Total Syntheses of (±)-Gusanlung A, (±)-Gusanlung D and 8-Oxyberberrubine and the Uncertainty Concerning the Structures of (-)-Gusanlung A, (-)-Gusanlung D and 8-Oxyberberrubine
Abstract
:Introduction

Results and Discussion
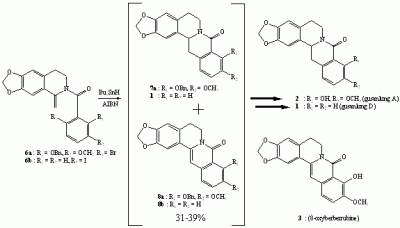
Syntheses of (±)-gusanlung A (2) and 8-oxyberberrubine (3)


Synthesis of (±)-gusanlung D
| (position) | (–)-gusanlung D CDCl3 [1] m.p. 250-251 °C | (–)-gusanlung DCDCl3 [4] m.p. 195-197 °C | (±)-gusanlung DCDCl3 [2] m.p. 175-177 °C | (±)-gusanlung D CDCl3 [this work] m.p. 175-176 °C |
| 1H | 1H | 1H | 1H | |
| 1 | 7.35 (s) | 6.71 (s) | 6.76 (d) | 6.72 (s) |
| 4 | 6.80 (s) | 6.67 (s) | 6.76 (d) | 6.67 (s) |
| 5α | 2.70-3.40 (m) | 2.7-2.8 (m) | 2.83-3.35 (m) | 2.70-2.82 (m) |
| 5β | 2.70-3.40 (m) | 2.82-3.02 (m) | 2.83-3.35 (m) | 2.87-3.07 (m) |
| 6α | 2.70-3.40 (m) | 2.82-3.02 (m) | 2.83-3.35 (m) | 2.87-3.07 (m) |
| 6β | 4.8 (m) | 4.93-4.99 (m) | 4.7-5.1 (m) | 4.88-4.99 (m) |
| 9 | 8.07 (d, 8.0) | 8.13 (d, 7.4) | 8.1-8.37 (m) | 8.13 (dd, 7.6, 1.4) |
| 10 | 7.29-7.41 (m) | 7.34-7.40 (m) | 7.25-7.65 (m) | 7.39 (br t, 7.4) |
| 11 | 7.29-7.41 (m) | 7.41-7.49 (m) | 7.25-7.65 (m) | 7.46 (dt, 7.4, 1.5) |
| 12 | 7.29-7.41 (m) | 7.24 (d, 7.4) | 7.25-7.65 (m) | 7.22-7.29 (m) |
| 13α | 2.70-3.40 (m) | 2.82-3.02 (m) | 2.83-3.35 (m) | 2.87-3.07 (m) |
| 13β | 2.70-3.40 (m) | 3.18 (dd, 15.3, 3.7) | 2.83-3.35 (m) | 3.18 (dd, 15.7, 3.7) |
| 14 | 3.95 (m) | 4.83 (dd, 13.3, 3.7) | 4.7-5.1 (m) | 4.84 (dd, 13.3, 3.7) |
| OCH2O | 6.20, 6.06 (s) | 5.96 (s) | 5.93 (s) | 5.96 (s) |
| (position) | (–)-gusanlung DCDCl3 [1] m.p. 250-251 °C | (–)-gusanlung DCDCl3 [4] m.p. 195-197 °C | (±)-gusanlung DCDCl3 [3] m.p. 175-177 °C | (±)-gusanlung DCDCl3 [this work] m.p. 175-176 °C |
| 13C | 13C | 13C | 13C | |
| 1 | 107.3 | 105.8 | 105.97 | 105.9 |
| 2 | 135.0 | 146.5b | 146.57 | 146.6c |
| 3 | 147.0 | 146.7b | 146.77 | 146.8c |
| 4 | 107.5 | 108.6 | 108.81 | 108.7 |
| 4a | 126.5 | 128.8 | 128.85 | 128.9 |
| 5 | 29.7 | 29.6 | 29.61 | 29.7 |
| 6 | 42.0 | 38.7 | 38.49 | 38.8 |
| 8 | 162.0 | 164.5 | 158.67 | 164.6 |
| 8a | 117.3 | 137.2 | 137.24 | 137.3 |
| 9 | 128.7a | 128.6 | 128.60 | 128.6 |
| 10 | 127.9a | 127.3 | 127.37 | 127.4* |
| 11 | 127.1a | 131.8 | 132.33 | 131.9* |
| 12 | 126.8a | 126.8 | 126.87 | 126.9 |
| 12a | 124.6 | 129.0 | 131.81 | 129.1 |
| 13 | 33.5 | 38.1 | 37.78 | 38.1 |
| 14 | 49.4 | 55.2 | 55.18 | 55.3 |
| 14a | 126.5 | 128.5 | 128.55 | 128.6 |
| OCH2O | 100.9 | 101.1 | 101.00 | 101.1 |
(position) | (-)-gusanlung A (DMSO-d6) [6] m.p. 260-262 °C | (±)-gusanlung A (DMSO-d6) [this work] m.p. 188-189 °C | (±)-gusanlung A (CDCl3) [this work] m.p. 188-189 °C |
| 1H | 1H | 1H | |
| 1 | 6.96 (s) | 7.00 (s) | 6.71 (s) |
| 4 | 6.80 (s) | 6.79 (s) | 6.66 (s) |
| 5 | 2.73-2.81 (m) | 2.75-2.89 (m) | 2.72-2.84 (m) |
| 6α | 2.73-2.81 (m) | 2.89-3.01 (m) | 2.94-3.40 (m) |
| 6β | 4.71 (m) | 4.69-4.59(m) | 4.80-4.87 (m) |
| 11 | 6.99 (d, 8.1) | 7.09 (d, 8.1) | 6.94 (d, 8.1) |
| 12 | 6.86 (d, 8.1) | 6.71 (d, 8.1) | 6.63 (d, 8.1) |
| 13α | 3.13 (dd, 15.3, 3.1) | 3.36 (dd, 15.2, 3.6) | 3.14 (dd, 15.2, 3.8) |
| 13β | 2.62 (dd, 15.3, 13.3) | 2.66-2.75 (m) | 2.80-2.94 (m) |
| 14 | 4.68 (dd, 13.3, 3.1) | 4.84 (dd, 13.3, 3.4) | 4.80 (dd, 13.6, 3.5) |
| C10-OCH3 | 3.76 (s) | 3.78 (s) | 3.90 (s) |
| OCH2O | 5.98, 5.99 (s) | 5.98, 6.00 (s) | 5.96 (s) |
| OH | - | 12.88 (s) | 12.83 (s) |
| (position) | (-)-gusanlung A (DMSO-d6) [6] m.p. 260-262 °C | (±)-gusanlung A (DMSO-d6) [this work] m.p. 188-189 °C | (±)-gusanlung A (CDCl3) [this work] m.p. 188-189 °C | (±)-gusanlung A (DMSO-d6) [this work] | |
| HMBC | |||||
| 13C | 13C | 13C | 2J | 3J | |
| 1 | 106.1 | 106.6 | 105.8 | C-2 | C-3, 4a, 14 |
| 2 | 145.9a | 146.7c | 146.8* | - | - |
| 3 | 147.7a | 146.5c | 146.7* | - | - |
| 4 | 107.8 | 108.7 | 108.6 | C-3 | C-2, 5, 14a |
| 4a | 129.1b | 128.3 | 128.1 | - | - |
| 5 | 29.0 | 28.9 | 29.4 | C-4a, 6 | C-4, 14a |
| 6 | 37.8 | 38.5 | 38.4 | C-5 | C-4a, 8, 14 |
| 8 | 161.4 | 168.4 | 168.6 | - | - |
| 8a | 122.3b | 111.4 | 111.4 | - | - |
| 9 | 149.7a | 151.4 | 151.8 | - | - |
| 10 | 145.7a | 147.2 | 147.5 | - | - |
| 11 | 118.9 | 116.7 | 115.4 | C-10 | C-9, 12a |
| 12 | 122.1 | 116.9 | 116.1 | C-11 | C-8a, 10, 13 |
| 12a | 128.2b | 129.6 | 128.7 | - | - |
| 13 | 37.7 | 35.9 | 37.1 | C-12a, 14 | C-8a, 12, 14a |
| 14 | 54.4 | 55.4 | 55.7 | C-13, 14a | - |
| 14a | 129.3b | 129.1 | 128.4 | - | - |
| C10-OCH3 | 60.5 | 56.3 | 56.3 | - | C-10 |
| OCH2O | 100.5 | 101.3 | 101.2 | - | C-2, 3 |
| OH | C-9 | C-8a, 10 | |||
(position) | natural 8-oxy-berberrubine (3) CDCl3 [1] m.p. 240-241 °C | synthetic 8-oxy-berberrubine (3) CDCl3 [this work] m.p. 238-239 °C | natural 8-oxy-berberrubine (3) CDCl3 [1] m.p. 240-241 °C | synthetic 8-oxy-berberrubine (3) CDCl3 [this work] m.p. 238-239 °C | synthetic 8-oxyberberrubine (3) (CDCl3) [this work] | |
| HMBC | ||||||
| 1H | 1H | 13C | 13C | 2J | 3J | |
| 1 | 6.83 (s) | 7.21 (s) | 104.0 | 104.8 | C-2 | C-3, 4a, 14 |
| 2 | 141.6 | 147.5* | - | - | ||
| 3 | 146.4 | 148.6* | - | - | ||
| 4 | 6.72 (s) | 6.71 (s) | 107.1 | 108.0 | C-3 | C-2, 5, 14a |
| 4a | 109.6 | 129.5 | - | - | ||
| 5 | 2.91 (t, 7.2) | 2.92 (t, 6.1) | 28.4 | 28.4 | C-4a, 6 | C-4, 14a |
| 6 | 4.27 (t, 7.2) | 4.27 (t, 6.1) | 39.1 | 39.1 | C-5 | C-4a, 8, 14 |
| 8 | 164.0 | 165.4 | - | - | ||
| 8a | 129.9 | 111.0 | - | - | ||
| 9 | 149.0 | 150.3 | - | - | ||
| 10 | 147.5 | 144.9 | - | - | ||
| 11 | 7.30 (AB q, 8.0) | 7.28 (d, 8.5) | 114.9 | 119.1 | C-10 | C-9, 12a |
| 12 | 7.00 (AB q, 8.0) | 6.99 (d, 8.5) | 120.0 | 115.3 | C-11 | C-8a, 10, 13 |
| 12a | 128.9 | 130.5 | - | - | ||
| 13 | 7.21 (s) | 6.83 (s) | 103.6 | 103.6 | C-14 | C-8a, 12, 14a |
| 14 | 133.6 | 134.6 | - | - | ||
| 14a | 122.1 | 123.5 | - | - | ||
| C10-OCH3 | 3.96 (s) | 3.97 (s) | 56.7 | 56.7 | - | C-10 |
| OCH2O | 6.02 (s) | 6.02 (s) | 100.6 | 101.5 | - | C-2, 3 |
| OH | - | 13.14 | - | - | - | - |
Antimicrobial activity
Conclusions
Experimental
General
Minimum inhibitory concentration (MIC)
References
- Zhang, J-S.; Men-Olivier, L.L.; Massiot, G. Isoquinoline alkaloids from Acangelisia gusanlung. Phytochemistry 1995, 39, 439–442. [Google Scholar] [CrossRef]
- Kessar, S.V.; Singh, P.; Vohra, R.; Kaur, N.P.; Venugopal, D. Facile generation and trapping of α-Oxo-o-quinodimethanes: Synthesis of 3-aryl-3,4-dihydroisocoumarins and protoberberines. J. Org. Chem. 1992, 57, 6716–6720. [Google Scholar] [CrossRef]
- Reimann, E.; Grasberger, F.; Polborn, K. Protoberberines from Reissert-Compounds VIII [1]. Oxazoloisoquinolines, new and efficient educts for the synthesis of 8-oxoprotoberberines. Monatsh. Chem. 2003, 134, 991–1014. [Google Scholar] [CrossRef]
- Chrzanowska, M.; Dreas, A.; Rozwadowska, M.D. Asymmetric synthesis of (R)-(+) - and (S)-(-)-2,3-methylenedioxy-8-oxoberbine (gusanlung D). Tetrahedron Asymmetry 2004, 15, 1113–1120. [Google Scholar] [CrossRef]
- Chang, J-K.; Chang, N-C. Total synthesis of (±)-gusanlung D. Tetrahedron 2008, 64, 3483–3487. [Google Scholar] [CrossRef]
- Zhang, J-S.; Chen, Z.-L. Two new 8-oxotetrahydroprotoberberine alkaloids, gusanlung A and B, from Acangelisia gusanlung. Planta Med. 1991, 57, 457–459. [Google Scholar] [CrossRef]
- Nimgirawath, S.; Lorpitthaya, R.; Wanbanjob, A.; Taechowisan, T.; Shen, Y-M. Total synthesis and the biological activities of (±)-norannuradhapurine. Molecules 2009, 14, 89–101. [Google Scholar]
- Ishiwata, S.; Nozaki, T. Syntheses of isoquinoline derivatives. V. Syntheses of alkyl derivertives of the pyridine nucleus. J. Pharm. Soc. Jpn. 1951, 71, 1257–1260. [Google Scholar]
- National Committee for Clinical Laboratory Standards, Reference method for broth dilution antifungal susceptibility testing of yeasts. Approved standard M27-A; National Committee for Clinical Laboratory Standards: Wayne, Pa, USA, 1997.
- Sample Availability: All stable products reported in this paper are available from the authors.
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Nimgirawath, S.; Udomputtimekakul, P.; Apornpisarn, T.; Wanbanjob, A.; Taechowisan, T. Total Syntheses of (±)-Gusanlung A, (±)-Gusanlung D and 8-Oxyberberrubine and the Uncertainty Concerning the Structures of (-)-Gusanlung A, (-)-Gusanlung D and 8-Oxyberberrubine. Molecules 2009, 14, 726-737. https://doi.org/10.3390/molecules14020726
Nimgirawath S, Udomputtimekakul P, Apornpisarn T, Wanbanjob A, Taechowisan T. Total Syntheses of (±)-Gusanlung A, (±)-Gusanlung D and 8-Oxyberberrubine and the Uncertainty Concerning the Structures of (-)-Gusanlung A, (-)-Gusanlung D and 8-Oxyberberrubine. Molecules. 2009; 14(2):726-737. https://doi.org/10.3390/molecules14020726
Chicago/Turabian StyleNimgirawath, Surachai, Phansuang Udomputtimekakul, Thitima Apornpisarn, Asawin Wanbanjob, and Thongchai Taechowisan. 2009. "Total Syntheses of (±)-Gusanlung A, (±)-Gusanlung D and 8-Oxyberberrubine and the Uncertainty Concerning the Structures of (-)-Gusanlung A, (-)-Gusanlung D and 8-Oxyberberrubine" Molecules 14, no. 2: 726-737. https://doi.org/10.3390/molecules14020726