The Influence of Initiator Concentration on Selected Properties of Thermosensitive Poly(Acrylamide-co-2-Acrylamido-2-Methyl-1-Propanesulfonic Acid) Microparticles
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis
2.3. Conductivity Measurements
2.4. Attenuated Total Reflection-Fourier Transformed Infrared (ATR-FTIR) Spectroscopy Measurements
2.5. Proton Nuclear Magnetic Resonance (1H NMR) Spectroscopy Measurements
2.6. Hydrodynamic Diameter (HD) and Polydispersity Index (PDI) Measurements
2.7. Zeta Potential (ZP) Measurements
2.8. Thermogravimetric Analysis (TGA)
2.9. Powder X-ray Diffraction (PXRD) Analysis
3. Results
3.1. Synthesis
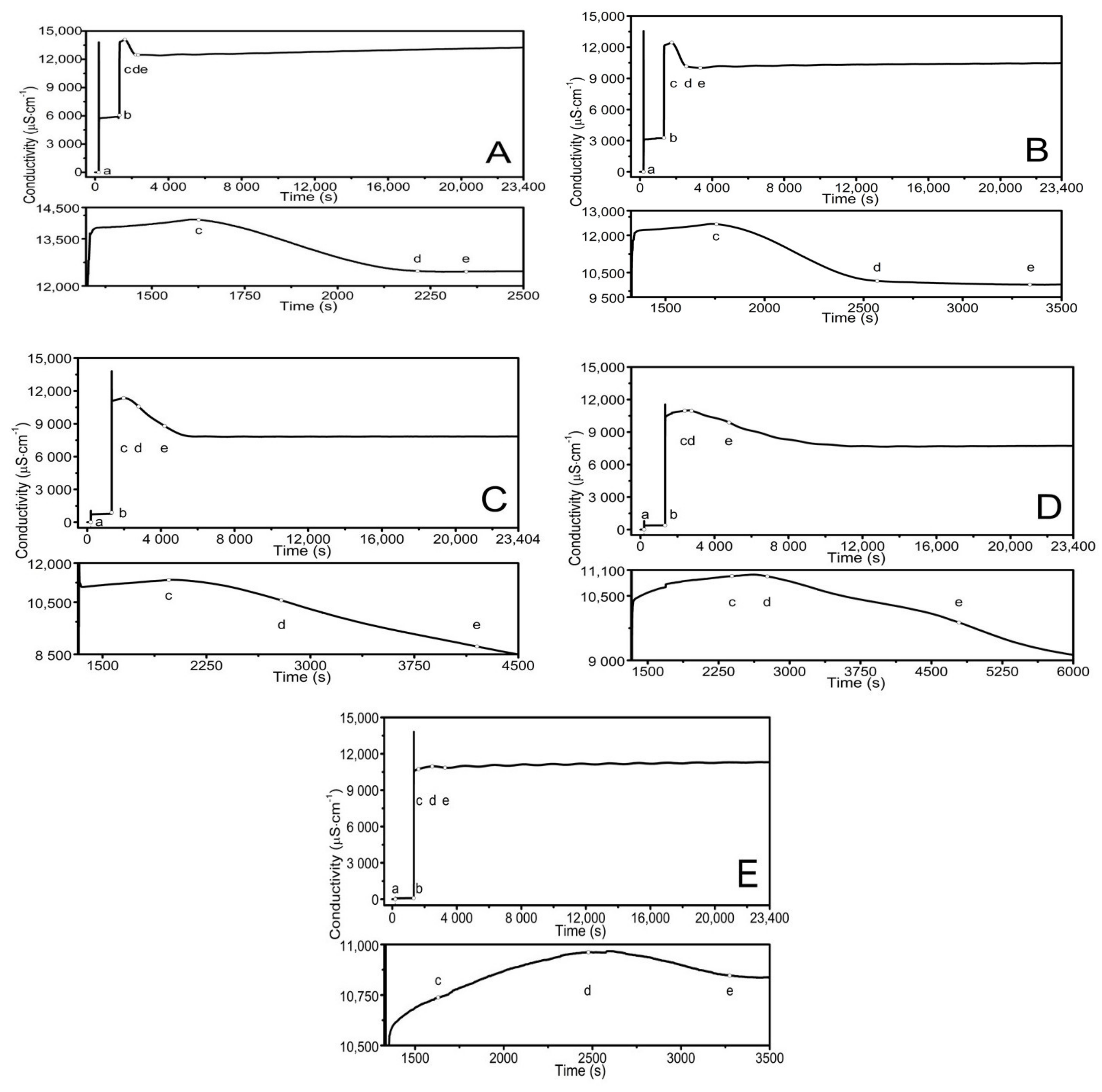
3.2. Conductivity Measurements
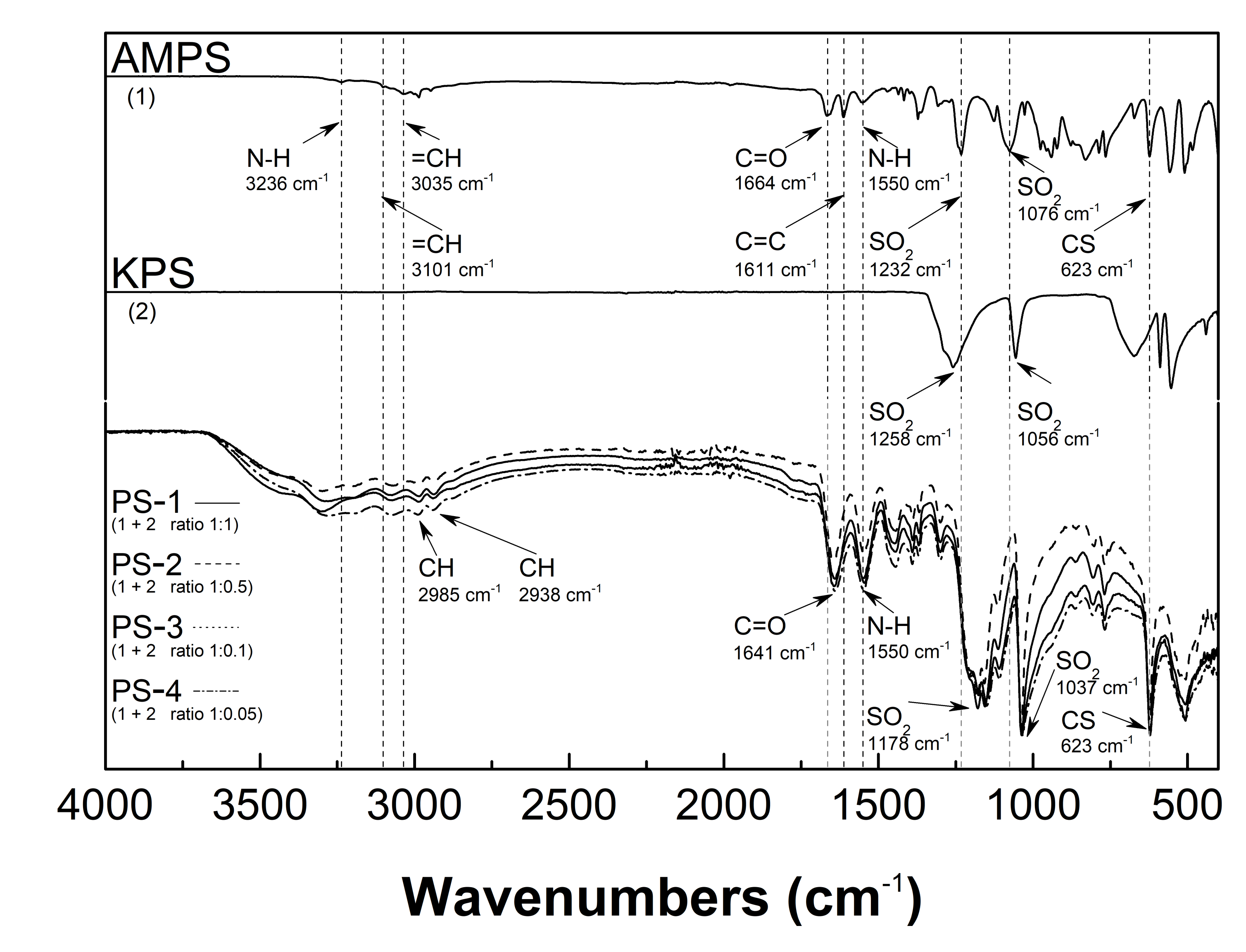
3.3. Attenuated Total Reflection-Fourier Transform Infrared (ATR-FTIR) Spectroscopy Analysis
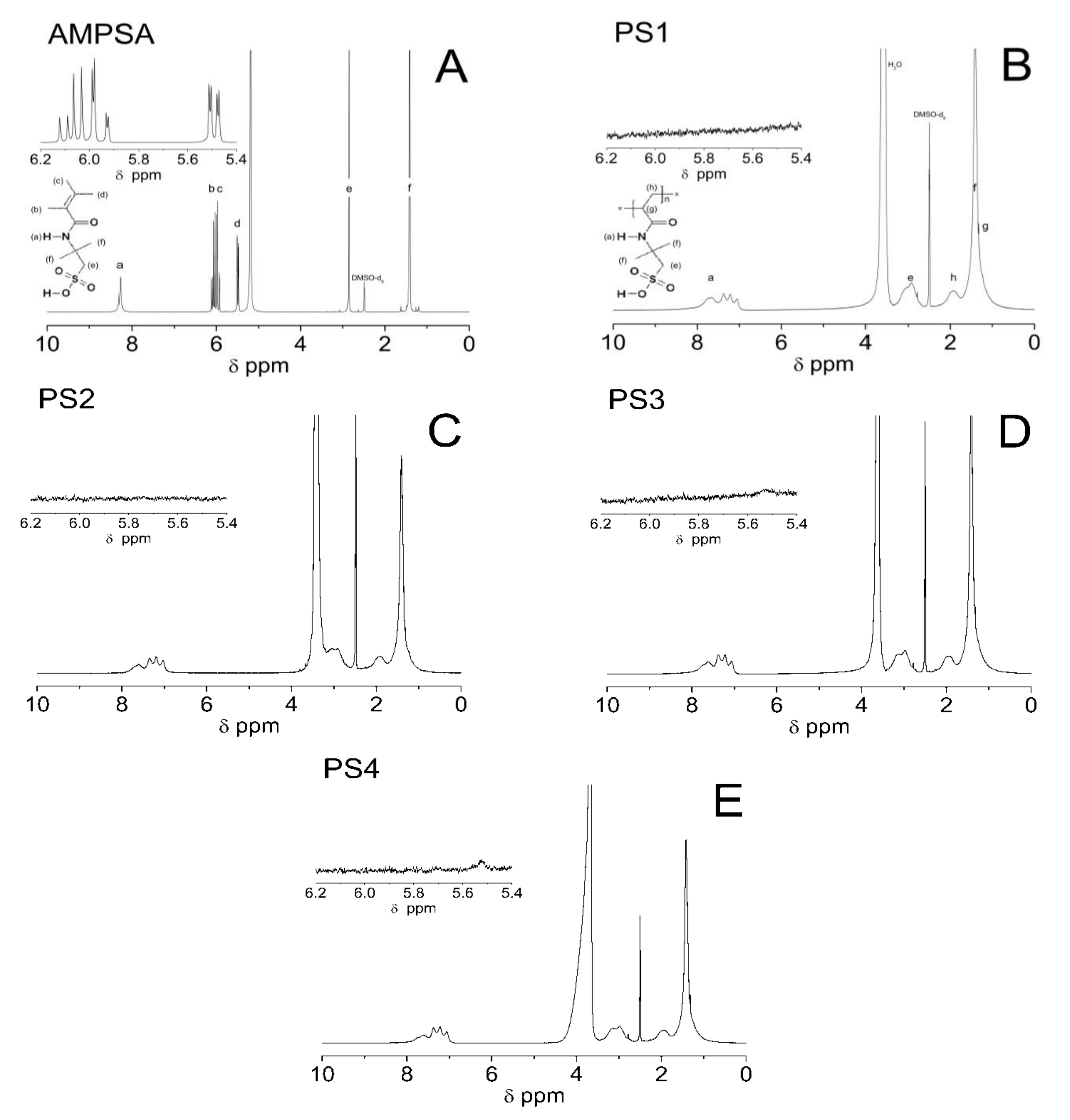
3.4. Proton Nuclear Magnetic Resonance (1H NMR) Spectroscopy Analysis
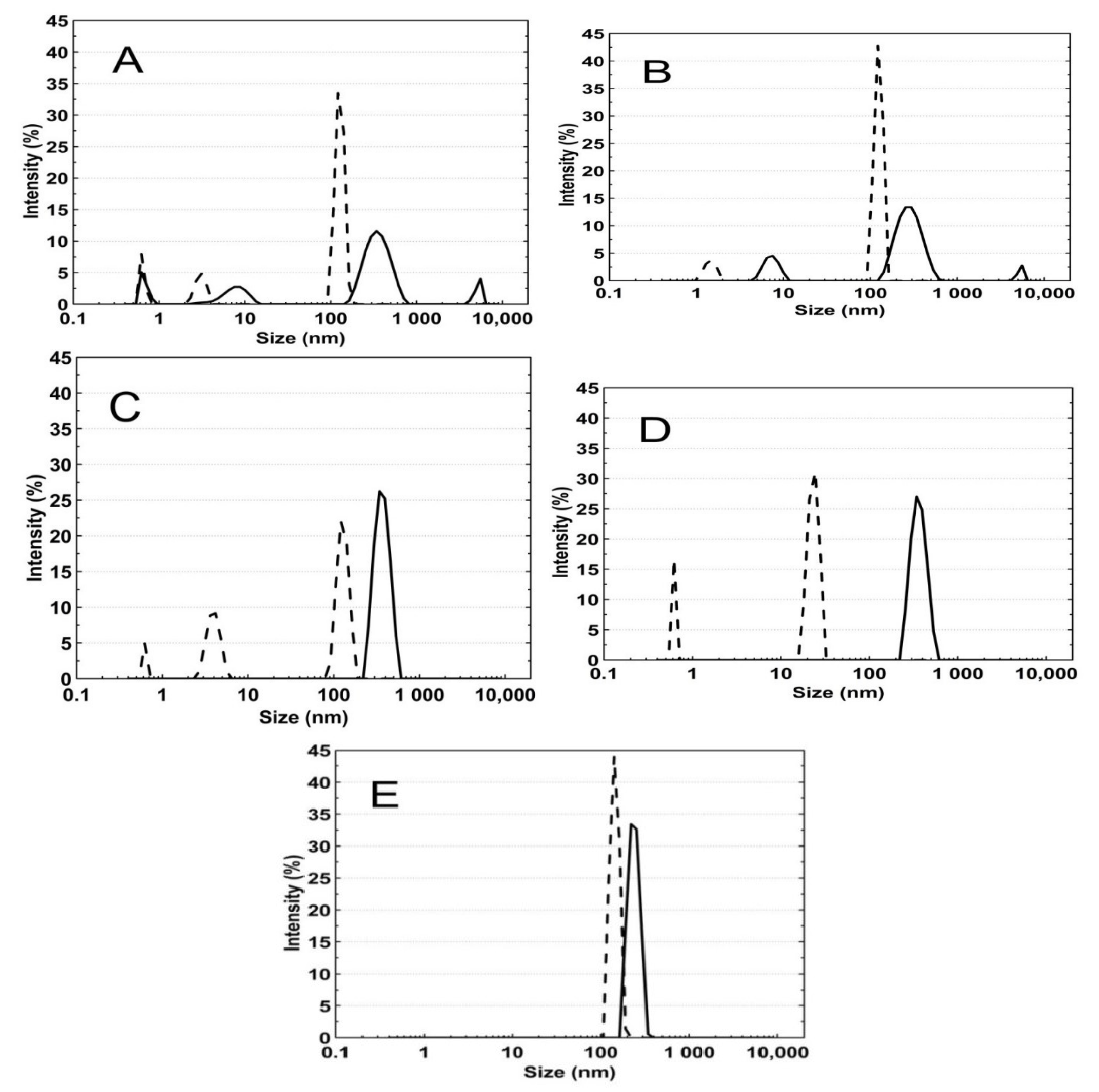
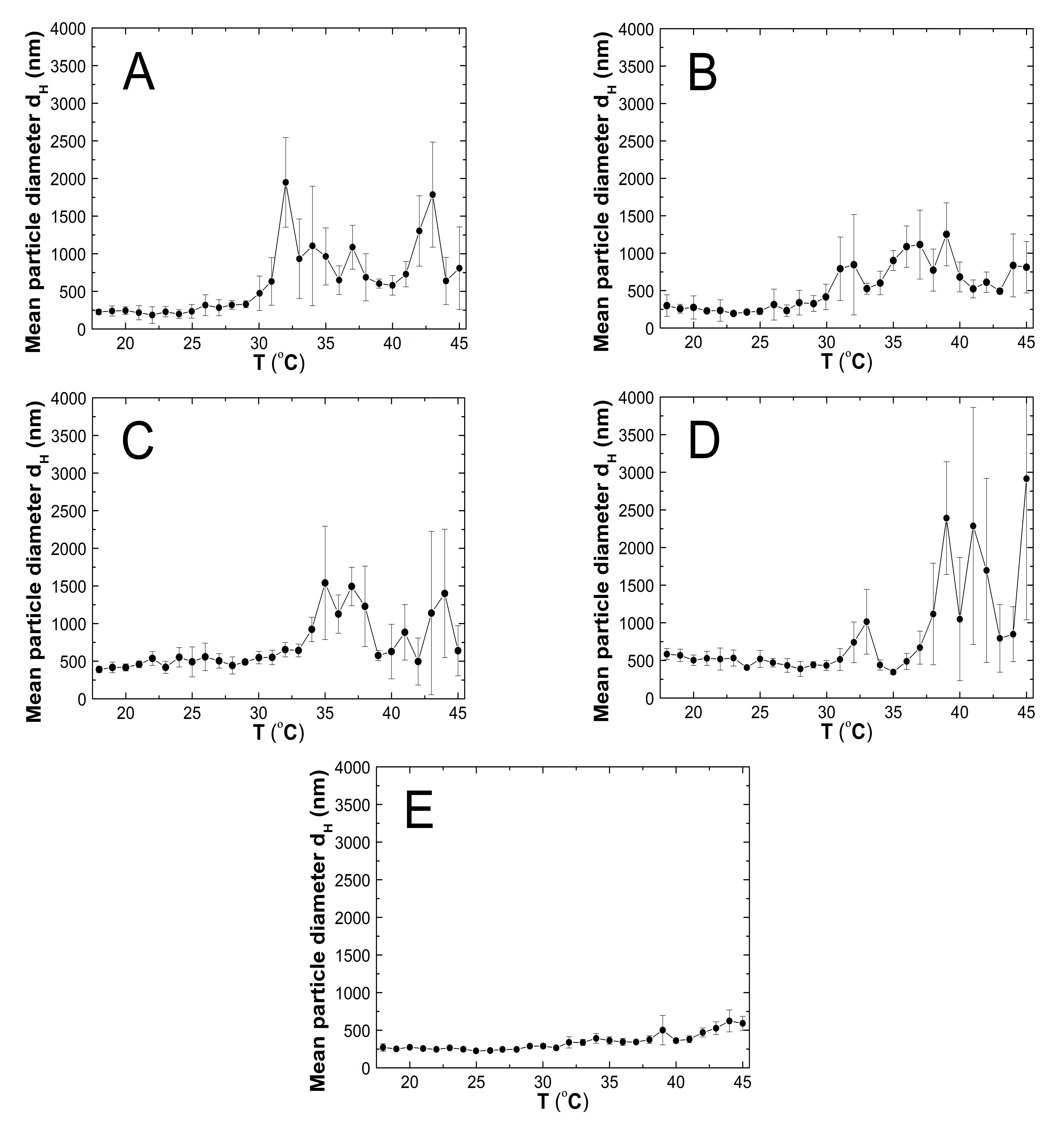
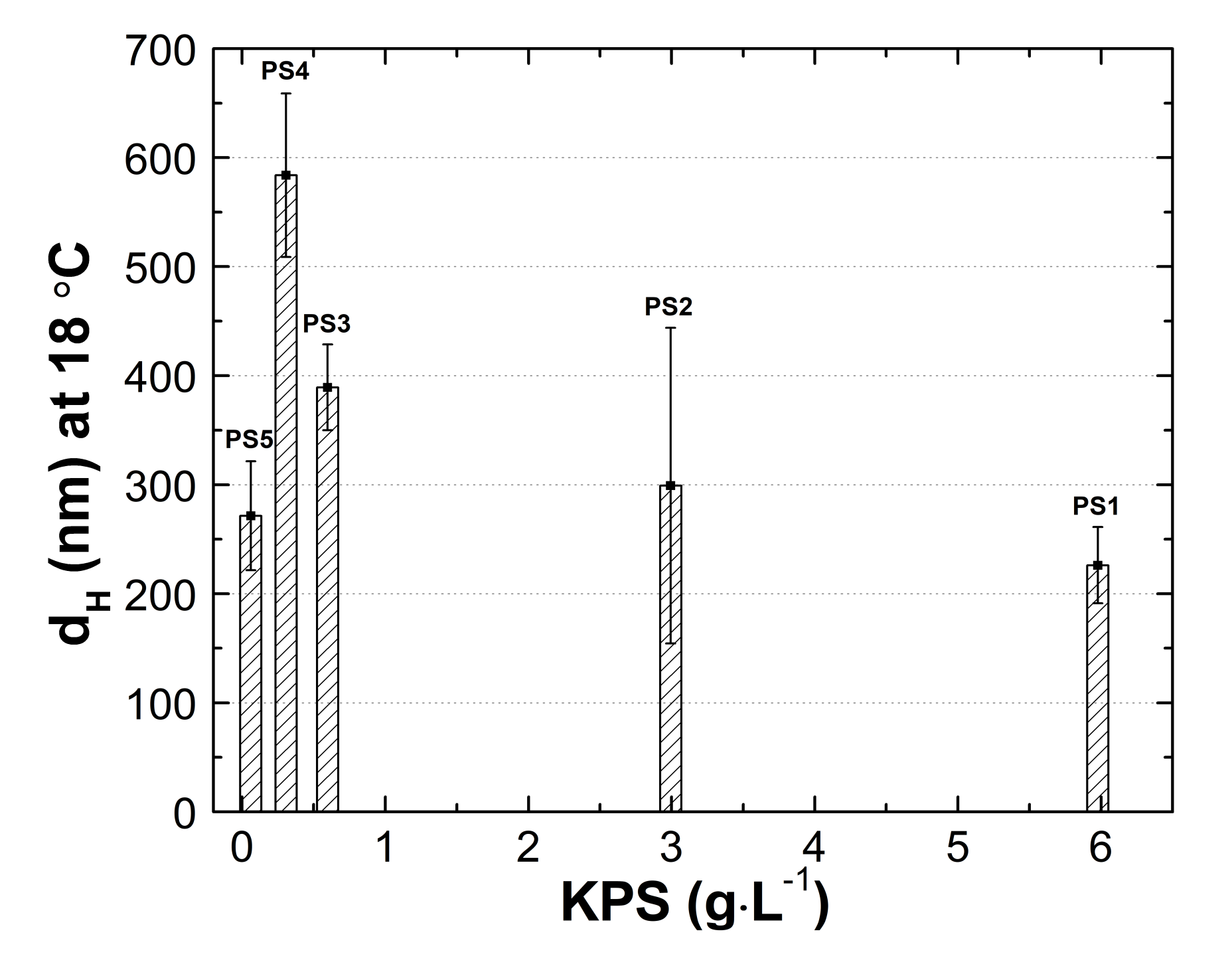
3.5. Hydrodynamic Diameter (HD)
3.5.1. Thermo-Responsivity
3.5.2. The pH-Responsivity
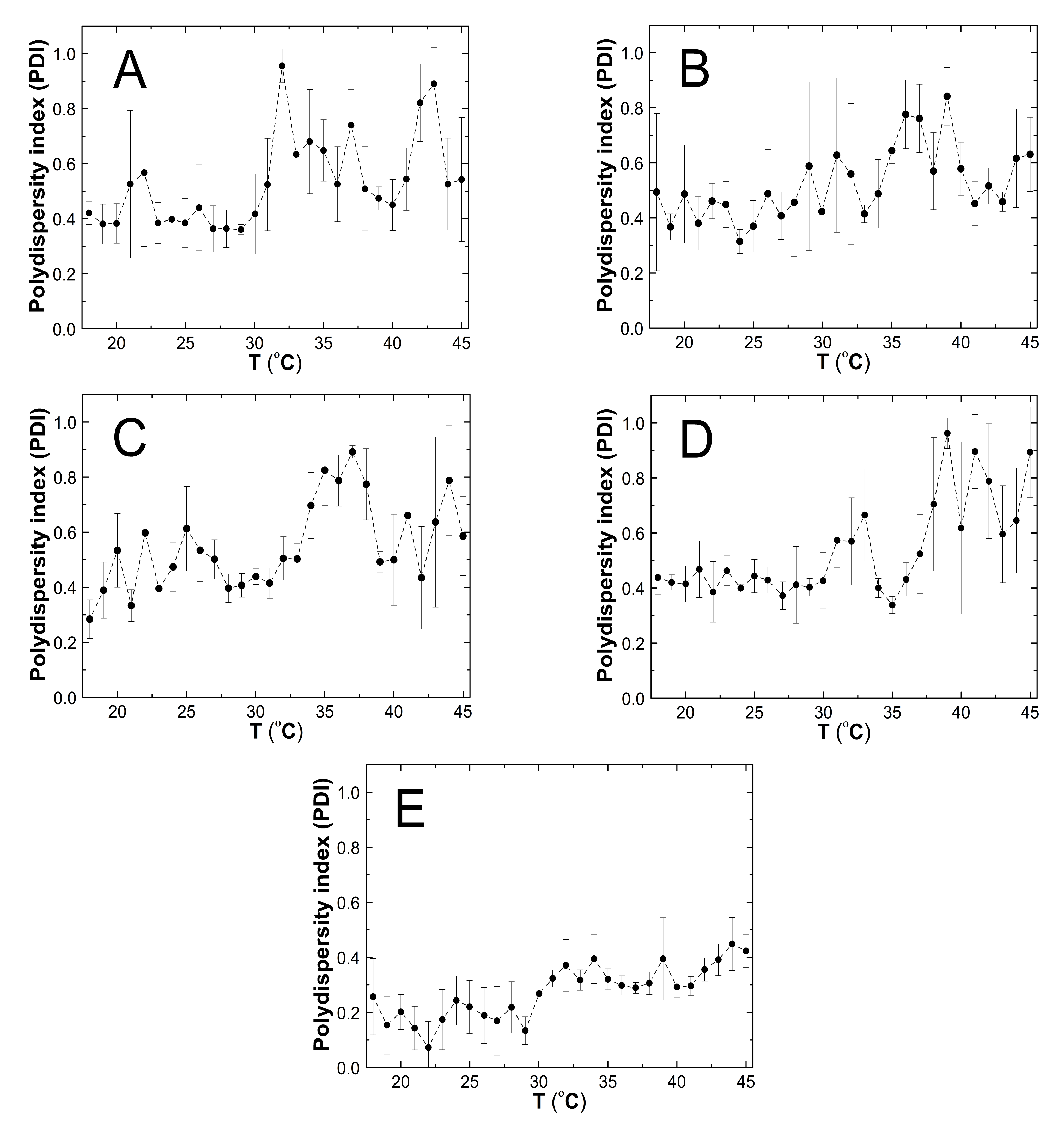
3.6. Polydispersity Index (PDI)
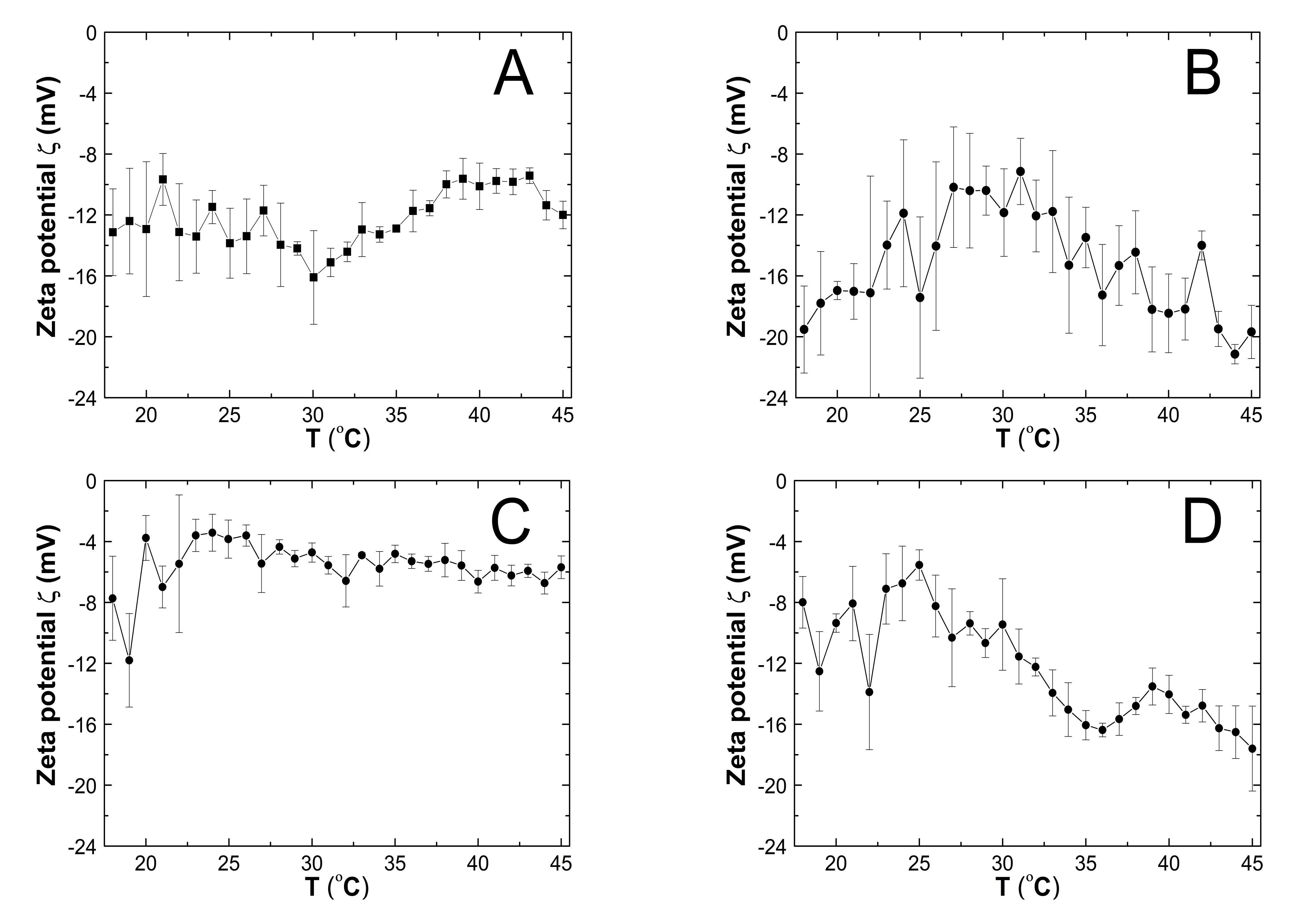
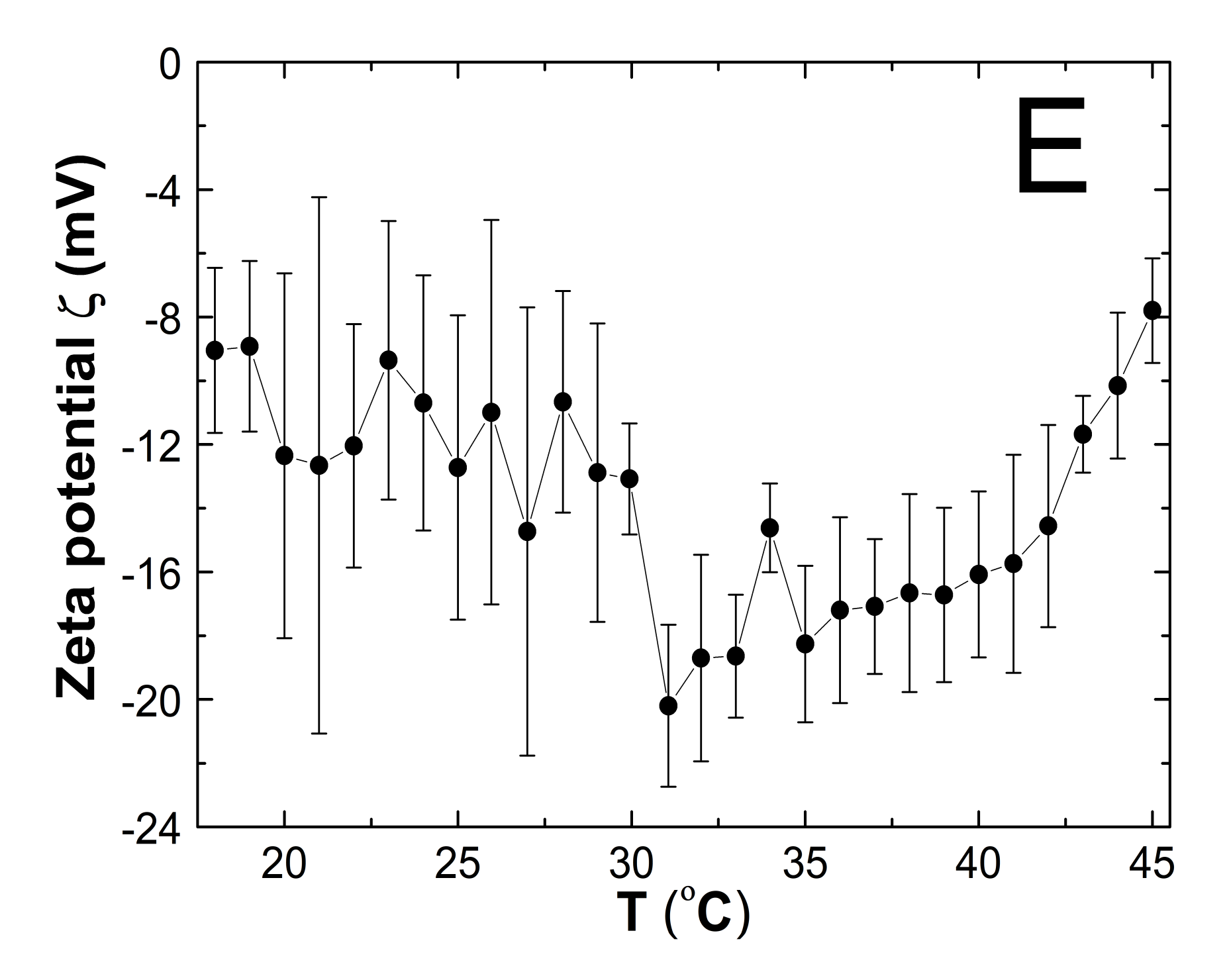
3.7. Zeta Potential (ZP)
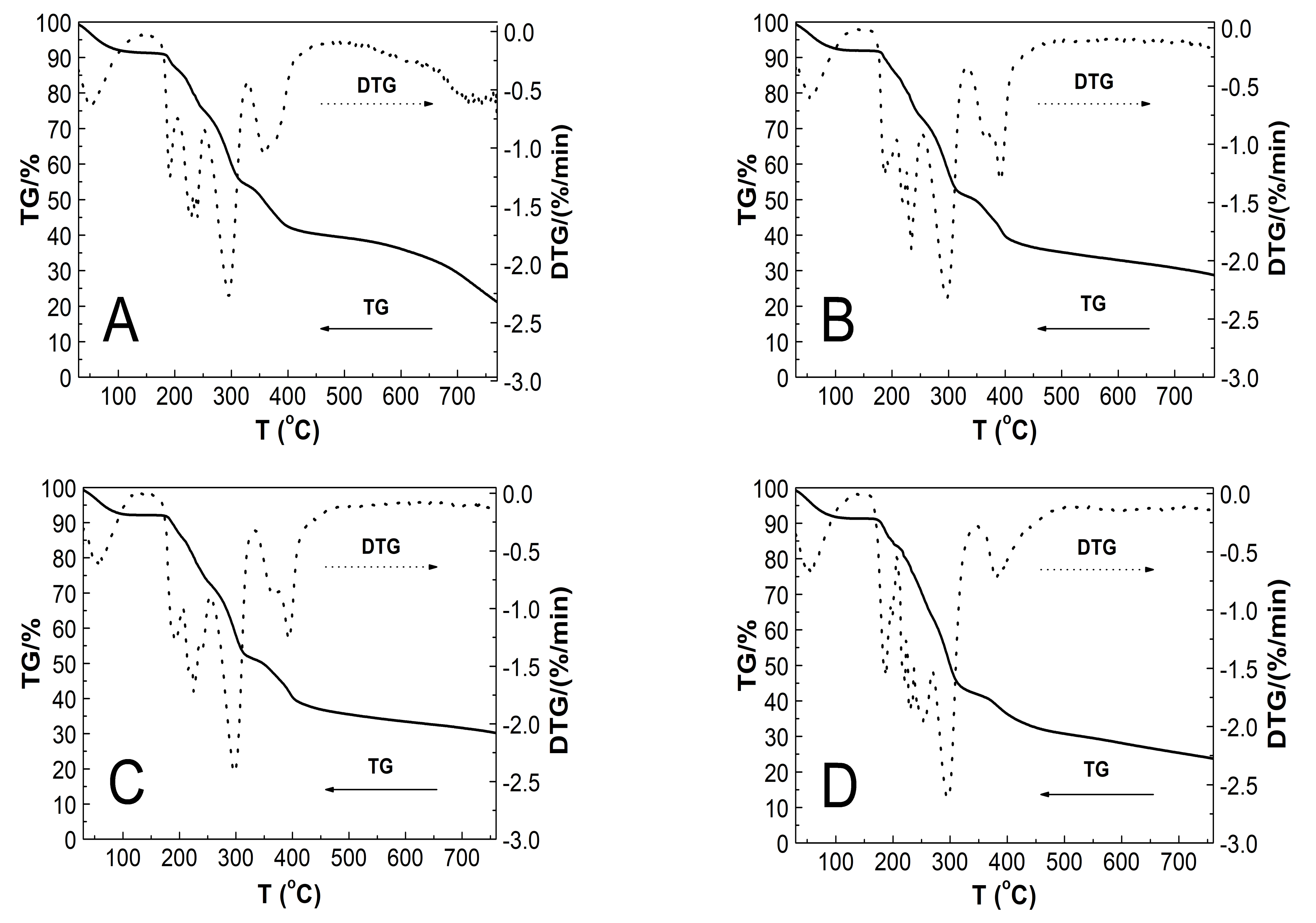
3.8. Thermogravimetric Analysis (TGA)
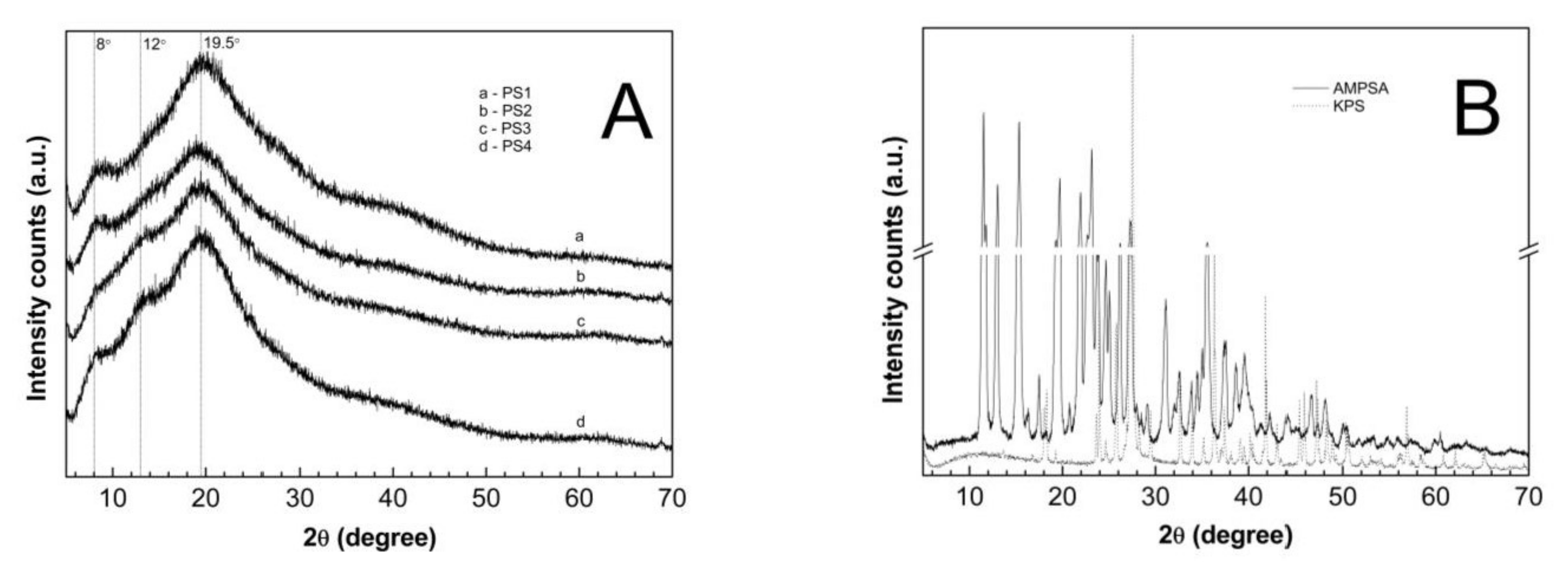
3.9. Powder X-ray Diffraction (PXRD) Analysis
4. Discussion
4.1. Synthesis
4.2. Conductivity
4.3. ATR-FTIR
4.4. 1H NMR
4.5. HD
4.5.1. Thermo-Responsivity
4.5.2. pH-Responsivity
4.6. PDI
4.7. ZP
4.8. TG
4.9. PXRD
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Srivastava, A.; Yadav, T.; Sharma, S.; Nayak, A.; Kumari, A.A.; Mishra, N. Polymers in Drug Delivery. J. Biosci. Med. 2015, 4, 69–84. [Google Scholar] [CrossRef] [Green Version]
- Oh, J.K.; Lee, D.I.; Park, J.M. Biopolymer-based microgels/nanogels for drug delivery applications. Prog. Polym. Sci. 2009, 34, 1261–1282. [Google Scholar] [CrossRef]
- Neamtu, I.; Rusu, A.G.; Diaconu, A.; Nita, L.E.; Chiriac, A.P. Basic concepts and recent advances in nanogels as carriers for medical applications. Drug Deliv. 2017, 24, 539–557. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Molavi, F.; Barzegar-Jalali, M.; Hamishehkar, H. Polyester based polymeric nano and microparticles for pharmaceutical purposes: A review on formulation approaches. J. Control. Release 2020, 320, 265–282. [Google Scholar] [CrossRef] [PubMed]
- Castanheira, E.J.; Correia, T.R.; Rodrigues, J.M.M.; Mano, J.F. Novel Biodegradable Laminarin Microparticles for Biomedical Applications. Bull. Chem. Soc. Jpn. 2020, 93, 713–719. [Google Scholar] [CrossRef]
- Galaev, I. ’Smart’ polymers and what they could do in biotechnology and medicine. Trends Biotechnol. 1999, 17, 335–340. [Google Scholar] [CrossRef]
- Galaev, I.; Mattiasson, B. Smart Polymers for Bioseparation and Bioprocessing; CRC Press: New York, NY, USA, 2001. [Google Scholar]
- Iurciuc-Tincu, C.-E.; Cretan, M.S.; Purcar, V.; Popa, M.; Daraba, O.M.; Atanase, L.I.; Ochiuz, L. Drug Delivery System Based on pH-Sensitive Biocompatible Poly(2-vinyl pyridine)-b-poly(ethylene oxide) Nanomicelles Loaded with Curcumin and 5-Fluorouracil. Polymers 2020, 12, 1450. [Google Scholar] [CrossRef]
- Winninger, J.; Iurea, D.M.; Atanase, L.I.; Salhi, S.; Delaite, C.; Riess, G. Micellization of novel biocompatible thermo-sensitive graft copolymers based on poly(ε-caprolactone), poly(N-vinylcaprolactam) and poly(N-vinylpyrrolidone). Eur. Polym. J. 2019, 119, 74–82. [Google Scholar] [CrossRef]
- Atanase, L.I.; Riess, G. Micellization of pH-stimulable poly(2-vinylpyridine)-b-poly(ethylene oxide) copolymers and their complexation with anionic surfactants. J. Colloid Interface Sci. 2013, 395, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Gao, G.H.; Lee, J.W.; Nguyen, M.K.; Im, G.H.; Yang, J.; Heo, H.; Jeon, P.; Park, T.G.; Lee, J.H.; Lee, D.S. pH-responsive polymeric micelle based on PEG-poly(β-amino ester)/(amido amine) as intelligent vehicle for magnetic resonance imaging in detection of cerebral ischemic area. J. Control. Release 2011, 155, 11–17. [Google Scholar] [CrossRef]
- Huh, K.M.; Kang, H.C.; Lee, Y.J.; Bae, Y.H. pH-sensitive polymers for drug delivery. Macromol. Res. 2012, 20, 224–233. [Google Scholar] [CrossRef]
- Zhang, J.; Shen, B.; Chen, L.; Chen, L.; Meng, Y.; Feng, J. A dual-sensitive mesoporous silica nanoparticle based drug carrier for cancer synergetic therapy. Colloids Surf. B Biointerfaces 2019, 175, 65–72. [Google Scholar] [CrossRef]
- Ghaz-Jahanian, M.A.; Abbaspour-Aghdam, F.; Anarjan, N.; Berenjian, A.; Jafarizadeh-Malmiri, H. Application of Chitosan-Based Nanocarriers in Tumor-Targeted Drug Delivery. Mol. Biotechnol. 2015, 57, 201–218. [Google Scholar] [CrossRef]
- Kundakci, S.; Üzüm, Ö.B.; Karadağ, E. Swelling and dye sorption studies of acrylamide/2-acrylamido-2-methyl-1-propanesulfonic acid/bentonite highly swollen composite hydrogels. React. Funct. Polym. 2008, 68, 458–473. [Google Scholar] [CrossRef]
- Amjad, Z. Influence of Polymer Architecture on the Stabilization of Iron and Manganese Ions in Aqueous Systems. Tenside Surfactants Deterg. 2007, 44, 202–208. [Google Scholar] [CrossRef] [Green Version]
- Nalampang, K.; Suebsanit, N.; Witthayaprapakorn, C.; Molloy, R. Design and preparation of AMPS-based hydrogels for biomedical use as wound dressings. Chiang Mai J. Sci. 2007, 34, 183–189. [Google Scholar] [CrossRef]
- Wang, W.; Li, A.; Zhang, J.; Wang, A. Study on superabsorbent composite. XI. Effect of thermal treatment and acid activation of attapulgite on water absorbency of poly(acrylic acid)/attapulgite superabsorbent composite. Polym. Compos. 2007, 28, 397–404. [Google Scholar] [CrossRef]
- Stile, R.A.; Burghardt, W.R.; Healy, K.E. Synthesis and Characterization of Injectable Poly(N-isopropylacrylamide)-Based Hydrogels That Support Tissue Formation in Vitro. Macromolecules 1999, 32, 7370–7379. [Google Scholar] [CrossRef]
- International Standard. ISO 13321:1996. Methods for Determination of Particle Size Distribution; Photon Correlation Spectroscopy; International Organization for Standardization (ISO): Geneva, Switzerland, 1997; pp. 3406–3408. [Google Scholar]
- International Standard. ISO22412 Particle Size Analysis–Dynamic Light Scattering; International Organisation for Standardisation (ISO): Geneva, Switzerland, 2008. [Google Scholar]
- Worldwide, M.I. Dynamic Light Scattering, Common Terms Defined; Inform White Paper; Malvern Instruments Limited: Malvern, UK, 2011; pp. 1–6. [Google Scholar]
- Van Berkel, K.Y.; Russell, G.T.; Gilbert, R.G. The dissociation rate coefficient of persulfate in emulsion polymerization systems. Polymer 2006, 47, 4667–4675. [Google Scholar] [CrossRef]
- Beylerian, N.M.; Vardanyan, L.R.; Harutyunyan, R.S.; Vardanyan, R.L. Kinetics and Mechanism of Potassium Persulfate Decomposition in Aqueous Solutions Studied by a Gasometric Method. Macromol. Chem. Phys. 2002, 203, 212–218. [Google Scholar] [CrossRef]
- House, D.A. Kinetics and Mechanism of Oxidations by Peroxydisulfate. Chem. Rev. 1962, 62, 185–203. [Google Scholar] [CrossRef]
- Bednarz, S.; Błaszczyk, A.; Błażejewska, D.; Bogdał, D. Free-radical polymerization of itaconic acid in the presence of choline salts: Mechanism of persulfate decomposition. Catal. Today 2015, 257, 297–304. [Google Scholar] [CrossRef]
- Rastogi, A.; Al-Abed, S.R.; Dionysiou, D.D. Sulfate radical-based ferrous–peroxymonosulfate oxidative system for PCBs degradation in aqueous and sediment systems. Appl. Catal. B Environ. 2009, 85, 171–179. [Google Scholar] [CrossRef]
- Okubo, M.; Mori, T. The decomposition of potassium persulphate used as initiator in emulsion polymerization. Makromol. Chemie. Macromol. Symp. 1990, 31, 143–156. [Google Scholar] [CrossRef]
- Bhardwaj, P.; Singh, V.; Aggarwal, S.; Mandal, U.K. Poly(acrylamide-co-2-acrylamido-2-methyl-1-propanesulfonic Acid) Nanogels made by Inverse Microemulsion Polymerization. J. Macromol. Sci. Part A 2009, 46, 1083–1094. [Google Scholar] [CrossRef]
- Demirci, S.; Zekoski, T.; Sahiner, N. The preparation and use of p(2-acrylamido-2-methyl-1-propanesulfonic acid)-tris(dioxa-3,6-heptyl)amine (p(AMPS)-TDA-1) ionic liquid microgel in hydrogen production. Polym. Bull. 2018, 76, 1717–1735. [Google Scholar] [CrossRef]
- Qiao, J.; Hamaya, T.; Okada, T. New highly proton-conducting membrane poly(vinylpyrrolidone)(PVP) modified poly(vinyl alcohol)/2-acrylamido-2-methyl-1-propanesulfonic acid (PVA–PAMPS) for low temperature direct methanol fuel cells (DMFCs). Polymer 2005, 46, 10809–10816. [Google Scholar] [CrossRef]
- Greesh, N.; Hartmann, P.C.; Cloete, V.; Sanderson, R.D. Adsorption of 2-acrylamido-2-methyl-1-propanesulfonic acid (AMPS) and related compounds onto montmorillonite clay. J. Colloid Interface Sci. 2008, 319, 2–11. [Google Scholar] [CrossRef]
- Çavuş, S.; Yaşar, G.; Kaya, Y.; Gönder, Z.B.; Gürdağ, G.; Vergili, I. Synthesis and characterization of gel beads based on ethyleneglycol dimethacrylate and 2-acrylamido-2-methyl-1-propane sulfonic acid: Removal of Fe(II), Cu(II), Zn(II), and Ni(II) from metal finishing wastewater. Process. Saf. Environ. Prot. 2016, 103, 227–236. [Google Scholar] [CrossRef]
- Kabiri, K.; Lashani, S.; Zohuriaan-Mehr, M.J.; Kheirabadi, M. Super alcohol-absorbent gels of sulfonic acid-contained poly(acrylic acid). J. Polym. Res. 2010, 18, 449–458. [Google Scholar] [CrossRef]
- Gola, A.; Sacharczuk, M.; Musiał, W. Synthesis of AMPSA Polymeric Derivatives Monitored by Electrical Conductivity and Evaluation of Thermosensitive Properties of Resulting Microspheres. Molecules 2019, 24, 1164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clara, I.; Lavanya, R.; Natchimuthu, N. pH and temperature responsive hydrogels of poly(2-acrylamido-2-methyl-1-propanesulfonic acid-co-methacrylic acid): Synthesis and swelling characteristics. J. Macromol. Sci. Part A 2016, 53, 492–499. [Google Scholar] [CrossRef]
- Sinirlioglu, D.; Muftuoglu, A.E.; Bozkurt, A. Preparation of Thin Films from New Azolic Copolymers and Investigation of Their Membrane Properties. J. Macromol. Sci. Part A 2014, 51, 420–434. [Google Scholar] [CrossRef]
- Çavuş, S.; Çakal, E. Poly(2-acrylamido-2-methyl-1-propane sulfonic acid-co-1-vinyl-2-pyrrolidone) Hydrogel and its use in the Removal of Cd(II), Pb(II) and Cu(II). Acta Phys. Pol. A 2017, 132, 505–508. [Google Scholar] [CrossRef]
- Abdelrahman, E.A.; Abdel-Salam, E.T.; El Rayes, S.M.; Mohamed, N.S. Facile synthesis of graft copolymers of maltodextrin and chitosan with 2-acrylamido-2-methyl-1-propanesulfonic acid for efficient removal of Ni(II), Fe(III), and Cd(II) ions from aqueous media. J. Polym. Res. 2019, 26, 251. [Google Scholar] [CrossRef]
- Li, H.Q.; Liu, X.J.; Wang, H.; Yang, H.; Wang, Z.; He, J. Proton exchange membranes with cross-linked interpenetrating network of sulfonated polyvinyl alcohol and poly(2-acrylamido-2-methyl-1-propanesulfonic acid): Excellent relative selectivity. J. Membr. Sci. 2020, 595, 117511. [Google Scholar] [CrossRef]
- Coletta, C.; Cui, Z.; Dazzi, A.; Guigner, J.-M.; Néron, S.; Marignier, J.-L.; Remita, S. A pulsed electron beam synthesis of PEDOT conducting polymers by using sulfate radicals as oxidizing species. Radiat. Phys. Chem. 2016, 126, 21–31. [Google Scholar] [CrossRef]
- Sharifi, M.; Wark, M.; Freude, D.; Haase, J. Highly proton conducting sulfonic acid functionalized mesoporous materials studied by impedance spectroscopy, MAS NMR spectroscopy and MAS PFG NMR diffusometry. Microporous Mesoporous Mater. 2012, 156, 80–89. [Google Scholar] [CrossRef]
- Shestakova, P.; Willem, R.; Vassileva, E. Elucidation of the Chemical and Morphological Structure of Double-Network (DN) Hydrogels by High-Resolution Magic Angle Spinning (HRMAS) NMR Spectroscopy. Chem. A Eur. J. 2011, 17, 14867–14877. [Google Scholar] [CrossRef]
- Kolthoff, I.M.; Miller, I.K. The Chemistry of Persulfate. I. The Kinetics and Mechanism of the Decomposition of the Persulfate Ion in Aqueous Medium1. J. Am. Chem. Soc. 1951, 73, 3055–3059. [Google Scholar] [CrossRef]
- Gola, A.; Knysak, T.; Musial, W. The Influence of Anionic Initiator on the Selected Properties of Poly-N-Isopropyl Acrylamide Evaluated for Controlled Drug Delivery. Molecules 2016, 22, 23. [Google Scholar] [CrossRef] [Green Version]
- Gola, A.; Niżniowska, A.; Musiał, W. The Influence of Initiator Concentration on Selected Properties on Poly-N-Vinylcaprolactam Nanoparticles. Nanomaterials 2019, 9, 1577. [Google Scholar] [CrossRef] [Green Version]
- Dixon, K.W. Decomposition Rates of Organic Free Radical Polymerization in Polymer Handbook, 4th ed.; Wiley & Sons: New York, NY, USA, 1999; p. II/1. [Google Scholar]
- Siegrist, R.L.; Crimi, M.; Simpkin, T.J. In Situ Chemical Oxidation for Groundwater Remediation, 1st ed.; Springer: New York, NY, USA, 2011. [Google Scholar]
- Peppas, N. Hydrogels in pharmaceutical formulations. Eur. J. Pharm. Biopharm. 2000, 50, 27–46. [Google Scholar] [CrossRef]
- Baumgartner, A. Statics and dynamics of the freely jointed polymer chain with Lennard-Jones interaction. J. Chem. Phys. 1980, 72, 871–879. [Google Scholar] [CrossRef]
- Cieśla, M.; Pawłowicz, J.; Longa, L. Molecular dynamics simulation of the Lennard–Jones polymers in a good solvent. Acta Phys. Pol. B 2007, 38, 1727–1736. [Google Scholar]
- Kratz, K.; Hellweg, T.; Eimer, W. Influence of charge density on the swelling of colloidal poly(N-isopropylacrylamide-co-acrylic acid) microgels. Colloids Surfaces A Physicochem. Eng. Asp. 2000, 170, 137–149. [Google Scholar] [CrossRef]
- Turan, E.; Demirci, S.; Caykara, T. Thermo- and pH-induced phase transitions and network parameters of poly(N-isopropylacrylamide-co-2-acrylamido-2-methyl-propanosulfonic acid) hydrogels. J. Polym. Sci. Part B Polym. Phys. 2008, 46, 1713–1724. [Google Scholar] [CrossRef]
- Gupta, P.; Vermani, K.; Garg, S. Hydrogels: From controlled release to pH-responsive drug delivery. Drug Discov. Today 2002, 7, 569–579. [Google Scholar] [CrossRef]
- Burmistrova, A.; Klitzing, R. Control of number density and swelling/shrinking behavior of P(NIPAM–AAc) particles at solid surfaces. J. Mater. Chem. 2010, 20, 3502–3507. [Google Scholar] [CrossRef]
- FDA. Liposome Drug Products; Chemistry, Manufacturing, and Controls; Human Pharmacokinetics and Bioavailability; Labeling Documentation. In Guidance for Industry; April 2018 Pharmaceutical Quality/CMC; U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER): Silver Spring, MD, USA, 2018. [Google Scholar]
- Greenwood, R.; Kendall, K. Selection of Suitable Dispersants for Aqueous Suspensions of Zirconia and Titania Powders using Acoustophoresis. J. Eur. Ceram. Soc. 1999, 19, 479–488. [Google Scholar] [CrossRef]
- Hanaor, D.; Michelazzi, M.; Leonelli, C.; Sorrell, C.C. The effects of carboxylic acids on the aqueous dispersion and electrophoretic deposition of ZrO2. J. Eur. Ceram. Soc. 2012, 32, 235–244. [Google Scholar] [CrossRef] [Green Version]
- Tauer, K.; Deckwer, R.; Kühn, I.; Schellenberg, C. A comprehensive experimental study of surfactant-free emulsion polymerization of styrene. Colloid Polym. Sci. 1999, 277, 607–626. [Google Scholar] [CrossRef]
- Pelton, R.H.; Pelton, H.M.; Morphesis, A.; Rowell, R.L. Particle sizes and electrophoretic mobilities of poly(N-isopropylacrylamide) latex. Langmuir 1989, 5, 816–818. [Google Scholar] [CrossRef]





| Type of Polymer Microparticle System | Monomer (g) | Anionic Initiator (g) | AMPSA to KPS Radical Molar Ratio |
|---|---|---|---|
| AMPSA 1 | KPS 2 | ||
| PS1 | 9.1520 | 5.9771 | 1:1 |
| PS2 | 9.1651 | 2.9937 | 1:0.5 |
| PS3 | 9.1570 | 5.974 × 10−1 | 1:0.1 |
| PS4 | 9.1592 | 3.076 × 10−1 | 1:0.05 |
| PS5 | 9.1563 | 6.081 × 10−2 | 1:0.01 |
| Type of Polymer Microparticle System | HD (nm) | ||||
|---|---|---|---|---|---|
| pH 3.0 | pH 4.0 | pH 5.0 | pH 6.0 | pH 7.0 | |
| Citric Acid/ Sodium Hydroxide/ Hydrogen Chloride | Potassium Hydrogen Phthalate | Citric Acid/ Sodium Hydroxide | Citric Acid/ Sodium Hydroxide | Potassium Dihydrogen Phosphate/ Disodium Hydrogen Phosphate | |
| PS1 | 229 ± 26 | 526 ± 78 | 789 ± 42 | 661 ± 78 | 883 ± 41 |
| PS2 | 280 ± 12 | 226 ± 15 | 491 ± 30 | 681 ± 69 | 1030 ± 61 |
| PS3 | 271 ± 30 | 394 ± 35 | 253 ± 50 | 811 ± 64 | 819.0 ± 53 |
| PS4 | 565 ± 42 | 387 ± 50 | 437 ± 42 | 763 ± 52 | 894 ± 64 |
| PS5 | 599 ± 53 | 435 ± 72 | 583 ± 115 | 1250 ± 141 | 743 ± 87 |
| Type of Polymer Microparticle System | T1 (°C) | Rate of Mass Loss 1 (% min−1) | T2 (°C) | Rate of Mass Loss 2 (% min−1) | T3 (°C) | Rate of Mass Loss 3 (% min−1) | TOnset (°C) | TEndset (°C) | Res (%) | T1.0wt% (°C) |
|---|---|---|---|---|---|---|---|---|---|---|
| PS1 | 50.7 | 0.64 | 293.6 | 2.27 | 357.1 | 1.06 | 185.1 | 396.2 | 22.30 | 33.4 |
| PS2 | 54.2 | 0.60 | 295.6 | 2.33 | 392.7 | 1.32 | 180.6 | 403.3 | 29.08 | 33.8 |
| PS3 | 56.8 | 0.63 | 296.4 | 2.43 | 393.3 | 1.27 | 180.6 | 404.2 | 30.20 | 33.2 |
| PS4 | 55.3 | 0.68 | 297.1 | 2.62 | 381.7 | 0.72 | 176.4 | 405.0 | 23.76 | 32.8 |
| Sample | Peak 1 2θ (°) | I1 (Arbitrary Units) | Peak 2 2θ (°) | I2 (Arbitrary Units) | Peak 3 2θ (°) | I3 (Arbitrary Units) | Peak 4 2θ (°) | I4 (Arbitrary Units) |
|---|---|---|---|---|---|---|---|---|
| PS1 | 8.19 | 906 | - | - | 19.68 | 1627 | - | - |
| PS2 | 8.21 | 745 | - | - | 19.18 | 1221 | - | - |
| PS3 | 7.95 | 528 | 12.93 | 918 | 19.44 | 1281 | - | - |
| PS4 | 8.02 | 816 | 12.94 | 1190 | 19.57 | 1617 | - | - |
| AMPSA | - | - | 11.52 | 22,674 | 15.34 | 21,291 | 23.14 | 17,315 |
| KPS | - | - | - | - | - | - | 27.53 | 34,071 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gola, A.; Bernardi, A.; Pasut, G.; Musiał, W. The Influence of Initiator Concentration on Selected Properties of Thermosensitive Poly(Acrylamide-co-2-Acrylamido-2-Methyl-1-Propanesulfonic Acid) Microparticles. Polymers 2021, 13, 996. https://doi.org/10.3390/polym13070996
Gola A, Bernardi A, Pasut G, Musiał W. The Influence of Initiator Concentration on Selected Properties of Thermosensitive Poly(Acrylamide-co-2-Acrylamido-2-Methyl-1-Propanesulfonic Acid) Microparticles. Polymers. 2021; 13(7):996. https://doi.org/10.3390/polym13070996
Chicago/Turabian StyleGola, Agnieszka, Andrea Bernardi, Gianfranco Pasut, and Witold Musiał. 2021. "The Influence of Initiator Concentration on Selected Properties of Thermosensitive Poly(Acrylamide-co-2-Acrylamido-2-Methyl-1-Propanesulfonic Acid) Microparticles" Polymers 13, no. 7: 996. https://doi.org/10.3390/polym13070996







