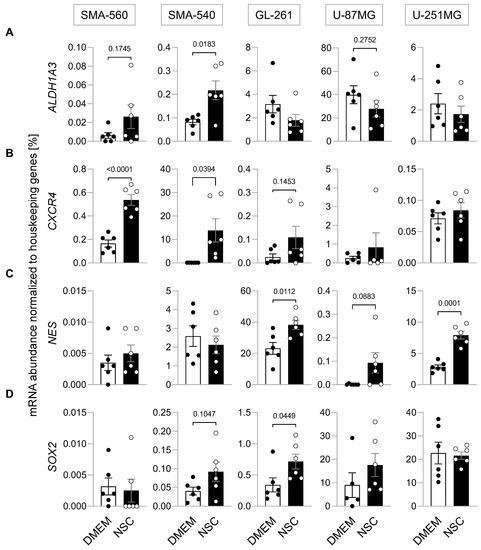
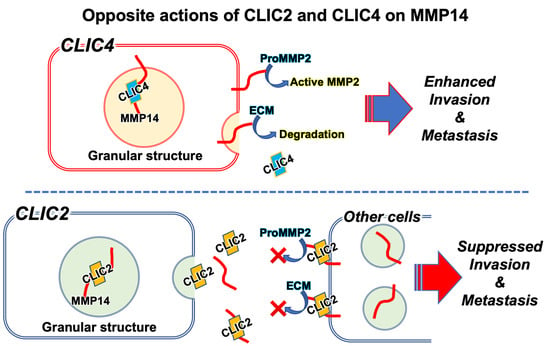
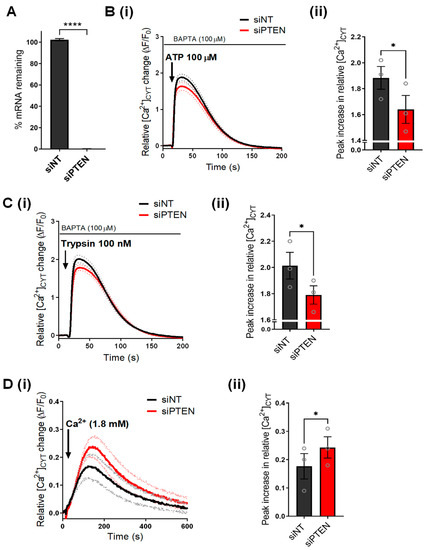
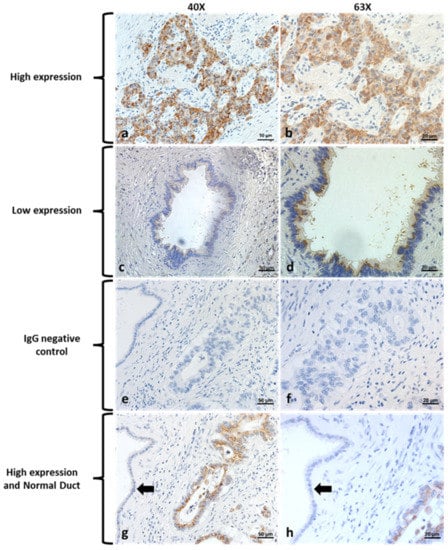
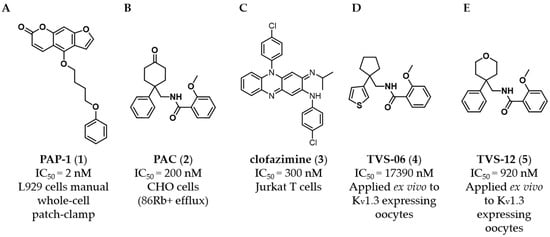
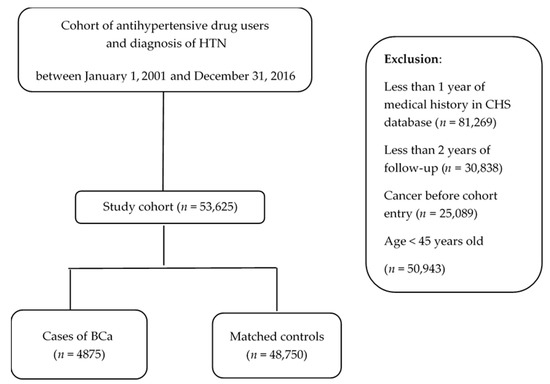
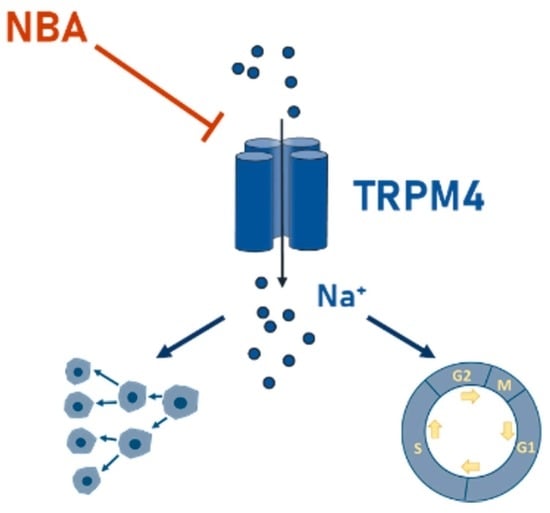
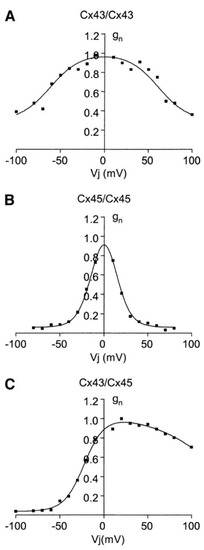
Ion Channels in Cancer Therapies
A topical collection in Cancers (ISSN 2072-6694). This collection belongs to the section "Cancer Therapy".
Viewed by 92186Editor
Interests: ion channels; voltage-gated potassium channels; membrane potential; cell biology; neuroscience; targeted therapies
Topical Collection Information
Dear Colleagues,
The idea that ion channels play crucial roles in the onset and development of all types of cancer has gained overwhelming support in the last several decades. Besides the purely scientific interest that this concept has for cancer biologists, the available extensive knowledge of the physiology and pharmacology of ion channels opens a window for therapeutic interventions that can correct or modify the behavior of tumor cells and their microenvironment and result in a benefit for the patients.
There are many aspects related to ion channels in the context of tumor therapy. First, not only channels in the tumor cells but also those in cells of the microenvironment can be targeted. Moreover, ion channels play relevant roles in some of the undesired effects of more classical treatment regimes for cancer patients, and can thus be addressed to diminish toxicity. The strategies to target channels are also very diverse. In some cases, the function of ion channels is directly tackled by pharmacological modulators, which can be either repurposed drugs in current or past clinical use for other indications or newly identified natural or synthetic compounds with improved selectivity profile and potency. Other strategies take advantage of the aberrant expression of particular ion channels to direct other agents to the tumor cells or aim to correct the pathways altered by pathological channel expression.
In summary, this Topical Collection aims to offer an overview of the present and potential importance of ion channels in cancer therapy.
Prof. Dr. Luis A. Pardo
Collection Editor
Manuscript Submission Information
Manuscripts should be submitted online at www.mdpi.com by registering and logging in to this website. Once you are registered, click here to go to the submission form. All submissions that pass pre-check are peer-reviewed. Accepted papers will be published continuously in the journal (as soon as accepted) and will be listed together on the collection website. Research articles, review articles as well as communications are invited. For planned papers, a title and short abstract (about 250 words) can be sent to the Editorial Office for assessment.
Submitted manuscripts should not have been published previously, nor be under consideration for publication elsewhere (except conference proceedings papers). All manuscripts are thoroughly refereed through a single-blind peer-review process. A guide for authors and other relevant information for submission of manuscripts is available on the Instructions for Authors page. Cancers is an international peer-reviewed open access semimonthly journal published by MDPI.
Please visit the Instructions for Authors page before submitting a manuscript. The Article Processing Charge (APC) for publication in this open access journal is 2900 CHF (Swiss Francs). Submitted papers should be well formatted and use good English. Authors may use MDPI's English editing service prior to publication or during author revisions.
Keywords
- cancer treatment
- ion channels
- ion channel pharmacology
- toxins
- immunotherapy
- drug resistance
- targeted therapies