The Accuracy of Three-Dimensional Soft Tissue Simulation in Orthognathic Surgery—A Systematic Review
Abstract
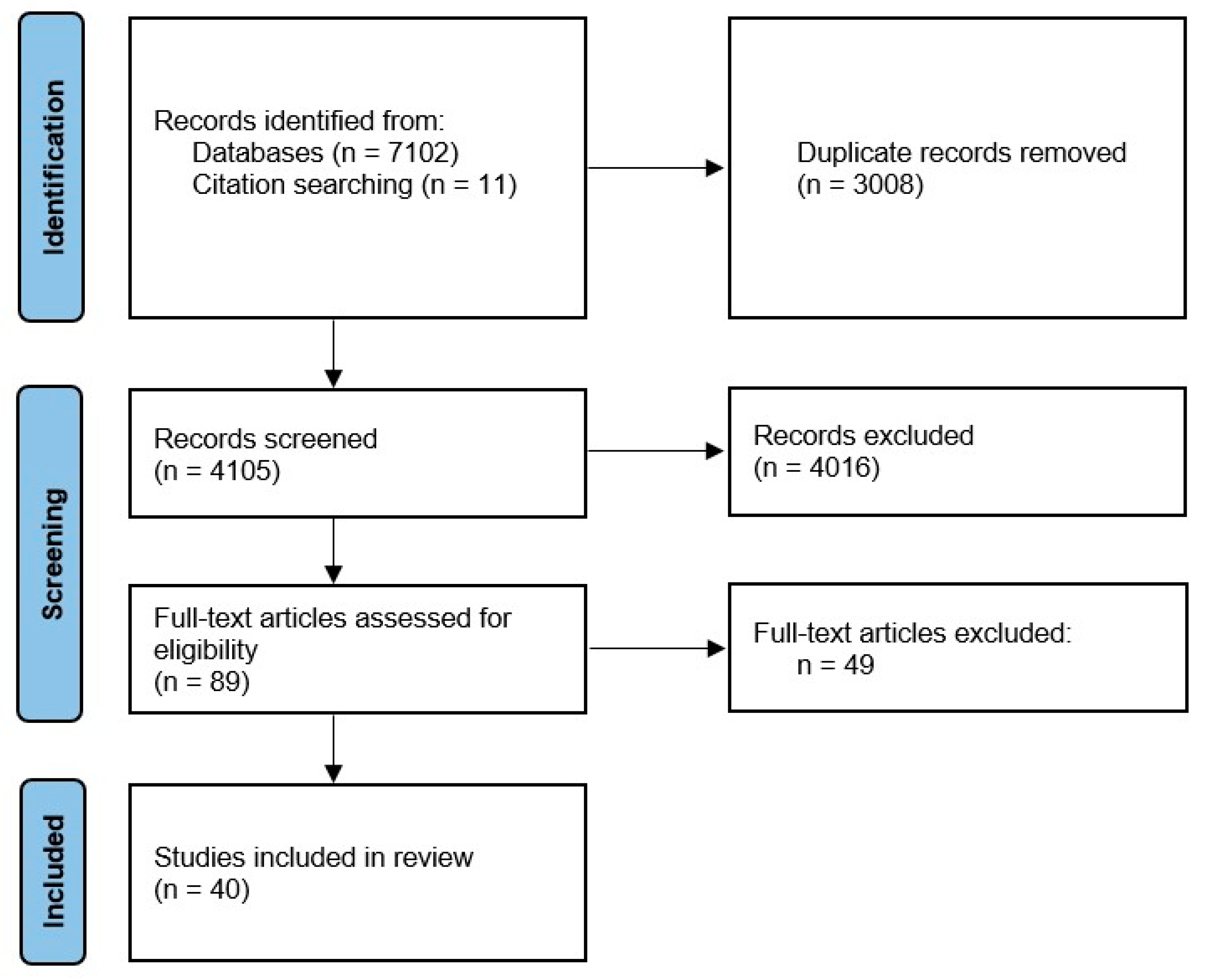
1. Introduction
2. Materials and Methods
Study Quality and Risk of Bias Assessment
3. Results
| Year, First Author | Sample Size | CBCT/MSCT a | Three-Dimensional Photo a | Software Package and/or Algorithm | Type of Surgery | Results b |
|---|---|---|---|---|---|---|
| 2004, Chabanas [42] | 3 | MSCT ** | No | FEM | NR | ME range: 1–1.5 mm, MaxE: 3–6 mm |
| 2007, Mollemans [3] | 10 | MSCT ** | Yes | (1) Linear FEM; (2) non-linear FEM; (3) MSM; (4) MTM | TRIMAX, BIMAX, BSSO, BSSO + Ch, LFI + Ch | Average median distance for MTM: 0.60 mm, FEM: 0.60 mm, MSM: 0.64 mm, NFEM: 0.63 mm; average 90th percentile distance for MTM: 1.48 mm, FEM: 1.51 mm, MSM: 1.67 mm, NFEM: 1.71 mm; highest accuracy: FEM and MTM |
| 2007, Marchetti [46] | 25 | MSCT | No | VISU system | LFI, BSSO, LFI + Ch, BSSO + Ch, BIMAX, TRIMAX | Error < 2 mm in 80% (20 of 25) of the patients |
| 2010, Bianchi [28] | 10 | CBCT | No | SurgiCase CMF Pro v.1.2 | BIMAX/TRIMAX | ME: 0.94 mm; error < 2 mm in 86.8% of the simulations; 90th percentile: 2.24 mm; 95th percentile: 2.81 mm |
| 2010, Ulusoy [43] | 6 | MSCT ** | No | Dynamic volume spline | BIMAX * | ME: 1.8 mm |
| 2011, Centenero [26] | 16 | MSCT/CBCT | No | SimPlant ProOMS v.10.1 | BIMAX, TRIMAX, BSSO + Ch | 5 of 8 ST measurements: high degree of correlation; 3 measurements: medium degree of correlation |
| 2011, Marchetti [29] | 10 | MSCT | No | SurgiCase CMF Pro v.1.2 | BIMAX, TRIMAX | ME: 0.75 +/− 0.78 mm; error < 2 mm in 91% of the simulations; 90th percentile: 1.94 mm; 95th percentile: 2.47 mm |
| 2013, Schendel [38] | 23 | CBCT | Yes c | 3dMDVultus—MSM | LFI, BSSO, Ch | Entire face ME: 0.27 mm, ComR: 1.10 mm, ComL: 0.99 mm, Pog: 0.79 mm |
| 2013, Shafi [19] | 13 | CBCT | No | Maxilim v.2.2.0—MTM | LFI | ME: 0.97 mm; all anatomical regions with error significantly < 3.0 mm, exception UL error: 2.73 +/− 1.72; overprediction of UL |
| 2013, Nadjmi [11] | 13 | CBCT ** lat ceph | Yes ** | (1) 2D Dolphin v.10—fixed hard-tissue-to-soft-tissue ratios; (2) Maxilim—MTM | LFI, LFI+Ch, BIMAX, TRIMAX | Dolphin range of error in horizontal position: −1.41 to 1.20 mm, in vertical position: −1.85 to 1.55 mm; Maxilim range of error in horizontal position: −1.60 to 1.50 mm, in vertical position: −4.25 to 2.42 mm. No statistical differences between software, exception SA in Maxilim |
| 2014, Terzic [44] | 13 | MSCT/CBCT | Yes c | 3dMDvultus v.2.2.0.8—MSM | BSSO, BSSO+Ch, BIMAX, TRIMAX | ME for the upper part: +0.27 mm, the lower part: –0.64 mm; in the lower part, error < +/− 1 mm 26.9%, >+/− 1 mm 73.1%, >+/− 2 mm 49.5%, and >+/− 3 mm 29.8% |
| 2014, Nadjmi [24] | 20 | CBCT | No | Maxilim—MTM | BSSO, BIMAX, TRIMAX | ME: 1.18 mm; 84% of errors between −2 mm and +2 mm |
| 2015, Ullah [37] | 13 | CBCT | No | 3dMDVultus v.2.2.0—MSM | LFI | ME: 0.92 mm (0.3–2.4 mm); 90th percentile from 0.65 mm (chin) to 1.17 mm (UL); ME significantly < 3 mm; the 95% CI in all regions < 2 mm |
| 2015, Khambay [45] | 10 | CBCT ** | No | 3dMDvultus v.2.2.0—MSM | LFI | ME for 95th percentile: 0.98–0.56 mm, for 90th percentile: 0.91–0.50 mm; error < 2 mm: 94.4%—85.2% points; RMS error: 2.49–0.94 mm; RMS difference for all measurements: 1.3 mm |
| 2015, Nam [27] | 29 | MSCT | No | Simplant Pro | BIMAX, TRIMAX | ME in all landmarks: 2.03 mm; error < 2 mm: 52.8%; absolute error values in the x-axis: 0.73 mm, y-axis: 1.39 mm, z-axis 0.85 mm; error significantly > 2 mm: ChR, ChL, LL, Pog; MaxE: 2.38 mm in ChL, MinE: 0.84 mm in pronasale |
| 2015a, Liebregts [23] | 60 | CBCT | No | Maxilim—MTM | BIMAX | Landmarks: MaxE at LI: 3.1 +/− 1.4 mm, MinE at SN: 1.5 +/− 0.6 mm; surfaces: entire face ME: 0.81 +/− 0.22 mm, for UL: 1.2 +/− 0.6 mm, LL: 1.4 +/− 0.5 mm, chin: 1.1 +/− 0.6 mm; error equal to or < 1 mm: 83.3%, < 2 mm: 100%; ME among patients who had a V-Y closure was significantly smaller than those without a V-Y closure |
| 2015b, Liebregts [22] | 100 | CBCT | No | Maxilim v.2.2.2.1—MTM | BSSO | Landmarks: ME at SN: 1.1 +/− 0.5 mm, at LS: 1.5 +/− 0.7 mm, at LI: 2.0 +/− 1.0 mm, at sublabial: 1.7 +/− 1.1 mm, at Pog: 1.5 +/− 0.9 mm; surface: entire face ME: 0.9 +/− 0.3 mm; error equal to or <2 mm: 100%, <1 mm: 78%; ME for UL: 0.9+/− 0.5, LL: 1.2+/− 0.5, and chin: 0.8 +/− 0.5 mm; average absolute error less or equal to 2 mm for UL: 98%, for LL: 94%, and for the chin: 97% |
| 2015, Van Hemelen [25] | 31 | CBCT | No | Maxilim—MTM | BSSO, BSSO + Ch, LFI, LFI + Ch, BIMAX, TRIMAX | ME in the horizontal direction: 1.48 mm, in the vertical direction: 1.46 mm |
| 2016, Liebregts [21] | 60 | CBCT | No | Maxilim—MTM | BIMAX | ME: 1.0 +/− 0.9 mm in alar width |
| 2016, Resnick [15] | 7 | CBCT | Yes c | Dolphin 3D v.11.8—sparse landmark-based algorithm | LFI | ME: 2.91 +/− 2.16 mm, for midline points: 1.66 +/− 1.82 mm, for lateral points: 3.84 +/− 1.92 mm; 2 (33%) midline points with error > 2 mm (SN, SA), 6 (75%) lateral points > 2 mm; ME at NLA: 8.1 +/− 5.6 degrees |
| 2017, Kim [9] | 40 | MSCT ** | Yes c | FEM with the mucosa sliding effect | BIMAX, TRIMAX | Quantitative: entire face ME: 1.1 +/− 0.3 mm, UL: 1.2 +/− 0.7 mm, LL: 1.5 +/− 0.7 mm, chin: 1.3 +/− 0.7; qualitative: 80% (32/40) clinically acceptable |
| 2021, Kim [41] | 35 | MSCT ** | Yes c | FEM with the sliding effect of the lip and the mucosa | BSSO, BIMAX, TRIMAX | Quantitative: entire face ME: 1.03 +/− 0.30 mm, UL: 0.86 +/− 0.36 mm, LL: 1.10 +/− 0.41 mm, chin: 1.08 +/− 0.51 mm; qualitative: improvement in lips compared with previous FEM methods |
| 2017, Mundluru [20] | 13 | CBCT | No | Maxilim—MTM | BIMAX, BSSO, BSSO+Ch | Underprediction of ST changes; signed ME from −0.55 to 0.43 mm; absolute ME from 0.6 to 1.3 mm |
| 2018, Holzinger [35] | 16 | MSCT | No | SOTIRIOS | NR—surgery first | ME: 1.46 +/− 1.53 mm; 50% < 1.03 mm, 80% < 2.20 mm, and 95% up to 4.34 mm |
| 2019, Knoops [16] | 7 | CBCT | No | (1) Dolphin 3D v.11.95—sparse landmark-based algorithm; (2) ProPlan CMF v.3.0.1—FDM; (3) PFEM | LFI | RMSDolphin = 1.8 +/− 0.8 mm, RMSPro-Plan = 1.2+/− 0.4 mm, and RMSPFEM = 1.3+/− 0.4 mm; average percentage of points < 2 mm: PDolphin = 83+/− 12%, PProPlan = 91+/− 9%, and PPFEM = 88+/− 10%; better results for ProPlan and PFEM compared to Dolphin |
| 2019, Elshebiny [14] | 20 | CBCT | No | Dolphin 3D v.11.9—sparse landmark-based algorithm | BIMAX/TRIMAX | Statistically significant differences in 2 angular measurements (FNA and NLA) and in 3 linear measurements (SA, UL length, and subalar width) |
| 2021, Cunha [32] | 16 | MSCT ** | No | OrtogOnBlender-OOB—MSM | BIMAX/TRIMAX | ME for all landmarks < 2 mm, entire face ME: 1.07 mm; MaxE: ChR, ChL, and SB |
| 2021, Willinger [18] | 19 | MSCT/CBCT ** | No | (1) IPS Case Designer—MTM; (2) Dolphin 3D v.11.95—sparse landmark-based algorithm | Modified IQLFIIO +/− BSSO | IR level: Dolphin ME: 2.90 +/− 2.1 mm, IPS ME: 1.70 +/− 1.3 mm; SF level: Dolphin ME: 3.57 +/− 2.0 mm, IPS ME: 1.34 +/− 0.9 mm; Li level: Dolphin ME: 2.48 +/− 1.9 mm, IPS ME: 2.25 +/− 1.6 mm; MaxE for Dolphin at SF level |
| 2021, Tanikawa [36] | 72 | No—lat ceph | Yes | Geometric morphometric methods (GMMs), DL | BIMAX | System error: 0.89 ± 0.30 mm; MaxE of 0.8–1.2 mm in the nasal ala, chin, corner of the mouth; total success rate at <1 mm: 54%, and at <2 mm: 100% |
| 2021, ter Horst [33] | 14 | CBCT | Yes c | DL; IPS CaseDesigner—MTM | BSSO | DL-based: lower face ME: 1.0 +/− 0.6 mm, simulations with MaxE of 1 mm: 64.3% and of 2 mm: 92.9%; RMS: 1.2 +/− 0.6 mm; ME: for LL 1.1 +/− 0.9 mm; for the chin: 1.4 +/− 0.9 mm. MTM-based: lower face ME: 1.5 +/− 0.5 mm, simulations with MaxE of 1 mm: 21.4% and of 2 mm: 85.7%; RMS: 2.0 +/− 0.7 mm; ME for LL: 1.7 +/− 0.9 mm; chin: 2.0 +/− 1.0 mm; DL model had higher accuracy |
| 2021, Alcañiz [39] | 10 | CBCT ** | Yes ** | FEM | LFI, LFII, BSSO, USSO, Ch, BIMAX | Surface with error < 3 mm with coarse meshes: 92%, with fine meshes: 95% |
| 2022, Lee [10] | 10 | CBCT ** | Yes **c | ProPlan CMF—FDM | BIMAX | Entire face ME: 0.73 +/− 0.21 mm, for LL: 1.42 +/− 0.77 mm, for UL: 1.14 +/− 0.80 mm, for chin: 0.95 +/− 0.58 mm; error < 2 mm: 90.9% |
| 2022, Gutiérrez [40] | 10 | CBCT ** | Yes ** | FEM | LFI, LFII, BSSO, USSO, Ch, BIMAX | All distances for both meshes and their mean distances significantly < 2 mm, except LL, RGo, and LGo; distances for all landmarks significantly < 3 mm, except for LL of the fine mesh |
| 2022, Yamashita [17] | 88 | CBCT | No | Dolphin 3D v.11.95—sparse landmark-based algorithm | BIMAX, TRIMAX | C II: underprediction with downward direction in S-Y, S-Z, LI-Y, SB-Y, Pog-Y, Pog-Z, Gn-Y, Gn-Z, Me-Y, Me-Z, values > 2 mm: LI-Y, SB-Y, Pog-Y, Gn-Y, Gn-Z, Me-Y; MaxE LI-Y: 2.73 mm. C III: overprediction and downward direction in Pog-Z, Gn-Y, Gn-Z, Me-Y, and Me-Z, all discrepancies < 2 mm |
| 2022, Ma [8] | 40 | MSCT ** | No | FSC-Net, point cloud DL | NR | Qualitative: FSC-Net comparable with FEM-RLSE; quantitative: landmarks entire face ME: 2.95 +/− 0.61 mm; surface entire face ME: 1.55 +/− 0.30 mm, lips: 1.58 +/− 0.26 mm, chin: 2.11 +/− 0.77 mm; FSC-Net comparable with FEM-RLSE |
| 2022, Awad [34] | 20 | CBCT | Yes | IPS CaseDesigner v.2.1.4.4—MTM | BIMAX | Entire face ME: −1.5 to 1.4 mm, UL: −2.5 to 1.3 mm, LL: −2.1 to 2.5 mm, chin: −1.8 to 2.6 mm |
| 2022, Hou [31] | 58 | CBCT | Yes c | ProPlan CMF—FDM | BIMAX | Entire face ME: 1.43 +/− 0.40 mm; error of UL, LL, chin, right external buccal, and left external buccal > 2.0 mm; LL the least predictable: 2.69 ± 1.25 mm |
| 2023, Şenyürek [30] | 16 | CBCT ** | No | ProPlan CMF v.3.0—FDM | LFI | Error in UL and LL: 1.49 +/− 0.77 mm, in cheeks: 0.98 +/− 0.34 mm, nose: 0.86 +/− 0.23 mm, and eyes: 0.76 +/− 0.32 mm |
| 2023, Ruggiero [47] | 5 | CBCT + MRI | No | FEM with patient-specific model generated from CBCT and MRI | BIMAX | Midface ME: 0.55 +/− 2.29 mm |
| 2024, Fang [48] | 40 | MSCT ** | No | DL, ACMT-Net with the CPSA module | BIMAX | Quantitative: surface entire face ME: 1.06 +/− 0.43 mm, UL: 1.13 +/− 0.71 mm, LL: 1.23 +/− 0.48 mm, chin: 1.13 +/− 0.62 mm; landmarks entire face: ME 2.44 +/− 0.45 mm, upper face: 1.23 +/− 0.47 mm, lower face: 3.25 +/− 0.66 mm Qualitative: 77.5% (31/40) of the simulations clinically acceptable |
4. Discussion
4.1. Image Acquisition (Pre- and Postoperative)
4.2. Virtual Osteotomies and VTP
4.3. Considerations Regarding Additional Surgical Procedures
4.4. Soft Tissue Simulation Algorithms
4.5. Rigid Registration of Preoperative/Simulated and Postoperative Data
4.6. Postprocessing and Analysis
4.7. Accuracy Cut-Off
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Swennen, G.R.J.; Schutyser, F.; Barth, E.-L.; De Groeve, P.; De Mey, A. A New Method of 3-D Cephalometry Part I. J. Craniofacial Surg. 2006, 17, 314–325. [Google Scholar] [CrossRef]
- Swennen, G.R.J.; Schutyser, F.; Hausamen, J.-E. Three-Dimensional Cephalometry; Springer: Berlin/Heidelberg, Germany, 2006; Chapter 8; p. 291. [Google Scholar]
- Mollemans, W.; Schutyser, F.; Nadjmi, N.; Maes, F.; Suetens, P. Predicting soft tissue deformations for a maxillofacial surgery planning system: From computational strategies to a complete clinical validation. Med. Image Anal. 2007, 11, 282–301. [Google Scholar] [CrossRef]
- Arnett, G.W.; Gunson, M.J. Facial planning for orthodontists and Oral surgeons. Am. J. Orthod. Dentofac. Orthop. 2004, 126, 290–295. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Kuang, T.; Rodrigues, Y.L.; Gateno, J.; Shen, S.G.F.; Wang, X.; Deng, H.; Yuan, P.; Alfi, D.M.; Liebschner, M.A.K.; et al. A New Approach of Predicting Facial Changes Following Orthognathic Surgery Using Realistic Lip Sliding Effect; Springer: Berlin/Heidelberg, Germany, 2019; pp. 336–344. [Google Scholar]
- Tondin, G.M.; de Oliveira Coelho Dutra Leal, M.; Costa, S.T.; Grillo, R.; Jodas, C.R.P.; Teixeira, R.G. Evaluation of the accuracy of virtual planning in bimaxillary orthognathic surgery: A systematic review. Br. J. Oral Maxillofac. Surg. 2022, 60, 412–421. [Google Scholar] [CrossRef]
- Gaber, R.M.; Shaheen, E.; Falter, B.; Araya, S.; Politis, C.; Swennen, G.R.J.; Jacobs, R. A Systematic Review to Uncover a Universal Protocol for Accuracy Assessment of 3-Dimensional Virtually Planned Orthognathic Surgery. J. Oral Maxillofac. Surg. 2017, 75, 2430–2440. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Xiao, D.; Kim, D.; Lian, C.; Kuang, T.; Liu, Q.; Deng, H.; Yang, E.; Liebschner, M.A.K.; Gateno, J.; et al. Simulation of Postoperative Facial Appearances via Geometric Deep Learning for Efficient Orthognathic Surgical Planning. IEEE Trans. Med. Imaging 2022, 42, 336–345. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Ho, D.C.; Mai, H.; Zhang, X.; Shen, S.G.F.; Shen, S.; Yuan, P.; Liu, S.; Zhang, G.; Zhou, X.; et al. A clinically validated prediction method for facial soft-tissue changes following double-jaw surgery. Med. Phys. 2017, 44, 4252–4261. [Google Scholar] [CrossRef]
- Lee, K.J.C.; Tan, S.L.; Low, H.Y.A.; Chen, L.J.; Yong, C.W.; Chew, M.T. Accuracy of 3-dimensional soft tissue prediction for orthognathic surgery in a Chinese population. J. Stomatol. Oral Maxillofac. Surg. 2022, 123, 551–555. [Google Scholar] [CrossRef] [PubMed]
- Nadjmi, N.; Tehranchi, A.; Azami, N.; Saedi, B.; Mollemans, W. Comparison of soft-tissue profiles in Le Fort I osteotomy patients with Dolphin and Maxilim softwares. Am. J. Orthod. Dentofac. Orthop. 2013, 144, 654–662. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Whiting, P.F.; Rutjes, A.W.S.; Westwood, M.E.; Mallett, S.; Deeks, J.J.; Reitsma, J.B.; Leeflang, M.M.G.; Sterne, J.A.C.; Bossuyt, P.M.M.; QUADAS-2 Group. QUADAS-2: A revised tool for the quality assessment of diagnostic accuracy studies. Ann. Intern. Med. 2011, 155, 529–536. [Google Scholar] [CrossRef]
- Elshebiny, T.; Morcos, S.; Mohammad, A.; Quereshy, F.; Valiathan, M. Accuracy of Three-Dimensional Soft Tissue Prediction in Orthognathic Cases Using Dolphin Three-Dimensional Software. J. Craniofacial. Surg. 2019, 30, 525–528. [Google Scholar] [CrossRef] [PubMed]
- Resnick, C.M.; Dang, R.R.; Glick, S.J.; Padwa, B.L. Accuracy of three-dimensional soft tissue prediction for Le Fort I osteotomy using Dolphin 3D software: A pilot study. Int. J. Oral Maxillofac. Surg. 2017, 46, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Knoops, P.G.M.; Borghi, A.; Breakey, R.W.F.; Ong, J.; Jeelani, N.U.O.; Bruun, R.; Schievano, S.; Dunaway, D.J.; Padwa, B.L. Three-dimensional soft tissue prediction in orthognathic surgery: A clinical comparison of Dolphin, ProPlan CMF, and probabilistic finite element modelling. Int. J. Oral Maxillofac. Surg. 2019, 48, 511–518. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, A.L.; Iwaki Filho, L.; Ferraz, F.W.d.S.; Ramos, A.L.; Previdelli, I.T.d.S.; Pereira, O.C.N.; Tolentino, E.d.S.; Chicarelli, M.; Iwaki, L.C.V. Accuracy of three-dimensional soft tissue profile prediction in orthognathic surgery. Oral Maxillofac. Surg. 2022, 26, 271–279. [Google Scholar] [CrossRef]
- Willinger, K.; Guevara-Rojas, G.; Cede, J.; Schicho, K.; Stamm, T.; Klug, C. Accuracy of Soft Tissue Prediction of 2 Virtual Planning Systems in Patients Undergoing Intraoral Quadrangular Le Fort II Osteotomy. Plast. Reconstr. Surg. Glob. Open 2021, 9, e3326. [Google Scholar] [CrossRef]
- Shafi, M.I.; Ayoub, A.; Ju, X.; Khambay, B. The accuracy of three-dimensional prediction planning for the surgical correction of facial deformities using Maxilim. Int. J. Oral Maxillofac. Surg. 2013, 42, 801–806. [Google Scholar] [CrossRef] [PubMed]
- Mundluru, T.; Almukhtar, A.; Ju, X.; Ayoub, A. The accuracy of three-dimensional prediction of soft tissue changes following the surgical correction of facial asymmetry: An innovative concept. Int. J. Oral Maxillofac. Surg. 2017, 46, 1517–1524. [Google Scholar] [CrossRef]
- Liebregts, J.; Xi, T.; Schreurs, R.; van Loon, B.; Bergé, S.; Maal, T. Three-dimensional virtual simulation of alar width changes following bimaxillary osteotomies. Int. J. Oral Maxillofac. Surg. 2016, 45, 1315–1321. [Google Scholar] [CrossRef]
- Liebregts, J.H.F.; Timmermans, M.; de Koning, M.J.J.; Bergé, S.J.; Maal, T.J.J. Three-Dimensional Facial Simulation in Bilateral Sagittal Split Osteotomy: A Validation Study of 100 Patients. J. Oral Maxillofac. Surg. 2015, 73, 961–970. [Google Scholar] [CrossRef]
- Liebregts, J.; Xi, T.; Timmermans, M.; de Koning, M.; Bergé, S.; Hoppenreijs, T.; Maal, T. Accuracy of three-dimensional soft tissue simulation in bimaxillary osteotomies. J. Cranio-Maxillofac. Surg. 2015, 43, 329–335. [Google Scholar] [CrossRef]
- Nadjmi, N.; Defrancq, E.; Mollemans, W.; Hemelen, G.; Bergé, S. Quantitative validation of a computer-aided maxillofacial planning system, focusing on soft tissue deformations. Ann. Maxillofac. Surg. 2014, 4, 171. [Google Scholar] [CrossRef]
- van Hemelen, G.; van Genechten, M.; Renier, L.; Desmedt, M.; Verbruggen, E.; Nadjmi, N. Three-dimensional virtual planning in orthognathic surgery enhances the accuracy of soft tissue prediction. J. Cranio-Maxillofac. Surg. 2015, 43, 918–925. [Google Scholar] [CrossRef]
- Aboul-Hosn Centenero, S.; Hernández-Alfaro, F. 3D planning in orthognathic surgery: CAD/CAM surgical splints and prediction of the soft and hard tissues results—Our experience in 16 cases. J. Cranio-Maxillofac. Surg. 2012, 40, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Nam, K.-U.; Hong, J. Is Three-Dimensional Soft Tissue Prediction by Software Accurate? J. Craniofacial. Surg. 2015, 26, e729–e733. [Google Scholar] [CrossRef]
- Bianchi, A.; Muyldermans, L.; di Martino, M.; Lancellotti, L.; Amadori, S.; Sarti, A.; Marchetti, C. Facial Soft Tissue Esthetic Predictions: Validation in Craniomaxillofacial Surgery with Cone Beam Computed Tomography Data. J. Oral Maxillofac. Surg. 2010, 68, 1471–1479. [Google Scholar] [CrossRef] [PubMed]
- Marchetti, C.; Bianchi, A.; Muyldermans, L.; di Martino, M.; Lancellotti, L.; Sarti, A. Validation of new soft tissue software in orthognathic surgery planning. Int. J. Oral Maxillofac. Surg. 2011, 40, 26–32. [Google Scholar] [CrossRef]
- Şenyürek, S.A.; Ajami, S.; Ruggiero, F.; van de Lande, L.; Caron, C.J.J.M.; Schievano, S.; Dunaway, D.J.; Padwa, B.; Koudstaal, M.J.; Borghi, A. The Accuracy of Computer-Assisted Surgical Planning in Predicting Soft Tissue Responses After Le Fort I Osteotomy: Retrospective Analysis. J. Craniofacial. Surg. 2023, 34, 131–138. [Google Scholar] [CrossRef]
- Hou, L.; He, Y.; Yi, B.; Wang, X.; Liu, X.; Zhang, Y.; Li, Z. Evaluation of soft tissue prediction accuracy for orthognathic surgery with skeletal class III malocclusion using maxillofacial regional aesthetic units. Clin. Oral Investig. 2022, 27, 173–182. [Google Scholar] [CrossRef]
- Cunha, H.S.; da Costa Moraes, C.A.; de Faria Valle Dornelles, R.; da Rosa, E.L.S. Accuracy of three-dimensional virtual simulation of the soft tissues of the face in OrtogOnBlender for correction of class II dentofacial deformities: An uncontrolled experimental case-series study. Oral Maxillofac. Surg. 2021, 25, 319–335. [Google Scholar] [CrossRef]
- ter Horst, R.; van Weert, H.; Loonen, T.; Bergé, S.; Vinayahalingam, S.; Baan, F.; Maal, T.; de Jong, G.; Xi, T. Three-dimensional virtual planning in mandibular advancement surgery: Soft tissue prediction based on deep learning. J. Cranio-Maxillofac. Surg. 2021, 49, 775–782. [Google Scholar] [CrossRef] [PubMed]
- Awad, D.; Reinert, S.; Kluba, S. Accuracy of Three-Dimensional Soft-Tissue Prediction Considering the Facial Aesthetic Units Using a Virtual Planning System in Orthognathic Surgery. J. Pers. Med. 2022, 12, 1379. [Google Scholar] [CrossRef] [PubMed]
- Holzinger, D.; Juergens, P.; Shahim, K.; Reyes, M.; Schicho, K.; Millesi, G.; Perisanidis, C.; Zeilhofer, H.-F.; Seemann, R. Accuracy of soft tissue prediction in surgery-first treatment concept in orthognathic surgery: A prospective study. J. Cranio-Maxillofac. Surg. 2018, 46, 1455–1460. [Google Scholar] [CrossRef] [PubMed]
- Tanikawa, C.; Yamashiro, T. Development of novel artificial intelligence systems to predict facial morphology after orthognathic surgery and orthodontic treatment in Japanese patients. Sci. Rep. 2021, 11, 15853. [Google Scholar] [CrossRef]
- Ullah, R.; Turner, P.J.; Khambay, B.S. Accuracy of three-dimensional soft tissue predictions in orthognathic surgery after Le Fort I advancement osteotomies. Br. J. Oral Maxillofac. Surg. 2015, 53, 153–157. [Google Scholar] [CrossRef] [PubMed]
- Schendel, S.A.; Jacobson, R.; Khalessi, S. 3-Dimensional Facial Simulation in Orthognathic Surgery: Is It Accurate? J. Oral Maxillofac. Surg. 2013, 71, 1406–1414. [Google Scholar] [CrossRef] [PubMed]
- Alcañiz, P.; Pérez, J.; Gutiérrez, A.; Barreiro, H.; Villalobos, Á.; Miraut, D.; Illana, C.; Guiñales, J.; Otaduy, M.A. Soft-Tissue Simulation for Computational Planning of Orthognathic Surgery. J. Pers. Med. 2021, 11, 982. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez Venturini, A.; Guiñales Díaz de Cevallos, J.; del Castillo Pardo de Vera, J.L.; Alcañiz Aladrén, P.; Illana Alejandro, C.; Cebrián Carretero, J.L. A Quantitative and Qualitative Clinical Validation of Soft Tissue Simulation for Orthognathic Surgery Planning. J. Pers. Med. 2022, 12, 1460. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Kuang, T.; Rodrigues, Y.L.; Gateno, J.; Shen, S.G.F.; Wang, X.; Stein, K.; Deng, H.H.; Liebschner, M.A.K.; Xia, J.J. A novel incremental simulation of facial changes following orthognathic surgery using FEM with realistic lip sliding effect. Med. Image Anal. 2021, 72, 102095. [Google Scholar] [CrossRef]
- Chabanas, M.; Marécaux, C.; Chouly, F.; Boutault, F.; Payan, Y. Evaluating soft tissue simulation in maxillofacial surgery using preoperative and postoperative CT scans. Int. Congr. Ser. 2004, 1268, 419–424. [Google Scholar] [CrossRef]
- Ulusoy, İ.; Akagunduz, E.; Sabuncuoglu, F.; Gorgulu, S.; Ucok, O. Use of the dynamic volume spline method to predict facial soft tissue changes associated with orthognathic surgery. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2010, 110, e17–e23. [Google Scholar] [CrossRef]
- Terzic, A.; Combescure, C.; Scolozzi, P. Accuracy of Computational Soft Tissue Predictions in Orthognathic Surgery from Three-Dimensional Photographs 6 Months After Completion of Surgery: A Preliminary Study of 13 Patients. Aesthetic Plast. Surg. 2014, 38, 184–191. [Google Scholar] [CrossRef]
- Khambay, B.; Ullah, R. Current methods of assessing the accuracy of three-dimensional soft tissue facial predictions: Technical and clinical considerations. Int. J. Oral Maxillofac. Surg. 2015, 44, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Marchetti, C.; Bianchi, A.; Bassi, M.; Gori, R.; Lamberti, C.; Sarti, A. Mathematical Modeling and Numerical Simulation in Maxillofacial Virtual Surgery. J. Craniofacial. Surg. 2007, 18, 826–832. [Google Scholar] [CrossRef] [PubMed]
- Ruggiero, F.; Borghi, A.; Bevini, M.; Badiali, G.; Lunari, O.; Dunaway, D.; Marchetti, C. Soft tissue prediction in orthognathic surgery: Improving accuracy by means of anatomical details. PLoS ONE 2023, 18, e0294640. [Google Scholar] [CrossRef]
- Fang, X.; Kim, D.; Xu, X.; Kuang, T.; Lampen, N.; Lee, J.; Deng, H.H.; Liebschner, M.A.K.; Xia, J.J.; Gateno, J.; et al. Correspondence attention for facial appearance simulation. Med. Image Anal. 2024, 93, 103094. [Google Scholar] [CrossRef]
- Swennen, G.R.J. 3D Virtual Treatment Planning of Orthognathic Surgery: A Step-by-Step Approach for Orthodontists and Surgeons; Springer: Berlin/Heidelberg, Germany, 2017. [Google Scholar]
- Iblher, N.; Gladilin, E.; Stark, B.G. Soft-Tissue Mobility of the Lower Face Depending on Positional Changes and Age. Plast. Reconstr. Surg. 2013, 131, 372–381. [Google Scholar] [CrossRef] [PubMed]
- Guijarro-Martínez, R.; Swennen, G.R.J. Cone-beam computerized tomography imaging and analysis of the upper airway: A systematic review of the literature. Int. J. Oral Maxillofac. Surg. 2011, 40, 1227–1237. [Google Scholar] [CrossRef]
- Stratemann, S.; Huang, J.; Maki, K.; Miller, A.; Hatcher, D. Comparison of cone beam computed tomography imaging with physical measures. Dentomaxillofacial Radiol. 2008, 37, 80–93. [Google Scholar] [CrossRef]
- Eidson, L.; Cevidanes, L.H.S.; de Paula, L.K.; Hershey, H.G.; Welch, G.; Rossouw, P.E. Three-dimensional evaluation of changes in lip position from before to after orthodontic appliance removal. Am. J. Orthod. Dentofac. Orthop. 2012, 142, 410–418. [Google Scholar] [CrossRef]
- Kim, Y.-K.; Lee, N.-K.; Moon, S.-W.; Jang, M.-J.; Kim, H.-S.; Yun, P.-Y. Evaluation of soft tissue changes around the lips after bracket debonding using three-dimensional stereophotogrammetry. Angle Orthod. 2015, 85, 833–840. [Google Scholar] [CrossRef]
- van der Vlis, M.; Dentino, K.M.; Vervloet, B.; Padwa, B.L. Postoperative Swelling After Orthognathic Surgery: A Prospective Volumetric Analysis. J. Oral Maxillofac. Surg. 2014, 72, 2241–2247. [Google Scholar] [CrossRef]
- Baan, F.; Liebregts, J.; Xi, T.; Schreurs, R.; de Koning, M.; Bergé, S.; Maal, T. A New 3D Tool for Assessing the Accuracy of Bimaxillary Surgery: The OrthoGnathicAnalyser. PLoS ONE 2016, 11, e0149625. [Google Scholar] [CrossRef]
- Singh, G.D.; Singh, M. Virtual Surgical Planning: Modeling from the Present to the Future. J. Clin. Med. 2021, 10, 5655. [Google Scholar] [CrossRef] [PubMed]
- Lampen, N.; Kim, D.; Fang, X.; Xu, X.; Kuang, T.; Deng, H.H.; Barber, J.C.; Gateno, J.; Xia, J.; Yan, P. Deep learning for biomechanical modeling of facial tissue deformation in orthognathic surgical planning. Int. J. Comput. Assist. Radiol. Surg. 2022, 17, 945–952. [Google Scholar] [CrossRef] [PubMed]
- Knoops, P.G.M.; Papaioannou, A.; Borghi, A.; Breakey, R.W.F.; Wilson, A.T.; Jeelani, O.; Zafeiriou, S.; Steinbacher, D.; Padwa, B.L.; Dunaway, D.J.; et al. A machine learning framework for automated diagnosis and computer-assisted planning in plastic and reconstructive surgery. Sci. Rep. 2019, 9, 13597. [Google Scholar] [CrossRef] [PubMed]
- Bouletreau, P.; Makaremi, M.; Ibrahim, B.; Louvrier, A.; Sigaux, N. Artificial Intelligence: Applications in orthognathic surgery. J. Stomatol Oral Maxillofac. Surg. 2019, 120, 347–354. [Google Scholar] [CrossRef]
- Choi, R.Y.; Coyner, A.S.; Kalpathy-Cramer, J.; Chiang, M.F.; Campbell, J.P. Introduction to Machine Learning, Neural Networks, and Deep Learning. Transl. Vis. Sci. Technol. 2020, 9, 14. [Google Scholar]
- Almukhtar, A.; Ju, X.; Khambay, B.; McDonald, J.; Ayoub, A. Comparison of the Accuracy of Voxel Based Registration and Surface Based Registration for 3D Assessment of Surgical Change following Orthognathic Surgery. PLoS ONE 2014, 9, e93402. [Google Scholar] [CrossRef]
- Holte, M.B.; Sæderup, H.; Pinholt, E.M. Comparison of surface- and voxel-based registration on the mandibular ramus for long-term three-dimensional assessment of condylar remodelling following orthognathic surgery. Dentomaxillofac. Radiol. 2022, 51, 20210499. [Google Scholar] [CrossRef]
- Andriola, F.d.O.; Haas Junior, O.L.; Guijarro-Martínez, R.; Hernández-Alfaro, F.; Oliveira, R.B.D.; Pagnoncelli, R.M.; Swennen, G.R. Computed tomography imaging superimposition protocols to assess outcomes in orthognathic surgery: A systematic review with comprehensive recommendations. Dentomaxillofac. Radiol. 2022, 51, 20210340. [Google Scholar] [CrossRef] [PubMed]
- Maal, T.J.J.; Plooij, J.M.; Rangel, F.A.; Mollemans, W.; Schutyser, F.A.C.; Bergé, S.J. The accuracy of matching three-dimensional photographs with skin surfaces derived from cone-beam computed tomography. Int. J. Oral Maxillofac. Surg. 2008, 37, 641–646. [Google Scholar] [CrossRef] [PubMed]
- Plooij, J.M.; Swennen, G.R.J.; Rangel, F.A.; Maal, T.J.J.; Schutyser, F.A.C.; Bronkhorst, E.M.; Kuijpers–Jagtman, A.M.; Bergé, S.J. Evaluation of reproducibility and reliability of 3D soft tissue analysis using 3D stereophotogrammetry. Int. J. Oral Maxillofac. Surg. 2009, 38, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Marlière, D.A.A.; Demétrio, M.S.; Verner, F.S.; Asprino, L.; Chaves Netto, H.D.d.M. Feasibility of iterative closest point algorithm for accuracy between virtual surgical planning and orthognathic surgery outcomes. J. Cranio-Maxillofac. Surg. 2019, 47, 1031–1040. [Google Scholar] [CrossRef] [PubMed]
- Almukhtar, A.; Ayoub, A.; Khambay, B.; McDonald, J.; Ju, X. State-of-the-art three-dimensional analysis of soft tissue changes following Le Fort I maxillary advancement. Br. J. Oral Maxillofac. Surg. 2016, 54, 812–817. [Google Scholar] [CrossRef]
- Cheung, M.Y.; Almukhtar, A.; Keeling, A.; Hsung, T.-C.; Ju, X.; McDonald, J.; Ayoub, A.; Khambay, B.S. The Accuracy of Conformation of a Generic Surface Mesh for the Analysis of Facial Soft Tissue Changes. PLoS ONE 2016, 11, e0152381. [Google Scholar] [CrossRef]
| Systematic Search Strategy/PICOS Format | |
|---|---|
| (P) Population | Patients with class I, class II, class III, or asymmetric dentoskeletal deformities who underwent orthognathic surgery (Le Fort I, II osteotomy, bilateral sagittal split osteotomy, bimaxillary osteotomy, genioplasty) |
| (I) Intervention | Three-dimensional soft tissue simulation in VTP |
| (C) Comparison | Comparison of different methods or approaches for assessing the accuracy of 3D soft tissue simulation. Comparison of various software platforms or algorithms utilized for 3D soft tissue simulation |
| (O) Outcome | Accuracy of 3D soft tissue simulation in VTP |
| (S) Study design | Pro- and retrospective studies with a minimum sample size of 3 subjects |
| Inclusion Criteria | Exclusion Criteria |
|---|---|
|
|
| Software | References | |
|---|---|---|
| Dolphin 3D software | (Dolphin Imaging & Management Solutions, Chatsworth, CA, USA) | [14,15,16,17,18] |
| Maxilim | (Medicim NV, Mechelen, Belgium) | [11,19,20,21,22,23,24,25] |
| SimPlant ProOMS | (Materialise, Leuven, Belgium) | [26,27] |
| SurgiCase CMF Pro | (Materialise, Leuven, Belgium) | [28,29] |
| ProPlan CMF | (Dentsply-Sirona, York, PA, USA; Materialise, Leuven, Belgium) | [10,16,30,31] |
| OrtogOnBlender-OOB | (Blender Foundation) | [32] |
| IPS Case Designer | (KLS Martin Group, Tuttlingen, Germany) | [18,33,34] |
| SOTIRIOS software | (University of Basel, Switzerland) | [35] |
| Year, Author | Image Acquisition | Real Bony Movement | Additional Procedures | Type of Rigid Registration Method for Soft Tissue Evaluation (VOI for Superimposition) | Method of Analysis | Fixed Point of Accuracy | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Patient Position | Lip Position | CR/Wax Bite/CO | Postop Imaging Time Interval | a. Landmark-Based, LB; b. Surface-Based, SB; c. Voxel-Based, VB; d. Registration-Free Method, RF | Landmarks | Surface to Surface | ||||
| 2004, Chabanas [42] | H | NR | NR | NR | Yes | NR | * | Entire face; closest point; signed Euclidian distances | NR | |
| 2007, Mollemans [3] | H | NR | NR | 4 mos | Yes | NR | VB (top of skull) | Entire face—region of interest; corresponding points; signed Euclidean distances; mean, variance, 50%, 90%, and 95% percentiles of distance distributions; qualitative validation: surgeons’ visual inspection | NR | |
| 2007, Marchetti [46] | H | NR | NR | 3–6 mos | No | NR | VB * | Face surface—region of interest; mean distance, % of simulations with error < 2 mm | 2 mm | |
| 2010, Bianchi [28] | H | NR | NR | 6 mos a | Yes | NR | SB (soft tissue—forehead and eyes) | Entire face; closest point; average absolute error, SD and max, and 90th and 95th percentiles; % with error equal to or < 2 mm | 2 mm | |
| 2010, Ulusoy [43] | H | NR | NR | NR | No | NR | SB | Entire face; closest point; mean differences | NR | |
| 2011, Centenero [26] | H/V | NR | CR, wax bite | 3 mos | No | NR | RF | Landmarks; difference between linear and angular measurements within each face; ICC between measurements (“high”, “medium”, and “low” correlation) | NR | |
| 2011, [29] Marchetti | H | NR | NR | 6 mos | Yes | NR | SB (soft tissue—forehead and eyes) | Entire face; closest point; mean absolute error, SD, max, and 90th and 95th percentile; % of simulations with error equal to or <2 mm | 2 mm | |
| 2013, Schendel [38] | V | Relaxed | CR * | 6 mos | Yes | Reconstruction of nasolabial muscles * | SB * | Eighteen landmarks (10 midline, 8 lateral); signed mean values, absolute mean values, SD, RMS difference for all measurements | 0.5 mm | |
| 2013, Shafi [19] | V | Relaxed | CO, wax bite | 6–12 mos | Yes | ANS plasty; alar cinch suture; V–Y closure | SB (soft tissue—forehead) | Eight regions; mean absolute error, SD, 95% CI | 3 mm | |
| 2013, Nadjmi [11] | NR | NR | CR, wax bite | 4 mos | Yes | NR | Two-dimensional best fit and superimposition of SNL and OCSNL | Fifteen midline landmarks; differences across x-axis and y-axis: signed mean, SD, min, max, and frequency of clinically acceptable error (%) +/− 0.5 mm; nasolabial and mentolabial angles | 0.5 mm | |
| 2014, Terzic [44] | H/V | Relaxed | CO * | 6 mos | Yes | NR | SB * | Upper and lower half of face (pupil line), closest point, signed mean difference, SD, % of mass spring points with error < ±1 mm and exceeding ±1, ±2, and ±3 mm | 1 mm | |
| 2014, Nadjmi [24] | V | Relaxed | CR, wax bite | 4 mos | Yes | NR | VB (between infraorbital rim and rest of viscerocranium) | Entire face; closest point; mean absolute difference; mean signed distance, 25–75% distance range, 5–95% distance range | 2 mm | |
| 2015, Ullah [37] | V | Relaxed | CO; wax bite | 6–12 mos | Yes | ANS plasty; alar cinch suture; V–Y closure | SB (anterior cranial base, skull vault) | Eight regions; 90th percentile mean absolute error, SD, 95% CI | 3 mm | |
| 2015, Khambay [45] | NR | NR | NR | Min 6 mos | Yes | NR | SB (skull base) | Ten landmarks (six midline; four lateral); closest distance between two surface meshes at that point; arithmetic mean, absolute mean; SD | Entire face and 8 regions; closest point; max and absolute mean, 95th and 90th percentiles; SD; % of 3D points equal to or <2 mm; RMS error | 2 mm |
| 2015, Nam [27] | H | NR | NR | 6 mos | No | Alar cinch suture | * | Ten landmarks (six midline, four lateral); corresponding; means, SD; absolute values and vector values using x, y, and z coordinates; accuracy rate: <2 mm | 2 mm | |
| 2015a, Liebregts [23] | V—seated | Relaxed | NR | Min 6 mos a | Yes | Alar cinch suture; V-Y closure | VB (cranial base, forehead, zygomatic arches) | Six midline landmarks; corresponding; Euclidean distances; mean absolute error; SD; max and min absolute error; 95% CI | Entire face and 3 regions; closest point, mean absolute error, SD, range and 95th percentile; % of simulations with error equal to or <1 mm and 2 mm | 2 mm |
| 2015b, Liebregts [22] | V—seated | Relaxed | CR, wax bite | Min 6 mos a | Yes | NR | VB (cranial base, forehead, zygomatic arches) | Six midline landmarks; corresponding; Euclidean distances; mean absolute difference, SD | Entire face and 3 regions; closest point, mean absolute error, SD, 90th percentile, % of simulations with error equal to or <1 mm and 2 mm | 2 mm |
| 2015, Van Hemelen [25] | V—seated | NR | CR, wax bite | 4 mos | No | NR | VB (between infraorbital rim and rest of viscerocranium) | Nine landmarks in midsagittal plane; corresponding; difference in depth (Y), in height (Z), and 2D distance in sagittal plane (NR) | 2 mm | |
| 2016, Liebregts [21] | V—seated | Relaxed | NR | Min 6 mosa | Yes | ANS plasty, nasal base plasty, alar cinch suture; V-Y closure | VB (cranial base, forehead, zygomatic arches) | Three landmarks; corresponding; Euclidean distances; mean absolute error, SD, range | NR | |
| 2016, Resnick [15] | V | Relaxed | CO | Min 6 mos a | Yes | Alar cinch suture | LB * | Fourteen landmarks * (six midline, eight lateral) and nasolabial angle; mean error, % of average absolute error <2 mm | 2 mm | |
| 2017, Kim [9] | H | NR | NR | MSCT 6 weeks/3D photo min 6 mos | Yes | NR | SB (forehead and nasal bridge) | Entire face and 8 subregions; mean errors, SD, and max errors (absolute mean Euclidean distances along normal vectors); clinicians’ qualitative evaluation: binary visual scoring scale (Unacceptable; Acceptable) | Mean: 1.5 mm; max: 3 mm | |
| 2021, Kim [41] | H | NR | NR | MSCT 6 weeks/3D photo min 6 mos | Yes | NR | SB (forehead and nasal bridge) | Entire face and 6 regions; mean error (absolute Euclidean distances along surface normal vectors); qualitative evaluation: lip shape analysis | NR | |
| 2017, Mundluru [20] | V | NR | NR | 6–12 mos | Yes | Alar cinch suture; V–Y closure; condylectomy | VB (skull base) | Eight anatomical regions; min, max, mean, SD, absolute max, absolute mean, and absolute SD of 90% of points; directional discrepancies at each vertex in x, y, and z dimensions separately—conformed generic meshes—corresponding points | 2 mm | |
| 2018, Holzinger [35] | H | Relaxed | CR | 6 mos | Yes | NR | LB | Entire face; corresponding points; mean error, SD, median, 80th, 95th, 99th, and 99.9th percentiles | 2 mm | |
| 2019, Knoops [16] | V | Relaxed | CO | 12 mos a | Yes | Alar cinch suture | SB (skull base) | Midface: upper lip and paranasal regions; closest point; RMS distance; % of points <2 mm | 2 mm | |
| 2019, Elshebiny [14] | V | NR | CO | 6–12 mos | Yes | NR | VB (cranial base) | Landmarks; difference between 12 linear and 3 angular measurements within each face; means, SD | 1.5 mm | |
| 2021, Cunha [32] | H | Relaxed | CR, wax bite | Min 6 mos | No | Alar cinch sutures, V-Y closure | SB *(skull base, nasal bones, frontal bone, zygomatic arches) | Seventeen landmarks (five midline, twelve lateral); corresponding; Euclidean distance; mean deviation, 95% CI, max, min, SD | 2 mm | |
| 2021, Willinger [18] | NR | NR | NR | Min 4 mos | Yes | Camouflage of infraorbital step with milled bone and fibrin glue | SB* (maxilla) | Six landmarks; three lateral landmarks at both sides along MFAL—Midfacial Advancement Line technique; mean error, SD, median, 95% CI, variants, min, max, range, interquartile range, skewness, and kurtosis | 2 mm | |
| 2021, Tanikawa [36] | V | NR | NR | NR | NR | NR | Common coordinate system based on landmarks | Entire face; corresponding points; differences in z-axis; average error, SD, min and max; % of cases with average error <1 mm or <2 mm | 1 mm, 2 mm | |
| 2021, ter Horst [33] | V | Relaxed | CO | CBCT: 12 mos; 3D photo 5–19 mos | Yes | NR | SB * (soft tissue) | Three anatomical regions; closest point; mean absolute error, RMS error, SD, 95th percentile; % of simulations with a max error of 1 mm and 2 mm | 1 mm, 2 mm | |
| 2021, Alcañiz [39] | NR | NR | NR | NR | Yes | NR | NR | Entire face; signed closest-point distance; cumulative surface % with error < 3 mm; comparison of fine and coarse meshes | 3 mm | |
| 2022, Lee [10] | NR | NR | NR | Min 6 mos a | Yes | No | SB * (forehead and nose bridge) | Entire face (with upper third removed), 6 anatomical regions; closest point; mean absolute difference, SD; % of 3D points with error equal to or <2 mm; absolute mean, RMS for 95th percentile | 2 mm | |
| 2022, Gutiérrez [40] | NR | NR | NR | NR | Yes | NR | NR | Nine landmarks (seven midline, two lateral); closest point; 25th, median, 75th; surgeons’ qualitative evaluation: Likert scale and binary questions; comparison of fine and coarse meshes | 2 mm, 3 mm | |
| 2022, Yamashita [17] | V—seated | Relaxed | CR, wax bite * | 6–12 mos | No | Alar cinch suture, suspension of mentalis muscle | VB (cranial base) | Landmarks (midsagittal plane); corresponding, signed distances in coordinate y (anteroposterior direction) and z (superoinferior direction) | 2 mm | |
| 2022, Ma [8] | H | NR | NR | NR | No | NR | NR | Landmarks; corresponding *, Euclidean distance; mean error, SD, max error | Entire face and 5 subregions; Chamfer distance and Hausdorff distance; mean error, SD, max error; clinicians’ qualitative evaluation | NR |
| 2022, Awad [34] | V—seated | Relaxed | CR * | 4 mos | No | No | LB | Entire face and 6 regions; absolute discrepancies, unsigned mean absolute discrepancies (RMS), SD, % of surface with error < 2 mm = % of surface congruence (IO%) | 2 mm | |
| 2022, Hou [31] | NR | Relaxed | * | Min 6 mos a | Yes | NR | SB * (soft tissue forehead, nose bridge) | Landmarks (8 midline, 6 lateral); corresponding; differences on x, y, and z planes | Entire face and 10 regions; RMS distance, SD, 95% CI | 2 mm |
| 2023, Şenyürek [30] | NR | NR | NR | 11–15 mos | Yes | NR | LB | 6 anatomical regions; mean error, SD | 2 mm | |
| 2023, Ruggiero [47] | CBCT: V standing; MRI: H | NR | CO, wax cast | 6 mos | Yes | NR | SB | Midface; the closest point; mean error, SD | NR | |
| 2024, Fang [48] | H | NR | NR | NR | Yes | NR | * | Twenty landmarks (eight upper face, twelve lower face); Euclidean distance, mean error, SD | Entire face and 6 regions; mean error (average surface deviation error, SD); qualitative evaluation: lip-shape analysis | NR |
| Workflow | Guidelines |
|---|---|
| 1. Image acquisition (pre- and postoperative) | Report the details of the CBCT apparatus and the pre- and postoperative image acquisition protocol; CBCT in a vertical position (seated or standing) without deformation of the facial soft tissue mask, with the mandible in “centric relation”; extended FOV; the same CBCT device pre- and postoperatively; postoperative CBCT at least at 6 months after surgery to evaluate soft tissue changes and skeletal relapse; fixed orthodontic appliances should be in place |
| 2. Image data processing | |
| Three-dimensional rendering of DICOM, STL, and OBJ files | Perform and evaluate 3D soft tissue simulation of the CBCT facial soft tissues; avoid the superimposition of the 3D photograph for analysis |
| Virtual osteotomies and VTP | Determine the exact skeletal changes that occurred after the surgery and any potential relapse (postoperative long-term CBCT); use the postoperative hard tissues as a template for analysis |
| Additional surgical procedures | Report additional surgical procedures, such as septoplasty, rhinoplasty, bony reshaping (ANS, nasal base, lateral nasal walls, chin, gonial angles, zygomas), bone augmentation (grafts, PSIs), soft tissue closure method after Le Fort I (V-Y closure, alar base cinch suture, paranasal cross sutures), lipofilling, and/or liposuction |
| 3. Three-dimensional soft tissue simulation | Report the 3D soft tissue algorithm used for simulation |
| 4. Rigid registration of preoperative/simulated data and postoperative data | Use voxel-based superimposition protocol using only one software, fully automated (user-independent) on a stable VOI (e.g., anterior cranial base, total cranial base, both zygomatic arches); report the software that was used |
| 5. Postprocessing and analysis | |
| Quantitative analysis | Report absolute mean values or Euclidean distances and root mean square error |
| Three-dimensional landmark-based analysis | Use reliable and accurately definable corresponding points; automatic 3D landmark identification is recommended |
| Surface mesh-based analysis | Recommended true correspondences: generic/conformable mesh; only analyze facial surface that is affected by surgery; division of face into anatomical regions and separate analysis |
| Qualitative analysis | Objective evaluation method in addition to quantitative analysis is recommended |
| Algorithm | Characteristics | Software |
|---|---|---|
| Sparse landmark-based algorithm |
| Dolphin® |
| Mass spring model (MSM) | OrtogOnBlender® 3dMDVultus® | |
| Mass tensor model (MTM) | Maxilim® IPS Case Designer® | |
| Finite element model (FEM) | ||
| Artificial intelligence (AI) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olejnik, A.; Verstraete, L.; Croonenborghs, T.-M.; Politis, C.; Swennen, G.R.J. The Accuracy of Three-Dimensional Soft Tissue Simulation in Orthognathic Surgery—A Systematic Review. J. Imaging 2024, 10, 119. https://doi.org/10.3390/jimaging10050119
Olejnik A, Verstraete L, Croonenborghs T-M, Politis C, Swennen GRJ. The Accuracy of Three-Dimensional Soft Tissue Simulation in Orthognathic Surgery—A Systematic Review. Journal of Imaging. 2024; 10(5):119. https://doi.org/10.3390/jimaging10050119
Chicago/Turabian StyleOlejnik, Anna, Laurence Verstraete, Tomas-Marijn Croonenborghs, Constantinus Politis, and Gwen R. J. Swennen. 2024. "The Accuracy of Three-Dimensional Soft Tissue Simulation in Orthognathic Surgery—A Systematic Review" Journal of Imaging 10, no. 5: 119. https://doi.org/10.3390/jimaging10050119
APA StyleOlejnik, A., Verstraete, L., Croonenborghs, T.-M., Politis, C., & Swennen, G. R. J. (2024). The Accuracy of Three-Dimensional Soft Tissue Simulation in Orthognathic Surgery—A Systematic Review. Journal of Imaging, 10(5), 119. https://doi.org/10.3390/jimaging10050119










