Synthesis, Reactions and Biological Evaluation of Some New Naphtho[2,1-b]furan Derivatives Bearing a Pyrazole Nucleus
Abstract
:Introduction
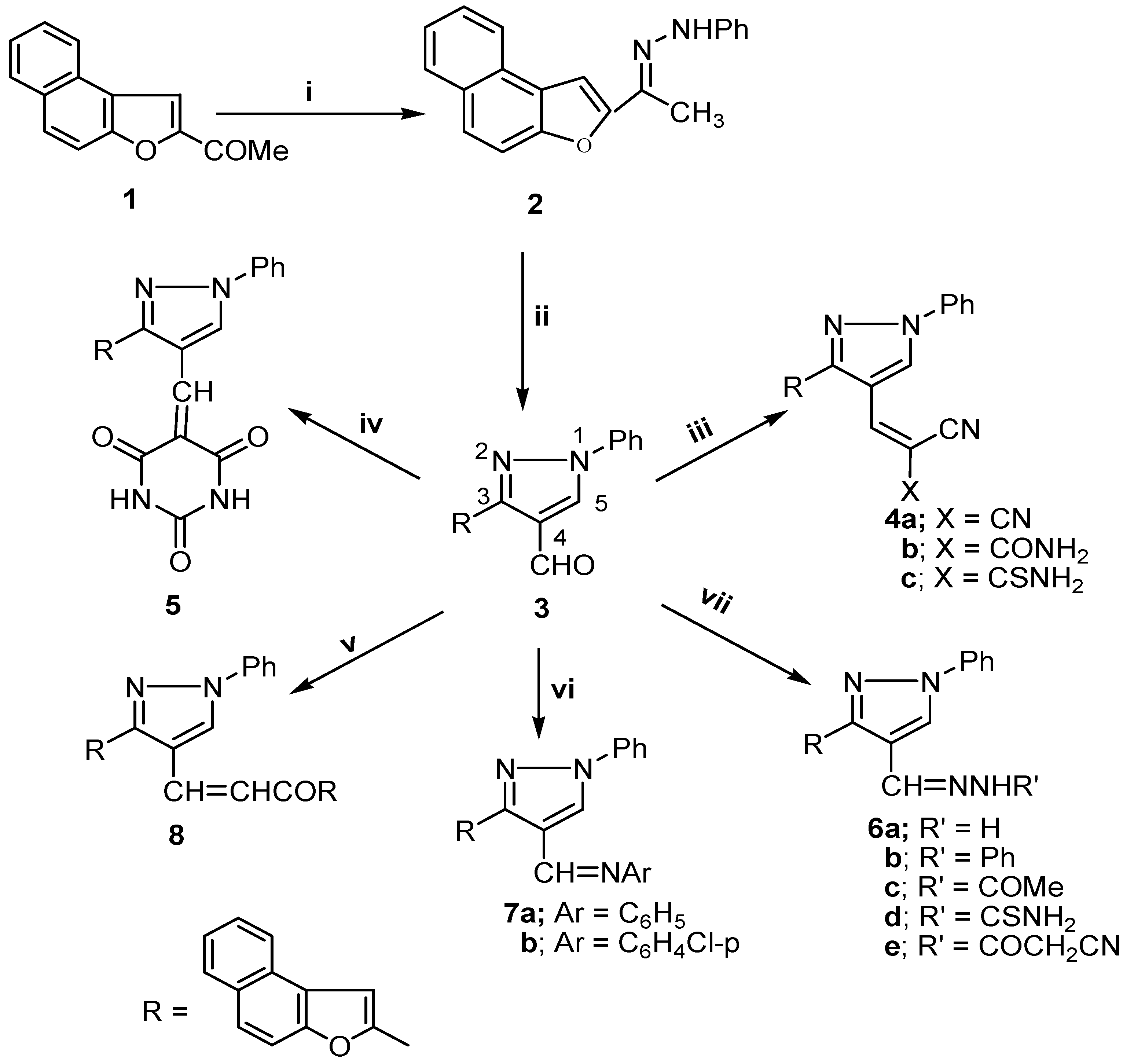
Results and Discussion
Biological activities
Experimental
General
Antibacterial activity
Conclusions
References
- Abd El-Wahab, F.H.A.; Ali, M.F.; El-Agrody, M.A.; Bedear, H.A.; Halawa, A.A.; El-Sherbiny, G.M. Binary Heterocyclic Systems Containing Ethylidine amino Likage:-Synthesis Of Some New Heterocyclic Compounds Bearing Naphtho[2,1,b] furan Moiety. J. Serb. Chem. Soc. 2006, 71, 459–469. [Google Scholar]
- Cavier, R.; Buisson, J.P.; Lemoine, J.; Royer, R. Comparaison des activités antibactériennes et protozoocides de dérivés méthoxylés ou halogénés des nitrobenzofurannes et des nitronaphthofurannes. Eur. J. Med. Chem. 1981, 16, 73–76. [Google Scholar]
- Royer, R.; Buisson, J.P. Recherche sur les dérivés nitrés d’intérêt biologique: XX. Modalités de synthèse des dérivés méthoxylés ou halogénés des nitro-2 naphthofurannes. Eur. J. Med. Chem. 1980, 18, 79–84. [Google Scholar]
- Arnaise, S.; Boeuf, H.; Buisson, J.P.; Cantat, N.; Demerseman, P.; Einhorn, J.; Lamotte, G.; Lemelin, M.; Brimer, P.A.; Perdue, S.W.; Hsie, A.W.; Royer, R.; Kelly, F.; Hofnung, M. Genotoxic activity of 2-nitronaphthofurans and related molecules. Mutagenesis 1986, 3, 217–229. [Google Scholar] [CrossRef]
- Quillardet, P.; Touati, E.; Hofnung, M. Influence of the uvr-dependent nucleotide excision repair on DNA adducts formation and mutagenic spectrum of a potent genotoxic agent: 7-Methoxy-2-nitronaphtho[2,1-b]furan (R7000). Mutat. Res. 1996, 358, 113–122. [Google Scholar] [CrossRef]
- Weill-Thévenet, N.; Buisson, J.P.; Royer, R.; Hofnung, M. Genetic toxicology studies with 2-nitrobenzofurans and 2-nitronaphthofurans. Mutat. Res. 1982, 104, 1–8. [Google Scholar] [CrossRef]
- Touati, E.; Krin, E.; Quillardet, P.; Hofnung, M. 7-Methoxy-2-nitronaphtho[2,1-b]furan (R7000)-induced mutation spectrum in the lacI gene of Escherichia coli: Influence of SOS mutagenesis. Carcinogenesis 1996, 17, 2543–2550. [Google Scholar] [CrossRef] [PubMed]
- Salmon, R.J.; Buisson, J.P.; Aussepe, L.; Royer, R. Induction of sarcomas in rats by subcutaneous injection of 7-methoxy-2-nitro-naphtho[2,1-b]furan (R7000). Carcinogenesis 1985, 6, 109–111. [Google Scholar] [CrossRef] [PubMed]
- Salmon, R.J.; Buisson, J.P.; Vielh, P.; Aussepe, L.; Royer, R. Cancer épidermoïde du rumen induit chez la souris C3H par le méthoxy-7-nitro-2 naphtho [2,1-b] furanne (R7000). C. R. Soc. Biol. 1986, 180, 622–624. [Google Scholar]
- Quillardet, P.; Michel, V.; Arrault, X.; Hofnung, M.; Touati, E. Mutagenic properties of a nitrofuran, 7-methoxy-2-nitro-naphtho[2,1-b]furan (R7000), in lacI transgenic mice. Mutat. Res. 2000, 470, 177–188. [Google Scholar] [CrossRef]
- Touati, E.; Phillips, D.H.; Buisson, J.P.; Quillardet, P.; Royer, R.; Hofnung, M. DNA adduct formation by 7-methoxy-2-nitro-naphtho[2,1-b]furan (R7000), an extremely potent mutagen. Mutagenesis 1989, 4, 254–258. [Google Scholar] [CrossRef] [PubMed]
- Touati, E.; Phillips, D.H.; Quillardet, P.; Hofnung, M. Determination of target nucleotides involved in 7-methoxy-2-nitro-naphtho[2,1-b]furan (R7000)–DNA adduct formation. Mutagenesis 1993, 8, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Weill-Thévenet, N.; Buisson, J.P.; Royer, R.; Hofnung, M. Mutagenic activity of benzofurans and naphthofurans in the Salmonella/microsome assay: 2-Nitro-7-methoxy-naphtho[2,1-b]furan (R7000), a new highly potent mutagenic agent. Mutat. Res. 1981, 88, 355–362. [Google Scholar] [CrossRef]
- Maurizis, J.C.; Madelmont, J.C.; Parry, D.; Meyniel, G. Disposition in rats and mice of 7-methoxy-2-nitronaphtho[2,1-b]furan. Xenobiotica 1985, 15, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Maurizis, J.C.; Madelmont, J.C.; Parry, D.; Dauzonne, D.; Royer, R.; Chabard, J.L. Main excretion metabolites of 7-methoxy-2-nitronaphtho[2,1-b]furan. Xenobiotica 1986, 16, 635–643. [Google Scholar] [CrossRef] [PubMed]
- Latha, K.P.; Vaidya, V.P.; Keshvayya, J.; Vijay Kumar, M.L.; Shreedhara, C. Synthesis of 2-acetylnaphtho[2,1-b]furan. Nat. Acad. Sci. Lett. 2002, 25, 153. [Google Scholar]
- Attaryan, O.; Antanosyan, S.; Panosyan, G.; Asratyan, G.; Matsoyan, S. Vilsmeier formyatiom of 3,5-dimethylpyrazoles. Rus. J. Chem. 2006, 76, 1817–1819. [Google Scholar] [CrossRef]
- El-Agrody, A.M. Activated nitriles in hetorcyclic synthesis: Synthesis of several new naphtho [2, 1-b] pyran-3-one derivatives. J. Chem. Res. 1994, 50–51. [Google Scholar]
- Carrod, L.P.; Grady, F.D. Antibiotic and Chemotherapy, 3rd ed.; Churchill Livingstone: Edinburgh, UK, 1972; p. 47. [Google Scholar]
- Hewitt, W.; Vincent, S. Theory and Application of Microbiological Assay; Academic Press: New York, NY, USA, 1989. [Google Scholar]
Sample Availability: Samples are available from the authors. |



| Comp. No. | M.P. (T/oC)a | Molecular Formula (MW) | Analysis (%) Found/calculated | ||
|---|---|---|---|---|---|
| C | H | N | |||
| 1 | 108-110a | C14H10O2 (210.23) | 79.95 (79.98) | 4.78 (4.79) | - |
| 2 | 172-174a | C20H16N2O (300.35) | 79.96 (79.98) | 5.35 (5.37) | 9.30 (9.33) |
| 3 | 180-182a | C22H14N2O2 (338.36) | 78.00 (78.09) | 4.12 (4.17) | 8.20 (8.28) |
| 4a | 185-187a | C25H14N4O (386.40) | 77.65 (77.71) | 3.60 (3.65) | 14.47 (14.50) |
| 4b | 292-294b | C25H16N4O2 (404.42) | 74.20 (74.25) | 3.95 (3.99) | 13.78 (13.85) |
| 4c | 265-267a | C25H16N4OS (420.49) | 71.35 (71.41) | 3.80 (3.84) | 13.25 (13.32) |
| 5 | 350-352a | C26H16N4O4 (448.43) | 69.60 (69.64) | 3.55 (3.60) | 12.35 (12.49) |
| 6a | 280-282c | C22H16N4O (352.39) | 74.90 (74.98) | 4.50 (4.58) | 15.85 (15.90) |
| 6b | 190-192d | C29H24N4O (444.53) | 78.29 (78.36) | 5.35 (5.44) | 12.55 (12.60) |
| 6c | 220-222d | C25H22N4O2 (410.47) | 73.10 (73.15) | 5.32 (5.40) | 13.60 (13.65) |
| 6d | 228-230b | C23H17N5OS (411.48) | 67.09 (67.13) | 4.12 (4.16) | 16.95 (17.02) |
| 6e | 345-347e | C25H17N5O2 (419.43) | 71.50 (71.59) | 4.05 (4.09) | 16.67 (16.70) |
| 7a | 174-176d | C29H23N3O (429.51) | 81.00 (81.09) | 5.32 (5.40) | 9.70 (9.78) |
| 7b | 170-172d | C29H22ClN3O (463.96) | 75.00 (75.07) | 4.70 (4.78) | 9.00 (9.06) |
| 8 | 260-262b | C36H22N2O3 (530.57) | 81.40 (81. 49) | 4.12 (4.18) | 5.25 (5.28) |
| 10 | 218-220c | C32H22N4O2 (494.54) | 77.70 (77.72) | 4.35 (4.48) | 11.30 (11.33) |
| 12 | 208-210a | C31H24N4O4 (516.55) | 72.00 (72.08) | 4.60 (4.68) | 10.80 (10.85) |
| 13 | 260-262b | C34H20N44O4 (548.55) | 74.40 (74.44) | 3.60 (3.67) | 10.18 (10.21) |
| 14 | 271-273c | C25H17N5O2S (451.50) | 66.45 (66.50) | 3.72 (3.80) | 15.45 (15.51) |
| 15 | 220-222c | C26H19N5OS (449.53) | 69.42 (69.47) | 4.23 (4.26) | 15.54 (15.85) |
| 16 | 255-257c | C27H21N5O3S (495.55) | 65.38 (65.44) | 4.23 (4.27) | 14.00 (14.13) |
| Comp. No. | m/z (intensity/%) | |||||||
|---|---|---|---|---|---|---|---|---|
| M+ | 336 | 310 | 232 | 193 | 139 | 77 | Other peaks | |
| 1 | 210 (32) | 139(100) | 168(3), 113(11), 89(15), 63(38). | |||||
| 3 | 338 (100) | 310 (20) | 139 (15) | 77 (62) | 234 (1), 208 (1), 167 (1) | |||
| 4b | 404 (100) | 232 (1) | 193 (3) | 139 (12) | 77 (67) | 360 (25), 283 (4), 257 (3), | ||
| 4c | 420 (35) | 310 (1) | 193 (8) | 139 (15) | 77 (58) | 386(100), 281(2) | ||
| 5 | 448 (100) | 139 (7) | 77 (34) | 338 (14), 290 (11), 22 4(5), 181 (3) | ||||
| 6a | 352 (100) | 336 (66) | 139 (30) | 77 (44) | 168 (20) | |||
| 6b | 428 (100) | 336 (60) | 139 (13) | 77 (64) | 351 (1), 284 (15), 176 (11) | |||
| 6c | 394 (100) | 336 (29) | 139 (5) | 77 (20) | 352 (10), 309 (2), 232 (3), 180 (1) | |||
| 6d | 411 (1) | 336 (100) | 77 (32) | 308 (2), 233 (2), 20 (1), 181 (1) | ||||
| 7b | 530 (65) | 336 (100) | 232 (5) | 139 (41) | 77 (28) | 258 (4), 195 (16) | ||
| 8 | 447 (100) | 336 (7) | 310 (2) | 233 (3), 207 (5), 141 (15), 76 (64) | ||||
| 10 | 494 (100) | 310 (2) | 193 (5) | 139 (8) | 77 (31) | 466 (2), 386 (25), 233 (4) | ||
| 12 | 516 (2) | 193 (13) | 139 (5) | 77 (16) | 386 (100), 323 (12), 254 (4), | |||
| 13 | 548 (2) | 310 (5) | 139 (11) | 482 (15), 414 (9), 268 (12), 215 (25), 162 (23), 71 (60) | ||||
| 16 | 495 (45) | 336(100) | 232 (3) | 193(4) | 77 (40) | 453 (18), 394 (29), 306 (4) | ||
| Comp. No. | Inhibition zone diameter (mm per 200 mcg sample) | |||||
|---|---|---|---|---|---|---|
| (BS) ACTC 10400 | (SA) NCTC 7447 | (EC) NCTC 10410 | (PA) ATCC 10415 | (CA) IMRU 3669 | (AN) ATCC 6275 | |
| 2 | ++ ve | ++ ve | ++ ve | ++ ve | - ve | - ve |
| 14 | +++ ve | +++ ve | +++ ve | +++ ve | +++ ve | +++ ve |
| Neomycin (30 µg mL–1) | +++ ve | +++ ve | +++ ve | +++ ve | +++ ve | +++ ve |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
El-Wahab, A.H.F.A.; Al-Fifi, Z.I.A.; Bedair, A.H.; Ali, F.M.; Halawa, A.H.A.; El-Agrody, A.M. Synthesis, Reactions and Biological Evaluation of Some New Naphtho[2,1-b]furan Derivatives Bearing a Pyrazole Nucleus. Molecules 2011, 16, 307-318. https://doi.org/10.3390/molecules16010307
El-Wahab AHFA, Al-Fifi ZIA, Bedair AH, Ali FM, Halawa AHA, El-Agrody AM. Synthesis, Reactions and Biological Evaluation of Some New Naphtho[2,1-b]furan Derivatives Bearing a Pyrazole Nucleus. Molecules. 2011; 16(1):307-318. https://doi.org/10.3390/molecules16010307
Chicago/Turabian StyleEl-Wahab, Ashraf H. F. Abd, Zarrag Isa A. Al-Fifi, Ahmed H. Bedair, Fawzy M. Ali, Ahmed H. A. Halawa, and Ahemed M. El-Agrody. 2011. "Synthesis, Reactions and Biological Evaluation of Some New Naphtho[2,1-b]furan Derivatives Bearing a Pyrazole Nucleus" Molecules 16, no. 1: 307-318. https://doi.org/10.3390/molecules16010307




