The Potential Use of Remote Underwater Video (RUV) to Evaluate Small-Bodied Fish Assemblages
Abstract
:1. Introduction
2. Methods
2.1. Study Sites
2.1.1. Silver Creek
2.1.2. West Credit
2.1.3. Lower Credit
2.2. Fish Observation Methods
- Establish baseline pixel-to-length (PTL) scale using a video clip with a known filming distance from the rod (di) and a known rod measurement increment (li) (i.e., l is equal to x number of pixels at distance d; Figure 3). This will differ between camera models and lens types.
- Measure pixel length of known rod increment (lmeas) using an on-screen pixel ruler.
- Using the inverse square law, lmeas, and the baseline PTL scale, determine the distance the rod is away from the camera (dmeas). This step may be skipped if distance dmeas is precisely measured in situ.
- Estimate the unitless proportional distance (p) that the fish is from the camera to the rod. For example, if the fish were directly adjacent to the rod, p would equal 1. If the fish were halfway between the rod and the camera, p would equal 0.5. Proportional distance estimates are used instead of actual distance estimates to reduce user bias.
- Convert p to a real length by multiplying with dmeas and, using the inverse square law, establish a new PTL scale for the fish distance location using Equation (1):where C = dili2.PTL = C ∗ (pdmeas)−0.5
- Measure the pixel length of the fish (f) when it is perpendicular to the camera viewing direction and convert to a real length with the new PTL scale.
- Repeat steps 4–6 at least three times per fish, preferably at multiple distances from the camera, and average the resulting estimates. It is imperative that each video clip is analyzed using uniform screen and video player dimensions to avoid alterations in the PTL scales.
2.3. Water Quality Monitoring
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- King, A.J.; Gawne, B.; Beesley, L.; Koehn, J.D.; Nielsen, D.L.; Price, A. Improving Ecological Response Monitoring of Environmental Flows. Environ. Manag. 2015, 55, 991–1005. [Google Scholar] [CrossRef]
- King, A.J.; George, A.; Buckle, D.J.; Novak, P.A.; Fulton, C.J. Efficacy of Remote Underwater Video Cameras for Monitoring Tropical Wetland Fishes. Hydrobiologia 2018, 807, 145–164. [Google Scholar] [CrossRef]
- Mallet, D.; Pelletier, D. Underwater Video Techniques for Observing Coastal Marine Biodiversity: A Review of Sixty Years of Publications (1952–2012). Fish. Res. 2014, 154, 44–62. [Google Scholar] [CrossRef]
- Ebner, B.C.; Morgan, D.L. Using Remote Underwater Video to Estimate Freshwater Fish Species Richness: Video-Based Survey of Freshwater Fishes. J. Fish. Biol. 2013, 82, 1592–1612. [Google Scholar] [CrossRef] [PubMed]
- Ebner, B.C.; Starrs, D.; Morgan, D.L.; Donaldson, J.A.; Doody, J.S.; Cousins, S.; Butler, G.; Tonkin, Z.; Beatty, S.; Broadhurst, B.; et al. Emergence of Field-Based Underwater Video for Understanding the Ecology of Freshwater Fishes and Crustaceans in Australia. J. R. Soc. W. Aust. 2014, 97, 287–296. [Google Scholar]
- Ebner, B.; Thiem, J.; Lintermans, M.; Gilligan, D. Brendan Video Monitoring in Upland Streams. In An Ecological Approach to Re-establishing Australian Freshwater Cod Populations: An Application to Trout Cod in the Murrumbidgee Catchment; Canberra Parks, Conservation & Lands: Canberra, Australia, 2006; pp. 95–104. [Google Scholar]
- Schmid, K.; Reis-Filho, J.A.; Harvey, E.; Giarrizzo, T. Baited Remote Underwater Video as a Promising Nondestructive Tool to Assess Fish Assemblages in Clearwater Amazonian Rivers: Testing the Effect of Bait and Habitat Type. Hydrobiologia 2017, 784, 93–109. [Google Scholar] [CrossRef]
- Sloat, M.R.; Baker, P.F.; Ligon, F.K. Estimating Habitat-Specific Abundances of PIT-Tagged Juvenile Salmonids Using Mobile Antennas: A Comparison with Standard Electrofishing Techniques in a Small Stream. N. Am. J. Fish. Manag. 2011, 31, 986–993. [Google Scholar] [CrossRef]
- Ellender, B.R.; Becker, A.; Weyl, O.L.F.; Swartz, E.R. Underwater Video Analysis as a Non-Destructive Alternative to Electrofishing for Sampling Imperilled Headwater Stream Fishes: Underwater video analysis as an alternative stream sampling technique. Aquatic. Conserv. Mar. Freshw. Ecosyst. 2012, 22, 58–65. [Google Scholar] [CrossRef]
- Wilson, K.L.; Allen, M.S.; Ahrens, R.N.M.; Netherland, M.D. Use of Underwater Video to Assess Freshwater Fish Populations in Dense Submersed Aquatic Vegetation. Mar. Freshw. Res. 2015, 66, 10. [Google Scholar] [CrossRef]
- Davis, L.; Cockburn, J.; Villard, P.V. Deploying Action Cameras to Observe Fish in Shallow, Ice-Covered Streams. J. Freshw. Ecol. 2016, 32, 193–198. [Google Scholar] [CrossRef]
- Tweedie, J. Evaluating Juvenile Salmonid Physical Habitat Use and Characteristics Using a Spatially Explicit Bioenergetics Model and Remote Underwater Video Surveys. Master’s Thesis, University of Guelph, Guelph, ON, USA, 2018. [Google Scholar]
- Lowry, M.; Folpp, H.; Gregson, M.; Suthers, I. Comparison of Baited Remote Underwater Video (BRUV) and Underwater Visual Census (UVC) for Assessment of Artificial Reefs in Estuaries. J. Exp. Mar. Biol. Ecol. 2012, 416–417, 243–253. [Google Scholar] [CrossRef]
- Dibble, E.D.; Eric, D.; Killgore, K.J.; Harrel, S.L. Assessment of Fish-Plant Interactions; Environmental Laboratory (U.S.): Washington, DC, USA, 1997. [Google Scholar]
- Fulton, C.; Starrs, D.; Ruibal, M.; Ebner, B. Counting Crayfish: Active Searching and Baited Cameras Trump Conventional Hoop Netting in Detecting Euastacus Armatus. Endang. Species. Res. 2012, 19, 39–45. [Google Scholar] [CrossRef]
- Harvey, E.; Shortis, M.; Stadler, M.; Cappo, M. A Comparison of the Accuracy and Precision of Measurements from Single and Stereo-Video Systems. Mar. Technol. Soc. J. 2002, 36, 38–49. [Google Scholar] [CrossRef]
- Shortis, M.R.; Ravanbakskh, M.; Shaifat, F.; Harvey, E.S.; Mian, A.; Seager, J.W.; Culverhouse, P.F.; Cline, D.E.; Edgington, D.R. A Review of Techniques for the Identification and Measurement of Fish in Underwater Stereo-Video Image Sequences; Remondino, F., Shortis, M.R., Beyerer, J., Puente León, F., Eds.; SPIE Privacy Policy: Munich, Germany, 2013; p. 87910G. [Google Scholar]
- Cappo, M.; Speare, P.; De’ath, G. Comparison of Baited Remote Underwater Video Stations (BRUVS) and Prawn (Shrimp) Trawls for Assessments of Fish Biodiversity in Inter-Reefal Areas of the Great Barrier Reef Marine Park. J. Exp. Mar. Biol. Ecol. 2004, 302, 123–152. [Google Scholar] [CrossRef]
- Credit Valley Conservation Authority Fishes of the Credit River Watershed 2002. Available online: https://files.cvc.ca/cvc/uploads/2011/02/FishoftheCRW.pdf (accessed on 16 April 2023).
- Allen, B.; Mandrak, N.E. Historical Changes in the Fish Communities of the Credit River Watershed. Aehm 2019, 22, 316–328. [Google Scholar] [CrossRef]
- Ministry of Natural Resources and Credit Valley Conservation Credit River Fisheries Management Plan: A Cooperative Management Planning Initiative for the Credit River Fishery 2002. Available online: https://files.cvc.ca/cvc/uploads/2012/06/credit-river-fisheries-mgmplan.pdf (accessed on 16 April 2023).
- Newson, M.D.; Newson, C.L. Geomorphology, Ecology and River Channel Habitat: Mesoscale Approaches to Basin-Scale Challenges. Prog. Phys. Geogr. 2000, 24, 195–217. [Google Scholar] [CrossRef]
- Huntingford, F.A.; Metcalfe, N.B.; Thorpe, J.E.; Graham, W.D.; Adams, C.E. Social Dominance and Body Size in Atlantic Salmon Parr, Salmo Solar L. J. Fish. Biol. 1990, 36, 877–881. [Google Scholar] [CrossRef]
- Metcalfe, N.B. The Interaction between Behavior and Physiology in Determining Life History Patterns in Atlantic Salmon (Salmo Salar). Can. J. Fish. Aquat. Sci. 1998, 55, 93–103. [Google Scholar] [CrossRef]
- Thornton, E.J.; Duda, J.J.; Quinn, T.P. Influence of Species, Size and Relative Abundance on the Outcomes of Competitive Interactions between Brook Trout and Juvenile Coho Salmon. Ethol. Ecol. Evol. 2017, 29, 157–169. [Google Scholar] [CrossRef]
- Freeman, M.C.; Grossman, G.D. Group Foraging by a Stream Minnow: Shoals or Aggregations? Anim. Behav. 1992, 44, 393–403. [Google Scholar] [CrossRef]
- Peuhkuri, N. Size-Assortative Shoaling in Fish: The Effect of Oddity on Foraging Behaviour. Anim. Behav. 1997, 54, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Butler, G.L.; Rowland, S.J. Using Underwater Cameras to Describe the Reproductive Behaviour of the Endangered Eastern Freshwater Cod Maccullochella Ikei. Ecol. Freshw. Fish 2009, 18, 337–349. [Google Scholar] [CrossRef]
- Bovee, K.D.; Cochnauer, T. Development and Evaluation of Weighted Criteria, Probability-of-Use Curves for Instream Flow Assessments: Fisheries; Cooperative Instream Flow Service Group, Western Energy and Land Use Team, Office of Biological Services, Fish and Wildlife Service, U.S. Department of the Interior Report: Washington, DC, USA, 1978.
- Hayes, J.W.; Goodwin, E.; Shearer, K.A.; Hay, J.; Kelly, L. Can Weighted Useable Area Predict Flow Requirements of Drift-Feeding Salmonids? Comparison with a Net Rate of Energy Intake Model Incorporating Drift–Flow Processes. Trans. Am. Fish. Soc. 2016, 145, 589–609. [Google Scholar] [CrossRef]
- Rosenfeld, J.S.; Bouwes, N.; Wall, C.E.; Naman, S.M. Successes, Failures, and Opportunities in the Practical Application of Drift-Foraging Models. Environ. Biol. Fish. 2014, 97, 551–574. [Google Scholar] [CrossRef]



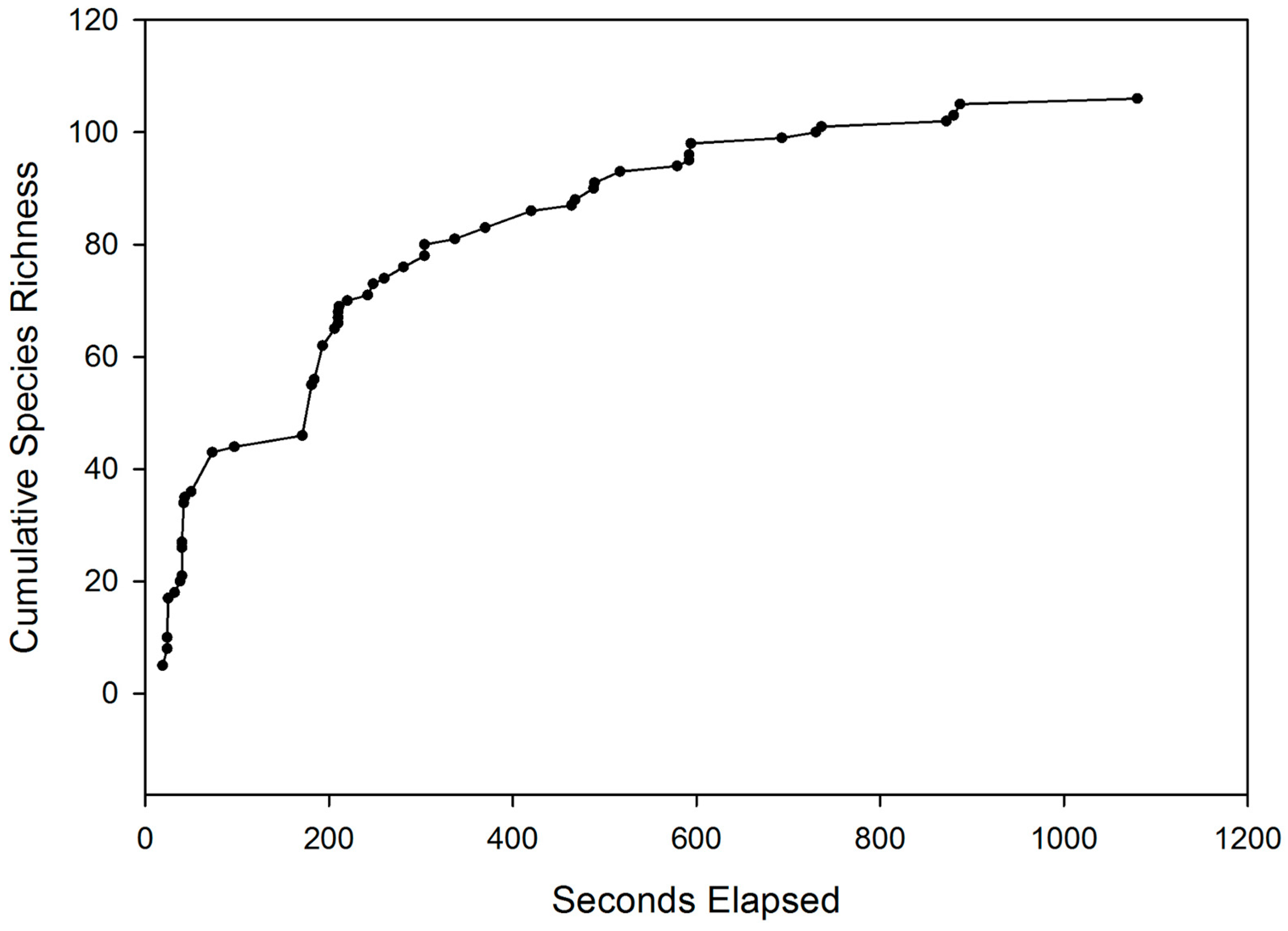

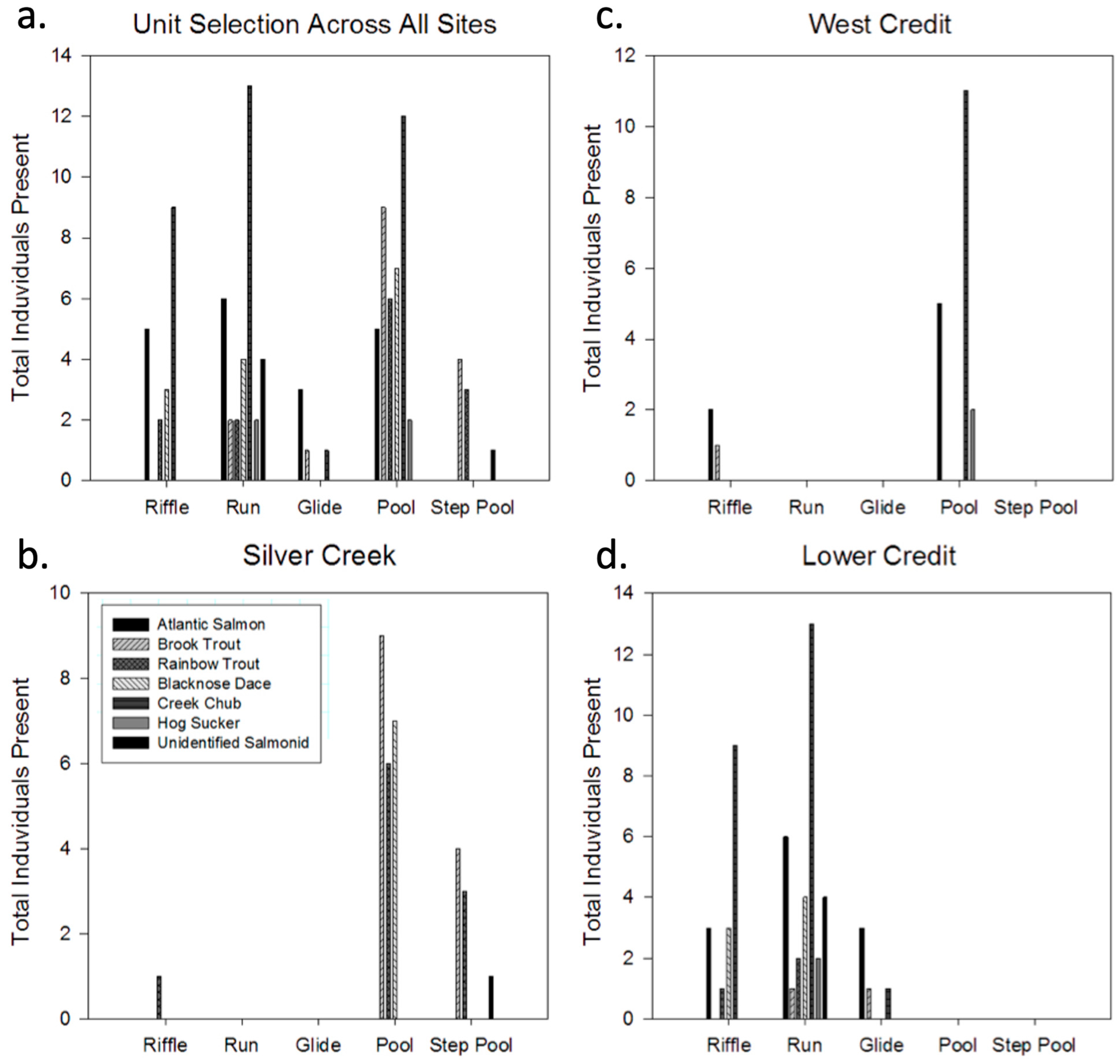
| West Credit | Lower Credit | Silver Creek | All Sites | |
|---|---|---|---|---|
| Video capture success | ||||
| Total video clips | 22 | 56 | 16 | 94 |
| Successful clips | 6 | 22 | 9 | 37 |
| Success rate | 27% | 39% | 56% | 39% |
| Species richness | ||||
| Atlantic salmon | 7 | 12 | 0 | 19 |
| Blacknose dace | 0 | 7 | 7 | 14 |
| Brook trout | 1 | 2 | 13 | 16 |
| Creek chub | 11 | 23 | 1 | 35 |
| Northern hog sucker | 2 | 2 | 0 | 4 |
| Rainbow trout | 0 | 3 | 10 | 13 |
| Unidentified salmonid | 0 | 4 | 1 | 5 |
| Total | 21 | 53 | 32 | 106 |
| Simpson’s Diversity Index | ||||
| 0.367 | 0.293 | 0.310 | 0.210 | |
| MaxN averages | Estimated Average Fish Length (cm) | |||||||
|---|---|---|---|---|---|---|---|---|
| Species | West Credit | Lower Credit | Silver Creek | All Sites | West Credit | Lower Credit | Silver Creek | All Sites |
| Atlantic salmon | 1.40 | 1.22 | n/a | 1.29 | 9.79 | 12.94 | n/a | 11.73 |
| Blacknose dace | n/a | 1.75 | 7.00 | 2.80 | n/a | 6.90 | 4.64 | 6.45 |
| Brook trout | 1.00 | 1.00 | 2.17 | 1.78 | 10.47 | 9.82 | 8.29 | 8.87 |
| Creek chub | 5.50 | 4.60 | 1.00 | 4.38 | 7.00 | 7.22 | 8.76 | 7.38 |
| Northern hog sucker | 2.00 | 2.00 | n/a | 2.00 | 10.97 | n/a | n/a | 10.97 |
| Rainbow trout | n/a | 1.50 | 2.00 | 1.86 | n/a | 11.93 | 7.94 | 9.08 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tweedie, J.B.; Cockburn, J.M.H.; Villard, P.V. The Potential Use of Remote Underwater Video (RUV) to Evaluate Small-Bodied Fish Assemblages. Hydrobiology 2023, 2, 507-520. https://doi.org/10.3390/hydrobiology2030034
Tweedie JB, Cockburn JMH, Villard PV. The Potential Use of Remote Underwater Video (RUV) to Evaluate Small-Bodied Fish Assemblages. Hydrobiology. 2023; 2(3):507-520. https://doi.org/10.3390/hydrobiology2030034
Chicago/Turabian StyleTweedie, John B., Jaclyn M.H. Cockburn, and Paul V. Villard. 2023. "The Potential Use of Remote Underwater Video (RUV) to Evaluate Small-Bodied Fish Assemblages" Hydrobiology 2, no. 3: 507-520. https://doi.org/10.3390/hydrobiology2030034
APA StyleTweedie, J. B., Cockburn, J. M. H., & Villard, P. V. (2023). The Potential Use of Remote Underwater Video (RUV) to Evaluate Small-Bodied Fish Assemblages. Hydrobiology, 2(3), 507-520. https://doi.org/10.3390/hydrobiology2030034







