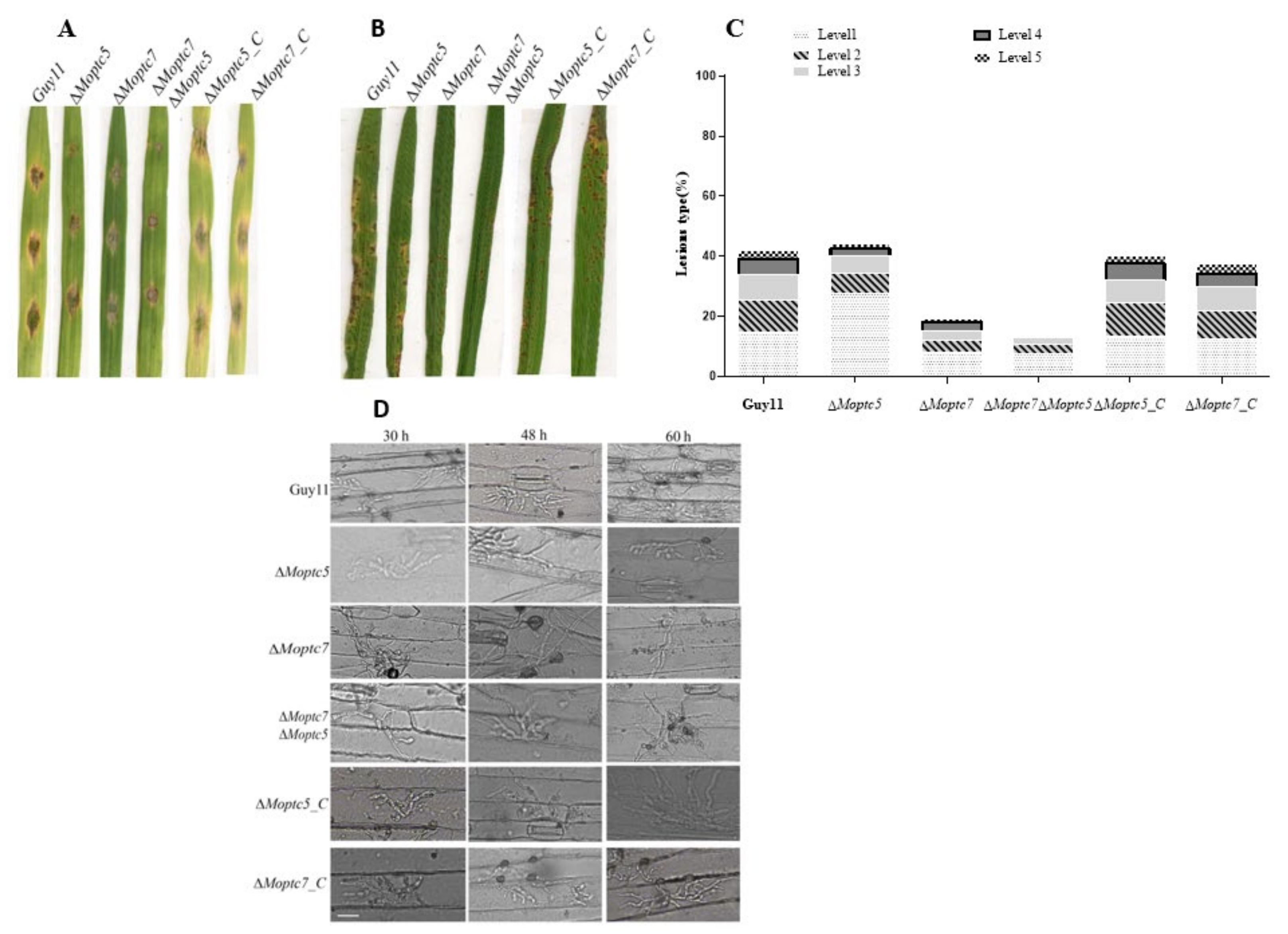
3.9. MoPtc5 and MoPtc7 Are Involved in M. oryzae Pathogenicity
3.10. Subcellular Localization of MoPtc5 and MoPtc7 Genes
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Talbot, N. Exploring the Biology of Magnaporthe grisea, on the Trail of a Cereal Killer. Annu. Rev. Microbiol. 2003, 57, 177–202. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dean, R.A.; Talbot, N.J.; Ebbole, D.J.; Farman, M.L.; Mitchell, T.K.; Orbach, M.J.; Thon, M.; Kulkarni, R.; Xu, J.-R.; Pan, H. The genome sequence of the rice blast fungus Magnaporthe grisea. Nature 2005, 434, 980–986. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ebbole, D.J. Magnaporthe as a model for understanding host-pathogen interactions. Annu. Rev. Phytopathol. 2007, 45, 437–456. [Google Scholar] [CrossRef] [PubMed]
- Cooper, A.J.; Jeitner, T.M. Central role of glutamate metabolism in the maintenance of nitrogen homeostasis in normal and hyperammonemic brain. Biomolecules 2016, 6, 16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, K.S.; Lee, Y.-H. Gene expression profiling during conidiation in the rice blast pathogen Magnaporthe oryzae. PLoS ONE 2012, 7, e43202. [Google Scholar] [CrossRef]
- Tucker, S.L.; Talbot, N.J. Surface attachment and pre-penetration stage development by plant pathogenic fungi. Annu. Rev. Phytopathol. 2001, 39, 385–417. [Google Scholar] [CrossRef]
- Howard, R.J.; Ferrari, M.A.; Roach, D.H.; Money, N.P. Penetration of hard substrates by a fungus employing enormous turgor pressures. Proc. Natl. Acad. Sci. USA 1991, 88, 11281–11284. [Google Scholar] [CrossRef] [Green Version]
- Adachi, O.; Kawai, T.; Takeda, K.; Matsumoto, M.; Tsutsui, H.; Sakagami, M.; Nakanishi, K.; Akira, S. Targeted disruption of the MyD88 gene results in loss of IL-1-and IL-18-mediated function. Immunity 1998, 9, 143–150. [Google Scholar] [CrossRef] [Green Version]
- Xu, J.-R.; Hamer, J.E. MAP kinase and cAMP signaling regulate infection structure formation and pathogenic growth in the rice blast fungus Magnaporthe grisea. Genes Dev. 1996, 10, 2696–2706. [Google Scholar] [CrossRef] [Green Version]
- Seshacharyulu, P.; Pandey, P.; Datta, K.; Batra, S.K. Phosphatase: PP2A structural importance, regulation and its aberrant expression in cancer. Cancer Lett. 2013, 335, 9–18. [Google Scholar] [CrossRef]
- Rodriguez, P.L. Protein phosphatase 2C (PP2C) function in higher plants. Plant Mol. Biol. 1998, 38, 919–927. [Google Scholar] [CrossRef] [PubMed]
- Ariño, J.; Velázquez, D.; Casamayor, A. Ser/Thr protein phosphatases in fungi: Structure, regulation and function. Microb. Cell 2019, 6, 217. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, X. Defining the Molecular Function of Mitochondrial Phosphatases; The University of Wisconsin-Madison: Madison, WI, USA, 2017. [Google Scholar]
- Tasdelen, I.; van Beekum, O.; Gorbenko, O.; Fleskens, V.; van den Broek, N.J.; Koppen, A.; Hamers, N.; Berger, R.; Coffer, P.J.; Brenkman, A.B. The serine/threonine phosphatase PPM1B (PP2Cβ) selectively modulates PPARγ activity. Biochem. J. 2013, 451, 45–53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Warmka, J.; Hanneman, J.; Lee, J.; Amin, D.; Ota, I. Ptc1, a type 2C Ser/Thr phosphatase, inactivates the HOG pathway by dephosphorylating the mitogen-activated protein kinase Hog1. Mol. Cell. Biol. 2001, 21, 51–60. [Google Scholar] [CrossRef] [Green Version]
- Ariño, J.; Casamayor, A.; González, A. Type 2C protein phosphatases in fungi. Eukaryot. Cell 2011, 10, 21–33. [Google Scholar] [CrossRef] [Green Version]
- Du, Y.; Walker, L.; Novick, P.; Ferro-Novick, S. Ptc1p regulates cortical ER inheritance via Slt2p. EMBO J. 2006, 25, 4413–4422. [Google Scholar] [CrossRef]
- Leroy, C.; Lee, S.E.; Vaze, M.B.; Ochsenbien, F.; Guerois, R.; Haber, J.E.; Marsolier-Kergoat, M.-C. PP2C phosphatases Ptc2 and Ptc3 are required for DNA checkpoint inactivation after a double-strand break. Mol. Cell 2003, 11, 827–835. [Google Scholar] [CrossRef]
- Cheng, M.; Olivier, P.; Diehl, J.A.; Fero, M.; Roussel, M.F.; Roberts, J.M.; Sherr, C.J. The p21Cip1 and p27Kip1 CDK ‘inhibitors’ are essential activators of cyclin D-dependent kinases in murine fibroblasts. EMBO J. 1999, 18, 1571–1583. [Google Scholar] [CrossRef] [PubMed]
- Shitamukai, A.; Hirata, D.; Sonobe, S.; Miyakawa, T. Evidence for antagonistic regulation of cell growth by the calcineurin and high osmolarity glycerol pathways in Saccharomyces cerevisiae. J. Biol. Chem. 2004, 279, 3651–3661. [Google Scholar] [CrossRef] [Green Version]
- Markovich, S.; Yekutiel, A.; Shalit, I.; Shadkchan, Y.; Osherov, N. Genomic approach to identification of mutations affecting caspofungin susceptibility in Saccharomyces cerevisiae. Antimicrob. Agents Chemother. 2004, 48, 3871–3876. [Google Scholar] [CrossRef]
- Krause-Buchholz, U.; Gey, U.; Wünschmann, J.; Becker, S.; Rödel, G. YIL042c and YOR090c encode the kinase and phosphatase of the Saccharomyces cerevisiae pyruvate dehydrogenase complex. FEBS Lett. 2006, 580, 2553–2560. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brock, A.K.; Willmann, R.; Kolb, D.; Grefen, L.; Lajunen, H.M.; Bethke, G.; Lee, J.; Nürnberger, T.; Gust, A.A. The Arabidopsis mitogen-activated protein kinase phosphatase PP2C5 affects seed germination, stomatal aperture, and abscisic acid-inducible gene expression. Plant Physiol. 2010, 153, 1098–1111. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- González, A.; Ruiz, A.; Casamayor, A.; Arino, J. Normal function of the yeast TOR pathway requires the type 2C protein phosphatase Ptc1. Mol. Cell. Biol. 2009, 29, 2876–2888. [Google Scholar] [CrossRef] [Green Version]
- Talbot, N.J.; Ebbole, D.J.; Hamer, J.E. Identification and characterization of MPG1, a gene involved in pathogenicity from the rice blast fungus Magnaporthe grisea. Plant Cell 1993, 5, 1575–1590. [Google Scholar]
- Hou, Z.; Xue, C.; Peng, Y.; Katan, T.; Kistler, H.C.; Xu, J.-R. A mitogen-activated protein kinase gene (MGV1) in Fusarium graminearum is required for female fertility, heterokaryon formation, and plant infection. Mol. Plant Microbe Interact. 2002, 15, 1119–1127. [Google Scholar] [CrossRef] [Green Version]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Aron, O.; Wang, M.; Lin, L.; Batool, W.; Lin, B.; Shabbir, A.; Wang, Z.; Tang, W. MoGLN2 is important for vegetative growth, conidiogenesis, maintenance of cell wall integrity and pathogenesis of Magnaporthe oryzae. J. Fungi 2021, 7, 463. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Liu, H.; Du, D.; Yu, Y.; Ma, C.; Jiao, F.; Yao, H.; Lu, C.; Zhang, W. Identification of novel laminin-and fibronectin-binding proteins by far-western blot: Capturing the adhesins of Streptococcus suis type 2. Front. Cell. Infect. Microbiol. 2015, 5, 82. [Google Scholar] [CrossRef]
- Liu, X.; Qian, B.; Gao, C.; Huang, S.; Cai, Y.; Zhang, H.; Zheng, X.; Wang, P.; Zhang, Z. The putative protein phosphatase MoYvh1 functions upstream of MoPdeH to regulate the development and pathogenicity in Magnaporthe oryzae. Mol. Plant-Microbe Interact. 2016, 29, 496–507. [Google Scholar] [CrossRef] [Green Version]
- Lin, L.; Cao, J.; Du, A.; An, Q.; Chen, X.; Yuan, S.; Batool, W.; Shabbir, A.; Zhang, D.; Wang, Z. eIF3k Domain-Containing Protein Regulates Conidiogenesis, Appressorium Turgor, Virulence, Stress Tolerance, and Physiological and Pathogenic Development of Magnaporthe oryzae Oryzae. Front. Plant Sci. 2021, 12, 748120. [Google Scholar] [CrossRef]
- Novaković, L.; Guo, T.; Bacic, A.; Sampathkumar, A.; Johnson, K.L. Hitting the wall—Sensing and signaling pathways involved in plant cell wall remodeling in response to abiotic stress. Plants 2018, 7, 89. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Chen, Y.; Li, B.; Chen, T.; Tian, S. Reactive oxygen species: A generalist in regulating development and pathogenicity of phytopathogenic fungi. Comput. Struct. Biotechnol. J. 2020, 18, 3344–3349. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.-X.; Miller, L.A.; Hartman, G.L. Melanin-independent accumulation of turgor pressure in appressoria of Phakopsora pachyrhizi. Phytopathology 2014, 104, 977–984. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, S.-R.; Hao, Z.-M.; Wang, L.-H.; Shen, S.; Cao, Z.-Y.; Xin, Y.-Y.; Hou, M.-L.; Gu, S.-Q.; Han, J.-M.; Dong, J.-G. StRas2 regulates morphogenesis, conidiation and appressorium development in Setosphaeria turcica. Microbiol. Res. 2012, 167, 478–486. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Biregeya, J.; Anjago, W.M.; Pan, S.; Zhang, R.; Yang, Z.; Chen, M.; Felix, A.; Xu, H.; Lin, Y.; Nkurikiyimfura, O.; et al. Type 2C Protein Phosphatases MoPtc5 and MoPtc7 Are Crucial for Multiple Stress Tolerance, Conidiogenesis and Pathogenesis of Magnaporthe oryzae. J. Fungi 2023, 9, 1. https://doi.org/10.3390/jof9010001
Biregeya J, Anjago WM, Pan S, Zhang R, Yang Z, Chen M, Felix A, Xu H, Lin Y, Nkurikiyimfura O, et al. Type 2C Protein Phosphatases MoPtc5 and MoPtc7 Are Crucial for Multiple Stress Tolerance, Conidiogenesis and Pathogenesis of Magnaporthe oryzae. Journal of Fungi. 2023; 9(1):1. https://doi.org/10.3390/jof9010001
Chicago/Turabian StyleBiregeya, Jules, Wilfred M. Anjago, Shu Pan, Ruina Zhang, Zifeng Yang, Meilian Chen, Abah Felix, Huxiao Xu, Yaqi Lin, Oswald Nkurikiyimfura, and et al. 2023. "Type 2C Protein Phosphatases MoPtc5 and MoPtc7 Are Crucial for Multiple Stress Tolerance, Conidiogenesis and Pathogenesis of Magnaporthe oryzae" Journal of Fungi 9, no. 1: 1. https://doi.org/10.3390/jof9010001






