Neuroprotection by Radical Avoidance: Search for Suitable Agents
Abstract
Introduction
The Multiple Levels of Enhanced Radical Formation and Radical Avoidance
Nitrones

Leptin
Thiazolidinediones

Glutamatergic Modulators

Resveratrol and Other Sirtuin Activators

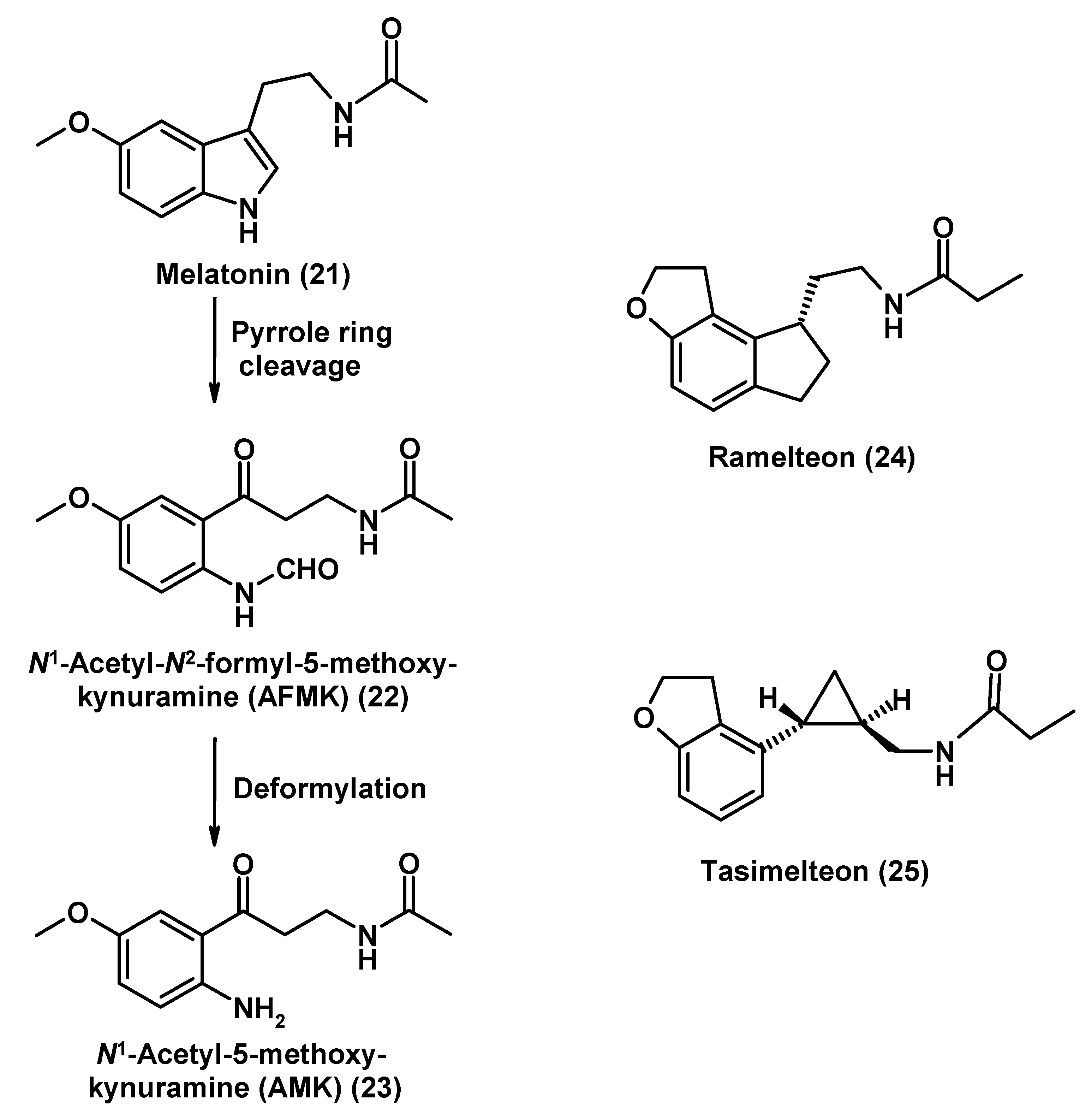
Melatonin, Its Analogs and Metabolites
Conclusions
- Samples Availability: Not available.
References and Notes
- Grundman, M.; Grundman, M.; Delaney, P. Antioxidant strategies for Alzheimer's disease. Proc. Nutr. Soc. 2002, 61, 191–202. [Google Scholar] [CrossRef]
- Frank, B.; Gupta, S. A review of antioxidants and Alzheimer's disease. Ann. Clin. Psychiatry 2005, 17, 269–286. [Google Scholar] [CrossRef]
- Boothby, L.A.; Doering, P.L. Vitamin C and vitamin E for Alzheimer's disease. Ann. Pharmacother. 2005, 39, 2073–2080. [Google Scholar] [CrossRef]
- Bhat, V.; Weiner, W.J. Parkinson's disease. Diagnosis and the initiation of therapy. Minerva Med. 2005, 96, 145–154. [Google Scholar]
- Isaac, M.G.E.K.N.; Quinn, R.; Tabet, N. Vitamin E for Alzheimer's disease and mild cognitive impairment. Cochrane Database Syst. Rev. 2008, 3, CD002854. [Google Scholar]
- Poeggeler, B.; Reiter, R.J.; Tan, D.X.; Chen, L.D.; Manchester, L.C. Melatonin, hydroxyl radical-mediated oxidative damage, and aging: a hypothesis. J. Pineal Res. 1993, 14, 151–168. [Google Scholar] [CrossRef]
- Poeggeler, B. Melatonin, aging, and age-related diseases: Perspectives for prevention, intervention, and therapy. Endocrine 2005, 27, 201–212. [Google Scholar] [CrossRef]
- Dröge, W.; Schipper, H.M. Oxidative stress and aberrant signaling in aging and cognitive decline. Aging Cell 2007, 6, 361–370. [Google Scholar] [CrossRef]
- Figueiredo, P.A.; Mota, M.P.; Appell, H.J.; Duarte, J.A. The role of mitochondria in aging of skeletal muscle. Biogerontology 2008, 9, 67–84. [Google Scholar] [CrossRef]
- Rattan, S.I. Increased molecular damage and heterogeneity as the basis of aging. Biol. Chem. 2008, 389, 267–272. [Google Scholar]
- Wei, Y.H.; Wu, S.B.; Ma, Y.S.; Lee, H.C. Respiratory function decline and DNA mutation in mitochondria, oxidative stress and altered gene expression during aging. Chang Gung Med. J. 2009, 32, 113–132. [Google Scholar]
- Calabrese, V.; Cornelius, C.; Mancuso, C.; Pennisi, G.; Calafato, S.; Bellia, F.; Bates, T.E.; Giuffrida Stella, A.M.; Schapira, T.; Dinkova Kostova, A.T.; Rizzarelli, E. Cellular stress response: A novel target for chemoprevention and nutritional neuroprotection in aging, neurodegenerative disorders and longevity. Neurochem. Res. 2008, 33, 2444–2471. [Google Scholar]
- Chigurupati, S.; Wie, Z.; Belal, C.; Vandermey, M.; Kyriazis, G.A.; Arumugam, T.V.; Chan, S.L. The homocysteine-inducible endoplasmic reticulum stress protein counteracts calcium store depletion and induction of CCAAT enhancer-binding protein homologous protein in a neurotoxin model of Parkinson disease. J. Biol. Chem. 2009, 284, 18323–18333. [Google Scholar]
- Hardeland, R. Antioxidative protection by melatonin: Multiplicity of mechanisms from radical detoxification to radical avoidance. Endocrine 2005, 27, 119–130. [Google Scholar] [CrossRef]
- Halliwell, B.B.; Gutteridge, J.M.C. Free Radicals in Biology and Medicine, 4th ed; Oxford University Press: Oxford, UK, 2007. [Google Scholar]
- Kanki, T.; Nakayama, H.; Sasaki, N.; Takio, K.; Alam, T.I.; Hamasaki, N.; Kang, D. Mitochondrial nucleoid and transcription factor A. Ann. N.Y. Acad. Sci. 2004, 1011, 61–68. [Google Scholar]
- Ekstrand, M.I.; Falkenberg, M.; Rantanen, A.; Park, C.B.; Gaspari, M.; Hultenby, K.; Rustin, P.; Gustafsson, C.M.; Larsson, N.G. Mitochondrial transcription factor A regulates mtDNA copy number in mammals. Hum. Mol. Genet. 2004, 13, 935–944. [Google Scholar] [CrossRef]
- Kang, D.; Hamasaki, N. Mitochondrial transcription factor A in the maintenance of mitochondrial DNA: Overview of its multiple roles. Ann. N.Y. Acad. Sci. 2005, 1042, 101–108. [Google Scholar]
- Kienhöfer, J.; Häussler, D.J.; Ruckelshausen, F.; Muessig, E.; Weber, K.; Pimentel, D.; Ullrich, V.; Bürkle, A.; Bachschmid, M.M. Association of mitochondrial antioxidant enzymes with mitochondrial DNA as integral nucleoid constituents. FASEB J. 2009, 23, 2034–2044. [Google Scholar] [CrossRef]
- Fukui, H.; Moraes, C.T. The mitochondrial impairment, oxidative stress and neurodegeneration connection: reality or just an attractive hypothesis? Trends Neurosci. 2008, 31, 251–256. [Google Scholar] [CrossRef]
- Thompson, L.V. Oxidative stress, mitochondria and mtDNA-mutator mice. Exp. Gerontol. 2006, 41, 1220–1222. [Google Scholar] [CrossRef] [Green Version]
- Reilly, T.; Waterhouse, J.; Atkinson, G. Aging, rhythms of physical performance, and adjustment to changes in the sleep-activity cycle. Occup. Environ. Med. 1997, 54, 812–816. [Google Scholar] [CrossRef]
- Touitou, Y.; Bogdan, A.; Haus, E.; Touitou, C. Modifications of circadian and circannual rhythms with aging. Exp. Gerontol. 1997, 32, 603–614. [Google Scholar] [CrossRef]
- Weinert, D. Age-dependent changes of the circadian system. Chronobiol. Int. 2000, 17, 261–283. [Google Scholar] [CrossRef]
- Van Someren, E.J. Circadian rhythms and sleep in human aging. Chronobiol. Int. 2000, 17, 233–243. [Google Scholar] [CrossRef]
- Yamazaki, S.; Straume, M.; Tei, H.; Sakaki, Y.; Menaker, M.; Block, G.D. Effects of aging on central and peripheral mammalian clocks. Proc. Natl. Acad. Sci. USA 2002, 99, 10801–10806. [Google Scholar]
- Weinert, D. The temporal order of mammals. Evidence for multiple central and peripheral control mechanisms and for endogenous and exogenous components: some implications for research on aging. Biol. Rhythm Res. 2005, 36, 293–308. [Google Scholar]
- Morin, L.P. Age-related changes in hamster circadian period, entrainment and rhythm splitting. Biol. Rhythm Res. 1988, 3, 237–248. [Google Scholar] [CrossRef]
- Penev, P.D.; Zee, P.C.; Turek, F.W. Quantitative analysis of the age-related fragmentation of hamster 24-h activity rhythms. Am. J. Physiol. 1997, 273, R2132–R2137. [Google Scholar]
- Weinert, H.; Weinert, D.; Waterhouse, J. The circadian activity and body temperature rhythms of mice during their last days of life. Biol. Rhythm Res. 2002, 33, 199–212. [Google Scholar] [CrossRef]
- Humbert, W.; Pévet, P. The decrease of pineal melatonin production with age. Causes and consequences. Ann. N.Y. Acad. Sci. 1994, 719, 43–63. [Google Scholar] [CrossRef]
- Magri, F.; Locatelli, M.; Balza, G.; Molla, G.; Cuzzoni, G.; Fioravanti, M.; Solerte, S.B.; Ferrari, E. Changes in endocrine circadian rhythms as markers of physiological and pathological brain aging. Chronobiol. Int. 1997, 14, 385–396. [Google Scholar] [CrossRef]
- Touitou, Y. Human aging and melatonin. Clinical relevance. Exp. Gerontol. 2001, 36, 1083–1100. [Google Scholar] [CrossRef]
- Karasek, M.; Reiter, R.J. Melatonin and aging. Neuroendocrinol. Lett. 2002, 23 (Suppl. 1), 14–16. [Google Scholar]
- Karasek, M. Melatonin, human aging, and age-related diseases. Exp. Gerontol. 2004, 39, 1723–1729. [Google Scholar] [CrossRef]
- Coto-Montes, A.; Hardeland, R. Diurnal rhythm of protein carbonyl as an indicator of oxidative damage in Drosophila melanogaster: Influence of clock gene alleles and deficiencies in the formation of free-radical scavengers. Biol. Rhythm Res. 1999, 30, 383–391. [Google Scholar] [CrossRef]
- Coto-Montes, A.; Tomás-Zapico, C.; Rodríguez-Colunga, M.J.; Tolivia-Cadrecha, D.; Martínez-Fraga, J.; Hardeland, R.; Tolivia, D. Effects of the circadian mutation 'tau' on the Harderian glands of Syrian hamsters. J. Cell. Biochem. 2001, 83, 426–434. [Google Scholar] [CrossRef]
- Hardeland, R.; Coto-Montes, A.; Poeggeler, B. Circadian rhythms, oxidative stress and antioxidative defense mechanisms. Chronobiol. Int. 2003, 20, 921–962. [Google Scholar] [CrossRef]
- Krishnan, N.; Davis, A.J.; Giebultowicz, J.M. Circadian regulation of response to oxidative stress in Drosophila melanogaster. Biochem. Biophys. Res. Commun. 2008, 374, 299–303. [Google Scholar] [CrossRef]
- Coto-Montes, A.; Boga, J.A.; Tomás-Zapico, C.; Rodríguez-Colunga, M.J.; Martínez-Fraga, J.; Tolivia-Cadrecha, D.; Menéndez, G.; Hardeland, R.; Tolivia, D. Physiological oxidative stress model: Syrian hamster Harderian gland—Sex differences in antioxidant enzymes. Free Radic. Biol. Med. 2001, 30, 785–792. [Google Scholar] [CrossRef]
- Arundine, M.; Tymianski, M. Molecular mechanisms of calcium-dependent neurodegeneration in excitotoxicity. Cell Calcium 2003, 34, 325–337. [Google Scholar] [CrossRef]
- Dawson, V.L.; Dawson, T.M. Deadly conversations: Nuclear-mitochondrial cross-talk. J. Bioenerg. Biomembr. 2004, 36, 287–294. [Google Scholar] [CrossRef]
- Gardoni, F.; di Luca, M. New targets for pharmacological intervention in the glutamatergic synapse. Eur. J. Pharmacol. 2006, 545, 2–10. [Google Scholar] [CrossRef]
- Dong, X.X.; Wang, Y.; Qin, Z.H. Molecular mechanisms of excitotoxicity and their relevance to pathogenesis of neurodegenerative diseases. Acta Pharmacol. Sin. 2009, 30, 379–387. [Google Scholar] [CrossRef]
- Tabrizi, S.J.; Cleeter, M.W.; Xuereb, J.; Taanman, J.W.; Cooper, J.M.; Schapira, A.H. Biochemical abnormalities and excitotoxicity in Huntington’s disease brain. Ann. Neurol. 1999, 45, 25–32. [Google Scholar] [CrossRef]
- Hilditch-Maguire, P.; Trettel, F.; Passani, L.A.; Auerbach, A.; Persichetti, F.; MacDonald, M.E. Huntingtin: an iron-regulated protein essential for normal nuclear and perinuclear organelles. Hum. Mol. Genet. 2000, 9, 2789–2797. [Google Scholar] [CrossRef]
- Panov, A.V.; Gutekunst, C.A.; Leavitt, B.R.; Hayden, M.R.; Burke, J.R.; Strittmatter, W.J.; Greenamyre, J.T. Early mitochondrial calcium defects in Huntington’s disease are direct effects of polyglutamines. Nat. Neurosci. 2002, 5, 731–736. [Google Scholar]
- Ellerby, L.M. Hunting for excitement: NMDA receptors in Huntington’s disease. Neuron 2002, 33, 841–842. [Google Scholar] [CrossRef]
- Panov, A.V.; Burke, J.R.; Strittmatter, W.J.; Greenamyre, J.T. In vitro effects of polyglutamine tracts on Ca2+-dependent depolarization of rat and human mitochondria: Relevance to Huntington’s disease. Arch. Biochem. Biophys. 2003, 410, 1–6. [Google Scholar] [CrossRef]
- Estrada Sánchez, A.M.; Mejía-Toiber, J.; Massieu, L. Excitotoxic neuronal death and the pathogenesis of Huntington's disease. Arch. Med. Res. 2008, 39, 265–276. [Google Scholar] [CrossRef]
- Quintanilla, R.A.; Jin, Y.N.; Fuenzalida, K.; Bronfman, M.; Johnson, G.V. Rosiglitazone treatment prevents mitochondrial dysfunction in mutant huntingtin-expressing cells: Possible role of peroxisome proliferator-activated receptor-γ (PPARγ) in the pathogenesis of Huntington disease. J. Biol. Chem. 2008, 283, 25628–25637. [Google Scholar] [CrossRef]
- Damiano, M.; Galvan, L.; Déglon, N.; Brouillet, E. Mitochondria in Huntington's disease. Biochim. Biophys. Acta. 2009. [Google Scholar] [CrossRef]
- Hensley, K.; Maidt, M.L.; Yu, Z.; Sang, H.; Markesbery, W.R.; Floyd, R.A. Electrochemical analysis of protein nitrotyrosine and dityrosine in the Alzheimer brain indicates region-specific accumulation. J. Neurosci. 1998, 18, 8126–8132. [Google Scholar]
- Srinivasan, V.; Pandi-Perumal, S.R.; Cardinali, D.P.; Poeggeler, B.; Hardeland, R. Melatonin in Alzheimer’s disease and other neurodegenerative disorders. Behav. Brain Funct. 2006, 2. [Google Scholar] [CrossRef]
- Rogers, J.; Webster, S.; Lue, L.F.; Brachova, W.; Civin, W.H.; Emmerling, M.; Shivers, B.; Walker, D.; McGeer, P. Inflammation and Alzheimer's disease pathogenesis. Neurobiol. Aging 1996, 17, 681–686. [Google Scholar] [CrossRef]
- Pereira, H.A.; Kumar, P.; Gramma, P. Expression of CAP37, a novel inflammatory mediator, in Alzheimer's disease. Neurobiol. Aging 1996, 17, 753–759. [Google Scholar]
- Sheng, J.G.; Ito, K.; Skinner, R.D.; Mrak, R.E.; Rovnaghi, C.R.; van Eldik, L.J.; Griffin, W.S.T. In vivo and in vitro evidence supporting a role for the inflammatory cytokine interleukin-1 as a driving force in Alzheimer pathogenesis. Neurobiol. Aging 1996, 17, 761–766. [Google Scholar] [CrossRef]
- Yan, S.D.; Zhu, H.; Fu, J.; Yan, S.F.; Roher, A.; Tourtellotte, W.W.; Rajavashisth, T.; Chen, X.; Godman, G.C.; Stern, D.; Schmidt, A.M. Amyloid-β peptide-receptor for advanced glycation endproduct interaction elicits neuronal expression of macrophage-colony stimulating factor: A proinflammatory pathway in Alzheimer disease. Proc. Natl. Acad. Sci. USA 1997, 94, 5296–5301. [Google Scholar]
- Smith, M.A.; Richey Harris, P.L.; Sayre, L.M.; Beckman, J.S.; Perry, G. Widespread peroxynitrite-mediated damage in Alzheimer’s disease. J. Neurosci. 1997, 17, 2653–2657. [Google Scholar]
- Carpenter, A.F.; Carpenter, P.W.; Markesbery, W.R. Morphometric analysis of microglia in Alzheimer's disease. J. Neuropathol. Exp. Neurol. 1993, 52, 601–608. [Google Scholar] [CrossRef]
- MacKenzie, I.R.A.; Hao, C.; Muñoz, D.G. Role of microglia in senile plaque formation. Neurobiol. Aging 1995, 16, 797–804. [Google Scholar] [CrossRef]
- McGeer, E.G.; McGeer, P.L. Neuroinflammation in Alzheimer's disease and mild cognitive impairment: A field in its infancy. J. Alzheimers Dis. 2009. [Google Scholar] [CrossRef]
- Landreth, G.E. Microglia in central nervous dystem diseases. J. Neuroimmune Pharmacol. 2009. [Google Scholar] [CrossRef] [Green Version]
- Alberati-Giani, D.; Ricciardi-Castagnoli, P.; Köhler, C.; Cesura, A.M. Regulation of the kynurenine metabolic pathway by interferon-γ in murine cloned macrophages and microglial cells. J. Neurochem. 1996, 66, 996–1004. [Google Scholar]
- Heyes, M.P.; Achim, C.L.; Wiley, C.A.; Major, E.O.; Saito, K.; Markey, S.P. Human microglia convert l-tryptophan into the neurotoxin quinolinic acid. Biochem. J. 1996, 320, 595–597. [Google Scholar]
- Guillemin, G.J.; Smythe, G.A.; Veas, L.A.; Takikawa, O.; Brew, B.J. Aβ1-42 induces production of quinolinic acid by human macrophages and microglia. Neuroreport 2003, 14, 2311–2315. [Google Scholar] [CrossRef]
- Kwidzinski, E.; Bunse, J.; Kovac, A.D.; Ullrich, O.; Zipp, F.; Nitsch, R.; Bechmann, I. IDO (indolamine 2,3-dioxygenase) expression and function in the CNS. Adv. Exp. Med. Biol. 2003, 527, 113–118. [Google Scholar] [CrossRef]
- Guillemin, G.J.; Williams, K.R.; Smith, D.G.; Smythe, G.A.; Croitoru-Lamoury, J.; Brew, B.J. Quinolinic acid in the pathogenesis of Alzheimer's disease. Adv. Exp. Med. Biol. 2003, 527, 167–176. [Google Scholar] [CrossRef]
- Guillemin, G.J.; Brew, B.J.; Noonan, C.E.; Takikawa, O.; Cullen, K.M. Indoleamine 2,3 dioxygenase and quinolinic acid immunoreactivity in Alzheimer's disease hippocampus. Neuropathol. Appl. Neurobiol. 2005, 31, 395–404. [Google Scholar] [CrossRef]
- Yadav, M.C.; Burudi, E.M.; Alirezaei, M.; Flynn, C.C.; Watry, D.D.; Lanigan, C.M.; Fox, H.S. IFN-γ-induced IDO and WRS expression in microglia is differentially regulated by IL-4. Glia 2007, 55, 1385–1396. [Google Scholar] [CrossRef]
- Foster, A.C.; Collins, J.F.; Schwarcz, R. On the excitotoxic properties of quinolinic acid, 2,3-piperidine dicarboxylic acids and structurally related compounds. Neuropharmacology 1983, 22, 1331–1342. [Google Scholar] [CrossRef]
- Schurr, A.; West, C.A.; Rigor, B.M. Neurotoxicity of quinolinic acid and its derivatives in hypoxic rat hippocampal slices. Brain Res. 1991, 568, 199–204. [Google Scholar] [CrossRef]
- Misztal, M.; Skangiel-Kramska, J.; Niewiadomska, G.; Danysz, W. Subchronic intraventricular infusion of quinolinic acid produces working memory impairment—A model of progressive excitotoxicity. Neuropharmacology 1996, 35, 449–458. [Google Scholar] [CrossRef]
- Braidy, N.; Grant, R.; Adams, S.; Brew, B.J.; Guillemin, G.J. Mechanism for quinolinic acid cytotoxicity in human astrocytes and neurons. Neurotox. Res. 2009, 16, 77–86. [Google Scholar] [CrossRef]
- Guidetti, P.; Luthi-Carter, R.E.; Augood, S.J.; Schwarcz, R. Neostriatal and cortical quinolinate levels are increased in early grade Huntington's disease. Neurobiol. Dis. 2004, 17, 455–461. [Google Scholar] [CrossRef]
- Guidetti, P.; Bates, G.P.; Graham, R.K.; Hayden, M.R.; Leavitt, B.R.; MacDonald, M.E.; Slow, E.J.; Wheeler, V.C.; Woodman, B.; Schwarcz, R. Elevated brain 3-hydroxykynurenine and quinolinate levels in Huntington disease mice. Neurobiol. Dis. 2006, 23, 190–197. [Google Scholar] [CrossRef]
- Park, L.; Anrather, J.; Girouard, H.; Zhou, P.; Iadecola, C. Nox2-derived reactive oxygen species mediate neurovascular dysregulation in the aging mouse brain. J. Cereb. Blood Flow Metab. 2007, 27, 1908–1918. [Google Scholar] [CrossRef]
- McCann, S.K.; Dusting, G.J.; Roulston, C.L. Early increase of Nox4 NADPH oxidase and superoxide generation following endothelin-1-induced stroke in conscious rats. J. Neurosci. Res. 2008, 86, 2524–2534. [Google Scholar] [CrossRef]
- Chéret, C.; Gervais, A.; Lelli, A.; Colin, C.; Amar, L.; Ravassard, P.; Mallet, J.; Cumano, A.; Krause, K.H.; Mallat, M. Neurotoxic activation of microglia is promoted by a nox1-dependent NADPH oxidase. J. Neurosci. 2008, 28, 12039–12051. [Google Scholar] [CrossRef]
- Collins-Underwood, J.R.; Zhao, W.; Sharpe, J.G.; Robbins, M.E. NADPH oxidase mediates radiation-induced oxidative stress in rat brain microvascular endothelial cells. Free Radic. Biol. Med. 2008, 45, 929–938. [Google Scholar] [CrossRef]
- Chen, H.; Song, Y.S.; Chan, P.H. Inhibition of NADPH oxidase is neuroprotective after ischemia-reperfusion. J. Cereb. Blood Flow Metab. 2009, 29, 1262–1272. [Google Scholar] [CrossRef]
- Schiavone, S.; Sorce, S.; Dubois-Dauphin, M.; Jaquet, V.; Colaianna, M.; Zotti, M.; Cuomo, V.; Trabace, L.; Krause, K.H. Involvement of NOX2 in the development of behavioral and pathologic alterations in isolated rats. Biol. Psychiatry 2009, 66, 384–392. [Google Scholar] [CrossRef]
- Block, K.; Gorin, Y.; Abboud, H.E. Subcellular localization of Nox4 and regulation in diabetes. Proc. Natl. Acad. Sci. USA 2009, 106, 14385–14390. [Google Scholar] [CrossRef]
- Hardeland, R.; Poeggeler, B.; Pappolla, M.A. New vistas on mitochondrial electron flux rates and aging. Cell. 2008. Available online: http://www.cell.com/content/article/comments?uid= PIIS0092867408000627/ accessed on 4 December 2009.
- Hardeland, R. Melatonin, mitochondrial electron flux and leakage: Recent findings and resolution of contradictory results. Adv. Stud. Biol. 2009, 1, 207–230. [Google Scholar]
- Hardeland, R.; Poeggeler, B.; Pappolla, M.A. Mitochondrial actions of melatonin — an endeavor to identify their adaptive and cytoprotective mechanisms. Abh. Sächs. Akad. Wiss. Math. Nat. Kl. 2009, 65(4), 14–31. [Google Scholar]
- Wang, W.; Fang, H.; Groom, L.; Cheng, A.; Zhang, W.; Liu, J.; Wang, X.; Li, K.; Han, P.; Zheng, M.; Yin, J.; Wang, W.; Mattson, M.P.; Kao, J.P.; Lakatta, E.G.; Sheu, S.S.; Ouyang, K.; Chen, J.; Dirksen, R.T.; Cheng, H. Superoxide flashes in single mitochondria. Cell 2008, 134, 279–290. [Google Scholar] [CrossRef]
- Sheu, S.S.; Wang, W.; Cheng, H.; Dirksen, R.T. Superoxide flashes: Illuminating new insights into cardiac ischemia/reperfusion injury. Future Cardiol. 2008, 4, 551–554. [Google Scholar] [CrossRef]
- Genova, M.L.; Ventura, B.; Giuliano, G.; Bovina, C.; Formiggini, G.; Parenti Castelli, G.; Lenaz, G. The site of production of superoxide radical in mitochondrial Complex I is not a bound ubisemiquinone but presumably iron-sulfur cluster N2. FEBS Lett. 2001, 505, 364–368. [Google Scholar] [CrossRef]
- Genova, M.L.; Merlo Pich, M.; Bernacchia, A.; Bianchi, C.; Biondi, A.; Bovina, C.; Falasca, A.I.; Formiggini, G.; Parenti Castelli, G.; Lenaz, G. The mitochondrial production of reactive oxygen species in relation to aging and pathology. Ann. N.Y. Acad. Sci. 2004, 1011, 86–100. [Google Scholar]
- Lenaz, G.; Bovina, C.; D’Aurelio, M.; Fato, R.; Formiggini, G.; Genova, M.L.; Giuliano, G.; Merlo Pich, M.; Paolucci, U.; Parenti Castelli, G.; Ventura, B. Role of mitochondria in oxidative stress and aging. Ann. N.Y. Acad. Sci. 2002, 959, 199–213. [Google Scholar]
- Ohnishi, S.T.; Ohnishi, T.; Muranaka, S.; Fujita, H.; Kimura, H.; Uemura, K.; Yoshida, K.; Utsumi, K. A possible site of superoxide generation in the complex I segment of rat heart mitochondria. J. Bioenerg. Biomembr. 2005, 37, 1–15. [Google Scholar] [CrossRef]
- Lenaz, G.; Fato, R.; Genova, M.L.; Bergamini, C.; Bianchi, C.; Biondi, A. Mitochondrial complex I: Structural and functional aspects. Biochim. Biophys. Acta 2006, 1757, 1406–1420. [Google Scholar] [CrossRef]
- Genova, M.L.; Merlo Pich, M.; Biondi, A.; Bernacchia, A.; Falasca, A.; Bovina, C.; Formiggini, G.; Parenti Castelli, G.; Lenaz, G. Mitochondrial production of oxygen radical species and the role of coenzyme Q as an antioxidant. Exp. Biol. Med. Maywood 2003, 228, 506–513. [Google Scholar]
- Staniek, K.; Gille, L.; Kozlov, A.V.; Nohl, H. Mitochondrial superoxide radical formation is controlled by electron bifurcation to the high and low potential pathways. Free Radic. Res. 2002, 36, 381–387. [Google Scholar] [CrossRef]
- Gong, X.; Yu, L.; Xia, D.; Yu, C.A. Evidence for electron equilibrium between the two hemes bL in the dimeric cytochrome bc1 complex. J. Biol. Chem. 2005, 280, 9251–9257. [Google Scholar] [Green Version]
- Miwa, S.; Brand, M.D. The topology of superoxide production by complex III and glycerol 3-phosphate dehydrogenase in Drosophila mitochondria. Biochim. Biophys. Acta 2005, 1709, 214–219. [Google Scholar]
- Tyagi, N.; Moshal, K.S.; Sen, U.; Lominadze, D.; Ovechkin, A.V.; Tyagi, S.C. Ciglitazone ameliorates homocysteine-mediated mitochondrial translocation and matrix metalloproteinase-9 activation in endothelial cells by inducing peroxisome proliferator activated receptor-γ activity. Cell. Mol. Biol. (Noisy-le-grand) 2006, 52, 21–27. [Google Scholar]
- Wang, X.; Su, B.; Zheng, L.; Perry, G.; Smith, M.A.; Zhu, X. The role of abnormal mitochondrial dynamics in the pathogenesis of Alzheimer's disease. J. Neurochem. 2009, 109 (Suppl. 1), 153–159. [Google Scholar]
- Wang, X.; Su, B.; Lee, H.G.; Li, X.; Perry, G.; Smith, M.A.; Zhu, X. Impaired balance of mitochondrial fission and fusion in Alzheimer's disease. J. Neurosci. 2009, 29, 9090–9103. [Google Scholar] [CrossRef]
- Su, B.; Wang, X.; Zheng, L.; Perry, G.; Smith, M.A.; Zhu, X. Abnormal mitochondrial dynamics and neurodegenerative diseases. Biochim. Biophys. Acta 2009. [Google Scholar] [CrossRef]
- Wang, X.; Su, B.; Siedlak, S.L.; Moreira, P.I.; Fujioka, H.; Wang, Y.; Casadesus, G.; Zhu, X. Amyloid-β overproduction causes abnormal mitochondrial dynamics via differential modulation of mitochondrial fission/fusion proteins. Proc. Natl. Acad. Sci. USA 2008, 105, 19318–19323. [Google Scholar]
- Uo, T.; Dworzak, J.; Kinoshita, C.; Inman, D.M.; Kinoshita, Y.; Horner, P.J.; Morrison, R.S. Drp1 levels constitutively regulate mitochondrial dynamics and cell survival in cortical neurons. Exp. Neurol. 2009, 218, 274–285. [Google Scholar] [CrossRef]
- Radak, Z.; Chung, H.Y.; Koltai, E.; Taylor, A.W.; Goto, S. Exercise, oxidative stress and hormesis. Ageing Res. Rev. 2008, 7, 34–42. [Google Scholar] [CrossRef]
- Radak, Z.; Kumagai, S.; Taylor, A.W.; Naito, H.; Goto, S. Effects of exercise on brain function: role of free radicals. Appl. Physiol. Nutr. Metab. 2007, 32, 942–946. [Google Scholar] [CrossRef]
- Goto, S.; Naito, H.; Kaneko, T.; Chung, H.Y.; Radák, Z. Hormetic effects of regular exercise in aging: correlation with oxidative stress. Appl. Physiol. Nutr. Metab. 2007, 32, 948–953. [Google Scholar] [CrossRef]
- Radak, Z.; Chung, H.Y.; Goto, S. Systemic adaptation to oxidative challenge induced by regular exercise. Free Radic. Biol. Med. 2008, 44, 153–159. [Google Scholar] [CrossRef]
- Ames, B.N.; Shigenaga, M.K.; Hagen, T.M. Mitochondrial decay in aging. Biochim. Biophys. Acta 1995, 1271, 165–170. [Google Scholar]
- Ekstrand, M.I.; Terzioglu, M.; Galter, D.; Zhu, S.; Hofstetter, C.; Lindqvist, E.; Thams, S.; Bergstrand, A.; Hansson, F.S.; Trifunovic, A.; Hoffer, B.; Cullheim, S.; Mohammed, A.H.; Olson, L.; Larsson, N.G. Progressive parkinsonism in mice with respiratory-chain-deficient dopamine neurons. Proc. Natl. Acad. Sci. USA 2007, 104, 1325–1330. [Google Scholar]
- Terzioglu, M.; Larsson, N.G. Mitochondrial dysfunction in mammalian ageing. Novartis Found. Symp. 2007, 287, 197–208. [Google Scholar] [CrossRef]
- Boveris, A.; Alvarez, S.; Navarro, A. The role of mitochondrial nitric oxide synthase in inflammation and septic shock. Free Radic. Biol. Med. 2002, 33, 1186–1193. [Google Scholar] [CrossRef]
- Escames, G.; Acuña-Castroviejo, D.; López, L.C.; Tan, D.X.; Maldonado, M.D.; Sánchez-Hidalgo, M.; León, J.; Reiter, R.J. Pharmacological utility of melatonin in the treatment of septic shock: experimental and clinical evidence. J. Pharm. Pharmacol. 2006, 58, 1153–1165. [Google Scholar] [CrossRef]
- Alvarez, S.; Evelson, P.A. Nitric oxide and oxygen metabolism in inflammatory conditions: Sepsis and exposition to polluted ambients. Front. Biosci. 2007, 12, 964–974. [Google Scholar] [CrossRef] [Green Version]
- Brown, G.C.; Bal-Price, A. Inflammatory neurodegeneration mediated by nitric oxide, glutamate, and mitochondria. Mol. Neurobiol. 2003, 27, 325–355. [Google Scholar] [CrossRef]
- Dahm, C.C.; Moore, K.; Murphy, M.P. Persistent S-nitrosation of complex I and other mitochondrial membrane proteins by S-nitrosothiols but not nitric oxide or peroxynitrite: Implications for the interaction of nitric oxide with mitochondria. J. Biol. Chem. 2006, 281, 10056–10065. [Google Scholar] [CrossRef]
- Ducrocq, C.; Blanchard, B.; Pignatelli, B.; Ohshima, H. Peroxynitrite: An endogenous oxidizing and nitrating agent. Cell. Mol. Life Sci. 1999, 55, 1068–1077. [Google Scholar] [CrossRef]
- Blanchard, B.; Pompon, D.; Ducrocq, C. Nitrosation of melatonin by nitric oxide and peroxynitrite. J. Pineal Res. 2000, 29, 184–192. [Google Scholar]
- Radi, R.; Cassina, A.; Hodara, R.; Quijano, C.; Castro, L. Peroxynitrite reactions and formation in mitochondria. Free Radic. Biol. Med. 2002, 33, 1451–1464. [Google Scholar] [CrossRef]
- Guenther, A.L.; Schmidt, S.I.; Laatsch, H.; Fotso, S.; Ness, H.; Ressmeyer, A.R.; Poeggeler, B.; Hardeland, R. Reactions of the melatonin metabolite AMK (N1-acetyl-5-methoxykynuramine) with reactive nitrogen species: Formation of novel compounds, 3-acetamidomethyl-6-methoxycinnolinone and 3-nitro-AMK. J. Pineal Res. 2005, 39, 251–260. [Google Scholar] [CrossRef]
- Trujillo, M.; Folkes, L.; Bartesaghi, S.; Kalyanaraman, B.; Wardman, P.; Radi, R. Peroxynitrite-derived carbonate and nitrogen dioxide radicals readily react with lipoic and dihydrolipoic acid. Free Radic. Biol. Med. 2005, 39, 279–288. [Google Scholar] [CrossRef]
- Ferrer-Sueta, G.; Radi, R. Chemical biology of peroxynitrite: Kinetics, diffusion, and radicals. ACS Chem. Biol. 2009, 4, 161–177. [Google Scholar] [CrossRef]
- Celano, L.; Gil, M.; Carballal, S.; Durán, R.; Denicola, A.; Banerjee, R.; Alvarez, B. Inactivation of cystathionine β-synthase with peroxynitrite. Arch. Biochem. Biophys. 2009. [Google Scholar] [CrossRef]
- Klingen, A.R.; Palsdottir, H.; Hunte, C.; Ullmann, G.M. Redox-linked protonation state changes in cytochrome bc1 identified by Poisson-Boltzmann electrostatics calculations. Biochim. Biophys. Acta 2007, 1767, 204–221. [Google Scholar]
- Lesnefsky, E.J.; Hoppel, C.L. Cardiolipin as an oxidative target in cardiac mitochondria in the aged rat. Biochim. Biophys. Acta 2008, 1777, 1020–1027. [Google Scholar]
- Lesnefsky, E.J.; Minkler, P.; Hoppel, C.L. Enhanced modification of cardiolipin during ischemia in the aged heart. J. Mol. Cell. Cardiol. 2009, 46, 1008–1015. [Google Scholar] [CrossRef]
- Wenz, T.; Hielscher, R.; Hellwig, P.; Schägger, H.; Richers, S.; Hunte, C. Role of phospholipids in respiratory cytochrome bc1 complex catalysis and supercomplex formation. Biochim. Biophys. Acta 2009, 1787, 609–616. [Google Scholar]
- Petrosillo, G.; Matera, M.; Moro, N.; Ruggiero, F.M.; Paradies, G. Mitochondrial complex I dysfunction in rat heart with aging: critical role of reactive oxygen species and cardiolipin. Free Radic. Biol. Med. 2009, 46, 88–94. [Google Scholar] [CrossRef]
- Basova, L.V.; Kurnikov, I.V.; Wang, L.; Ritov, V.B.; Belikova, N.A.; Vlasova, I.I.; Pacheco, A.A.; Winnica, D.E.; Peterson, J.; Bayir, H.; Waldeck, D.H.; Kagan, V.E. Cardiolipin switch in mitochondria: Shutting off the reduction of cytochrome c and turning on the peroxidase activity. Biochemistry 2007, 46, 3423–3434. [Google Scholar]
- Ott, M.; Gogvadze, V.; Orrenius, S.; Zhivotovsky, B. Mitochondria, oxidative stress and cell death. Apoptosis 2007, 12, 913–922. [Google Scholar] [CrossRef]
- Bayir, H.; Tyurin, V.A.; Tyurina, Y.Y.; Viner, R.; Ritov, V.; Amoscato, A.A.; Zhao, Q.; Zhang, X.J.; Janesko-Feldman, K.L.; Alexander, H.; Basova, L.V.; Clark, R.S.; Kochanek, P.M.; Kagan, V.E. Selective early cardiolipin peroxidation after traumatic brain injury: an oxidative lipidomics analysis. Ann. Neurol. 2007, 62, 154–169. [Google Scholar] [CrossRef]
- Kagan, V.E.; Bayir, A.; Bayir, H.; Stoyanovsky, D.; Borisenko, G.G.; Tyurina, Y.Y.; Wipf, P.; Atkinson, J.; Greenberger, J.S.; Chapkin, R.S.; Belikova, N.A. Mitochondria-targeted disruptors and inhibitors of cytochrome c/cardiolipin peroxidase complexes: A new strategy in anti-apoptotic drug discovery. Mol. Nutr. Food Res. 2009, 53, 104–114. [Google Scholar] [CrossRef]
- Kagan, V.E.; Bayir, H.A.; Belikova, N.A.; Kapralov, O.; Tyurina, Y.Y.; Tyurin, V.A.; Jiang, J.; Stoyanovsky, D.A.; Wipf, P.; Kochanek, P.M.; Greenberger, J.S.; Pitt, B.; Shvedova, A.A.; Borisenko, G. Cytochrome c/cardiolipin relations in mitochondria: a kiss of death. Free Radic. Biol. Med. 2009, 46, 1439–1453. [Google Scholar] [CrossRef]
- Zoccarato, F.; Cavallini, L.; Alexandre, A. Succinate is the controller of O2–/H2O2 release at mitochondrial complex I: negative modulation by malate, positive by cyanide. J. Bioenerg. Biomembr. 2009, 41, 387–393. [Google Scholar] [CrossRef]
- Kim, S.C.; Sprung, R.; Chen, Y.; Xu, Y.; Ball, H.; Pei, J.; Cheng, T.; Kho, Y.; Xiao, H.; Xiao, L.; Grishin, N.V.; White, M.; Yang, X.J.; Zhao, Y. Substrate and functional diversity of lysine acetylation revealed by a proteomics survey. Mol. Cell 2006, 23, 607–618. [Google Scholar] [CrossRef]
- Schlicker, C.; Gertz, M.; Papatheodorou, P.; Kachholz, B.; Becker, C.F.; Steegborn, C. Substrates and regulation mechanisms for the human mitochondrial sirtuins Sirt3 and Sirt5. J. Mol. Biol. 2008, 382, 790–801. [Google Scholar] [CrossRef]
- Liu, Y.; He, J.; Ji, S.; Wang, Q.; Pu, H.; Jiang, T.; Meng, L.; Yang, X.; Ji, J. Comparative studies of early liver dysfunction in senescence-accelerated mouse using mitochondrial proteomics approaches. Mol. Cell. Proteomics 2008, 7, 1737–1747. [Google Scholar] [CrossRef]
- Okatani, Y.; Wakatsuki, A.; Reiter, R.J.; Miyahara, Y. Acutely administered melatonin restores hepatic mitochondrial physiology in old mice. Int. J. Biochem. Cell Biol. 2003, 35, 367–375. [Google Scholar] [CrossRef]
- Nishikawa, T.; Takahashi, J.A.; Fujibayashi, Y.; Fujisawa, H.; Zhu, B.; Nishimura, Y.; Ohnishi, K.; Higuchi, K.; Hashimoto, N.; Hosokawa, M. An early stage mechanism of the age-associated mitochondrial dysfunction in the brain of SAMP8 mice; an age-associated neurodegeneration animal model. Neurosci. Lett. 1998, 254, 69–72. [Google Scholar] [CrossRef]
- Omata, N.; Murata, T.; Fujibayashi, Y.; Waki, A.; Sadato, N.; Yoshimoto, M.; Wada, Y.; Yonekura, Y. Age-related changes in energy production in fresh senescence-accelerated mouse brain slices as revealed by positron autoradiography. Dement. Geriatr. Cogn. Disord. 2001, 12, 78–84. [Google Scholar] [CrossRef]
- Tomobe, K.; Nomura, Y. Neurochemistry, neuropathology, and heredity in SAMP8: A mouse model of senescence. Neurochem. Res. 2009, 34, 660–669. [Google Scholar] [CrossRef]
- Carretero, M.; Escames, G.; López, L.C.; Venegas, C.; Dayoub, J.C.; García, L.; Acuña-Castroviejo, D. Long-term melatonin administration protects brain mitochondria from aging. J. Pineal Res. 2009, 47, 192–200. [Google Scholar] [CrossRef]
- Boveris, A.; Navarro, A. Brain mitochondrial dysfunction in aging. IUBMB Life 2008, 60, 308–314. [Google Scholar] [CrossRef]
- Guarente, L. Sirtuins in aging and disease. Cold Spring Harb. Symp. Quant. Biol. 2007, 72, 483–488. [Google Scholar] [CrossRef]
- Dasgupta, B.; Milbrandt, J. Resveratrol stimulates AMP kinase activity in neurons. Proc. Natl. Acad. Sci. USA 2007, 104, 7217–7222. [Google Scholar] [CrossRef]
- Naudí, A.; Caro, P.; Jové, M.; Gómez, J.; Boada, J.; Ayala, V.; Portero-Otín, M.; Barja, G.; Pamplona, R. Methionine restriction decreases endogenous oxidative molecular damage and increases mitochondrial biogenesis and uncoupling protein 4 in rat brain. Rejuvenation Res. 2007, 10, 473–484. [Google Scholar] [CrossRef]
- Fontán-Lozano, A.; López-Lluch, G.; Delgado-García, J.M.; Navas, P.; Carrión, A.M. Molecular bases of caloric restriction regulation of neuronal synaptic plasticity. Mol. Neurobiol. 2008, 38, 167–177. [Google Scholar] [CrossRef]
- Guarente, L. Mitochondria — a nexus for aging, calorie restriction, and sirtuins? Cell 2008, 132, 171–176. [Google Scholar]
- Onyango, I.G.; Lu, J.; Rodova, M.; Lezi, E.; Crafter, A.B.; Swerdlow, R.H. Regulation of neuron mitochondrial biogenesis and relevance to brain health. Biochim. Biophys. Acta 2009. [Google Scholar] [CrossRef]
- Sen, S.; Phillis, J.W. α-Phenyl-tert-butyl-nitrone (PBN) attenuates hydroxyl radical production during ischemia-reperfusion injury of rat brain: an EPR study. Free Radic. Res. Commun. 1993, 19, 255–265. [Google Scholar] [CrossRef]
- Floyd, R.A.; Hensley, K. Nitrone inhibition of age-associated oxidative damage. Ann. N.Y. Acad. Sci. 2000, 899, 222–237. [Google Scholar]
- Floyd, R.A. Serendipitous findings while researching oxygen free radicals. Free Radic. Biol. Med. 2009, 46, 1004–1013. [Google Scholar] [CrossRef]
- Polovyanenko, D.N.; Plyusnin, V.F.; Reznikov, V.A.; Khramtsov, V.V.; Bagryanskaya, E.G. Mechanistic studies of the reactions of nitrone spin trap PBN with glutathiyl radical. J. Phys. Chem. B 2008, 112, 4841–4847. [Google Scholar] [CrossRef]
- Butterfield, D.A.; Howard, B.J.; Yatin, S.; Allen, K.L.; Carney, J.M. Free radical oxidation of brain proteins in accelerated senescence and its modulation by N-tert-butyl-α-phenylnitrone. Proc. Natl. Acad. Sci. 1997, 94, 674–678. [Google Scholar] [CrossRef]
- Floyd, R.A.; Hensley, K.; Forster, M.J.; Kelleher-Andersson, J.A.; Wood, P.L. Nitrones, their value as therapeutics and probes to understand aging. Mech. Ageing Dev. 2002, 30, 1021–1031. [Google Scholar]
- Floyd, R.A.; Hensley, K.; Forster, M.J.; Kelleher-Andersson, J.A.; Wood, P.L. Nitrones as neuroprotectants and antiaging drugs. Ann N.Y. Acad. Sci. 2002, 959, 321–329. [Google Scholar]
- Kelicen, P.; Cantuti-Castelvetri, I.; Pekiner, C.; Paulson, K.E. The spin trapping agent PBN stimulates H2O2-induced Erk and Src kinase activity in human neuroblastoma cells. Neuroreport 2002, 13, 1057–1061. [Google Scholar] [CrossRef]
- Li, P.A.; He, Q.P.; Nakamura, L.; Csiszar, K. Free radical spin trap α-phenyl-N-tert-butyl-nitron inhibits caspase-3 activation and reduces brain damage following a severe forebrain ischemic injury. Free Radic. Biol. Med. 2001, 31, 1191–1197. [Google Scholar] [CrossRef]
- Kotake, Y.; Sang, H.; Miyajima, T.; Wallis, G.L. Inhibition of NF-κB, iNOS mRNA, COX2 mRNA, and COX catalytic activity by phenyl-N-tert-butylnitrone (PBN). Biochim. Biophys. Acta 1998, 1448, 77–84. [Google Scholar]
- Floyd, R.A.; Hensley, K.; Jaffery, F.; Maidt, L.; Robinson, K.; Pye, Q.; Stewart, C. Increased oxidative stress brought on by pro-inflammatory cytokines in neurodegenerative processes and the protective role of nitrone-based free radical traps. Life Sci. 1999, 65, 1893–1899. [Google Scholar] [CrossRef]
- Sang, H.; Wallis, G.L.; Stewart, C.A.; Kotake, Y. Expression of cytokines and activation of transcription factors in lipopolysaccharide-administered rats and their inhibition by phenyl N-tert-butylnitrone (PBN). Arch. Biochem. Biophys. 1999, 363, 341–348. [Google Scholar] [CrossRef]
- Pieper, G.M.; Nilakantan, V.; Zhou, X.; Khanna, A.K.; Johnson, C.P.; Roza, A.M.; Adams, M.B.; Hilton, G.; Felix, C.C. Treatment with α-phenyl-N-tert-butylnitrone, a free radical-trapping agent, abrogates inflammatory cytokine gene expression during alloimmune activation in rat cardiac allografts. J. Pharmacol. Exp. Ther. 2005, 312, 774–779. [Google Scholar]
- Hensley, K.; Pye, Q.N.; Maidt, M.L.; Stewart, C.A.; Robinson, K.A.; Jaffrey, F.; Floyd, R.A. Interaction of α-phenyl-N-tert-butyl nitrone and alternate electron acceptors with complex I indicates a substrate reduction site upstream form the rotenone binding site. J. Neurochem. 1998, 71, 2549–2557. [Google Scholar]
- Hagen, T.M.; Wehr, C.M.; Ames, B.N. Mitochondrial decay in aging. Reversal through supplementation of acetyl-L-carnitine and N-tert-butyl-α-phenyl-nitrone. Ann. N.Y. Acad. Sci. 1998, 854, 214–223. [Google Scholar]
- Floyd, R.A.; Kotake, Y.; Hensley, K.; Nakae, D.; Konishi, Y. Reactive oxygen species in choline deficiency induced carcinogenesis and nitrone inhibition. Mol. Cell. Biochem. 2002, 234–235, 195–203. [Google Scholar]
- Xu, K.; Puchowicz, M.A.; Sun, X.; LaManna, J.C. Mitochondrial dysfunction in aging rat brain following transient global ischemia. Adv. Exp. Med. Biol. 2008, 614, 379–386. [Google Scholar] [CrossRef]
- Atamna, H.; Paler-Martínez, A.; Ames, B.N. N-t-butyl hydroxylamine, a hydrolysis product of α-phenyl-N-t-butyl nitrone, is more potent in delaying senescence in human lung fibroblasts. J. Biol. Chem. 2000, 275, 6741–6748. [Google Scholar]
- Liu, J.; Atamna, H.; Kuratsune, H.; Ames, B.N. Delaying brain mitochondrial decay and aging with mitochondrial antioxidants and metabolites. Ann. N.Y. Acad. Sci. 2002, 959, 133–166. [Google Scholar]
- Saito, K.; Yoshioka, H. Protective effect of spin trap agent, N-tert-butyl-α-phenylnitrone on hyperoxia-induced oxidative stress and its potential as a nitric oxide donor. Free Radic. Res. 2002, 36, 143–149. [Google Scholar] [CrossRef]
- Saito, K.; Yoshioka, H. ESR characterization of a novel spin-trapping agent, 15N-labeled N-tert-butyl-α-phenylnitrone, as a nitric oxide donor. Biosci. Biotechnol. Biochem. 2002, 66, 2189–2193. [Google Scholar] [CrossRef] [Green Version]
- Novakov, CP.; Stoyanovsky, D.A. Comparative metabolism of N-tert-butyl-N-[1-(1-oxy-pyridin-4-yl)-ethyl]- and N-tert-butyl-N-(1-phenyl-ethyl)-nitroxide by the cytochrome P450 monooxygenase system. Chem. Res. Toxicol. 2002, 15, 749–753. [Google Scholar] [CrossRef]
- Spooren, A.A.; Evelo, C.T. In vitro haematotoxic effects of three methylated hydroxylamines. Arch. Toxicol. 1997, 71, 299–305. [Google Scholar] [CrossRef]
- Killilea, D.W.; Wong, S.L.; Cahaya, H.S.; Atamna, H.; Ames, B.N. Iron accumulation during cellular senescence. Ann. N.Y. Acad. Sci. 2004, 1019, 365–367. [Google Scholar]
- Saito, K.; Ikeda, M.; Yoshioka, H.; Tomita, T. Nitric oxide and effect of a radical scavenger N-tert-butyl-α-phenylnitrone on stroke in a rat model. Pharmacology 2005, 73, 76–80. [Google Scholar] [CrossRef]
- Banerjee, R.; Saravanan, K.S.; Thomas, B.; Sindhu, K.M.; Mohanakumar, K.P. Evidence for hydroxyl radical scavenging action of nitric oxide donors in the protection against 1-methyl-4-phenylpyridinium-induced neurotoxicity in rats. Neurochem. Res. 2008, 33, 985–995. [Google Scholar] [CrossRef]
- Ha, K.S.; Kim, K.M.; Kwon, Y.G.; Bai, S.K.; Nam, W.D.; Yoo, Y.M.; Kim, P.K.; Chung, H.T.; Billiar, T.R.; Kim, Y.M. Nitric oxide prevents 6-hydroxydopamine-induced apoptosis in PC12 cells through cGMP-dependent PI3 kinase/Akt activation. FASEB J. 2003, 17, 1036–1047. [Google Scholar] [CrossRef]
- Farinelli, S.E.; Park, D.S.; Greene, L.A. Nitric oxide delays the death of trophic factor-deprived PC12 cells and sympathetic neurons by a cGMP-mediated mechanism. J. Neurosci. 1996, 16, 2325–2334. [Google Scholar] [Green Version]
- Estévez, A.G.; Spear, N.; Thompson, J.A.; Cornwell, T.L.; Radi, R.; Barbeito, L.; Beckman, J.S. Nitric oxide-dependent production of cGMP supports the survival of rat embryonic motor neurons cultured with brain-derived neurotrophic factor. J. Neurosci. 1998, 18, 3708–3714. [Google Scholar]
- Robb, S.J.; Connor, J.R. Nitric oxide protects astrocytes from oxidative stress. Ann. N.Y. Acad. Sci. 2002, 962, 93–102. [Google Scholar] [CrossRef]
- Borniquel, S.; Valle, I.; Cadenas, S.; Lamas, S.; Monsalve, M. Nitric oxide regulates mitochondrial oxidative stress protection via the transcriptional coactivator PGC-1α. FASEB J. 2006, 20, 1889–1891. [Google Scholar] [CrossRef]
- Paxinou, E.; Weisse, M.; Chen, Q.; Souza, J.M.; Hertkorn, C.; Selak, M.; Daikhin, E.; Yudkoff, M.; Sowa, G.; Sessa, W.C.; Ischiropoulos, H. Dynamic regulation of metabolism and respiration by endogenously produced nitric oxide protects against oxidative stress. Proc. Natl. Acad. Sci. USA 2001, 98, 11575–11580. [Google Scholar]
- Dubay, A.; Forster, M.J.; Sohal, R.S. Effect of the spin-trapping compound N-tert-butyl-α-phenylnitrone on protein oxidation and life span. Arch. Biochem. Biophys. 1995, 324, 249–254. [Google Scholar] [CrossRef]
- Poeggeler, B.; Durand, G.; Polidori, A.; Pappolla, M.A.; Vega-Naredo, I.; Coto-Montes, A.; Böker, J.; Hardeland, R.; Pucci, B. Mitochondrial medicine: neuroprotection and life extension by the new amphiphilic nitrone LPBNAH acting as a highly potent advanced antioxidant agent. J. Neurochem. 2005, 95, 962–973. [Google Scholar] [CrossRef]
- Durand, G.; Polidori, A.; Salles, J.P.; Pucci, B. Synthesis of a new family of glycolipidic nitrones as potential antioxidant drugs. Bioorg. Med. Chem. Lett. 2003, 13, 859–862. [Google Scholar] [CrossRef]
- Durand, G.; Polidori, A.; Ouari, O.; Tordo, P.; Gerome, V.; Rustin, P.; Pucci, B. Synthesis and preliminary biological evaluation of ionic and nonionic amphiphilic PBN. J. Med. Chem. 2003, 46, 5530–5535. [Google Scholar]
- Ortial, S.; Durand, G.; Poeggeler, B.; Polidori, A.; Pappolla, M.A.; Böker, J.; Hardeland, R.; Pucci, B. Fluorinated amphiphilic amino acid derivatives as antioxidant carriers: A new class of protective agents. J. Med. Chem. 2006, 49, 2812–2820. [Google Scholar] [CrossRef]
- Durand, G.; Poeggeler, B.; Böker, J.; Raynal, S. Fine-tuning the amphiphilicity: A crucial parameter in the design of potent α-phenyl-N-tert-butylnitrone analogues. J. Med. Chem. 2007, 50, 3976–3979. [Google Scholar] [CrossRef]
- Asanuma, T.; Yasui, H.; Inanami, O.; Waki, K.; Takahashi, M.; Iizuka, D.; Uemura, T.; Durand, G.; Polidori, A.; Kon, Y.; Pucci, B.; Kuwabara, M. A new amphiphilic derivative, N-{[4-(lactobionamido)methyl]benzylidene}-1,1-dimethyl-2-(octylsulfanyl)ethylamine N-oxide, has a protective effect against copper-induced fulminant hepatitis in Long-Evans Cinnamon rats at an extremely low concentration compared with its original form α-phenyl-N-(tert-butyl) nitrone. Chem. Biodivers. 2007, 4, 2253–2267. [Google Scholar] [CrossRef]
- Robertson, L.; Hartley, R.C. Synthesis of N-arylpyridinium salts bearing a nitrone spin trap as potential mitochondria-targeted antioxidants. Tetrahedron 2009, 65, 5284–5292. [Google Scholar] [CrossRef]
- El Fangour, S.; Marini, M.; Good, J.; McQuaker, S.J.; Shiels, P.G.; Hartley, R.C. Nitrones for understanding and ameliorating the oxidative stress associated with aging. Age (Dordr.) 2009. [Google Scholar] [CrossRef]
- Durand, G.; Poeggeler, B.; Ortial, S.; Polidori, A.; Böker, J.; Hardeland, R.; Pucci, B. Amphiphilic nitrone amines: A new class of protective agents acting as mitochondrial metabolism modifiers. Free Radic. Biol. Med. 2007, 43, S91–S92. [Google Scholar]
- Mastronardi, C.A.; Yu, W.H.; McCann, S.M. Resting and circadian release of nitric oxide is controlled by leptin in male rats. Proc. Natl. Acad. Sci. USA 2002, 99, 5721–5726. [Google Scholar] [CrossRef]
- Poeggeler, B.; Schulz, C.; Pappolla, M.A.; Bodó, E.; Tiede, S.; Lehnert, H.; Paus, R. Leptin and the skin: A new frontier. Exp. Dermatol. 2009. [Google Scholar] [CrossRef]
- Wang, C.H.; Lee, W.J.; Ghanta, V.K.; Wang, W.T.; Cheng, S.Y.; Hsueh, C.M. Molecules involve in the self-protection of neurons against glucose-oxygen-serum deprivation (GOSD)-induced cell damage. Brain Res. Bull. 2009, 79, 169–176. [Google Scholar] [CrossRef]
- Mercer, J.G.; Hoggard, N.; Williams, L.M.; Lawrence, C.B.; Hannah, L.T.; Trayhurn, P. Localization of leptin receptor mRNA and the long form splice variant (Ob-Rb) in mouse hypothalamus and adjacent brain regions by in situ hybridization. FEBS Lett. 1996, 387, 113–116. [Google Scholar] [CrossRef]
- Shioda, S.; Funahashi, H.; Nakajo, S.; Yada, T.; Maruta, O.; Nakai, Y. Immunohistochemical localization of leptin receptor in the rat brain. Neurosci. Lett. 1998, 243, 41–44. [Google Scholar] [CrossRef]
- Guo, Z.; Jiang, H.; Xu, X.; Duan, W.; Mattson, M.P. Leptin-mediated cell survival signaling in hippocampal neurons mediated by JAK STAT3 and mitochondrial stabilization. J. Biol. Chem. 2008, 283, 1754–1763. [Google Scholar]
- Mitchell, S.E.; Nogueiras, R.; Morris, A.; Tovar, S.; Grant, C.; Cruickshank, M.; Rayner, D.V.; Dieguez, C.; Williams, L.M. Leptin receptor gene expression and number in the brain are regulated by leptin level and nutritional status. J. Physiol. 2009, 587, 3573–3585. [Google Scholar] [CrossRef]
- Satoh, T.; Yoshino, S.; Katano, A.; Ishizuka, T.; Tomaru, T.; Shibusawa, N.; Hashimoto, K.; Yamada, M.; Mori, M. Isolation of a novel leptin receptor gene promoter preferentially functioning in neuronal cells. Biochem. Biophys. Res. Commun. 2009, 389, 673–677. [Google Scholar] [CrossRef]
- Li, X.L.; Aou, S.; Oomura, Y.; Hori, N.; Fukunaga, K.; Hori, T. Impairment of long-term potentiation and spatial memory in leptin receptor-deficient rodents. Neuroscience 2002, 113, 607–615. [Google Scholar] [CrossRef]
- Oomura, Y.; Hori, N.; Shiraishi, T.; Fukunaga, K.; Takeda, H.; Tsuji, M.; Matsumiya, T.; Ishibashi, M.; Aou, S.; Li, X.L.; Kohno, D.; Uramura, K.; Sougawa, H.; Yada, T.; Wayner, M.J.; Sasaki, K. Leptin facilitates learning and memory performance and enhances hippocampal CA1 long-term potentiation and CaMK II phosphorylation in rats. Peptides 2006, 27, 2738–2749. [Google Scholar] [CrossRef]
- Fulco, M.; Sartorelli, V. Comparing and contrasting the roles of AMPK and SIRT1 in metabolic tissues. Cell Cycle 2008, 7, 3669–3679. [Google Scholar] [CrossRef]
- López-Lluch, G.; Irusta, P.M.; Navas, P.; de Cabo, R. Mitochondrial biogenesis and healthy aging. Exp. Gerontol. 2008, 43, 813–819. [Google Scholar] [CrossRef]
- Myers, M.G. Jr. Leptin receptor signaling and the regulation of mammalian physiology. Recent Prog. Horm. Res. 2004, 59, 287–304. [Google Scholar] [CrossRef]
- Procopio, C.; Andreozzi, F.; Laratta, E.; Cassese, A.; Beguinot, F.; Arturi, F.; Hribal, M.L.; Perticone, F.; Sesti, G. Leptin-stimulated endothelial nitric-oxide synthase via an adenosine 5'-monophosphate-activated protein kinase/Akt signaling pathway is attenuated by interaction with C-reactive protein. Endocrinology 2009, 150, 3584–3593. [Google Scholar] [CrossRef]
- Ho, P.W.L.; Liu, H.F.; Ho, J.W.M.; Zhang, W.Y.; Chu, A.C.Y.; Kwok, K.H.H.; Ge, X.; Chan, K.H.; Ramsden, D.B.; Ho, S.L. Mitochondrial uncoupling protein-2 (UCP2) mediates leptin protection against MPP+ toxicity in neuronal cells. Neurotox. Res. 2009. [Google Scholar] [CrossRef]
- Horvath, T.L.; Warden, C.H.; Hajos, M.; Lombardi, A.; Goglia, F.; Diano, S. Brain uncoupling protein 2: Uncoupled neuronal mitochondria predict thermal synapses in homeostatic centers. J. Neurosci. 1999, 19, 10417–10427. [Google Scholar]
- Andrews, Z.B.; Horvath, T.L. Uncoupling protein-2 regulates lifespan in mice. Am. J. Physiol. Endocrinol. Metab. 2009, 296, E621–E627. [Google Scholar] [CrossRef]
- Dietrich, M.O.; Horvath, T.L. The role of mitochondrial uncoupling proteins in lifespan. Pflügers Arch. 2009. [Google Scholar] [CrossRef]
- Ceddia, R.B.; William, W.N. Jr.; Lima, F.B.; Flandin, P.; Curi, R.; Giacobino, J.P. Leptin stimulates uncoupling protein-2 mRNA expression and Krebs cycle activity and inhibits lipid synthesis in isolated rat white adipocytes. Eur. J. Biochem. 2000, 267, 5952–5958. [Google Scholar] [CrossRef]
- Tajima, D.; Masaki, T.; Hidaka, S.; Kakuma, T.; Sakata, T.; Yoshimatsu, H. Acute central infusion of leptin modulates fatty acid mobilization by affecting lipolysis and mRNA expression for uncoupling proteins. Exp. Biol. Med. (Maywood) 2005, 230, 200–206. [Google Scholar]
- Chu, A.C.; Ho, P.W.L.; Kwok, K.H.H.; Ho, J.W.M.; Chan, K.H.; Liu, H.F.; Kung, M.H.W.; Ramsden, D.B.; Ho, S.L. Mitochondrial UCP4 attenuates MPP+- and dopamine-induced oxidative stress, mitochondrial depolarization, and ATP deficiency in neurons and is interlinked with UCP2 expression. Free Radic. Biol. Med. 2009, 46, 810–820. [Google Scholar] [CrossRef]
- Deierborg, T.; Wieloch, T.; Diano, S.; Warden, C.H.; Horvath, T.L.; Mattiasson, G. Overexpression of UCP2 protects thalamic neurons following global ischemia in the mouse. J. Cereb. Blood Flow Metab. 2008, 28, 1186–1195. [Google Scholar] [CrossRef]
- Kalra, S.P. Global life-long health benefits of repression of hypothalamic NPY system by central leptin gene therapy. Curr. Top. Med. Chem. 2007, 7, 1675–1681. [Google Scholar] [CrossRef]
- Hooft van Huijsduijnen, R.; Wälchli, S.; Ibberson, M.; Harrenga, A. Protein tyrosine phosphatases as drug targets: PTP1B and beyond. Expert Opin. Ther. Targets 2002, 6, 637–647. [Google Scholar] [CrossRef]
- McCarty, M.F.; Barroso-Aranda, J.; Contreras, F. The "rejuvenatory" impact of lipoic acid on mitochondrial function in aging rats may reflect induction and activation of PPAR-γ coactivator-1α. Med. Hypotheses 2009, 72, 29–33. [Google Scholar] [CrossRef]
- Chiarelli, F.; Di Marzio, D. Peroxisome proliferator-activated receptor-γ agonists and diabetes: Current evidence and future perspectives. Vasc. Health Risk Manag. 2008, 4, 297–304. [Google Scholar]
- Leahy, J.L. Thiazolidinediones in prediabetes and early type 2 diabetes: What can be learned about that disease's pathogenesis. Curr. Diab. Rep. 2009, 9, 215–220. [Google Scholar] [CrossRef]
- Festuccia, W.T.; Oztezcan, S.; Laplante, M.; Berthiaume, M.; Michel, C.; Dohgu, S.; Denis, R.G.; Brito, M.N.; Brito, N.A.; Miller, D.S.; Banks, W.A.; Bartness, T.J.; Richard, D.; Deshaies, Y. Peroxisome proliferator-activated receptor-γ-mediated positive energy balance in the rat is associated with reduced sympathetic drive to adipose tissues and thyroid status. Endocrinology 2008, 149, 2121–2130. [Google Scholar] [CrossRef]
- Potula, R.; Ramirez, S.H.; Knipe, B.; Leibhart, J.; Schall, K.; Heilman, D.; Morsey, B.; Mercer, A.; Papugani, A.; Dou, H.; Persidsky, Y. Peroxisome proliferator-activated receptor-γ activation suppresses HIV-1 replication in an animal model of encephalitis. AIDS 2008, 22, 1539–1549. [Google Scholar] [CrossRef]
- Ramirez, S.H.; Heilman, D.; Morsey, B.; Potula, R.; Haorah, J.; Persidsky, Y. Activation of peroxisome proliferator-activated receptor γ (PPARγ) suppresses Rho GTPases in human brain microvascular endothelial cells and inhibits adhesion and transendothelial migration of HIV-1 infected monocytes. J. Immunol. 2008, 180, 1854–1865. [Google Scholar]
- Hyong, A.; Jadhav, V.; Lee, S.; Tong, W.; Rowe, J.; Zhang, J.H.; Tang, J. Rosiglitazone, a PPARγ agonist, attenuates inflammation after surgical brain injury in rodents. Brain Res. 2008, 1215, 218–224. [Google Scholar] [CrossRef] [Green Version]
- McTigue, D.M.; Tripathi, R.; Wie, P.; Lash, A.T. The PPARγ agonist Pioglitazone improves anatomical and locomotor recovery after rodent spinal cord injury. Exp. Neurol. 2007, 205, 396–406. [Google Scholar] [CrossRef]
- Roberts, T.J.; Chapman, A.C.; Cipolla, M.J. PPAR-γ agonist rosiglitazone reverses increased cerebral venous hydraulic conductivity during hypertension. Am. J. Physiol. Heart Circ. Physiol. 2009, 297, H1347–H1353. [Google Scholar] [CrossRef]
- Shimazu, T.; Inoue, I.; Araki, N.; Asano, Y.; Sawada, M.; Furuya, D.; Nagoya, H.; Greenberg, J.H. A peroxisome proliferator-activated receptor-γ agonist reduces infarct size in transient but not in permanent ischemia. Stroke 2005, 36, 353–359. [Google Scholar] [CrossRef]
- Kovács, T. Therapy of Alzheimer disease [in Hungarian]. Neuropsychopharmacol. Hung. 2009, 11, 27–33. [Google Scholar]
- Landreth, G.; Jiang, Q.; Mandrekar, S.; Heneka, M. PPARγ agonists as therapeutics for the treatment of Alzheimer's disease. Neurotherapeutics 2008, 5, 481–489. [Google Scholar] [CrossRef]
- Jiang, Q.; Heneka, M.; Landreth, G.E. The role of peroxisome proliferator-activated receptor-gamma (PPARgamma) in Alzheimer's disease: Therapeutic implications. CNS Drugs 2008, 22, 1–14. [Google Scholar]
- Escribano, L.; Simón, A.M.; Pérez-Mediavilla, A.; Salazar-Colocho, P.; Del Río, J.; Frechilla, D. Rosiglitazone reverses memory decline and hippocampal glucocorticoid receptor down-regulation in an Alzheimer's disease mouse model. Biochem. Biophys. Res. Commun. 2009, 379, 406–410. [Google Scholar] [CrossRef]
- Mogi, M.; Li, J.M.; Tsukuda, K.; Iwanami, J.; Min, L.J.; Sakata, A.; Fujita, T.; Iwai, M.; Horiuchi, M. Telmisartan prevented cognitive decline partly due to PPAR-γ activation. Biochem. Biophys. Res. Commun. 2008, 375, 446–449. [Google Scholar] [CrossRef]
- Tsukuda, K.; Mogi, M.; Iwanami, J.; Min, L.J.; Sakata, A.; Jing, F.; Iwai, M.; Horiuchi, M. Cognitive deficit in amyloid-β-injected mice was improved by pretreatment with a low dose of telmisartan partly because of peroxisome proliferator-activated receptor-γ activation. Hypertension 2009, 54, 782–787. [Google Scholar] [CrossRef]
- Stolar, M.W.; Hoogwerf, B.J.; Gorshow, S.M.; Boyle, P.J.; Wales, D.O. Managing type 2 diabetes: Going beyond glycemic control. J. Manag. Care Pharm. 2008, 14 (Suppl. B), s2–s19. [Google Scholar]
- Madsen, K.G.; Grönberg, G.; Skonberg, C.; Jurva, U.; Hansen, S.H.; Olsen, J. Electrochemical oxidation of troglitazone: Identification and characterization of the major reactive metabolite in liver microsomes. Chem. Res. Toxicol. 2008, 21, 2035–2041. [Google Scholar] [CrossRef]
- Tahara, K.; Nishikawa, T.; Hattori, Y.; Iijima, S.; Kouno, Y.; Abe, Y. Production of a reactive metabolite of troglitazone by electrochemical oxidation performed in nonaqueous medium. J. Pharm. Biomed. Anal. 2009, 50, 1030–1036. [Google Scholar] [CrossRef]
- Sapko, M.T.; Guidetti, P.; Yu, P.; Tagle, D.A.; Pellicciari, R.; Schwarcz, R. Endogenous kynurenate controls the vulnerability of striatal neurons to quinolinate: Implications for Huntington's disease. Exp. Neurol. 2006, 197, 31–40. [Google Scholar] [CrossRef]
- Ceresoli-Borroni, G.; Guidetti, P.; Schwarcz, R. Acute and chronic changes in kynurenate formation following an intrastriatal quinolinate injection in rats. J. Neural Transm. 1999, 106, 229–242. [Google Scholar] [CrossRef]
- Poeggeler, B.; Rassoulpour, A.; Wu, H.Q.; Guidetti, P.; Roberts, R.C.; Schwarcz, R. Dopamine receptor activation reveals a novel, kynurenate-sensitive component of striatal N-methyl-D-aspartate neurotoxicity. Neuroscience 2007, 148, 188–197. [Google Scholar] [CrossRef]
- Kessler, M.; Terramani, T.; Lynch, G; Baudry, M. A glycine site associated with N-methyl-D-aspartic acid receptors: Characterization and identification of a new class of antagonists. J. Neurochem. 1989, 52, 1319–1328. [Google Scholar]
- Parsons, C.G.; Danysz, W.; Quack, G.; Hartmann, S.; Lorenz, B.; Wollenburg, C.; Baran, L.; Przegalinski, E.; Kostowski, W.; Krzascik, P.; Chizh, B.; Headley, P.M. Novel systemically active antagonists of the glycine site of the N-methyl-D-aspartate receptor: Electrophysiological, biochemical and behavioral characterization. J. Pharmacol. Exp. Ther. 1997, 283, 1264–1275. [Google Scholar]
- Carpenedo, R.; Pittaluga, A.; Cozzi, A.; Attucci, S.; Galli, A.; Raiteri, M.; Moroni, F. Presynaptic kynurenate-sensitive receptors inhibit glutamate release. Eur. J. Neurosci. 2001, 13, 2141–2147. [Google Scholar] [CrossRef]
- Rassoulpour, A.; Wu, H.Q.; Ferré, S.; Schwarcz, R. Nanomolar concentrations of kynurenic acid reduce extracellular dopamine levels in the striatum. J. Neurochem. 2005, 93, 762–765. [Google Scholar] [CrossRef]
- Zsizsik, B.K.; Hardeland, R. Formation of kynurenic and xanthurenic acids from kynurenine and 3-hydroxykynurenine in the dinoflagellate Lingulodinium polyedrum: Role of a novel, oxidative pathway. Comp. Biochem. Physiol. 2002, 133C, 383–392. [Google Scholar]
- Politi, V.; D'Alessio, S.; Di Stazio, G.; De Luca, G. Antioxidant properties of indole-3-pyruvic acid. Adv. Exp. Med. Biol. 1996, 398, 291–298. [Google Scholar] [CrossRef]
- Lapin, I.P.; Politi, V. Anxiolytic effect of indole-3-pyruvic acid (IPA) in mice. Pharmacol. Res. 1993, 28, 129–134. [Google Scholar] [CrossRef]
- Lapin, I.P.; Politi, V. Antiethanol effects of indol-3-ylpyruvic acid in mice. Alcohol Alcohol. 1994, 29, 265–268. [Google Scholar]
- Bartolini, B.; Corniello, C.; Sella, A.; Somma, F.; Politi, V. The enol tautomer of indole-3-pyruvic acid as a biological switch in stress responses. Adv. Exp. Med. Biol. 2003, 527, 601–608. [Google Scholar] [CrossRef]
- Politi, V.; De Luca, G.; Gallai, V.; Puca, F.; Comin, M. Clinical experiences with the use of indole-3-pyruvic acid. Adv. Exp. Med. Biol. 1999, 467, 227–232. [Google Scholar] [CrossRef]
- Gardoni, F.; Di Luca, M. New targets for pharmacological intervention in the glutamatergic synapse. Eur. J. Pharmacol. 2006, 545, 2–10. [Google Scholar] [CrossRef]
- Rogawski, M.A.; Wenk, G.L. The neuropharmacological basis for the use of memantine in the treatment of Alzheimer's disease. CNS Drug Rev. 2003, 9, 275–308. [Google Scholar] [CrossRef]
- Areosa, S.A.; Sheriff, F.; McShane, R. Memantine for dementia. Cochrane Database Syst. Rev. 2005, 3, CD003154. [Google Scholar] [CrossRef]
- Kos, T.; Popik, P.; Pietraszek, M.; Schäfer, D.; Danysz, W.; Dravolina, O.; Blokhina, E.; Galankin, T.; Bespalov, A.Y. Effect of 5-HT3 receptor antagonist MDL 72222 on behaviors induced by ketamine in rats and mice. Eur. Neuropsychopharmacol. 2006, 16, 297–310. [Google Scholar] [CrossRef]
- Chipana, C.; Torres, I.; Camarasa, J.; Pubill, D.; Escubedo, E. Memantine protects against amphetamine derivatives-induced neurotoxic damage in rodents. Neuropharmacology 2008, 54, 1254–1263. [Google Scholar] [CrossRef]
- Seeman, P.; Caruso, C.; Lasaga, M. Memantine agonist action at dopamine D2High receptors. Synapse 2008, 62, 149–153. [Google Scholar] [CrossRef]
- Yokogoshi, H.; Kobayashi, M.; Mochizuki, M.; Terashima, T. Effect of theanine, γ-glutamylethylamide, on brain monoamines and striatal dopamine release in conscious rats. Neurochem. Res. 1998, 23, 667–673. [Google Scholar] [CrossRef]
- Nathan, P.J.; Lu, K.; Gray, M.; Oliver, C. The neuropharmacology of L-theanine (N-ethyl-L-glutamine): A possible neuroprotective and cognitive enhancing agent. J. Herb. Pharmacother. 2006, 6, 21–30. [Google Scholar]
- Yokogoshi, H.; Mochizuki, M.; Saitoh, K. Theanine-induced reduction of brain serotonin concentration in rats. Biosci. Biotechnol. Biochem. 1998, 62, 816–817. [Google Scholar] [CrossRef]
- Kakuda, T. Neuroprotective effects of the green tea components theanine and catechins. Biol. Pharm. Bull. 2002, 25, 1513–1518. [Google Scholar] [CrossRef]
- Egashira, N.; Hayakawa, K.; Mishima, K.; Kimura, H.; Iwasaki, K.; Fujiwara, M. Neuroprotective effect of γ-glutamylethylamide (theanine) on cerebral infarction in mice. Neurosci. Lett. 2004, 363, 58–61. [Google Scholar] [CrossRef]
- Cho, H.S.; Kim, S.; Lee, S.Y.; Park, J.A.; Kim, S.J.; Chun, H.S. Protective effect of the green tea component, L-theanine on environmental toxins-induced neuronal cell death. Neurotoxicology 2008, 29, 656–662. [Google Scholar] [CrossRef]
- de Mejia, E.G.; Ramirez-Mares, M.V.; Puangpraphant, S. Bioactive components of tea: Cancer, inflammation and behavior. Brain Behav. Immun. 2009, 23, 721–731. [Google Scholar] [CrossRef]
- Slotkin, T.A.; Seidler, F.J. Oxidative and excitatory mechanisms of developmental neurotoxicity: Transcriptional profiles for chlorpyrifos, diazinon, dieldrin, and divalent nickel in PC12 cells. Environ. Health Perspect. 2009, 117, 587–596. [Google Scholar]
- Hardeland, R.; Poeggeler, B. Actions of melatonin, its structural and functional analogs in the central nervous system and the significance of metabolism. Cent. Nerv. Syst. Agents Med. Chem. 2007, 7, 289–303. [Google Scholar] [CrossRef]
- Corton, J.C.; Brown-Borg, H.M. Peroxisome proliferator-activated receptor γ coactivator 1 in caloric restriction and other models of longevity. J. Gerontol. A Biol. Sci. Med. Sci. 2005, 60, 1494–1509. [Google Scholar] [CrossRef]
- Scarpulla, R.C. Transcriptional paradigms in mammalian mitochondrial biogenesis and function. Physiol. Rev. 2008, 88, 611–638. [Google Scholar] [CrossRef]
- Alaynick, W.A. Nuclear receptors, mitochondria and lipid metabolism. Mitochondrion 2008, 8, 329–337. [Google Scholar] [CrossRef]
- Chen, D.; Bruno, J.; Easlon, E.; Lin, S.J.; Cheng, H.L.; Alt, F.W.; Guarente, L. Tissue-specific regulation of SIRT1 by calorie restriction. Genes Dev. 2008, 22, 1753–1757. [Google Scholar] [CrossRef]
- Lombard, D.B.; Alt, F.W.; Cheng, H.L.; Bunkenborg, J.; Streeper, R.S.; Mostoslavsky, R.; Kim, J.; Yancopoulos, G.; Valenzuela, D.; Murphy, A.; Yang, Y.; Chen, Y.; Hirschey, M.D.; Bronson, R.T.; Haigis, M.; Guarente, L.P.; Farese, R.V., Jr.; Weissman, S.; Verdin, E.; Schwer, B. Mammalian Sir2 homolog SIRT3 regulates global mitochondrial lysine acetylation. Mol. Cell. Biol. 2007, 27, 8807–8814. [Google Scholar]
- Dilova, I.; Easlon, E.; Lin, S.J. Calorie restriction and the nutrient sensing signaling pathways. Cell. Mol. Life Sci. 2007, 64, 752–767. [Google Scholar] [CrossRef]
- Fulco, M.; Sartorelli, V. Comparing and contrasting the roles of AMPK and SIRT1 in metabolic tissues. Cell Cycle 2008, 7, 3669–3679. [Google Scholar] [CrossRef]
- Wareski, P.; Vaarmann, A.; Choubey, V.; Safiulina, D.; Liiv, J.; Kuum, M.; Kaasik, A. PGC-1α and PGC-1β regulate mitochondrial density in neurons. J. Biol. Chem. 2009, 284, 21379–21385. [Google Scholar] [CrossRef]
- Viña, J.; Gomez-Cabrera, M.C.; Borras, C.; Froio, T.; Sanchis-Gomar, F.; Martinez-Bello, V.E.; Pallardo, F.V. Mitochondrial biogenesis in exercise and in ageing. Adv. Drug Deliv. Rev. 2009. [Google Scholar] [CrossRef]
- Goldberg, D.M. Does wine work? Clin. Chem. 1995, 41, 14–16. [Google Scholar]
- Jang, M.; Cai, L.; Udeani, G.O.; Slowing, K.V.; Thomas, C.F.; Beecher, C.W.; Fong, H.H.; Farnsworth, N.R.; Kinghorn, A.D.; Mehta, R.G.; Moon, R.C.; Pezzuto, J.M. Cancer chemopreventive activity of resveratrol, a natural product derived from grapes. Science 1997, 275, 218–220. [Google Scholar]
- Wendler, J.; Hardeland, R. Phenolic food constituents as free-radical scavengers and luminophores: I. Resveratrol. In Studies on Antioxidants and their Metabolites; Hardeland, R., Ed.; Cuvillier: Göttingen, Germany, 2006; pp. 38–47. [Google Scholar]
- Burkhardt, S.; Reiter, R.J.; Tan, D.X.; Hardeland, R.; Cabrera, J.; Karbownik, M. DNA oxidatively damaged by chromium(III) and H2O2 is protected by melatonin, N1-acetyl-N2-formyl-5-methoxykynuramine, resveratrol and uric acid. Int. J. Biochem. Cell Biol. 2001, 33, 775–783. [Google Scholar] [CrossRef]
- Zini, R.; Morin, C.; Bertelli, A.; Bertelli, A.A.; Tillement, J.P. Effects of resveratrol on the rat brain respiratory chain. Drugs Exp. Clin. Res. 1999, 25, 87–97. [Google Scholar]
- Lagouge, M.; Argmann, C.; Gerhart-Hines, Z.; Meziane, H.; Lerin, C.; Daussin, F.; Messadeq, N.; Milne, J.; Lambert, P.; Elliott, P.; Geny, B.; Laakso, M.; Puigserver, P.; Auwerx, J. Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1α. Cell 2006, 127, 1109–1122. [Google Scholar] [CrossRef]
- Yousuf, S.; Atif, F.; Ahmad, M.; Hoda, N.; Ishrat, T.; Khan, B.; Islam, F. Resveratrol exerts its neuroprotective effect by modulating mitochondrial dysfunctions and associated cell death during cerebral ischemia. Brain Res. 2009, 1250, 242–253. [Google Scholar]
- Zhang, H.; Schools, G.P.; Lei, T.; Wang, W.; Kimelberg, H.K.; Zhou, M. Resveratrol attenuates early pyramidal neuron excitability impairment and death in acute rat hippocampal slices caused by oxygen-glucose deprivation. Exp. Neurol. 2008, 212, 44–52. [Google Scholar]
- Chong, Z.Z.; Maiese, K. Enhanced tolerance against early and late apoptotic oxidative stress in mammalian neurons through nicotinamidase and sirtuin mediated pathways. Curr. Neurovasc. Res. 2008, 5, 159–170. [Google Scholar] [CrossRef]
- Della-Morte, D.; Dave, K.R.; DeFazio, R.A.; Bao, Y.C.; Raval, A.P.; Perez-Pinzon, M.A. Resveratrol pretreatment protects rat brain from cerebral ischemic damage via a sirtuin 1-uncoupling protein 2 pathway. Neuroscience 2009, 159, 993–1002. [Google Scholar] [CrossRef]
- Pallàs, M.; Casadesús, G.; Smith, M.A.; Coto-Montes, A.; Pelegri, C.; Vilaplana, J.; Camins, A. Resveratrol and neurodegenerative diseases: Activation of SIRT1 as the potential pathway towards neuroprotection. Curr. Neurovasc. Res. 2009, 6, 70–81. [Google Scholar] [CrossRef]
- Borra, M.T.; Smith, B.C.; Denu, J.M. Mechanism of human SIRT1 activation by resveratrol. J. Biol. Chem. 2005, 280, 17187–17195. [Google Scholar] [CrossRef]
- Raval, A.P.; Lin, H.W.; Dave, K.R.; Defazio, R.A.; Della Morte, D.; Kim, E.J.; Perez-Pinzon, M.A. Resveratrol and ischemic preconditioning in the brain. Curr. Med. Chem. 2008, 15, 1545–1551. [Google Scholar] [CrossRef]
- Smith, J.J.; Kenney, R.D.; Gagne, D.J.; Frushour, B.P.; Ladd, W.; Galonek, H.L.; Israelian, K.; Song, J.; Razvadauskaite, G.; Lynch, A.V.; Carney, D.P.; Johnson, R.J.; Lavu, S.; Iffland, A.; Elliott, P.J.; Lambert, P.D.; Elliston, K.O.; Jirousek, M.R.; Milne, J.C.; Boss, O. Small molecule activators of SIRT1 replicate signaling pathways triggered by calorie restriction in vivo. BMC Syst. Biol. 2009, 3, article no. 31. [Google Scholar]
- Chaudhary, N.; Pfluger, P.T. Metabolic benefits from Sirt1 and Sirt1 activators. Curr. Opin. Clin. Nutr. Metab. Care 2009, 12, 431–437. [Google Scholar] [CrossRef]
- Beher, D.; Wu, J.; Cumine, S.; Kim, K.W.; Lu, S.C.; Atangan, L.; Wang, M. Resveratrol is not a direct activator of SIRT1 enzyme activity. Chem. Biol. Drug Des. 2009. [Google Scholar] [CrossRef]
- Xie, Z.; Dong, Y.; Zhang, M.; Cui, M.Z.; Cohen, R.A.; Riek, U.; Neumann, D.; Schlattner, U.; Zou, M.H. Activation of protein kinase Cζ by peroxynitrite regulates LKB1-dependent AMP-activated protein kinase in cultured endothelial cells. J. Biol. Chem. 2006, 281, 6366–6375. [Google Scholar]
- Shin, S.M.; Cho, I.J.; Kim, S.G. Resveratrol protects mitochondria against oxidative stress through AMP-activated protein kinase-mediated glycogen synthase kinase-3beta inhibition downstream of poly(ADP-ribose)polymerase-LKB1 pathway. Mol. Pharmacol. 2009, 76, 884–895. [Google Scholar] [CrossRef]
- Albani, D.; Polito, L.; Forloni, G. Sirtuins as novel targets for Alzheimer's disease and other neurodegenerative disorders: Experimental and genetic evidence. J. Alzheimers Dis. 2009, ahead . [Google Scholar] [CrossRef]
- Pirola, L.; Fröjdö, S. Resveratrol: One molecule, many targets. IUBMB Life 2008, 60, 323–332. [Google Scholar] [CrossRef]
- Allard, S.J.; Perez, E.; Zou, S.; de Cabo, R. Dietary activators of Sirt1. Mol. Cell. Endocrinol. 2009, 299, 58–63. [Google Scholar] [CrossRef]
- Alcaín, F.J.; Villalba, J.M. Sirtuin activators. Expert Opin. Ther. Pat. 2009, 19, 403–414. [Google Scholar] [CrossRef]
- Mai, A.; Valente, S.; Meade, S.; Carafa, V.; Tardugno, M.; Nebbioso, A.; Galmozzi, A.; Mitro, N.; De Fabiani, E.; Altucci, L.; Kazantsev, A. Study of 1,4-dihydropyridine structural scaffold: Discovery of novel sirtuin activators and inhibitors. J. Med. Chem. 2009, 52, 5496–5504. [Google Scholar]
- Pfister, J.A.; Ma, C.; Morrison, B.E.; D'Mello, S.R. Opposing effects of sirtuins on neuronal survival: SIRT1-mediated neuroprotection is independent of its deacetylase activity. PLoS One 2008, 3, article no. e4090. [Google Scholar]
- Allison, S.J.; Milner, J. SIRT3 is pro-apoptotic and participates in distinct basal apoptotic pathways. Cell Cycle 2007, 6, 2669–2677. [Google Scholar] [CrossRef]
- Sundaresan, N.R.; Samant, S.A.; Pillai, V.B.; Rajamohan, S.B.; Gupta, M.P. SIRT3 is a stress-responsive deacetylase in cardiomyocytes that protects cells from stress-mediated cell death by deacetylation of Ku70. Mol. Cell. Biol. 2008, 28, 6384–6401. [Google Scholar] [CrossRef]
- Rathaus, M.; Lerrer, B.; Cohen, H.Y. DeubiKuitylation: A novel DUB enzymatic activity for the DNA repair protein, Ku70. Cell Cycle 2009, 8, 1843–1852. [Google Scholar] [CrossRef]
- Rose, G.; Dato, S.; Altomare, K.; Bellizzi, D.; Garasto, S.; Greco, V.; Passarino, G.; Feraco, E.; Mari, V.; Barbi, C.; BonaFe, M.; Franceschi, C.; Tan, Q.; Boiko, S.; Yashin, A.I.; De Benedictis, G. Variability of the SIRT3 gene, human silent information regulator Sir2 homologue, and survivorship in the elderly. Exp. Gerontol. 2003, 38, 1065–1070. [Google Scholar] [CrossRef]
- Bellizzi, D.; Rose, G.; Cavalcante, P.; Covello, G.; Dato, S.; De Rango, F.; Greco, V.; Maggiolini, M.; Feraco, E.; Mari, V.; Franceschi, C.; Passarino, G.; De Benedictis, G. A novel VNTR enhancer within the SIRT3 gene, a human homologue of SIR2, is associated with survival at oldest ages. Genomics 2005, 85, 258–263. [Google Scholar]
- Hardeland, R. Melatonin: Signaling mechanisms of a pleiotropic agent. BioFactors 2009, 35, 183–192. [Google Scholar] [CrossRef]
- Hardeland, R. Melatonin, hormone of darkness and more – occurrence, control mechanisms, actions and bioactive metabolites. Cell. Mol. Life Sci. 2008, 65, 2001–2018. [Google Scholar] [CrossRef]
- Tan, D.X.; Manchester, L.C.; Hardeland, R.; Lopez-Burillo, S.; Mayo, J.C.; Sainz, R.M.; Reiter, R.J. Melatonin - a hormone, a tissue factor, an autocoid, a paracoid, and an antioxidant vitamin. J. Pineal Res. 2003, 34, 75–78. [Google Scholar] [CrossRef]
- Pandi-Perumal, S.R.; Srinivasan, V.; Maestroni, G.J.M.; Cardinali, D.P.; Poeggeler, B.; Hardeland, R. Melatonin - Nature’s most versatile biological signal? FEBS J. 2006, 273, 2813–2838. [Google Scholar] [CrossRef]
- Hardeland, R.; Pandi-Perumal, S.R. Melatonin, a potent agent in antioxidative defense: Actions as a natural food constituent, gastrointestinal factor, drug and prodrug. Nutr. Metab.(Lond.) 2005, 2, 22. [Google Scholar] [CrossRef]
- Matsubara, E.; Bryant-Thomas, T.; Pacheco Quinto, J.; Henry, T.L.; Poeggeler, B.; Herbert, D.; Cruz-Sanchez, F.; Chyan, Y.J.; Smith, M.A.; Perry, G.; Shoji, M.; Abe, K.; Leone, A.; Grundke-Ikbal, I.; Wilson, G.L.; Ghiso, J.; Williams, C.; Refolo, L.M.; Pappolla, M.A.; Chain, D.G.; Neria, E. Melatonin increases survival and inhibits oxidative and amyloid pathology in a transgenic model of Alzheimer's disease. J. Neurochem. 2003, 85, 1101–1108. [Google Scholar]
- Tricoire, H.; Locatelli, A.; Chemineau, P.; Malpaux, B. Melatonin enters the cerebrospinal fluid through the pineal recess. Endocrinology 2002, 143, 84–90. [Google Scholar] [CrossRef]
- Messner, M.; Hardeland, R.; Rodenbeck, A.; Huether, G. Tissue retention and subcellular distribution of continuously infused melatonin in rats under near physiological conditions. J. Pineal Res. 1998, 25, 251–259. [Google Scholar] [CrossRef]
- López, A.; García, J.A.; Escames, G.; Venegas, C.; Ortiz, F.; López, L.C.; Acuña-Castroviejo, D. Melatonin protects the mitochondria from oxidative damage reducing oxygen consumption, membrane potential, and superoxide anion production. J. Pineal Res. 2009, 46, 188–198. [Google Scholar] [CrossRef]
- Tan, D.X.; Chen, L.D.; Poeggeler, B.; Manchester, L.C.; Reiter, R.J. Melatonin: a potent, endogenous hydroxyl radical scavenger. Endocr. J. 1993, 1, 57–60. [Google Scholar]
- Reiter, R.J. Oxidative damage in the central nervous system: Protection by melatonin. Prog. Neurobiol. 1998, 56, 359–384. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.X.; Osuna, C.; Gitto, E. Actions of melatonin in the reduction of oxidative stress. A review. J. Biomed. Sci. 2000, 7, 444–458. [Google Scholar] [CrossRef]
- Tan, D.X.; Reiter, R.J.; Manchester, L.C.; Yan, M.T.; El-Sawi, M.; Sainz, R.M.; Mayo, J.C.; Kohen, R.; Allegra, M.; Hardeland, R. Chemical and physical properties and potential mechanisms: Melatonin as a broad spectrum antioxidant and free radical scavenger. Curr. Top. Med. Chem. 2002, 2, 181–197. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.X.; Mayo, J.C.; Sainz, R.M.; Leon, J.; Czarnocki, Z. Melatonin as an antioxidant: biochemical mechanisms and pathophysiological implications in humans. Acta Biochim. Pol. 2003, 50, 1129–1146. [Google Scholar]
- Reiter, R.J.; Tan, D.X.; Manchester, L.C.; Tamura, H. Melatonin defeats neurally-derived free radicals and reduces the associated neuromorphological and neurobehavioral damage. J. Physiol. Pharmacol. 2007, 58 (Suppl. 6), 5–22. [Google Scholar]
- Rosen, J.; Than, N.N.; Koch, D.; Poeggeler, B.; Laatsch, H.; Hardeland, R. Interactions of melatonin and its metabolites with the ABTS cation radical: extension of the radical scavenger cascade and formation of a novel class of oxidation products, C2-substituted 3-indolinones. J. Pineal Res. 2006, 41, 374–381. [Google Scholar] [CrossRef]
- Ressmeyer, A.R.; Mayo, J.C.; Zelosko, V.; Sáinz, R.M.; Tan, D.X.; Poeggeler, B.; Antolín, I.; Zsizsik, B.K.; Reiter, R.J.; Hardeland, R. Antioxidant properties of the melatonin metabolite N1-acetyl-5-methoxykynuramine (AMK): scavenging of free radicals and prevention of protein destruction. Redox Rep. 2003, 8, 205–213. [Google Scholar] [CrossRef]
- Hardeland, R.; Poeggeler, B. Melatonin beyond its classical functions. Open Physiol. J. 2008, 1, 1–23. [Google Scholar] [CrossRef]
- Golombek, D.A.; Escolar, E.; Cardinali, D.P. Melatonin-induced depression of locomotor activity in hamsters: Time-dependency and inhibition by the central-type benzodiazepine antagonist Ro 15-1788. Physiol. Behav. 1991, 49, 1091–1097. [Google Scholar] [CrossRef]
- Golombek, D.A.; Escolar, E.; Burin, L.J.; De Brito Sánchez, M.G.; Fernández Duque, D.; Cardinali, D.P. Chronopharmacology of melatonin: Inhibition by benzodiazepine antagonism. Chronobiol. Int. 1992, 9, 124–131. [Google Scholar] [CrossRef]
- Golombek, D.A.; Fernández Duque, D.; De Brito Sánchez, M.G.; Burin, L.; Cardinali, D.P. Time-dependent anticonvulsant activity of melatonin in hamsters. Eur. J. Pharmacol. 1992, 210, 253–258. [Google Scholar] [CrossRef]
- Muñoz-Hoyos, A.; Sánchez-Forte, M.; Molina-Carballo, A.; Escames, G.; Martin-Medina, E.; Reiter, R.J.; Molina-Font, J.A.; Acuña-Castroviejo, D. Melatonin's role as an anticonvulsant and neuronal protector: Experimental and clinical evidence. J. Child Neurol. 1998, 13, 501–509. [Google Scholar] [CrossRef]
- Molina-Carballo, A.; Muñoz-Hoyos, A.; Sánchez-Forte, M.; Uberos-Fernández, J.; Moreno-Madrid, F.; Acuña-Castroviejo, D. Melatonin increases following convulsive seizures may be related to its anticonvulsant properties at physiological concentrations. Neuropediatrics 2007, 38, 122–125. [Google Scholar] [CrossRef]
- Fenoglio-Simeone, K.; Mazarati, A.; Sefidvash-Hockley, S.; Shin, D.; Wilke, J.; Milligan, H.; Sankar, R.; Rho, J.M.; Maganti, R. Anticonvulsant effects of the selective melatonin receptor agonist ramelteon. Epilepsy Behav. 2009, 16, 52–57. [Google Scholar] [CrossRef]
- Solmaz, I.; Gürkanlar, D.; Gökçil, Z.; Göksoy, C.; Ozkan, M.; Erdoğan, E. Antiepileptic activity of melatonin in guinea pigs with pentylenetetrazol-induced seizures. Neurol. Res. 2009. [Google Scholar] [CrossRef]
- Pozo, D.; Reiter, J.R.; Calvo, J.R.; Guerrero, J.M. Physiological concentrations of melatonin inhibit nitric oxide synthase in rat cerebellum. Life Sci. 1994, 55, PL455–PL460. [Google Scholar] [CrossRef]
- León, J.; Vives, F.; Crespo, E.; Camacho, E.; Espinosa, A.; Gallo, M.A.; Escames, G.; Acuña-Castroviejo, D. Modification of nitric oxide synthase activity and neuronal response in rat striatum by melatonin and kynurenine derivatives. J. Neuroendocrinol. 1998, 10, 297–302. [Google Scholar]
- Chang, H.M.; Ling, E.A.; Chen, C.F.; Lue, J.H.; Wen, C.Y.; Shieh, J.Y. Melatonin attenuates the neuronal NADPH-d/NOS expression in the nodose ganglion of acute hypoxic rats. J. Pineal Res. 2002, 32, 65–73. [Google Scholar] [CrossRef]
- Chang, H.M.; Tseng, C.Y.; Wei, I.H.; Lue, J.H.; Wen, C.Y.; Shieh, J.Y. Melatonin restores the cytochrome oxidase reactivity in the nodose ganglia of acute hypoxic rats. J. Pineal Res. 2005, 39, 206–214. [Google Scholar] [CrossRef]
- Tjong, Y.W.; Li, M.F.; Hung, M.W.; Fung, M.L. Melatonin ameliorates hippocampal nitric oxide production and large conductance calcium-activated potassium channel activity in chronic intermittent hypoxia. J. Pineal Res. 2008, 44, 234–243. [Google Scholar] [CrossRef]
- Escames, G.; Acuña-Castroviejo, D.; López, L.C.; Tan, D.X.; Maldonado, M.D.; Sánchez-Hidalgo, M.; León, J.; Reiter, R.J. Pharmacological utility of melatonin in the treatment of septic shock: experimental and clinical evidence. J. Pharm. Pharmacol. 2006, 58, 1153–1165. [Google Scholar] [CrossRef]
- Escames, G.; López, L.C.; Ortiz, F.; López, A.; García, J.A.; Ros, E.; Acuña-Castroviejo, D. Attenuation of cardiac mitochondrial dysfunction by melatonin in septic mice. FEBS J. 2007, 274, 2135–2147. [Google Scholar] [CrossRef]
- Hardeland, R.; Backhaus, C.; Fadavi, A.; Hess, M. N1-acetyl-5-methoxykynuramine contrasts with other tryptophan metabolites by a peculiar type of NO scavenging: cyclization to a cinnolinone prevents formation of unstable nitrosamines. J. Pineal Res. 2007, 43, 104–105. [Google Scholar] [CrossRef]
- Hardeland, R.; Backhaus, C.; Fadavi, A. Reactions of the NO redox forms NO+, •NO and HNO (protonated NO–) with the melatonin metabolite N1-acetyl-5-methoxykynuramine. J. Pineal Res. 2007, 43, 382–388. [Google Scholar] [CrossRef]
- Hardeland, R.; Tan, D.X.; Reiter, R.J. Kynuramines, metabolites of melatonin and other indoles: the resurrection of an almost forgotten class of biogenic amines. J. Pineal Res. 2009, 47, 109–126. [Google Scholar] [CrossRef]
- León, J.; Escames, G.; Rodríguez, M.I.; López, L.C.; Tapias, V.; Entrena, A.; Camacho, E.; Carrión, M.D.; Gallo, M.A.; Espinosa, A.; Tan, D.X.; Reiter, R.J.; Acuña-Castroviejo, D. Inhibition of neuronal nitric oxide synthase activity by N1-acetyl-5-methoxykynuramine, a brain metabolite of melatonin. J. Neurochem. 2006, 98, 2023–2033. [Google Scholar] [CrossRef]
- Tapias, V.; Escames, G.; López, L.C.; López, A.; Camacho, E.; Carrión, M.D.; Entrena, A.; Gallo, M.A.; Espinosa, A.; Acuña-Castroviejo, D. Melatonin and its brain metabolite N1-acetyl-5-methoxykynuramine prevent mitochondrial nitric oxide synthase induction in parkinsonian mice. J. Neurosci. Res. 2009, 87, 3002–3010. [Google Scholar] [CrossRef]
- Srinivasan, V.; Pandi-Perumal, S.R.; Maestroni, G.J.M.; Esquifino, A.I.; Hardeland, R.; Cardinali, D.P. Role of melatonin in neurodegenerative diseases. Neurotox. Res. 2005, 7, 293–318. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.X.; Jou, M.J.; Korkmaz, A.; Manchester, L.C.; Paredes, S.D. Biogenic amines in the reduction of oxidative stress: Melatonin and its metabolites. Neuroendocrinol. Lett. 2008, 29, 391–398. [Google Scholar]
- Sharman, E.H.; Bondy, S.C. Effects of age and dietary antioxidants on cerebral electron transport chain activity. Neurobiol. Aging 2001, 22, 629–634. [Google Scholar] [CrossRef]
- Bondy, S.C.; Sharman, E.H. Melatonin and the aging brain. Neurochem. Int. 2007, 50, 571–580. [Google Scholar] [CrossRef]
- Petrosillo, G.; Fattoretti, P.; Matera, M.; Ruggiero, F.M.; Bertoni-Freddari, C.; Paradies, G. Melatonin prevents age-related mitochondrial dysfunction in rat brain via cardiolipin protection. Rejuvenation Res. 2008, 11, 935–943. [Google Scholar] [CrossRef]
- Rodríguez, M.I.; Escames, G.; López, L.C.; López, A.; García, J.A.; Ortiz, F.; Sánchez, V.; Romeu, M.; Acuña-Castroviejo, D. Improved mitochondrial function and increased life span after chronic melatonin treatment in senescent prone mice. Exp. Gerontol. 2008, 43, 749–756. [Google Scholar] [CrossRef]
- Andrabi, S.A.; Sayeed, I.; Siemen, D.; Wolf, G.; Horn, T.F. Direct inhibition of the mitochondrial permeability transition pore: a possible mechanism responsible for anti-apoptotic effects of melatonin. FASEB J. 2004, 18, 869–871. [Google Scholar]
- Acuña-Castroviejo, D.; Escames, G.; León, J.; Carazo, A.; Khaldy, H. Mitochondrial regulation by melatonin and its metabolites. Adv. Exp. Med. Biol. 2003, 527, 549–557. [Google Scholar] [CrossRef]
- Gutierrez-Cuesta, J.; Tajes, M.; Jiménez, A.; Coto-Montes, A.; Camins, A.; Pallàs, M. Evaluation of potential pro-survival pathways regulated by melatonin in a murine senescence model. J. Pineal Res. 2008, 45, 497–505. [Google Scholar] [CrossRef]
- Chang, H.M.; Wu, U.I.; Lan, C.T. Melatonin preserves longevity protein (sirtuin 1) expression in the hippocampus of total sleep-deprived rats. J. Pineal Res. 2009, 47, 211–220. [Google Scholar] [CrossRef]
- Tajes, M.; Gutierrez-Cuesta, J.; Ortuño-Sahagun, D.; Camins, A.; Pallàs, M. Anti-aging properties of melatonin in an in vitro murine senescence model: involvement of the sirtuin 1 pathway. J. Pineal Res. 2009, 47, 228–237. [Google Scholar] [CrossRef]
- Nakahata, Y.; Sahar, S.; Astarita, G.; Kaluzova, M.; Sassone-Corsi, P. Circadian control of the NAD+ salvage pathway by CLOCK-SIRT1. Science 2009, 324, 654–657. [Google Scholar] [CrossRef]
- Jung-Hynes, B.; Ahmad, N. SIRT1 controls circadian clock circuitry and promotes cell survival: a connection with age-related neoplasms. FASEB J. 2009, 23, 2803–2809. [Google Scholar] [CrossRef]
- Nowak, A.; Rahman, H.; Heer, C.; Schueth, A.; Laatsch, H.; Hardeland, R. Reactions of the melatonin metabolite N1-acetyl-5-methoxykynuramine (AMK) with the tyrosine side-chain fragment, 4-ethylphenol. Redox Rep. 2008, 13, 102–108. [Google Scholar] [CrossRef]
- Weishaupt, J.H.; Bartels, C.; Pölking, E.; Dietrich, J.; Rohde, G.; Poeggeler, B.; Mertens, N.; Sperling, S.; Bohn, M.; Huether, G.; Schneider, A.; Bach, A.; Sirén, A.L.; Hardeland, R.; Bähr, M.; Nave, K.A.; Ehrenreich, H. Reduced oxidative damage in ALS by high-dose enteral melatonin treatment. J. Pineal Res. 2006, 41, 313–321. [Google Scholar] [CrossRef]
- Hardeland, R. New approaches in the management of insomnia: weighing the advantages of prolonged release melatonin and synthetic melatoninergic agonists. Neuropsychiatr. Dis. Treat. 2009, 5, 341–354. [Google Scholar] [CrossRef]
- Hardeland, R. Tasimelteon, a melatonin agonist for the treatment of insomnia and circadian rhythm sleep disorders. Curr. Opin. Investig. Drugs 2009, 10, 691–701. [Google Scholar]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hardeland, R. Neuroprotection by Radical Avoidance: Search for Suitable Agents. Molecules 2009, 14, 5054-5102. https://doi.org/10.3390/molecules14125054
Hardeland R. Neuroprotection by Radical Avoidance: Search for Suitable Agents. Molecules. 2009; 14(12):5054-5102. https://doi.org/10.3390/molecules14125054
Chicago/Turabian StyleHardeland, Rüdiger. 2009. "Neuroprotection by Radical Avoidance: Search for Suitable Agents" Molecules 14, no. 12: 5054-5102. https://doi.org/10.3390/molecules14125054
APA StyleHardeland, R. (2009). Neuroprotection by Radical Avoidance: Search for Suitable Agents. Molecules, 14(12), 5054-5102. https://doi.org/10.3390/molecules14125054




