Sonochemical Preparation of Polymer Nanocomposites
Abstract
:Introduction
Polymer-Carbon Nanotube Nanocomposites
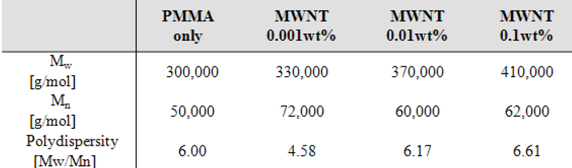 |




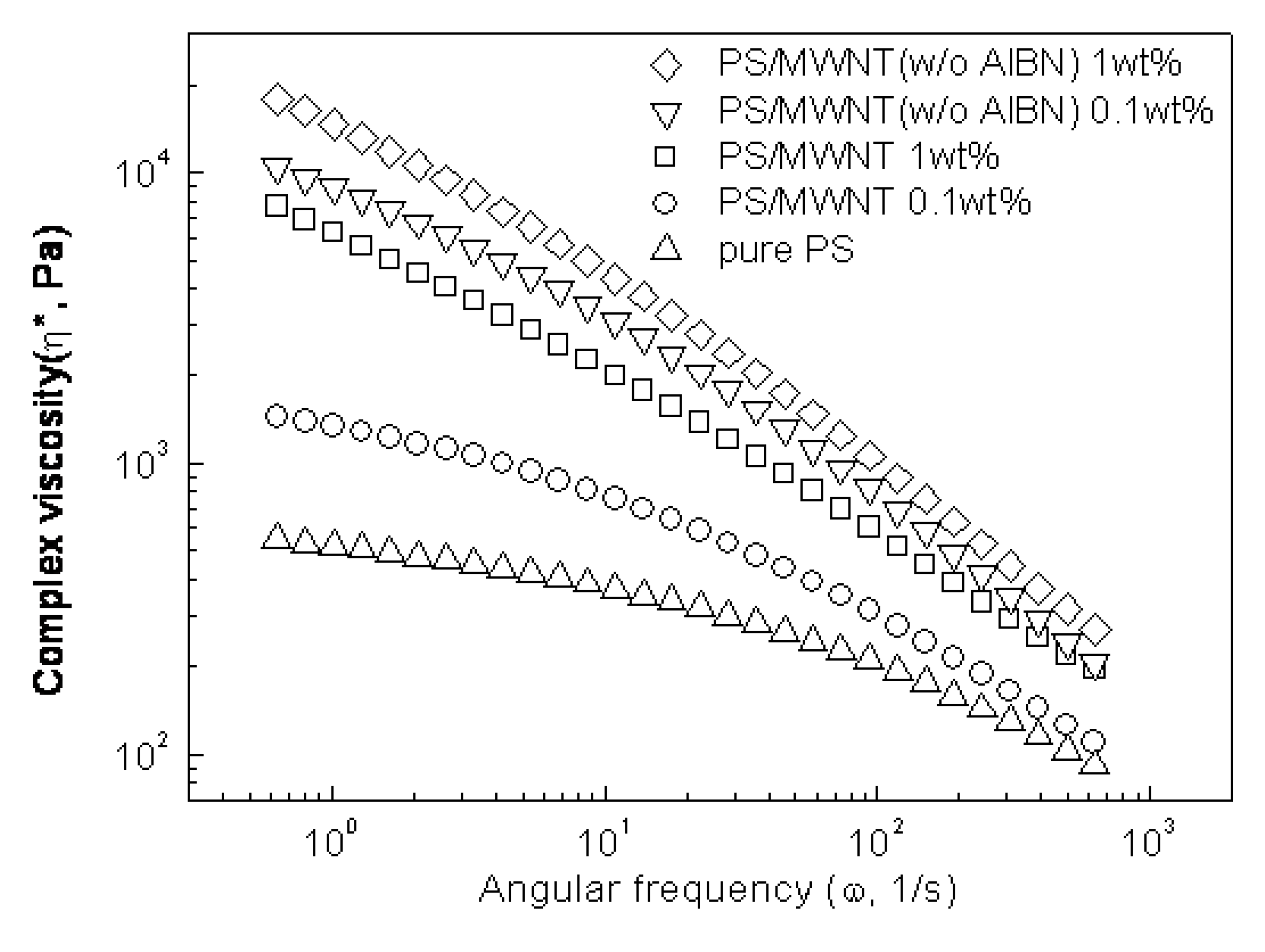
Conducting Nanocomposite Particles

 ) [45], it is thought that hydrogen bonding component (
) [45], it is thought that hydrogen bonding component (  ) may be the principal factor determining the dispersion states of MWNTs in Di-water, because the nanotubes have carboxylic acid functional groups on the surface of the side wall, which can help to form hydrogen bonding between MWNT and Di-water. Due to the benefit from having carboxylic acid functionalization on the surface of MWNT and the dispersive effect of ultrasonication, more individual nanotubes were well adsorbed onto the PMMA surface instead of aggregated ones (Figure 6). The conductivity of PMMA microspheres is found to be dramatically increased from 1.5×10-14 S/cm to 1.4×10-3 S/cm after addition of nanotubes. In addition, the ER fluids prepared using PMMA/MWNT microspheres dispersed in silicone oil (10 vol%) exhibit typical ER fibril structure under the applied electrical field of 0.2kV/mm and the structure remained stable as long as the field was applied. It is well known that the superior ER response is also due to the well-dispersed nanotubes on the PMMA surface. The ER fluids are in general suspensions of particles with a higher dielectric constant and/or conductivity than that of the medium oils with a low dielectric constant and a low shear viscosity. They demonstrate a drastic and reversible change in their rheological characteristics under an applied electric field, because the dispersed ER particles are attracted to each other to form fibrillar structure induced by electrostatic attraction of the polarized particles [46,47,48]. Recently, nanoparticles with MWNT and polyaniline synthesized by oxidative dispersion polymerization using PVA as a polymeric stabilizer and HCl as a dopant [49] were reported to exhibit ER characteristics.
) may be the principal factor determining the dispersion states of MWNTs in Di-water, because the nanotubes have carboxylic acid functional groups on the surface of the side wall, which can help to form hydrogen bonding between MWNT and Di-water. Due to the benefit from having carboxylic acid functionalization on the surface of MWNT and the dispersive effect of ultrasonication, more individual nanotubes were well adsorbed onto the PMMA surface instead of aggregated ones (Figure 6). The conductivity of PMMA microspheres is found to be dramatically increased from 1.5×10-14 S/cm to 1.4×10-3 S/cm after addition of nanotubes. In addition, the ER fluids prepared using PMMA/MWNT microspheres dispersed in silicone oil (10 vol%) exhibit typical ER fibril structure under the applied electrical field of 0.2kV/mm and the structure remained stable as long as the field was applied. It is well known that the superior ER response is also due to the well-dispersed nanotubes on the PMMA surface. The ER fluids are in general suspensions of particles with a higher dielectric constant and/or conductivity than that of the medium oils with a low dielectric constant and a low shear viscosity. They demonstrate a drastic and reversible change in their rheological characteristics under an applied electric field, because the dispersed ER particles are attracted to each other to form fibrillar structure induced by electrostatic attraction of the polarized particles [46,47,48]. Recently, nanoparticles with MWNT and polyaniline synthesized by oxidative dispersion polymerization using PVA as a polymeric stabilizer and HCl as a dopant [49] were reported to exhibit ER characteristics.Magnetic Particle Nanocomposites


Other Nanocomposite Systems
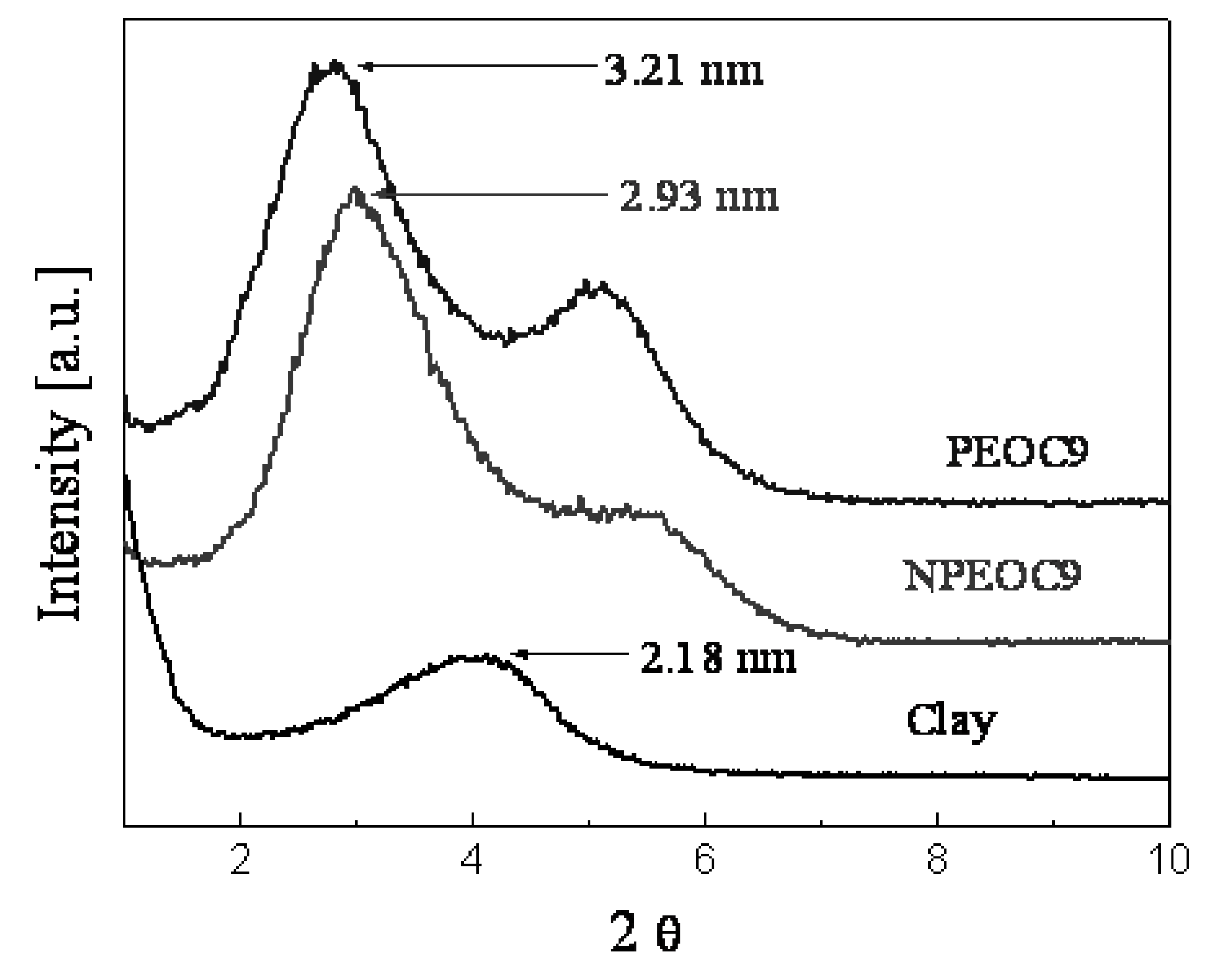


Conclusions
Acknowledgements
References
- Richards, W.T.; Loomis, A.L. The Chemical Effects of High Frequency Sound Waves I. A Preliminary Survey. J. Am. Chem. Soc. 1927, 49, 3086–3100. [Google Scholar] [CrossRef]
- Suslick, K.S.; Price, G.J. Applications of Ultrasound to Materials Chemistry. Annu. Rev. Mater. Sci. 1999, 29, 295–326. [Google Scholar] [CrossRef]
- Blaskovicova, M.; Gaplovsky, A.; Blasko, J. Synthesis and Photochemistry of 1-Iodocyclohexene: Influence of Ultrasound on Ionic vs. Radical Behaviour. Molecules 2007, 11, 188–193. [Google Scholar] [CrossRef]
- Wang, X.K.; Chen, G.H.; Guo, W.L. Sonochemical Degradation Kinetics of Methyl Violet in Aqueous Solutions. Molecules 2003, 8, 40–44. [Google Scholar] [CrossRef]
- Naddeoa, V.; Belgiornoa, V.; Napolib, R.M.A. Behaviour of Natural Organic Mater during Ultrasonic Irradiation. Desalination 2007, 210, 175–182. [Google Scholar] [CrossRef]
- Campos, M.D.; Muller, F.A.; Bressiani, A.H.A.; Bressiani, J.C.; Greil, P. Sonochemical Synthesis of Calcium Phosphate Powders. J. Mater. Sci. Mater. Med. 2007, 18, 669–675. [Google Scholar] [CrossRef]
- Gedanken, A. Using Sonochemistry for the Fabrication of Nanomaterials. Ultrason. Sonochem. 2004, 11, 47–55. [Google Scholar] [CrossRef]
- Adewuyi, Y.G. Sonochemistry: Environmental Science and Engineering Applications. Ind. Eng. Chem. Res. 2001, 40, 4681–4715. [Google Scholar]
- Kim, H.K.; Uchida, H.; Matyjaszewski, K. Sonochemical Synthesis of Polysilylenes by Reductive Coupling of Disubstituted Dichlorosilanes with Alkali Metals. Macromolecules 1995, 28, 59–72. [Google Scholar] [CrossRef]
- Price, G.J.; Norris, D.J.; Peter, J.W. Polymerization of Methyl Methacrylate Initiated by Ultrasound. Macromolecules 1992, 25, 6447–6454. [Google Scholar] [CrossRef]
- Biggs, S.; Grieser, F. Preparation of Polystyrene Latex with Ultrasonic Initiation. Macromolecules 1995, 28, 4877–4882. [Google Scholar] [CrossRef]
- Xia, H.S.; Wang, Q.; Liao, Y.Q.; Xu, X.; Baxter, S.M.; Slone, R.V.; Wu, S.G.; Swift, G.; Westmoreland, D. G. Polymerization Rate and Mechanism of Ultrasonically Initiated Emulsion Polymerization of n-butyl acrylate. Ultrason. Sonochem. 2002, 9, 151–158. [Google Scholar] [CrossRef]
- Chun, M.S.; Park, O.O. On the Intrinsic Viscosity of Anionic and Nonionic Rodlike Polysaccharide Solutions. Macromol. Chem. Phys. 1994, 195, 701–711. [Google Scholar]
- Kim, C.A.; Choi, H.J.; Kim, C.B.; Jhon, M.S. Drag Reduction Characteristics of Polysaccharide Xanthan Gum. Macromol. Rapid Commun. 1998, 19, 419–422. [Google Scholar] [CrossRef]
- Choi, H.J.; Kim, C.A.; Sohn, J.I.; Jhon, M.S. An Exponential Decay Function for Polymer Degradation in Turbulent Drag. Polym. Degrad. Stab. 2000, 69, 341–346. [Google Scholar] [CrossRef]
- Sohn, J.I.; Kim, C.A.; Choi, H.J.; Jhon, M.S. Drag-reduction Effectiveness of Xanthan Gum in a Rotating Disk Apparatus. Carbohydrate Polym. 2001, 45, 61–68. [Google Scholar] [CrossRef]
- Kim, C.A.; Lim, S.T.; Choi, H.J.; Sohn, J.I.; Jhon, M.S. Characterization of Drag Reducing Guar Gum in a Rotating Disk Flow. J. Appl. Polym. Sci. 2002, 83, 2938–2944. [Google Scholar] [CrossRef]
- Lim, S.T.; Choi, H.J.; Chan, C.K. Effect of Turbulent Flow on Coil-Globule Transition of DNA. Macromol. Rapid Commun. 2005, 26, 1237–1240. [Google Scholar] [CrossRef]
- Choi, H.J.; Lim, S.T.; Lai, P.Y.; Chan, C.K. Turbulent Drag Reduction and Degradation of DNA. Phys. Rev. Lett. 2002, 89. art. No. 088302. [Google Scholar]
- Lim, S.T.; Hong, C.H.; Choi, H.J.; Lai, P.Y.; Chan, C.K. Polymer Turbulent Drag Reduction near the Theta Point. EPL 200, 80. Art. No. 58003. [Google Scholar]
- Lim, S.T.; Choi, H.J.; Lee, S.Y.; So, J.S.; Chan, C.K. DNA Induced Turbulent Drag Reduction and Its Characteristics. Macromolecules 2003, 36, 5348–5354. [Google Scholar] [CrossRef]
- Fujiwara, H.; Kimura, T.; Segi, M.; Nakatuka, T.; Nakamura, H. Mechanochemical Block Copolymerization in Heterogeneous Systems of the Solid Poly (vinyl chloride) with Styrene by Ultrasonic Irradiation. Polym. Bull. 1992, 28, 189–196. [Google Scholar] [CrossRef]
- Fujiwara, H.; Ishida, T.; Taniguchi, N.; Wada, S. Mechanochemical Synthesis and Characterization of Poly (vinyl chloride)-block-Poly (vinyl alcohol) Copolymers by Ultrasonic Irradiation. Polym. Bull. 1999, 42, 197–204. [Google Scholar] [CrossRef]
- Fang, F.F.; Choi, H. J.; Joo, J. Conducting Polymer/Clay Nanocomposites and Their Applications. J. Nanosci. Nanotech. 2008, 8, 1559–1581. [Google Scholar] [CrossRef]
- Ray, S.S.; Vaudreuil, S.; Maazouz, A.; Bousmina, M. Dispersion of Multi-Walled Carbon Nanotubes in Biodegradable Poly(butylenes succinate) Matrix. J. Nanosci. Nanotech. 2006, 6, 2191–2195. [Google Scholar] [CrossRef]
- Xing, Y.C.; Li, L.; Chusuei, C.C.; Hull, R.V. Sonochemical Oxidation of Multiwalled Carbon Nanotubes. Langmuir 2005, 21, 4185–4190. [Google Scholar] [CrossRef]
- Chakraborty, S.; Raj, C.R. Mediated Electrocatalytic Oxidation of Bioanalytes and Biosensing of Glutamate using Functionalized Multiwall Carbon Nanotubes-Biopolymer Nanocomposite. J. Electroanal. Chem. 2007, 609, 155–162. [Google Scholar] [CrossRef]
- Xia, H.S.; Wang, Q.; Qiu, G.H. Polymer-Encapsulated Carbon Nanotubes Prepared through Ultrasonically Initiated In Situ Emulsion Polymerization. Chem. Mater. 2003, 15, 3879–3886. [Google Scholar]
- Park, C.; Ounaies, Z.; Watson, K.A.; Crooks, R.E.; Smith, J.; Lowther, S.E.; Connell, J.W.; Siochi, E.J.; Harrison, J.S.; St. Clair, T.L. Dispersion of Single Wall Carbon Nanotubes by In Situ Polymerization under Sonication. Chem. Phys. Lett. 2002, 364, 303–308. [Google Scholar] [CrossRef]
- Park, S.J.; Cho, M.S.; Lim, S.T.; Choi, H.J.; Jhon, M.S. Synthesis and Dispersion Characteristics of Multi-Walled Carbon Nanotube Composites with Poly(methyl methacrylate) Prepared by In-Situ Bulk Polymerization. Macromol. Rapid Commun. 2003, 24, 1070–1073. [Google Scholar] [CrossRef]
- Jia, Z.J.; Wang, Z.Y.; Xu, C.L.; Liang, J.; Wei, B.Q.; Wu, D.H.; Zhu, S.W. Study on Poly(methyl methacrylate):Carbon nanotube Composites. Mater. Sci. Eng. 1999, A271, 395–400. [Google Scholar]
- Kim, S.T.; Choi, H.J.; Hong, S.M. Bulk Polymerized Polystyrene in the Presence of Multiwalled Carbon Nanotubes. Colloid Polym. Sci. 2007, 285, 593–598. [Google Scholar] [CrossRef]
- Song, Y.S.; Youn, J.R. Influence of Dispersion States of Carbon Nanotubes on Physical Properties of Epoxy Nanocomposites. Carbon 2005, 43, 1378–1385. [Google Scholar] [CrossRef]
- Isayev, A.I.; Kumar, R.; Lewis, T.M. Ultrasound Assisted Twin Screw Extrusion of Polymer–Nanocomposites Containing Carbon Nanotubes. Polymer 2009, 50, 250–260. [Google Scholar] [CrossRef]
- Zou, J.H.; Liu, L.W.; Chen, H.; Khondaker, S.I.; McCullough, R.D.; Huo, Q.; Zhai, L. Dispersion of Pristine Carbon Nanotubes Using Conjugated Block Copolymers. Adv. Mater. 2008, 20, 2055–2060. [Google Scholar] [CrossRef]
- Kojima, Y.; Koda, S.; Nomura, H. Effect of Ultrasonic Frequency on Polymerization of Styrene under Sonication. Ultrason. Sonochem 2001, 8, 75–79. [Google Scholar] [CrossRef]
- Slobodian, P.; Lengálová, A.; Sáha, P.; Slouf, M. Poly (methyl methacrylate)/Multi-wall Carbon Nanotubes Composites Prepared by Solvent Cast Technique: Composites Electrical Percolation Threshold. J. Reinf. Plast. Compos. 2007, 26, 1705–1712. [Google Scholar] [CrossRef]
- Barraza, H.J.; Pompeo, F.; O’Rear, E.A.; Resasco, D.E. SWNT-filled Thermoplastic and Elastomeric Composites Prepared by Miniemulsion Polymerization. Nano Lett. 2002, 2, 797–802. [Google Scholar] [CrossRef]
- Choi, H.J.; Zhang, K.; Lim, J.Y. Multi-walled Carbon Nanotube/Polystyrene Composites Prepared by in-situ Bulk Sonochemical Polymerization. J. Nanosci. Nanotechnol. 2007, 7, 3400–3403. [Google Scholar] [CrossRef]
- Choi, C.S.; Park, B.J.; Choi, H.J. Electrical and Rheological Characteristics of Poly(vinyl acetate)/Multi-walled Carbon Nanotube Nanocomposites. Diamond Related Mater. 2007, 16, 1170–1173. [Google Scholar] [CrossRef]
- Sohn, J.I.; Lee, C.H.; Lim, T.H.; Choi, H.J.; Jhon, M.S. Viscoelasticity and Relaxation Characteristics of Polystyrene/Clay Nanocomposite. J. Mater. Sci. 2003, 38, 1849–1852. [Google Scholar] [CrossRef]
- Hyun, Y.H.; Lim, S.T.; Choi, H.J.; Jhon, M.S. Rheology of Poly(ethylene oxide)/Organoclay Nanocomposites. Macromolecules 2001, 34, 8084–8093. [Google Scholar] [CrossRef]
- Li, Q.Y.; Wu, G.Z.; Ma, Y.L; Wu, C.F. Grafting Modification of Carbon Black by Ttrapping Macroradicals Formed by Sonochemical Degradation. Carbon 2007, 45, 2411–2416. [Google Scholar] [CrossRef]
- Zhang, K.; Lim, J.Y.; Park, B.J.; Jin, H.J.; Choi, H.J. Carboxylic Acid Functionalized Multi-Walled Carbon Nanotube-Adsorption onto Poly(methyl methacrylate) Microspheres. J. Nanosci. Nanotech. 2009, 9, 1050–1061. [Google Scholar]
- Ham, H.T.; Choi, Y.S.; Chung, I.J. An Explanation of Dispersion States of Single-Walled Carbon Nanotubes in Solvents and Aqueous Surfactant Solutions using Solubility Parameters. J. Colloid Interf. Sci. 2005, 286, 216–223. [Google Scholar] [CrossRef]
- Cho, M.S.; Choi, H.J.; Jhon, M.S. Shear Stress Analysis of a Semiconducting Polymer Based Electrorheological Fluid System. Polymer 2005, 46, 11484–11488. [Google Scholar] [CrossRef]
- Kim, J.W.; Kim, S.G.; Choi, H.J.; Jhon, M.S. Synthesis and Electrorheological Properties of Polyaniline-Na+-Montmorillonite Suspensions. Macromol. Rapid Commun. 1999, 20, 450–452. [Google Scholar] [CrossRef]
- Choi, H.J.; Jhon, M.S. Jhon, Electrorheology of Polymers and Nanocomposites. Soft Matter 2009, 5, 1562–1567. [Google Scholar] [CrossRef]
- Choi, H.J.; Park, S.J.; Kim, S.T.; Jhon, M.S. Electrorheological Application of Polyaniline/Multi-walled Carbon Nanotube Composites. Diamond Related Mater. 2005, 14, 766–769. [Google Scholar] [CrossRef]
- Bica, I.; Choi, H.J. Preparation and Electro-thermoconductive Characteristics of Magnetorheological Suspensions. Int. J. Modern Phys. B 2008, 22, 5041–5064. [Google Scholar] [CrossRef]
- Bossis, G.; Khuzir, P.; Lacis, S.; Volkova, O. Yield behavior of magnetorheological suspensions. J. Magn. Magn. Mater. 2003, 258-259, 456–458. [Google Scholar] [CrossRef]
- Cho, M.S.; Lim, S.T.; Jang, I.B.; Choi, H.J.; Jhon, M.S. Encapsulation of spherical iron-particle with PMMA and its magnetorheological particles. IEEE Trans. Magn. 2004, 40, 3036–3038. [Google Scholar] [CrossRef]
- Lim, S.T.; Choi, H.J.; Jhon, M.S. Magnetorheological characterization of carbonyl iron-organoclay suspensions. IEEE Trans. Magn. 2005, 41, 3745–3747. [Google Scholar] [CrossRef]
- Moniruzzaman, M.; Winey, K.I. Polymer nanocomposites containing carbon nanotubes. Macromolecules 2006, 39, 5194–5205. [Google Scholar] [CrossRef]
- Sano, M.; Kamino, A.; Okamura, J.; Shinkai, S. Noncovalent Self-Assembly of Carbon Nanotubes for Construction of “Cages”. Nano Lett. 2002, 2, 531–533. [Google Scholar] [CrossRef]
- Fang, F.F.; Choi, H.J. Non-Covalent Self-Assembly of Carbon Nanotube Wrapped Carbonyl Iron Particles and Their Magnetorheology. J. Appl. Phys. 2008, 103, 07A301-1–07A301-3. [Google Scholar]
- Kittipoomwong, D.; Klingenberg, D.J.; Ulicny, J.C. Dynamic Yield Stress Enhancement in Bidisperse Magnetorheological Fluids. J. Rheol. 2005, 49, 1521–1538. [Google Scholar] [CrossRef]
- Morel, A.L.; Nikitenko, S.I.; Gionnet, K.; Wattiaux, A.; Lai-Kee-Him, J.; Labrugere, C.; Chevalier, B.; Deleris, G.; Petibois, C.; Brisson, A.; Simonoff, M. Sonochemical Approach to the Synthesis of Fe3O4@SiO2 Core/Shell Nanoparticles with Tunable Properties. ACS Nano 2008, 2, 847–856. [Google Scholar] [CrossRef]
- Delozier, D.M.; Orwoll, R.A.; Cahoon, J.F.; Johnston, N.J.; Smith, J.G.; Connel, J.W. Spatial Gradients in Particle Reinforced Polymers Characterized by X-ray Attenuation and Laser Confocal Microscopy. Polymer 2002, 43, 803–812. [Google Scholar] [CrossRef]
- Kim, D.W.; Blumstein, A.; Tripathy, S.K. Nanocomposite Films Derived From Exfoliated Functional Aluminosilicate through Electrostatic Layer-by-Layer Assembly. Chem. Mater. 2001, 13, 1916–1922. [Google Scholar] [CrossRef]
- Lim, S.T.; Choi, H.J.; Jhon, M.S. Dispersion Quality and Rheological Property of Polymer/Clay Nanocomposites: Ultrasonification Effect. J. Ind. Eng. Chem. 2003, 9, 51–57. [Google Scholar]
- Manias, E.; Toung, A.; Wu, L.; Strawhecker, K.; Lu, B.; Chung, T.C. Polypropylene/Montmorillonite Nanocomposites. Review of the Synthetic Routes and Materials Properties. Chem. Mater. 2001, 13, 3516–3523. [Google Scholar] [CrossRef]
- Lim, S.K.; Kim, J.W.; Chin, I.J.; Choi, H.J. Rheological Properties of a New Rubbery Nanocomposite: Polyepichlorohydrin/Organoclay Nanocomposites. J. Appl. Polym. Sci. 2002, 86, 3735–3739. [Google Scholar] [CrossRef]
- Krishnamoorti, R.; Vaia, R.A.; Giannelis, E.P. Structure and Dynamics of Polymer-Layered Silicate Nanocomposites. Chem. Mater. 1996, 8, 1728–1734. [Google Scholar] [CrossRef]
- Ren, J.; Silva, A.S.; Krishnamoorti, R. Linear Viscoelasticity of Disordered Polystyrene−Polyisoprene Block Copolymer Based Layered-Silicate Nanocomposites. Macromolecules 2000, 33, 3739–3746. [Google Scholar] [CrossRef]
- Gu, F.; Li, C.Z.; Wang, S. F. Solution-Chemical Synthesis of Carbon Nanotube/ZnS Nanoparticle Core/Shell Heterostructures. Inorg. Chem. 2007, 46, 5343–5348. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are available from the authors.
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhang, K.; Park, B.-J.; Fang, F.-F.; Choi, H.J. Sonochemical Preparation of Polymer Nanocomposites. Molecules 2009, 14, 2095-2110. https://doi.org/10.3390/molecules140602095
Zhang K, Park B-J, Fang F-F, Choi HJ. Sonochemical Preparation of Polymer Nanocomposites. Molecules. 2009; 14(6):2095-2110. https://doi.org/10.3390/molecules140602095
Chicago/Turabian StyleZhang, Ke, Bong-Jun Park, Fei-Fei Fang, and Hyoung Jin Choi. 2009. "Sonochemical Preparation of Polymer Nanocomposites" Molecules 14, no. 6: 2095-2110. https://doi.org/10.3390/molecules140602095





