Combination Therapies in Cancers
A topical collection in Cancers (ISSN 2072-6694). This collection belongs to the section "Cancer Therapy".
Viewed by 24052Editors
2. Institute of Pharmaceutical Sciences of Western Switzerland, University of Geneva, 1211 Geneva, Switzerland
3. Translational Research Center in Oncohaematology, 1211 Geneva, Switzerland
Interests: combination therapy; tumor microenvirontment; targeted therapy; angiogenesis
Special Issues, Collections and Topics in MDPI journals
Interests: angiogenesis; immunology; cancer vaccines; immunotherapy
Topical Collection Information
Dear Colleagues,
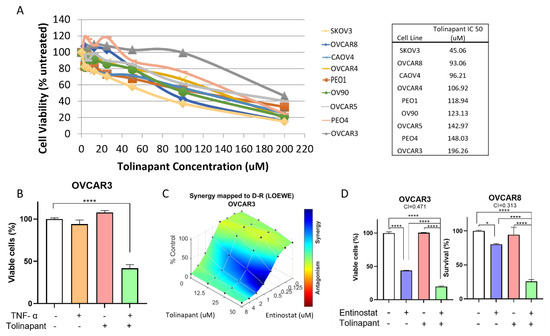
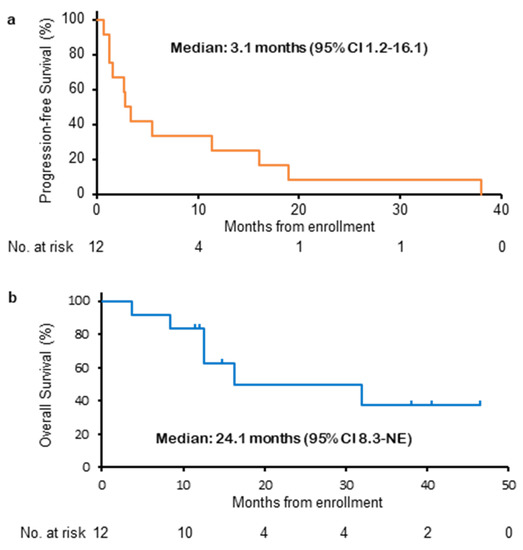
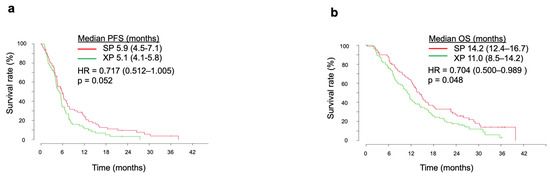
This Topical Collection on “Combination Therapies in Cancers” will address the current efforts and advances in preclinical and clinical studies for the development of combinatorial approaches for effective cancer treatment. Special attention will be on the translation of these findings towards the clinic. The Topical Collection will show different perspectives of improving the activity of cancer treatment through the design of combination therapies and the development of combinatorial strategies with other treatment options. Although many questions might remain unanswered, general interest in combination strategies seems to drive the whole field of cancer research. We hope that this Topical Collection facilitates recognition and understanding of the power of carefully designed combination treatments.
Prof. Dr. Patrycja Nowak-Sliwinska
Prof. Dr. Arjan W. Griffioen
Collection Editors
Manuscript Submission Information
Manuscripts should be submitted online at www.mdpi.com by registering and logging in to this website. Once you are registered, click here to go to the submission form. Manuscripts can be submitted until the deadline. All submissions that pass pre-check are peer-reviewed. Accepted papers will be published continuously in the journal (as soon as accepted) and will be listed together on the collection website. Research articles, review articles as well as communications are invited. For planned papers, a title and short abstract (about 100 words) can be sent to the Editorial Office for announcement on this website.
Submitted manuscripts should not have been published previously, nor be under consideration for publication elsewhere (except conference proceedings papers). All manuscripts are thoroughly refereed through a single-blind peer-review process. A guide for authors and other relevant information for submission of manuscripts is available on the Instructions for Authors page. Cancers is an international peer-reviewed open access semimonthly journal published by MDPI.
Please visit the Instructions for Authors page before submitting a manuscript. The Article Processing Charge (APC) for publication in this open access journal is 2900 CHF (Swiss Francs). Submitted papers should be well formatted and use good English. Authors may use MDPI's English editing service prior to publication or during author revisions.
Keywords
- angiogenesis
- combination therapy
- drug–drug interactions
- drug synergy
- immunology