Abstract
The organs of the male reproductive tract, including the testis, epididymis, prostate, seminal vesicles, and semen, must provide an immunoregulatory environment conducive to germ cell viability and successful fertilization. Many immune components, such as immune cells, have been investigated regarding reproductive immunology and function; however, the investigation of the role of complement in this system has only more recently been gaining traction in research. This review focuses on complement in the male reproductive tract, with the goal of compiling information currently known about complement components detected in male reproductive organs and identifying areas in need of further research. Considering the recent and upcoming research about the noncanonical functions of complement, this information is relevant and applicable in the fields of reproductive immunology, fertility, and immune regulation.
1. Introduction
The male reproductive tract must maintain a level of immune regulation for the maturation and transport of viable sperm. Male germ cells are stalled in the mitotic stages of gametogenesis at the time of birth. Upon adolescence, these germ cells then proceed through spermatogenesis. Spermatogenesis occurs after the development of central immune tolerance, during the onset of puberty, rendering the maturing gametes immunogenic and susceptible to autoimmune destruction. In the cases of orchitis, epididymitis, and/or epididymo-orchitis, the immune system can create anti-sperm antibodies (ASAs) that target meiotic germ cells for destruction and can even cause immune infertility [1]. Indeed, ASAs were detected in over 18% of infertile men, while no ASAs were detected in fertile men [1].
Most males are fertile, demonstrating robust immune regulation within the reproductive tract. Regulation of the immune response occurs through a variety of mechanisms in the various organs of the male reproductive tract. These mechanisms include (1) the physical separation of gamete-containing tracts from systemic access via the blood–testis barrier and the blood–epididymal barrier, (2) cellular modulation by testicular macrophages and regulatory T cells, and (3) the expression of immunomodulatory proteins such as transforming growth factor beta (TGF-β) and complement components (extensively reviewed in [2]). The focus of this review is to identify the current state of understanding regarding the presence and expression of complement components in male reproductive organs, their potential roles in immunology and reproduction, and the knowledge gaps in this context. To date, the last review on complement in the entirety of the male reproductive tract was conducted by Harris et al. in 2006 [3], so this paper will serve as a much-needed review for the field. As an evolving area of study, complement in reproduction is a relevant topic that will contribute to the better understanding of fertility, development, and immune regulation.
2. A Brief Overview of Complement
Immunologically, complement is an innate system of proteins involved in pathogen recognition, inflammation, immune cell recruitment and activation, and cell lysis. Complement consists of over 50 proteins that participate in the immunological cascade, the regulation of the cascade, and cell modulation (Figure 1).
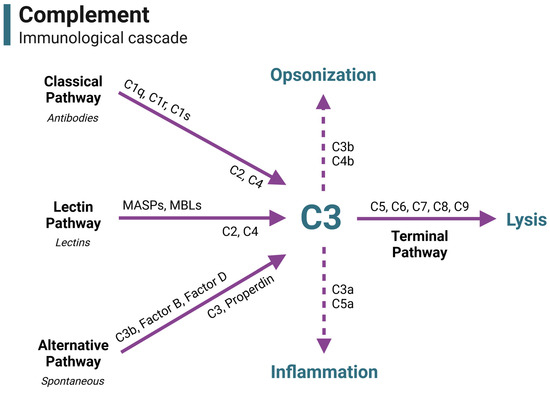
Figure 1.
The complement immunological cascade. In immunity, complement is activated through three primary pathways: the classical pathway (by antibody–antigen complexes), the lectin pathway (by bacterial lectins), and the alternative pathway (spontaneous activation). Each pathway is characterized by different pattern recognition receptors and the initial development of a C3 convertase. All activation pathways converge on the C3 component, which is a central point of complement activation. C3 is cleaved to feedback and amplify the cascade, opsonize pathogens for phagocytosis, initiate inflammation, and begin the terminal pathway. Activation of the terminal pathway leads to the creation and insertion of the membrane attack complex (MAC), leading to cell lysis. This figure is a guide to visually portray where complement components functionally reside in the immune cascade throughout the duration of this review. Acronyms and abbreviations are defined in Table 1. Figure was made with BioRender.
Antibody–antigen complexes (classical pathway), bacterial lectins (lectin pathway), and spontaneous action (alternative pathway) can activate the immunological complement cascade. Each pathway forms an enzyme (C3 convertase) that cleaves the complement component C3 into C3a and C3b—the central point of complement. C3a plays a role in inflammation, while C3b can participate in opsonization for phagocytosis or modify the C3 convertase enzyme into C5 convertase, beginning the terminal pathway. In the terminal pathway, C5 is cleaved into the anaphylatoxin C5a and the membrane attack complex (MAC) component C5b. C5b binds C6, C7, C8, and multiple C9 proteins to form the fully assembled MAC intermembrane pore. The formation and insertion of multiple MAC pores allows for a massive influx of fluid into the cell, leading to cell lysis.
The complement immunological cascade is a positive feedback look that is amplified by the action of the alternative pathway proteins Factor D, C3, and Properdin. Due to response amplification, complement must be tightly regulated to prevent excess collateral damage and chronic inflammation. Complement regulation is accomplished by a variety of inhibitory proteins and receptors (Figure 2). Complement inhibitors for nearly every step in the cascade have been identified. Thus, complement can be shut down at various and specific stages, allowing inhibitor-expressing cells to tailor local complement action. A cell’s specific complement signature may in fact be used to regulate immune responses by controlling which complement products are present in the environment, expressed by the host cells, or bound to pathogens.
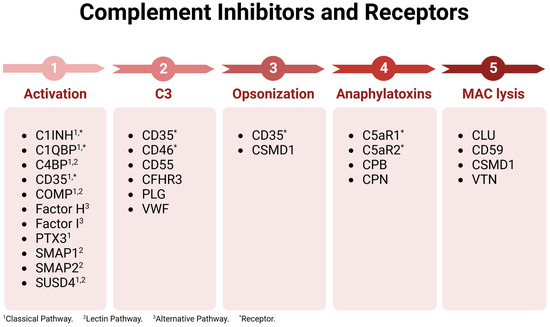
Figure 2.
Complement inhibitors and receptors. Complement regulatory proteins are listed based on the cascade point where they function. Acronyms and abbreviations are defined in Table 1. Figure was made with BioRender.
Complement’s modulation of immune responses goes further than just opsonizing pathogens for engulfment by phagocytes. The engagement of C3b and C4b with the complement receptors CD21, CD35, CR3, and CR4 on immune cells acts as an activation signal in the presence of specific cytokines such as interleukin-2 (IL-2) [4]. Moreover, C3b’s interaction with the complement inhibitors CD35, CD46, and CD55 have been implicated in T cell differentiation to either proinflammatory T helper 1 (Th1) cells or suppressive T regulatory cells (Treg), again influenced by the cytokine milieu in the paracrine space [5,6,7]. In effect, CD46-C3b has been shown to be an additional signal in Th1 activation when IL-2 is low and to generate highly suppressive Tregs when IL-2 is high [8]. The C3b in these encounters is not serum-derived but produced and secreted in an autocrine fashion, indicating an important role for C3 production by the cells in the environment [9].
In addition to C3b and C4b, the anaphylatoxins C3a and C5a have powerful immunomodulatory actions [10]. The most obvious mechanism is the chemotaxic and inflammatory properties of these molecules. Anaphylatoxins bind to their receptors on endothelial cells to encourage vasodilation, reduce cellular adhesion molecule expression, and to release proinflammatory mediators like IL-1 [10,11]. On immune cells, anaphylatoxins recruit tissue and circulating immune cells to the site of complement activation [11] and activate these cells for maximum pathogen clearance capacity [9]. Interestingly, activation of Th1 cells is stunted when C3a and C5a are absent, indicating that the anaphylatoxins are important in Th1 activation [12]. Furthermore, in a high-anaphylatoxin environment, the suppressive capacity of Tregs is decreased, while in a low-anaphylatoxin environment, this capacity increases [4,13]. These are just a few examples demonstrating the regulatory potential of complement over immune responses.
Outside of its immunologic functions, complement plays important roles in other body systems, particularly in neurological development and cellular metabolism [14,15,16,17]. Regarding the subject of this review, complement has been implicated in many important functions in reproduction, which will be discussed here. A summary of the complement components, their location in the male reproductive tract, and their functions is contained in Table 1.

Table 1.
Expression of complement components and their potential functions in the male reproductive tract.
Table 1.
Expression of complement components and their potential functions in the male reproductive tract.
| Complement Component | Organ Expression (Male Reproductive) | Potential Functions in Male Reproductive Tract |
|---|---|---|
| C1 inhibitor (C1INH) 1,2 | Testis | Inhibition of female genital tract complement |
| C1q 1 | Testis | Classical pathway |
| C1q-binding protein (C1QBP) 1,2 | Testis | Inhibition of female genital tract complement |
| C1r 2 | Testis | Classical pathway |
| C1s 1,2 | Testis | Classical pathway |
| C2 1,2 | Testis | Classical and lectin pathways |
| C3 1,2,3 | Testis, semen | Alternative pathway and complement cascade, modulation of female reproductive immunity, sperm–oocyte interaction |
| C4 1,2,3 | Testis, semen | Classical and lectin pathways |
| C4-binding protein (C4BP) 1,4 | Testis, epididymis, semen | Inhibition of female genital tract complement, modulation of female reproductive immunity |
| C5 1 | Testis | Terminal pathway, immune cell modulation |
| C5a receptor 1 (C5aR1) 1 | Testis | Anaphylatoxin receptor, immune cell activation, inflammation |
| C5a receptor 2 (C5aR2) 1 | Testis | Anaphylatoxin receptor, immune cell modulation |
| C6 1 | Testis | Terminal pathway |
| C7 1,5 | Testis, prostate | Terminal pathway |
| C8a 1 | Testis | Terminal pathway |
| C8b 1,2 | Testis | Terminal pathway |
| C8c 1,2 | Testis | Terminal pathway |
| C9 1,3 | Testis, semen | Terminal pathway |
| CD35 1 | Testis | Inhibition of female genital tract complement, immune modulation |
| CD46 1,6,7,8 | Testis, semen | Inhibition of female genital tract complement, immune modulation, sperm–oocyte fusion |
| CD55 1,2,7,8 | Testis, prostate, semen | Inhibition of female genital tract complement, immune modulation |
| CD59 1,2,3,7,8 | Testis, semen | Inhibition of female genital tract complement |
| Complement Factor-H-related protein 3 (CFHR3)8 | Testis | Inhibition of female genital tract complement |
| Clusterin (CLU) 1,9 | Testis, epididymis, seminal vesicle | Inhibition of female genital tract complement, sperm capacitation, sperm viability, sperm maturation and development |
| Cartilage oligomeric matrix protein (COMP) 1 | Testis | Inhibition of female genital tract complement |
| Carboxypeptidase B (CPB) 1 | Testis | Inhibition of anaphylatoxins in female genital tract |
| Carboxypeptidase N (CPN) 1 | Testis | Inhibition of anaphylatoxins in female genital tract |
| Cub and Sushi Multiple Domains 1 (CSMD1) 1 | Testis | Inhibition of female genital tract complement |
| Factor D 1 | Testis | Alternative pathway |
| Factor H 1,10 | Testis, epididymis, seminal vesicles, semen | Inhibition of female genital tract complement |
| Factor I 1 | Testis | Inhibition of female genital tract complement |
| Ficolin 1/2 (FCN1/2) 1 | Testis | Lectin pathway |
| MBL-associated serine protease 1/2 (MASP1/2) 1,2 | Testis | Lectin pathway |
| Mannose-binding lectin 1/2 (MBL1/2) 1 | Testis | Lectin pathway |
| Plasminogen (PLG) 1 | Testis | Inhibition of female genital tract complement |
| Properdin 1 | Testis | Alternative pathway |
| Pentraxin (PTX3) 1,11 | Testis, prostate, semen | Inhibition of female genital tract complement |
| Soluble MBL-associated protein 1/2 (SMAP1/2) 1 | Testis | Inhibition of female genital tract complement |
| Sushi domain-containing protein 4 (SUSD4) 1 | Testis | Inhibition of female genital tract complement, may play a role in immune tolerance |
| Vitronectin (VTN) 1,12 | Testis, semen | Inhibition of female genital tract complement, may influence acrosome reaction but needs investigation |
| Von Willebrand Factor (VWF) 1 | Testis | Inhibition of female genital tract complement |
Chart references: 1 [18]. 2 [19]. 3 [20,21,22,23]. 4 [24]. 5 [25]. 6 [26,27]. 7 [28,29]. 8 [28]. 9 [23,30,31,32,33]. 10 [34]. 11 [35]. 12 [36].
3. Complement in the Testis
The main reproductive organ in males is the testis, the site of testosterone production and spermatogenesis. The core of the testis are seminiferous tubules, where spermatogenesis occurs. Cells important in these testicular functions include Sertoli cells, Leydig cells, peritubular myoid cells, and testicular macrophages. Of these, complement expression has been confirmed in Sertoli cells, Leydig cells, macrophages, and germ cells [3,18,19,23,37,38,39] (Figure 3, Table 1). Germ cells will be discussed later with seminal plasma.
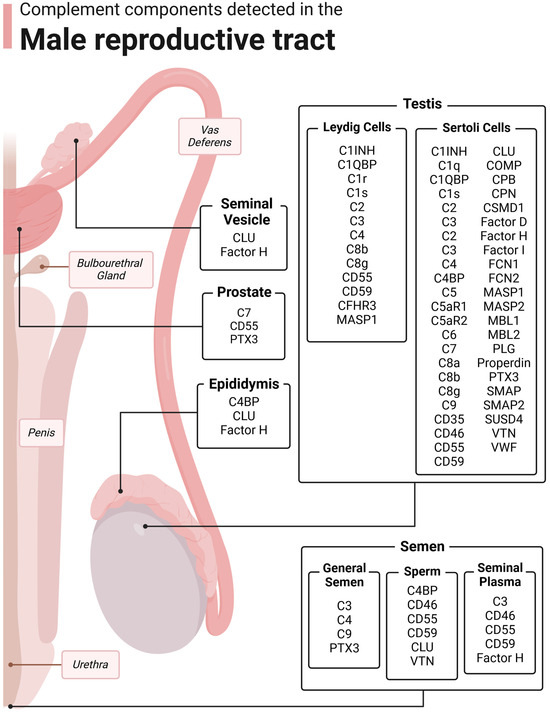
Figure 3.
Complement components detected in the male reproductive tract. The male reproductive tract consists of the testis, epididymis, vas deferens, prostate, seminal vesicle, bulbourethral glands, urethra, penis, and semen. Complement has been detected in many of these structures. Figure was made with BioRender.
3.1. Sertoli Cells
Sertoli cells are primarily responsible for establishing an immunoregulatory environment conducive for spermatogenesis. Along with peritubular myoid cells, Sertoli cells form the blood–testis barrier, which is a physical barrier that separates the lumen of the seminiferous tubules from the testis interstitium, where immune cells can operate [40]. This barrier is the first step in sequestering the maturing germ cells from the immune system. Sertoli cells also express and secrete various substances that have immune-modulatory properties including transforming growth factor beta (TGF-β) and indoleamine-2,3-dioxygenase ([41,42], reviewed in [2]). Significant research has been conducted into the importance of these individual factors in autoimmunity, transplantation, and immunity. Work from multiple investigators, including the Griswold [43], Korbutt [44], Chen [45], and Dufour [18,46,47,48] labs, has investigated the presence of complement in Sertoli cells.
The Dufour lab has identified the most detailed milieu of complement components expressed by Sertoli cells as of the writing of this review: 25 cascade factors, 21 inhibitors, and 2 receptors, for a total of 48 complement-related proteins (Figure 3, Table 1) [18]. These proteins have been described in detail by Washburn et al. [18] and extensively reviewed [37]. In brief, expression of nearly all complement cascade factors was detected in mouse, pig, and human Sertoli cells [18,47]. The identified regulators are known to inhibit all throughout the complement immunological cascade (Figure 2). Of interest, Sertoli cells express all but two of the complement proteins that have been detected in the different parts of the male genital tract (Table 1) [18,43,44,45,47].
The expression of such a wide repertoire of complement inhibitors by Sertoli cells is reasonable considering that a main function of Sertoli cells is to create and maintain an environment for spermatogenesis to thrive—one free from adverse immunological events. During acute infections of Zika virus, germ cells were shown to undergo complement-mediated damage, demonstrating that the complement system can be activated when the blood–testis barrier is disrupted [49]. However, the complement inhibitors expressed by Sertoli cells block all the major points within the immunological cascade, including activation, opsonization, C3 convergence, anaphylatoxin inflammatory activity, and MAC-mediated cytolysis. The presence of these complement regulatory factors may mitigate the damage in these cases. In this manner, Sertoli cells can prevent sustained deleterious complement cascade activation within the seminiferous tubules.
Sertoli cells also express two anaphylatoxin receptors (C5aR1 and C5aR2), both for the highly potent C5a anaphylatoxin. C5aR1 is associated with immune activation, while C5aR2 is associated with immune suppression under certain conditions [50]. An important function of C5a is the induction of inflammation and activation of pro-inflammatory immune cell types [11]. Along these lines, C5a has been shown to be a critical component in the differentiation and activation of Th1 T cells, which amount strong immune responses [6,51]. Moreover, C5aR1 signaling has been shown to inhibit the immune-suppressive functions of regulatory T cells, as blocking C5aR1 signaling increased numbers of regulatory T cell numbers and enhanced their suppressive capacity [13,52]. By expressing both C5a receptors, Sertoli cells may sequester any aberrant C5a molecules from activating inflammatory mechanisms in the seminiferous tubules and in the testicular interstitium. Though C5a is known to activate signaling cascades within immune and endothelial cells, its signaling potential has not yet been investigated in the realm of Sertoli cells.
A further study conducted by the Dufour lab analyzed Sertoli cell RNA expression after exposure to normal human serum (NHS), a model of robust complement-attack [53]. After incubation with NHS for only 90 min, 62 genes were differentially expressed [53]. Pathway analyses determined that many of these genes participate in immune suppression, angiogenesis, tolerance, cell proliferation, and apoptotic avoidance [53]. It appears that Sertoli cells respond to the presence of robust complement activation by expressing genes important in immune regulation, inhibition of inflammation, and cell survival, though functional studies need to be conducted to confirm these roles.
Nevertheless, these observations are supported through studies investigating the immunomodulatory functions of complement regulators. As previously discussed, immunomodulatory functions of CD46 have been demonstrated in T cell metabolism, differentiation, and survival [8,54,55]. Many of these functions have been shown to occur through metabolic reprogramming of both CD4 and CD8 T cells [14,54]. CD35 [5] and CD55 [7,56] have also been shown to have immunomodulatory effects on lymphocytes, and these mechanisms are currently under investigation. Indeed, complement inhibitor production by Sertoli cells may take advantage of these modulatory mechanisms to ensure an immune-protective environment in the seminiferous tubules, thus giving the male germ cells their best chance to develop and mature.
3.2. Leydig Cells
Leydig cells, another major cell type in the testis, are primarily responsible for testosterone production. Among systemic effects, testosterone hormonally regulates many processes in the male reproductive tract [40,57]. The gene expression of some complement components is also regulated by testosterone. Androgen synthesis is positively correlated with the expression of CD46, CD55, and CD59 by Leydig cells; however, a weak negative correlation was observed in autoimmune orchitis [19]. Along these lines, androgen synthesis is negatively correlated to the expression of the complement cascade factors C1r, C1s, C2, C3, C4, C8, and MASP1 [19].
It is reasonable to conclude that since CD46, CD55, and CD59 are major complement inhibitors, their expression indicates a regulated complement environment. Other complement components detected in this study include the inhibitors C1INH, C1QBP, and CFHR3 [19], which were not analyzed in depth. Of the Leydig cell complement repertoire, C1r and CFHR3 have not yet been detected in Sertoli cells, which may augment some regulatory functions within the seminiferous tubules, but a study of the roles of complement expressed by Leydig cells has not been conducted (Figure 3, Table 1).
3.3. Macrophages
Testicular macrophages, an important cell type in the testis, function like anti-inflammatory M2 macrophages and are likely to control inflammation around the blood–testis barrier. Testicular macrophages suppress inflammatory and cytotoxic immune responses in the delicate spermatogenic environment, clear cell debris, phagocytose potential pathogens, and perform testis-specific roles in homeostasis and development [58].
Though not specifically studied in testicular macrophages, the expression and influence of complement components have been established by macrophages in general (Table 1), so this review will draw complement-related data from studies over macrophages collected from bone marrow or the peritoneal cavity [39,59,60,61]. Since they are immune cells, their expression of complement proteins makes sense and coheres with this function. Macrophages are an intratissue source of the complement components C1q, C1r, C1s, C2-C9, Factor B, Factor D, and Properdin, which are important in eradicating infection [39]. They also express the complement regulators C1INH, C1QBP, CD35, CD46, CD55, CD59, CLU, Factor H, Factor I, PTX3, SMAP1, and SMAP2 [39], which inhibit complement at key points to prevent amplification and complement-mediated inflammation. Furthermore, macrophages express many complement receptors (CD35, CR3, CR4, CRIg, C3aR, C5aR1, and C5aR2), which initiate specific events in the macrophage, including activation of the macrophage, the enhancement of phagocytosis, macrophage differentiation (M1 or M2), chemotaxis and extravasation, and the suppression of macrophage function ([59,60,61], reviewed in [37]).
Overall, testicular macrophages may use complement to ensure effective pathogen removal while preserving the integrity of the blood–testis barrier and thus allow for the successful continuation of spermatogenesis, even during infection. Interestingly, this complement expression pattern is like that identified in Sertoli cells [18], supporting the notion that Sertoli cells may directly regulate complement and further immune response.
3.4. Germ Cells
Germ cells are located in the seminiferous tubules of the testis in association with Sertoli cells. The presence of various complement factors has been detected on male germ cells, and these will be discussed in-depth in Section 7.3 of this paper.
4. Complement in the Epididymis
The next organ in the male reproductive tract is the epididymis—the site of spermatozoa maturation accumulation [62]. The duct-like structure of the epididymis consists of four anatomically distinct segments starting at the end of the seminiferous tubules and proceeding to the beginning of the vas deferens: (1) the initial segment, (2) the caput, (3) the corpus, and (4) the cauda [62]. Spermatids disengage from their Sertoli cell nursery to become spermatozoa, which enter the initial segment of the epididymis and then the caput. Spermatozoa in the caput are non-motile cells, and as they progress through the various environments in the epididymis, they mature into motile, more functional spermatozoa [63]. Spermatozoa gain fertile functions in the female reproductive tract during the acrosome reaction [64] involving CD46, which will be discussed later. The presence of three complement components in the epididymis has been detected: C4BP, CLU, and Factor H (Figure 3, Table 1).
The lumen of the epididymis is lined with epithelial cells, and it is the epithelial cells within the distal caput, corpus, and cauda that synthesize C4-binding protein (C4BP) [24]. The epididymal C4BP transcription level, while constant in the liver, is regulated by androgens [24]. Since the lumen of the epididymis is also protected by a barrier, the blood–epididymal barrier, C4BP may serve complement-regulatory functions in preventing abhorrent complement activation in the environment. Spermatozoa may acquire C4BP secreted by epidydimal cells to prevent complement-mediated damage while in the female reproductive tract. Nonaka et al. also postulate that, due to the very low levels of C3 and C4 mRNA expressed by epididymal cells, C4BP secretion by epididymal cells and the subsequent uptake by spermatozoa may be more consequential in maturation, but this has yet to be investigated [24].
The epididymis also secretes CLU, termed secretory CLU (sCLU), an important inhibitor of the complement cytolytic cascade’s terminal pathway that has other functions in the reproductive tract outside of immunity. sCLU localization was detected by immunofluorescence and Western blot analysis [31,32]. CLU makes up a significant portion of the protein component of seminal plasma and will be discussed in Section 7.1.
Lastly, large amounts of Factor H transcripts were detected by PCR, but immunofluorescence detected only very low levels of Factor H protein in the epididymis limited to the caput section [34]. Thus, it is believed that protective Factor H comes from different sources in the male reproductive tract, such as the seminal vesicles. In support of this argument, spermatozoa extracted from the caput were significantly less resistant to MAC deposition (29%) than ejaculated sperm (77%) [34].
5. Complement in the Prostate
The epididymis is connected to the prostate via the vas deferens. As a tubuloalveolar gland, the prostate is responsible for producing most of the components of seminal plasma, including essential proteins and ions important in spermatozoa maintenance [65]. Some of these proteins include the complement factors C7, CD55, and PTX3 (Figure 3, Table 1).
C7 has been detected in prostate stromal cells, where its expression is regulated by androgens [25]. Long pentraxin 3 (PTX3) has been detected by immunohistochemistry in epithelial glandular cells of the prostate [35]. In prostate cancer, abnormal levels of C3, C7, and Factor B are detected, with increased levels of C7 being associated with positive clinical outcomes [25,29]. CD55 expression is significantly elevated in prostate cancer as compared to the healthy prostate [29]. Although these complement factors have been identified in the prostate, their investigation is mainly limited to malignant models [25,66]. Further research should study the role of CD55, PTX3, and C7 in normal prostate secretion and function.
6. Complement in the Seminal Vesicles
Another set of glands in the male reproductive system are the seminal vesicles, which are located behind the prostate. Seminal vesicles are blind-ended tubes tightly coiled on either side of the vas deferens that secrete most of the fluid that makes up seminal plasma [67]. Of note is the fact that fluid from the seminal vesicles is alkaline and contains fructose, vitamins, proteins, and prostaglandins [68]. Fructose is an essential source of energy for sperm motility, the alkalinity of seminal vesicle fluid neutralizes the normally acidic vaginal environment, and the proteins include immunomodulatory factors [68]. Together, these secretory products, along with those secreted by the prostate and bulbourethral glands, are critical in the maintenance and functionality of ejaculated sperm.
In addition to other immunomodulatory factors such as prostaglandins, Factor H and CLU have been detected in the fluid and epithelial cells of the seminal vesicles (Figure 3, Table 1) [31,32,34]. CLU feeds into the seminal plasma of semen and will be discussed next. Regarding Factor H, immunoblot analyses indicate that this expression is significantly higher in the seminal vesicles than in other organs of the male reproductive tract [34]. The function of Factor H in this organ has yet to be investigated; however, complement inhibition in the female reproductive tract is an attractive hypothesis.
7. Complement in Semen
The male reproductive tract is physiologically structured to deliver sperm through the female reproductive tract for the fertilization of oocytes. To accomplish this, the male germ cells must (1) progress through spermatogenesis, (2) mature into motile sperm, (3) reside in a nutrient-rich and protective fluid (seminal plasma), and (4) be able to survive ejaculation and transit through the hostile environment of the female reproductive tract [69]. The organs discussed previously cultivate spermatogenesis and spermatozoa maturation and secrete the necessary substances, including several complement components (Figure 3, Table 1), to create the ejaculation product of semen. Semen is primarily composed of seminal plasma and motile spermatozoa.
7.1. Whole Semen
Garcia et al. found that the sperm from infertile men had significantly decreased total count, count per mL, motility, viability, and normal morphology from that of fertile men [1]. Semen from infertile men had a significantly increased number of white blood cells than fertile men, which had no detectible white blood cell counts [1]. As previously discussed, many complement factors either increase or inhibit immune cell infiltration (reviewed in [2]). Moreover, some complement factors have been shown to affect the fertilization ability of sperm, which will be discussed shortly.
CD59 is expressed throughout the spermatozoa surface and in seminal plasma, both bound to prostasomes and as free proteins. Studies by Qin et al. indicate that CD59 may play an important part in male murine fertility, since the fertilization capacity of CD59-knockout mice dropped rapidly as the mice aged [70]. The mechanism behind this effect has not yet been elucidated. Along these lines, PTX3 has been detected in seminal fluid and on ejaculated sperm [35]. In motility experiments, PTX3 had no effect on sperm motility and did not correlate with fertilization capacity. Doni et al. suggest that PTX3 may play a role in normal sperm maturation, and this is an area in need of further research [35].
7.2. Seminal Plasma
Seminal plasma contains nutrients important for spermatozoa maintenance (metabolites, proteins, fructose, cholesterol, etc. [67,71]) and prostasomes (vesicles) [72]. Seminal plasma provides a nutrient-rich, immunoprotective environment for ejaculated sperm as they traverse through the female genital tract. The glycosylphosphatidylinositol (GPI)-anchored complement inhibitors CD46, CD55, and CD59 have been detected on the membranes of prostasomes as well as in a stable membrane-free configuration ([73,74], reviewed in [3]) (Table 1, Figure 3). The presence of free complement proteins may allow for easy transfer to cells, specifically to spermatozoa, and it is thought that through this they can confer protection against complement-mediated killing [3,73]. Prostasome-associated complement inhibitors may also serve complement-regulatory functions both in the seminal plasma and also through the transportation of the inhibitors to other cells [3]. Sakaue et al. determined the presence of Factor H in the seminal plasma of pigs [34]. Of particular interest was their data indicating that this Factor H had about two times the affinity for complement inhibition than the Factor H did in serum [34].
In kind, CLU is detected at increased levels in seminal fluid as compared to blood plasma, particularly sCLU [23,30,31,32]. Besides inhibiting MAC in the immunological complement cascade, levels of clusterin in seminal fluid correlate with spermatozoa viability and fertility [75]. The function of CLU outside of complement regulation has been extensively reviewed by Janiszewska and Kratz 2019 [33]. Indeed, CLU’s complement-associated function was discovered after its roles in reproduction were discovered [33]. CLU is essential in sperm capacitation, when the sperm within the female reproductive tract undergo the final physiologic changes critical for oocyte penetration and successful fertilization [76]. Additionally, CLU has been shown to assist in establishing a temporary immune tolerance of sperm in the female genital tract during ejaculation and subsequent fertilization events [33,76,77]. Though much has been uncovered regarding CLU in reproduction, investigation into potential non-canonical CLU functions in seminal plasma, sperm motility, and fertilization is worthy of continued study.
Complement cascade components (C3, C4, and C9) have been detected in human seminal plasma [20,21,22,23] and have been used in various studies to measure the efficacy of complement inhibition [23]. In healthy semen samples, concentrations of C3 and C9 are very low, at less than 5% and 0.3% of blood plasma levels [23]. Even with these cascade components present, high levels of complement inhibition are detected in seminal plasma, contributing to its immunosuppressive aspect [78,79]. In some infertile males [38], decreased complement-inhibitory activity is correlated with infertility and abnormal sperm. Interestingly, these complement components can become activated in the presence of HIV-1, which is opsonized by C3b, even in the presence of so many inhibitors [80], indicating that an immune response could potentially be executed when necessary.
Ultimately, two other functions of complement components in seminal fluid have been proposed. The first is the contribution to the clearance of apoptotic and dead cells, which could potentially inhibit sperm motility and health [81]. The second is as a mechanism to ensure that the strongest sperm reach the oocyte for fertilization by attacking and clearing the weak sperm.
7.3. Sperm/Germ Cells
Factor H, C4BP, CD46, CD55, CD59, and CLU have been detected on human sperm and germ cells (Figure 3, Table 1) [23,31,34]. Factor H was detected by immunofluorescence on the sperm outer acrosome [34], and hyperglycosylated isoforms of CD59 and CD55 are present throughout the spermatozoa plasma membrane [28,82]. The inner plasma membrane of spermatozoa contains testicular-derived naïve CLU, which is similar in form to the sCLU found in the seminal fluid [31,83,84].
The presence of C4BP was also only detected on the outer acrosome of spermatozoa via indirect immunofluorescence [24]. As discussed previously, C4BP is secreted from epididymal epithelial cells, where it is taken up by the maturing spermatozoa [24]. C4BP is structurally similar to the acrosomal protein sp56, which is thought to participate in sperm recognition of the oocyte [24]. C4BP is also expressed on the spermatozoa plasma membrane [3], but its role in the male reproductive tract aside from complement inhibition is unclear.
Complement activity within the female genital tract is strong [34,85] and has been shown to cause immobilization of sperm moving through ovarian fluid [3]. Along these lines, it is reasonable to conclude that the presence of so many complement regulators in semen (in the seminal plasma and on the sperm) assures redundant levels of inhibition to protect sperm from complement-mediated lysis. Though C3b, created through complement activation, is present in the female genital tract [26,86], it can be readily shut down due to the presence of these inhibitors. In addition to its role in fertilization, CD46 acts as a complement inhibitor in this context, serving as a cofactor of female-derived Factor I [26].
Another function for complement expression in semen is under study—the role of complement activation and binding in the acrosome reaction during sperm–egg fusion. The acrosome reaction allows the sperm to penetrate the oocyte [87] and is so critical to reproduction that sperm without this ability are not able to fertilize oocytes [88]. CD46 and vitronectin are present on the inner acrosome of sperm, but their expression on the rest of the sperm surface is absent or minimal, indicating a potential role in this reaction [36,89]. As of now, a role for vitronectin on the acrosome has not been investigated. CD46 has been most studied in its role in fertilization, which will be discussed next.
8. Clinical Applications for Complement
Complement’s presence in the reproductive tract and on the sperm indicates potential direct action in reproduction. The main complement components studied in sperm health and fertilization outside of immunological protection are CD59, CSDM1, C3, and CD46. Current investigations of complement have led to various clinical applications, particularly in autoimmune research and drug development, but these have not yet been looked at in reproduction.
8.1. Complement Action in Sperm Health and Fertilization
As touched upon previously, complement components may contribute to sperm health and fertilization. CD59, a strong inhibitor of the terminal pathway, has been studied with regards to fertility. Sperm from male CD59b−/− mice had two significant phenotypic differences from wild-type ones: (1) immobile sperm and (2) a decreased number of sperm [70]. These issues made the CD59b−/− mice increasingly infertile over the five-month study [90]. CSMD1 is detected strongly in regions where germ cells and somatic cells interact, particularly near Sertoli cells, so knockout mice were generated for the complement inhibitor CSMD1. Infertility in CSMD1−/− males was significantly increased as compared to wild-type mice [90]. A histological examination of testicular tissue displayed increased deposition of C3, consistent with the degradation of testicular tissue, indicating that CSDM1 is an important guardian protein in the blood–testis barrier [90]. However, fertility was still significantly decreased in CSMD1−/− and C3−/− double-knockout mice, implying that complement may have a further function in reproductive success [90]. Taking these studies into account, CD59 and CSMD1 have the potential to be used diagnostically for infertility.
Anderson et al. suggest that CD46 bound to dimerized C3b physically connects the sperm acrosome to a C3b receptor on oocytes [26]. This has been shown in hamsters, as C3b increased the adherence of sperm to oocytes [26]. Further evidence includes an investigation of acrosome CD46 in mouse models and clinical sampling. When antibodies were used to disrupt the first short consensus repeat (SCR) segment of the entire CD46 structure, the binding of sperm to oocytes was significantly reduced [27]. Sperm from 11% of infertile men participating in a Japanese study had significantly decreased interaction with SCR1-specific antibodies [91]. This defect was not detected in leukocytes, so it was limited to the sperm itself.
However, in subsequent mouse studies, CD46-knockout (CD46−/−) mice were hyperfertile [92]. The CD46−/− sperm underwent the acrosome reaction spontaneously, implying that CD46 may serve more of a regulatory role than a direct role by stabilizing the acrosome reaction, ensuring that the fittest sperm would fertilize the oocyte. A point to consider is that, in rodents, CD46 is only expressed on germ cells [93], whereas in humans, CD46 is expressed nearly ubiquitously [94]. Thus, knocking out CD46 in mice would not account for any potential effects in other parts of the murine male reproductive tract. Until further research is conducted on the mechanism behind the ability of CD46−/− sperm to spontaneously undergo the acrosome reaction, particularly in non-rodent models, the role of C3b and CD46 in fertilization is still debatable. Either way, CD46 and C3b seem to influence sperm reproductive success in some manner, thus these complement proteins may also be worthy of analysis as future diagnostics for male infertility.
8.2. Complement Therapeutics in Brief
Twelve complement-inhibitor drugs have been approved by the FDA to date, most with the express purpose of treating various autoimmune and inflammatory disorders where complement-mediated inflammation causes pathology (Figure 4, Table 2) [95,96]. These drugs target C1r, C1s, MASPs, Factor B, C3, C5, and C5aR1. Twenty-seven other complement inhibitory drugs are in clinical trials. These drugs target the above-listed complement components along with C1q, C2, Factor D, Factor H, and Properdin.
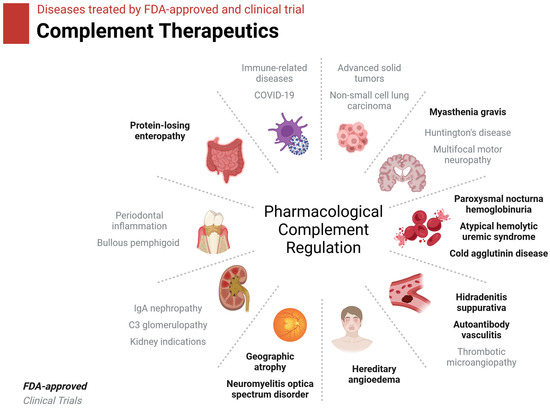
Figure 4.
Diseases treated by current complement therapeutics. FDA-approved therapies are in black text. Therapies currently in clinical trials are in gray text. Figure was created with BioRender.

Table 2.
Complement therapeutics.
Another option besides inhibiting complement action by pharmaceutical intervention is to knock-in complement genes. Regarding xenotransplantation, transgenic pigs are used as donors. These pigs have ten genetic modifications intended to prolong graft viability [98]. Among these are the knock-ins of human CD46 and CD55. With the aid of novel immunosuppressive treatments, the first xenotransplant of a pig heart occurred in January of 2022 and survived for two months—a major feat in xenotransplantation [98]. This clinical application demonstrates that the knock-in of complement components could be an effective therapeutic, and its application should be investigated in the realm of infertility—particularly of CSMD1 and CD46.
Considering these current developments, an investigation of complement therapeutics on sperm health has not yet occurred. Since many of these complement inhibitors and complement genetic knock-ins work to control deleterious inflammation, these therapeutics have the potential to address this issue in infertility. With the wealth of complement-related therapeutics being developed, there is much opportunity to investigate their application in reproduction.
9. Discussion
The presence of complement has been detected in the testis, epididymis, prostate, and semen of the male reproductive tract (Figure 3, Table 1). Of these, complement expression has been most robustly investigated in the testis, particularly regarding Sertoli cells, and semen. Studies indicate that complement may play a role in the establishment and/or maintenance of the immune privilege awarded to the seminiferous tubules of the testis, both by directly inhibiting cascade activation and through potential modulation of immune cells [18,45,46,47,53]. The presence of complement inhibitors in the epididymis and prostate support the inhibition of complement’s inflammatory functions [62,63,83,84].
Complement inhibitors are detected in semen, both on the spermatozoa and in the seminal plasma as free proteins or bound to prostasome membranes. Clusterin makes up the largest protein component of semen, and it is critical in maintaining spermatozoa health [76]. CD55 and CD59 are also detected on spermatozoa, but aside from inhibiting complement-mediated lysis and inflammation, their further function is unknown [3,28,73,89]. Considering the current research on the immune-modulatory functions of complement regulators such as CD46 and CD55, these factors may serve to encourage and maintain an immune-protective milieu conducive to sperm success. The prostasome-associated complement inhibitors, most studied regarding Factor H, have been found to interact with spermatozoa, allowing for the transfer of Factor H to the spermatozoa [34]. This brings up an interesting notion—utilizing vesicle-bound complement regulators, or even other proteins, in the development of new fertility therapies. Surely, in cases of infertile males, supplementing receptor deficiency with exogenous prostasome-bound proteins may be worth investigating.
Pharmacological intervention regarding the complement system may be a potential treatment for male infertility (Figure 4, Table 2). Additionally, in men with aberrant testicular inflammation, the utilization of complement inhibitors may aid in maintaining the blood–testis barrier, thus protecting spermatogenesis. Furthermore, as some complement components seem to be important in sperm health, such as CD59 [70,90] and CSMD1, it could be beneficial to analyze sperm from men with fertility issues to assess if these components are missing. In this manner, new therapeutics could be developed to replace these important proteins and increase sperm motility.
Along the lines of fertilization capacity, sperm from CD46−/− mice demonstrated spontaneous acrosome reaction activity [92], though the mechanism behind the phenotype is not currently known. Still, this knowledge may lead to the development of CD46-deficent fertility treatments, as increasing the likelihood of a spontaneous acrosome reaction in the case of disrupted sperm activity may be beneficial. CD46 may not play the most critical role in ensuring fertilization; its place in this reaction seems redundant to that of other acrosome proteins [99]. This redundancy of function is appropriate since the acrosome reaction is essential to fertilization and needs every opportunity to succeed in ensuring the reproductive success of the species.
A significant limitation in the study of complement in the male reproductive tract, and in general, is that mouse models do not translate well to humans, particularly regarding CD46 [100,101]. In rodents, CD46 is only expressed on the acrosome of sperm and in Sertoli cells [37]. In humans, CD46 is expressed ubiquitously, and it is particularly important in T cell metabolism, clonal expansion, and survival [101]. As rodent T cells do not express CD46, it is then understood that CD46 does not play the same role, or at least does not have the same importance, as it does in human T cells. At this time, humanized mouse models with human CD46 knock-ins are available [102] and are being integrated into complement studies to account for this limitation.
Overall, there is still much to be discovered regarding complement in the male reproductive tract, including:
- The roles of anaphylatoxin signaling in the gonads;
- Functional investigation into genes affected by robust complement exposure;
- Complement expressed by testicular macrophages;
- The effects of C4BP secreted by epididymal cells on sperm maturation;
- Complement in the healthy prostate;
- The specific function of seminal vesicle-derived Factor H;
- A role for PTX3 in sperm maturation;
- Noncanonical CLU functions in seminal plasma, sperm motility, and fertilization;
- The effects of C4BP on sperm in fertilization and sperm health;
- The function of vitronectin on the acrosome.
10. Conclusions
Though much information has been uncovered regarding the presence and function of complement in male reproduction, many areas warrant further study, including complement’s role regarding the prostate and other glands, its function in fertilization, and its importance in testis immune regulation. Garnering a solid understanding of immunological responses and functions in reproduction has many applications in fertility assessment and treatment in humans, agriculture-related reproduction, autoimmune disease pathology, and cancer response.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The information described in this paper is from research articles using publicly available data sets: Sertoli cell bulk RNA sequencing (NCBI GEO database, accession number GSE221711) [18] and Leydig cell scRNA sequencing (NCBI GEO database, accession numbers GSM5563668 and GSM5563669) [19].
Acknowledgments
I would like to acknowledge BioRender.com for their figure-making software and the Jannette M. Dufour lab for her technical training and previous support.
Conflicts of Interest
The author declares no conflicts of interest.
References
- Garcia, P.C.; Rubio, E.M.; Pereira, O.C.M. Antisperm antibodies in infertile men and their correlation with seminal parameters. Reprod. Med. Biol. 2007, 6, 33–38. [Google Scholar] [CrossRef]
- Washburn, R.L.; Hibler, T.; Kaur, G.; Dufour, J.M. Sertoli Cell Immune Regulation: A Double-Edged Sword. Front. Immunol. 2022, 13, 913502. [Google Scholar] [CrossRef]
- Harris, C.L.; Mizuno, M.; Morgan, B.P. Complement and complement regulators in the male reproductive system. Mol. Immunol. 2006, 43, 57–67. [Google Scholar] [CrossRef]
- Dierich, M.P.; Erdei, A.; Huemer, H.; Petzer, A.; Stauder, R.; Schulz, T.F.; Gergely, J. Involvement of complement in B-cell, T-cell and monocyte/macrophage activation. Immunol. Lett. 1987, 14, 235–242. [Google Scholar] [CrossRef]
- Wagner, C.; Ochmann, C.; Schoels, M.; Giese, T.; Stegmaier, S.; Richter, R.; Hug, F.; Hänsch, G.M. The complement receptor 1, CR1 (CD35), mediates inhibitory signals in human T-lymphocytes. Mol. Immunol. 2006, 43, 643–651. [Google Scholar] [CrossRef]
- Kemper, C.; Atkinson, J.P. T-cell regulation: With complements from innate immunity. Nat. Rev. Immunol. 2007, 7, 9–18. [Google Scholar] [CrossRef]
- Heeger, P.S.; Lalli, P.N.; Lin, F.; Valujskikh, A.; Liu, J.; Muqim, N.; Xu, Y.; Medof, M.E. Decay-accelerating factor modulates induction of T cell immunity. J. Exp. Med. 2005, 201, 1523–1530. [Google Scholar] [CrossRef]
- Cardone, J.; Le Friec, G.; Vantourout, P.; Roberts, A.; Fuchs, A.; Jackson, I.; Suddason, T.; Lord, G.; Atkinson, J.P.; Cope, A.; et al. Complement regulator CD46 temporally regulates cytokine production by conventional and unconventional T cells. Nat. Immunol. 2010, 11, 862–871. [Google Scholar] [CrossRef]
- West, E.E.; Kolev, M.; Kemper, C. Complement and the Regulation of T Cell Responses. Annu. Rev. Immunol. 2018, 36, 309–338. [Google Scholar] [CrossRef]
- Merle, N.S.; Noe, R.; Halbwachs-Mecarelli, L.; Fremeaux-Bacchi, V.; Roumenina, L.T. Complement System Part II: Role in Immunity. Front. Immunol. 2015, 6, 257. [Google Scholar] [CrossRef]
- Guo, R.F.; Ward, P.A. Role of C5a in inflammatory responses. Annu. Rev. Immunol. 2005, 23, 821–852. [Google Scholar] [CrossRef]
- Peng, Q.; Li, K.; Patel, H.; Sacks, S.H.; Zhou, W. Dendritic cell synthesis of C3 is required for full T cell activation and development of a Th1 phenotype. J. Immunol. 2006, 176, 3330–3341. [Google Scholar] [CrossRef]
- Strainic, M.G.; Shevach, E.M.; An, F.; Lin, F.; Medof, M.E. Absence of signaling into CD4⁺ cells via C3aR and C5aR enables autoinductive TGF-β1 signaling and induction of Foxp3⁺ regulatory T cells. Nat. Immunol. 2013, 14, 162–171. [Google Scholar] [CrossRef]
- Arbore, G.; West, E.E.; Rahman, J.; Le Friec, G.; Niyonzima, N.; Pirooznia, M.; Tunc, I.; Pavlidis, P.; Powell, N.; Li, Y.; et al. Complement receptor CD46 co-stimulates optimal human CD8(+) T cell effector function via fatty acid metabolism. Nat. Commun. 2018, 9, 4186. [Google Scholar] [CrossRef]
- Stephan, A.H.; Barres, B.A.; Stevens, B. The Complement System: An Unexpected Role in Synaptic Pruning During Development and Disease. Annu. Rev. Neurosci. 2012, 35, 369–389. [Google Scholar] [CrossRef]
- Veerhuis, R.; Nielsen, H.M.; Tenner, A.J. Complement in the brain. Mol. Immunol. 2011, 48, 1592–1603. [Google Scholar] [CrossRef]
- West, E.E.; Kemper, C. Complosome—The intracellular complement system. Nat. Rev. Nephrol. 2023, 19, 426–439. [Google Scholar] [CrossRef]
- Washburn, R.L.; Martinez-Marin, D.; Korać, K.; Sniegowski, T.; Rodriguez, A.R.; Chilton, B.S.; Hibler, T.; Pruitt, K.; Bhutia, Y.D.; Dufour, J.M. The Sertoli Cell Complement Signature: A Suspected Mechanism in Xenograft Survival. Int. J. Mol. Sci. 2023, 24, 1890. [Google Scholar] [CrossRef]
- Li, Y.; Mi, P.; Wu, J.; Tang, Y.; Liu, X.; Cheng, J.; Huang, Y.; Qin, W.; Cheng, C.Y.; Sun, F. High Throughput scRNA-Seq Provides Insights Into Leydig Cell Senescence Induced by Experimental Autoimmune Orchitis: A Prominent Role of Interstitial Fibrosis and Complement Activation. Front. Immunol. 2021, 12, 771373. [Google Scholar] [CrossRef]
- Blenk, H.; Hofstetter, A. Complement C3, coeruloplasmin and PMN-elastase in the ejaculate in chronic prostato-adnexitis and their diagnostic value. Infection 1991, 19 (Suppl. S3), S138–S140. [Google Scholar] [CrossRef]
- Rahimi, A.; Sepehri, H.; Pakravesh, J.; Bahar, K. Quantification of C3 and C4 in infertile men with antisperm antibody in their seminal plasma. Am. J. Reprod. Immunol. 1999, 41, 330–336. [Google Scholar] [CrossRef]
- Sullivan, H.; Quinlivan, W.L.G. Immunoglobulins in the Semen of Men with Azoospermia, Oligospermia, or Self-Agglutination of Spermatozoa. Fertil. Steril. 1980, 34, 465–468. [Google Scholar] [CrossRef]
- Esther Bozas, S.; Kirszbaum, L.; Sparrow, R.L.; Walker, I.D. Several Vascular Complement Inhibitors are Present on Human Sperm1. Biol. Reprod. 1993, 48, 503–511. [Google Scholar] [CrossRef][Green Version]
- Nonaka, M.I.; Hishikawa, Y.; Moriyama, N.; Koji, T.; Ogata, R.T.; Kudo, A.; Kawakami, H.; Nonaka, M. Complement C4b-binding protein as a novel murine epididymal secretory protein. Biol. Reprod. 2003, 69, 1931–1939. [Google Scholar] [CrossRef]
- Ronca, R.; Alessi, P.; Coltrini, D.; Di Salle, E.; Giacomini, A.; Leali, D.; Corsini, M.; Belleri, M.; Tobia, C.; Garlanda, C.; et al. Long pentraxin-3 as an epithelial-stromal fibroblast growth factor-targeting inhibitor in prostate cancer. J. Pathol. 2013, 230, 228–238. [Google Scholar] [CrossRef]
- Anderson, D.J.; Abbott, A.F.; Jack, R.M. The role of complement component C3b and its receptors in sperm-oocyte interaction. Proc. Natl. Acad. Sci. USA 1993, 90, 10051–10055. [Google Scholar] [CrossRef]
- Hsu, E.C.; Dörig, R.E.; Sarangi, F.; Marcil, A.; Iorio, C.; Richardson, C.D. Artificial mutations and natural variations in the CD46 molecules from human and monkey cells define regions important for measles virus binding. J. Virol. 1997, 71, 6144–6154. [Google Scholar] [CrossRef]
- Cummerson, J.A.; Flanagan, B.F.; Spiller, D.G.; Johnson, P.M. The complement regulatory proteins CD55 (decay accelerating factor) and CD59 are expressed on the inner acrosomal membrane of human spermatozoa as well as CD46 (membrane cofactor protein). Immunology 2006, 118, 333–342. [Google Scholar] [CrossRef]
- Geller, A.; Yan, J. The Role of Membrane Bound Complement Regulatory Proteins in Tumor Development and Cancer Immunotherapy. Front. Immunol. 2019, 10, 1074. [Google Scholar] [CrossRef]
- Jenne, D.E.; Tschopp, J. Clusterin: The intriguing guises of a widely expressed glycoprotein. Trends Biochem. Sci. 1992, 17, 154–159. [Google Scholar] [CrossRef]
- Han, Z.; Wang, Z.; Cheng, G.; Liu, B.; Li, P.; Li, J.; Wang, W.; Yin, C.; Zhang, W. Presence, localization, and origin of clusterin in normal human spermatozoa. J. Assist. Reprod. Genet. 2012, 29, 751–757. [Google Scholar] [CrossRef]
- O’Bryan, M.K.; Baker, H.W.; Saunders, J.R.; Kirszbaum, L.; Walker, I.D.; Hudson, P.; Liu, D.Y.; Glew, M.D.; d’Apice, A.J.; Murphy, B.F. Human seminal clusterin (SP-40,40). Isolation and characterization. J. Clin. Investig. 1990, 85, 1477–1486. [Google Scholar] [CrossRef]
- Janiszewska, E.; Kratz, E.M. Could the glycosylation analysis of seminal plasma clusterin become a novel male infertility biomarker? Mol. Reprod. Dev. 2020, 87, 515–524. [Google Scholar] [CrossRef]
- Sakaue, T.; Takeuchi, K.; Maeda, T.; Yamamoto, Y.; Nishi, K.; Ohkubo, I. Factor H in porcine seminal plasma protects sperm against complement attack in genital tracts. J. Biol. Chem. 2010, 285, 2184–2192. [Google Scholar] [CrossRef]
- Doni, A.; Paffoni, A.; Nebuloni, M.; Ragni, G.; Pasqualini, F.; Valentino, S.; Bonetti, S.; Mantovani, A.; Somigliana, E.; Garlanda, C. The long pentraxin 3 is a soluble and cell-associated component of the human semen. Int. J. Androl. 2009, 32, 255–264. [Google Scholar] [CrossRef]
- Bronson, R.; Peresleni, T.; Golightly, M.; Preissner, K. Vitronectin is sequestered within human spermatozoa and liberated following the acrosome reaction. Mol. Hum. Reprod. 2000, 6, 977–982. [Google Scholar] [CrossRef][Green Version]
- Washburn, R.L.; Dufour, J.M. Complementing Testicular Immune Regulation: The Relationship between Sertoli Cells, Complement, and the Immune Response. Int. J. Mol. Sci. 2023, 24, 3371. [Google Scholar] [CrossRef]
- Chowdhury, N.A.; Kamada, M.; Takikawa, M.; Mori, H.; Gima, H.; Aono, T. Complement-inhibiting activity of human seminal plasma and semen quality. Arch. Androl. 1996, 36, 109–118. [Google Scholar] [CrossRef]
- Luo, C.; Chen, M.; Madden, A.; Xu, H. Expression of Complement Components and Regulators by Different Subtypes of Bone Marrow-Derived Macrophages. Inflammation 2012, 35, 1448–1461. [Google Scholar] [CrossRef]
- Kaur, G.; Wright, K.; Verma, S.; Haynes, A.; Dufour, J.M. The Good, the Bad and the Ugly of Testicular Immune Regulation: A Delicate Balance Between Immune Function and Immune Privilege. Adv. Exp. Med. Biol. 2021, 1288, 21–47. [Google Scholar] [CrossRef]
- Gualdoni, G.S.; Jacobo, P.V.; Sobarzo, C.M.; Pérez, C.V.; Matzkin, M.E.; Höcht, C.; Frungieri, M.B.; Hill, M.; Anegon, I.; Lustig, L.; et al. Role of indoleamine 2,3-dioxygenase in testicular immune-privilege. Sci. Rep. 2019, 9, 15919. [Google Scholar] [CrossRef]
- Skinner, M.K.; Moses, H.L. Transforming growth factor beta gene expression and action in the seminiferous tubule: Peritubular cell-Sertoli cell interactions. Mol. Endocrinol. 1989, 3, 625–634. [Google Scholar] [CrossRef]
- França, L.R.; Hess, R.A.; Dufour, J.M.; Hofmann, M.C.; Griswold, M.D. The Sertoli cell: One hundred fifty years of beauty and plasticity. Andrology 2016, 4, 189–212. [Google Scholar] [CrossRef]
- Dufour, J.M.; Hamilton, M.; Rajotte, R.V.; Korbutt, G.S. Neonatal Porcine Sertoli Cells Inhibit Human Natural Antibody-Mediated Lysis1. Biol. Reprod. 2005, 72, 1224–1231. [Google Scholar] [CrossRef]
- Yin, Z.; Wang, L.; Xiang, Y.; Ruan, Y.; Li, J.; Wang, X.; Ichim, T.E.; Chen, S.; Chen, G. Resistance of neonatal porcine Sertoli cells to human xenoantibody and complement-mediated lysis is associated with low expression of alpha-Gal and high production of clusterin and CD59. Xenotransplantation 2010, 17, 215–223. [Google Scholar] [CrossRef]
- Doyle, T.J.; Kaur, G.; Putrevu, S.M.; Dyson, E.L.; Dyson, M.; McCunniff, W.T.; Pasham, M.R.; Kim, K.H.; Dufour, J.M. Immunoprotective Properties of Primary Sertoli Cells in Mice: Potential Functional Pathways that Confer Immune Privilege1. Biol. Reprod. 2012, 86, 6. [Google Scholar] [CrossRef]
- Washburn, R.L.; Kaur, G.; Dufour, J.M. Mouse Sertoli Cells Inhibit Humoral-Based Immunity. Int. J. Mol. Sci. 2022, 23, 12760. [Google Scholar] [CrossRef]
- Wright, K.; Dziuk, R.; Mital, P.; Kaur, G.; Dufour, J.M. Xenotransplanted Pig Sertoli Cells Inhibit Both the Alternative and Classical Pathways of Complement-Mediated Cell Lysis While Pig Islets Are Killed. Cell Transplant. 2016, 25, 2027–2040. [Google Scholar] [CrossRef]
- Yang, W.; Liu, L.-B.; Liu, F.-L.; Wu, Y.-H.; Zhen, Z.-D.; Fan, D.-Y.; Sheng, Z.-Y.; Song, Z.-R.; Chang, J.-T.; Zheng, Y.-T.; et al. Single-cell RNA sequencing reveals the fragility of male spermatogenic cells to Zika virus-induced complement activation. Nat. Commun. 2023, 14, 2476. [Google Scholar] [CrossRef]
- Pandey, S.; Maharana, J.; Li, X.X.; Woodruff, T.M.; Shukla, A.K. Emerging Insights into the Structure and Function of Complement C5a Receptors. Trends Biochem. Sci. 2020, 45, 693–705. [Google Scholar] [CrossRef]
- Rudilla, F.; Fayolle, C.; Casares, N.; Durantez, M.; Arribillaga, L.; Lozano, T.; Villanueva, L.; Pio, R.; Sarobe, P.; Leclerc, C.; et al. Combination of a TLR4 ligand and anaphylatoxin C5a for the induction of antigen-specific cytotoxic T cell responses. Vaccine 2012, 30, 2848–2858. [Google Scholar] [CrossRef]
- Kwan, W.H.; van der Touw, W.; Paz-Artal, E.; Li, M.O.; Heeger, P.S. Signaling through C5a receptor and C3a receptor diminishes function of murine natural regulatory T cells. J. Exp. Med. 2013, 210, 257–268. [Google Scholar] [CrossRef]
- Washburn, R.L.; Martinez-Marin, D.; Sniegowski, T.; Korać, K.; Rodriguez, A.R.; Miranda, J.M.; Chilton, B.S.; Bright, R.K.; Pruitt, K.; Bhutia, Y.D.; et al. Sertoli Cells Express Accommodation, Survival, and Immunoregulatory Factors When Exposed to Normal Human Serum. Biomedicines 2023, 11, 1650. [Google Scholar] [CrossRef]
- Kolev, M.; Le Friec, G.; Kemper, C. The role of complement in CD4⁺ T cell homeostasis and effector functions. Semin. Immunol. 2013, 25, 12–19. [Google Scholar] [CrossRef]
- Kemper, C.; Chan, A.C.; Green, J.M.; Brett, K.A.; Murphy, K.M.; Atkinson, J.P. Activation of human CD4+ cells with CD3 and CD46 induces a T-regulatory cell 1 phenotype. Nature 2003, 421, 388–392. [Google Scholar] [CrossRef]
- Liu, J.; Miwa, T.; Hilliard, B.; Chen, Y.; Lambris, J.D.; Wells, A.D.; Song, W.C. The complement inhibitory protein DAF (CD55) suppresses T cell immunity in vivo. J. Exp. Med. 2005, 201, 567–577. [Google Scholar] [CrossRef]
- Zirkin, B.R.; Papadopoulos, V. Leydig cells: Formation, function, and regulation. Biol. Reprod. 2018, 99, 101–111. [Google Scholar] [CrossRef]
- Mossadegh-Keller, N.; Sieweke, M.H. Testicular macrophages: Guardians of fertility. Cell Immunol. 2018, 330, 120–125. [Google Scholar] [CrossRef]
- Bohlson, S.S.; O’Conner, S.D.; Hulsebus, H.J.; Ho, M.-M.; Fraser, D.A. Complement, C1q, and C1q-Related Molecules Regulate Macrophage Polarization. Front. Immunol. 2014, 5, 402. [Google Scholar] [CrossRef]
- Chaumonnot, K.; Masson, S.; Sikner, H.; Bouchard, A.; Baverel, V.; Bellaye, P.-S.; Collin, B.; Garrido, C.; Kohli, E. The HSP GRP94 interacts with macrophage intracellular complement C3 and impacts M2 profile during ER stress. Cell Death Dis. 2021, 12, 114. [Google Scholar] [CrossRef]
- Tao, J.; Zhao, J.; Qi, X.-M.; Wu, Y.-G. Complement-mediated M2/M1 macrophage polarization may be involved in crescent formation in lupus nephritis. Int. Immunopharmacol. 2021, 101, 108278. [Google Scholar] [CrossRef]
- James, E.R.; Carrell, D.T.; Aston, K.I.; Jenkins, T.G.; Yeste, M.; Salas-Huetos, A. The Role of the Epididymis and the Contribution of Epididymosomes to Mammalian Reproduction. Int. J. Mol. Sci. 2020, 21, 5377. [Google Scholar] [CrossRef]
- Ali Hassan, H.; Domain, G.; Luvoni, G.C.; Chaaya, R.; Van Soom, A.; Wydooghe, E. Canine and Feline Epididymal Semen—A Plentiful Source of Gametes. Animals 2021, 11, 2961. [Google Scholar] [CrossRef]
- Calvel, P.; Rolland, A.D.; Jégou, B.; Pineau, C. Testicular postgenomics: Targeting the regulation of spermatogenesis. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2010, 365, 1481–1500. [Google Scholar] [CrossRef][Green Version]
- Mann, T. Secretory function of the prostate, seminal vesicle and other male accessory organs of reproduction. J. Reprod. Fertil. 1974, 37, 179–188. [Google Scholar] [CrossRef]
- Zhou, Z.; Jia, D.; Kwon, O.; Li, S.; Sun, H.; Roudier, M.P.; Lin, D.W.; True, L.; Morrissey, C.; Creighton, C.J.; et al. Androgen-regulated stromal complement component 7 (C7) suppresses prostate cancer growth. Oncogene 2023, 42, 2428–2438. [Google Scholar] [CrossRef]
- Samanta, L.; Parida, R.; Dias, T.R.; Agarwal, A. The enigmatic seminal plasma: A proteomics insight from ejaculation to fertilization. Reprod. Biol. Endocrinol. 2018, 16, 41. [Google Scholar] [CrossRef]
- McKay, A.C.; Odeluga, N.; Jiang, J.; Sharma, S. Anatomy, Abdomen and Pelvis, Seminal Vesicle. In StatPearls; StatPearls Publishing LLC: Treasure Island, FL, USA, 2023. [Google Scholar]
- Mann, T.; Lutwak-Mann, C. Male Reproductive Function and the Composition of Semen: General Considerations. In Male Reproductive Function and Semen: Themes and Trends in Physiology, Biochemistry and Investigative Andrology; Mann, T., Lutwak-Mann, C., Eds.; Springer: London, UK, 1981; pp. 1–37. [Google Scholar] [CrossRef]
- Qin, X.; Krumrei, N.; Grubissich, L.; Dobarro, M.; Aktas, H.; Perez, G.; Halperin, J.A. Deficiency of the mouse complement regulatory protein mCd59b results in spontaneous hemolytic anemia with platelet activation and progressive male infertility. Immunity 2003, 18, 217–227. [Google Scholar] [CrossRef]
- Talluri, T.R.; Mal, G.; Ravi, S.K. Biochemical components of seminal plasma and their correlation to the fresh seminal characteristics in Marwari stallions and Poitou jacks. Vet. World 2017, 10, 214–220. [Google Scholar] [CrossRef]
- Cross, N.L.; Mahasreshti, P. Prostasome fraction of human seminal plasma prevents sperm from becoming acrosomally responsive to the agonist progesterone. Arch. Androl. 1997, 39, 39–44. [Google Scholar] [CrossRef]
- Rooney, I.A.; Heuser, J.E.; Atkinson, J.P. GPI-anchored complement regulatory proteins in seminal plasma. An analysis of their physical condition and the mechanisms of their binding to exogenous cells. J. Clin. Investig. 1996, 97, 1675–1686. [Google Scholar] [CrossRef]
- Seya, T.; Hara, T.; Matsumoto, M.; Kiyohara, H.; Nakanishi, I.; Kinouchi, T.; Okabe, M.; Shimizu, A.; Akedo, H. Membrane cofactor protein (MCP, CD46) in seminal plasma and on spermatozoa in normal and “sterile” subjects. Eur. J. Immunol. 1993, 23, 1322–1327. [Google Scholar] [CrossRef]
- Janiszewska, E.; Kokot, I.; Gilowska, I.; Faundez, R.; Kratz, E.M. The possible association of clusterin fucosylation changes with male fertility disorders. Sci. Rep. 2021, 11, 15674. [Google Scholar] [CrossRef]
- Milardi, D.; Grande, G.; Vincenzoni, F.; Messana, I.; Pontecorvi, A.; De Marinis, L.; Castagnola, M.; Marana, R. Proteomic approach in the identification of fertility pattern in seminal plasma of fertile men. Fertil. Steril. 2012, 97, 67–73.e1. [Google Scholar] [CrossRef]
- Merlotti, A.; Dantas, E.; Remes Lenicov, F.; Ceballos, A.; Jancic, C.; Varese, A.; Rubione, J.; Stover, S.; Geffner, J.; Sabatté, J. Fucosylated clusterin in semen promotes the uptake of stress-damaged proteins by dendritic cells via DC-SIGN. Hum. Reprod. 2015, 30, 1545–1556. [Google Scholar] [CrossRef]
- Brooks, G.F.; Lammel, C.J.; Petersen, B.H.; Stites, D.P. Human seminal plasma inhibition of antibody complement-mediated killing and opsonization of Neisseria gonorrhoeae and other gram-negative organisms. J. Clin. Investig. 1981, 67, 1523–1531. [Google Scholar] [CrossRef]
- Petersen, B.H.; Lammel, C.J.; Stites, D.P.; Brooks, G.F. Human seminal plasma inhibition of complement. J. Lab. Clin. Med. 1980, 96, 582–591. [Google Scholar]
- Bouhlal, H.; Chomont, N.; Haeffner-Cavaillon, N.; Kazatchkine, M.D.; Belec, L.; Hocini, H. Opsonization of HIV-1 by semen complement enhances infection of human epithelial cells. J. Immunol. 2002, 169, 3301–3306. [Google Scholar] [CrossRef]
- Tomlinson, M.J.; White, A.; Barratt, C.L.; Bolton, A.E.; Cooke, I.D. The removal of morphologically abnormal sperm forms by phagocytes: A positive role for seminal leukocytes? Hum. Reprod. 1992, 7, 517–522. [Google Scholar] [CrossRef]
- Baalasubramanian, S.; Harris, C.L.; Donev, R.M.; Mizuno, M.; Omidvar, N.; Song, W.C.; Morgan, B.P. CD59a is the primary regulator of membrane attack complex assembly in the mouse. J. Immunol. 2004, 173, 3684–3692. [Google Scholar] [CrossRef]
- Sylvester, S.R.; Morales, C.; Oko, R.; Griswold, M.D. Localization of sulfated glycoprotein-2 (clusterin) on spermatozoa and in the reproductive tract of the male rat. Biol. Reprod. 1991, 45, 195–207. [Google Scholar] [CrossRef][Green Version]
- O’Bryan, M.K.; Mallidis, C.; Murphy, B.F.; Baker, H.W. Immunohistological localization of clusterin in the male genital tract in humans and marmosets. Biol. Reprod. 1994, 50, 502–509. [Google Scholar] [CrossRef][Green Version]
- Perricone, R.; Pasetto, N.; De Carolis, C.; Vaquero, E.; Piccione, E.; Baschieri, L.; Fontana, L. Functionally active complement is present in human ovarian follicular fluid and can be activated by seminal plasma. Clin. Exp. Immunol. 1992, 89, 154–157. [Google Scholar] [CrossRef]
- Kabut, J.; Kondera-Anasz, Z.; Sikora, J.; Mielczarek-Palacz, A. Levels of complement components iC3b, C3c, C4, and SC5b-9 in peritoneal fluid and serum of infertile women with endometriosis. Fertil. Steril. 2007, 88, 1298–1303. [Google Scholar] [CrossRef]
- Brucker, C.; Lipford, G.B. The human sperm acrosome reaction: Physiology and regulatory mechanisms. An update. Hum. Reprod. Update 1995, 1, 51–62. [Google Scholar] [CrossRef]
- Morohoshi, A.; Miyata, H.; Tokuhiro, K.; Iida-Norita, R.; Noda, T.; Fujihara, Y.; Ikawa, M. Testis-enriched ferlin, FER1L5, is required for Ca(2+)-activated acrosome reaction and male fertility. Sci. Adv. 2023, 9, eade7607. [Google Scholar] [CrossRef]
- Cervoni, F.; Oglesby, T.J.; Fénichel, P.; Dohr, G.; Rossi, B.; Atkinson, J.P.; Hsi, B.L. Expression of decay-accelerating factor (CD55) of the complement system on human spermatozoa. J. Immunol. 1993, 151, 939–948. [Google Scholar] [CrossRef]
- Lee, A.S.; Rusch, J.; Lima, A.C.; Usmani, A.; Huang, N.; Lepamets, M.; Vigh-Conrad, K.A.; Worthington, R.E.; Mägi, R.; Wu, X.; et al. Rare mutations in the complement regulatory gene CSMD1 are associated with male and female infertility. Nat. Commun. 2019, 10, 4626. [Google Scholar] [CrossRef]
- Kitamura, M.; Matsumiya, K.; Yamanaka, M.; Takahara, S.; Hara, T.; Matsumoto, M.; Namiki, M.; Okuyama, A.; Seya, T. Possible association of infertility with sperm-specific abnormality of CD46. J. Reprod. Immunol. 1997, 33, 83–88. [Google Scholar] [CrossRef]
- Inoue, N.; Ikawa, M.; Nakanishi, T.; Matsumoto, M.; Nomura, M.; Seya, T.; Okabe, M. Disruption of mouse CD46 causes an accelerated spontaneous acrosome reaction in sperm. Mol. Cell Biol. 2003, 23, 2614–2622. [Google Scholar] [CrossRef]
- Mizuno, M.; Harris, C.L.; Johnson, P.M.; Morgan, B.P. Rat membrane cofactor protein (MCP; CD46) is expressed only in the acrosome of developing and mature spermatozoa and mediates binding to immobilized activated C3. Biol. Reprod. 2004, 71, 1374–1383. [Google Scholar] [CrossRef]
- Choileain, S.N.; Astier, A.L. CD46 processing: A means of expression. Immunobiology 2012, 217, 169–175. [Google Scholar] [CrossRef]
- Zelek, W.M.; Xie, L.; Morgan, B.P.; Harris, C.L. Compendium of current complement therapeutics. Mol. Immunol. 2019, 114, 341–352. [Google Scholar] [CrossRef] [PubMed]
- Mastellos, D.C.; Ricklin, D.; Lambris, J.D. Clinical promise of next-generation complement therapeutics. Nat. Rev. Drug Discov. 2019, 18, 707–729. [Google Scholar] [CrossRef]
- West, E.E.; Woodruff, T.; Fremeaux-Bacchi, V.; Kemper, C. Complement in human disease: Approved and up-and-coming therapeutics. Lancet 2023. [Google Scholar] [CrossRef]
- Griffith, B.P.; Goerlich, C.E.; Singh, A.K.; Rothblatt, M.; Lau, C.L.; Shah, A.; Lorber, M.; Grazioli, A.; Saharia, K.K.; Hong, S.N.; et al. Genetically Modified Porcine-to-Human Cardiac Xenotransplantation. N. Engl. J. Med. 2022, 387, 35–44. [Google Scholar] [CrossRef]
- Klinovska, K.; Sebkova, N.; Dvorakova-Hortova, K. Sperm-egg fusion: A molecular enigma of mammalian reproduction. Int. J. Mol. Sci. 2014, 15, 10652–10668. [Google Scholar] [CrossRef]
- Kemper, C.; Verbsky, J.W.; Price, J.D.; Atkinson, J.P. T-Cell stimulation and regulation: With complements from CD46. Immunol. Res. 2005, 32, 31–43. [Google Scholar] [CrossRef]
- Yamamoto, H.; Fara, A.F.; Dasgupta, P.; Kemper, C. CD46: The ‘multitasker’ of complement proteins. Int. J. Biochem. Cell Biol. 2013, 45, 2808–2820. [Google Scholar] [CrossRef]
- Verhaagh, S.; de Jong, E.; Goudsmit, J.; Lecollinet, S.; Gillissen, G.; de Vries, M.; van Leuven, K.; Que, I.; Ouwehand, K.; Mintardjo, R.; et al. Human CD46-transgenic mice in studies involving replication-incompetent adenoviral type 35 vectors. J. Gen. Virol. 2006, 87, 255–265. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).