Antioxidant and Cytotoxic Constituents from Wisteria sinensis
Abstract
:1. Introduction
2. Results and Discussion
| NO | δH | δC | HMBC correlation |
|---|---|---|---|
| 2 | 164.3 | ||
| 3 | 6.55 s | 103.7 | |
| 4 | 181.7 | ||
| 5 | 161.2 | ||
| 6 | 6.35 d (2.0) | 99.5 | H-8, 10 |
| 7 | 162.3 | ||
| 8 | 6.67 d (2.0) | 94.3 | H-6, 10 |
| 9 | 156.7 | ||
| 10 | 105.4 | ||
| 1' | 121.6 | ||
| 2' | 7.50 (brs) | 113.7 | H-6', 4' |
| 3' | 147.6 | ||
| 4' | 149.3 | ||
| 5' | 6.68 d (8.0) | 115.0 | H-3', 1' |
| 6' | 7.34 dd (8.0, 1.8) | 115.4 | H-2', 4' |
| OCH3 | 3.73 s | 55.5 | H-3' |
| 1'' | 5.19 d (7.5) | 99.1 | H-7, 3'' |
| 2'' | 3.78 dd (10.5, 7.5 ) | 75.8 | H-1''', 4'' |
| 3'' | 3.41 brd | 76.6 | H-5'', 1'' |
| 4'' | 3.20 t (10.5) | 69.8 | H-2'', 6'' |
| 5'' | 3.46 m | 77.5 | H-3'' |
| 6''a | 3.52 m | 60.5 | |
| 6''b | 3.70 brd (12.0) | 60.5 | |
| 1''' | 5.37 s | 108.2 | H-3''', 2'' |
| 2''' | 3.58* | 76.7 | H-4''' |
| 3''' | 3.65* | 77.0 | H-1''' |
| 4'''a | 3.75 d (9.5) | 73.8 | |
| 4'''b | 4.04 d (9.5) | 73.8 | |
| 5''' | 4.07 brs | 66.6 | H-2''' |
| 1'''' | 125.4 | ||
| 2'''' | 6.87 d (1.80) | 115.4 | H-6'''', 4'''', 7'''' |
| 3'''' | 145.0 | ||
| 4'''' | 149.3 | ||
| 5'''' | 6.67 d (8.0) | 115.9 | H-3'''', 1'''' |
| 6'''' | 6.84 d (8.0, 1.8) | 122.8 | H-2'''', 4'''', 7'''' |
| 7'''' | 7.29 d (16.0) | 144.9 | H-5'''', 2'''', 9'''' |
| 8'''' | 6.18 d (16.0) | 113.4 | H-1'''' |
| 9'''' | 167.5 |

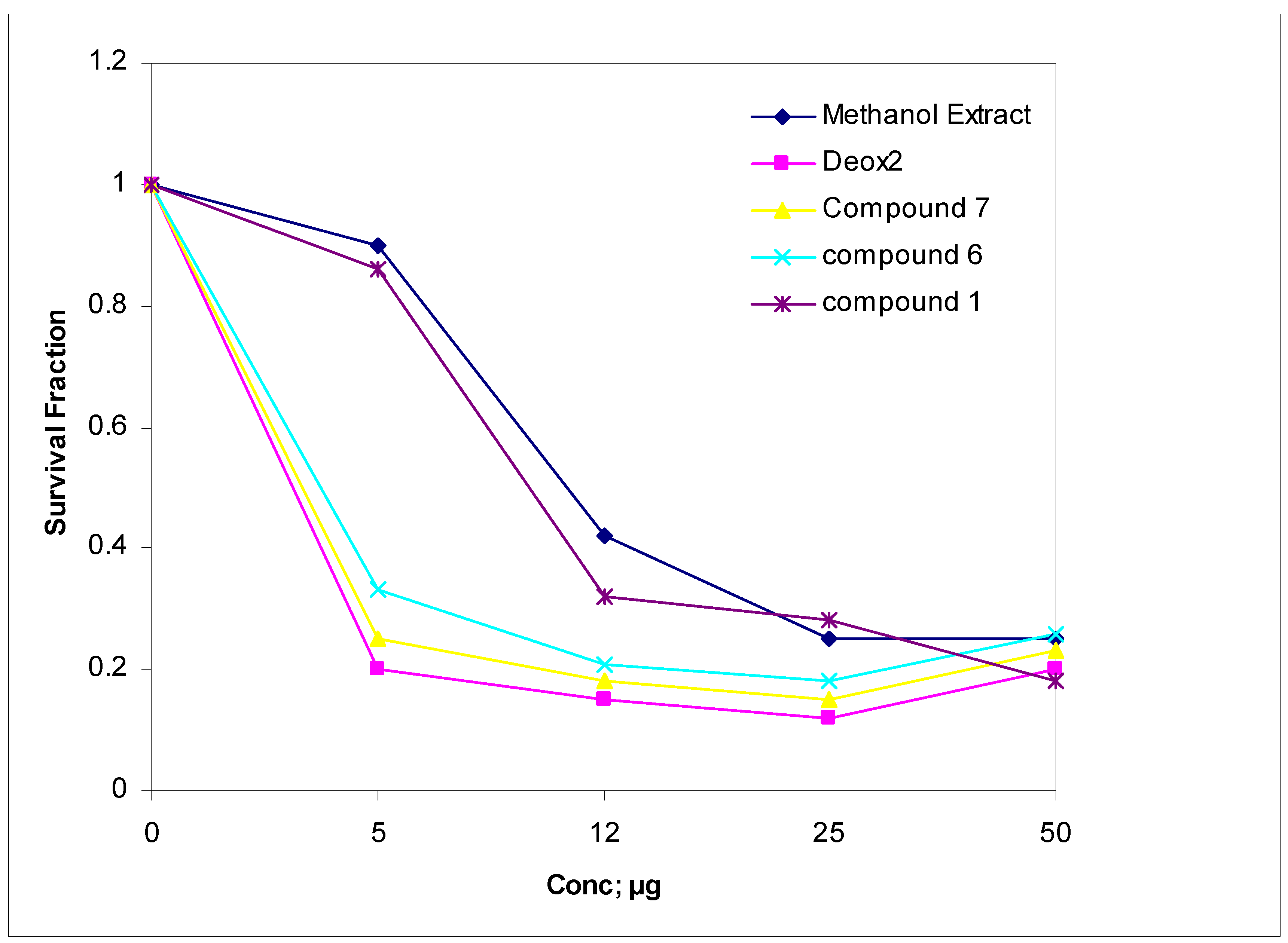
| Investigated materials | Maximum reactive reacting rate after 5 min concentration mL−1 | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 2 | 4 | 8 | 10 | 20 | 40 | 60 | 80 | 100 | |
| Methanol extract | - | - | - | - | 82.9 | 861 | 74.5 | 79.4 | 65.1 |
| 1 | - | - | - | - | 86.1 | 88.6 | 89.1 | 88.7 | 87.1 |
| 6 | - | - | - | - | 77.9 | 79.7 | 81.1 | 82.5 | 87.3 |
| 7 | 80.5 | 83.1 | 84.2 | 85.3 | 79.7 | 81.5 | 82. 9 | 82. 9 | 81.5 |

3. Experimental
3.1. General
3.2. Plant material
3.3. Extraction and isolation
3.4. Chrysoeriol -7-O-[2''-O-(5'''-O-caffeoyl)-β-D-apiofuranosyl]-β- D-glucopyranoside (1)
3.5. Measurement of potential cytotoxicity by SRB assay
3.6. Antioxidant activity
4. Conclusions
References and Notes
- Yoon, K.Y. Clinical Prescriptions of Korean Traditional Medical Books; Myung Bo Publishing: Seoul, Korea, 1992. [Google Scholar]
- Medicinal Herbs. Herbs: Chinese Wisteria. Available online: http://www.naturalmedicinalherbs.net/herbs/w/wisteria-sinensis=chinese-wisteria.php (accessed on 28 April 2010).
- Plants for A Future. Wisteria sinensis - (Sims.)DC. Available online: http://www.pfaf.org/user/Plant.aspx?LatinName=Wisteria%20sinensis (accessed on 28 April 2010).
- Yamazaki, K.; Sugiura, S. Arthropods associated with bacterium galls on Wisteria. Appl. Entomol. Zool. 2008, 43, 191–196. [Google Scholar] [CrossRef]
- The organicist conception in which every phenomenon was connected with every other according to a hierarchical order was universal amongst Chinese thinkers. In other words, the Chinese were a priori inclined to field theories Needham, J. Science in Traditional China; Harvard University Press: Cambridge, MA, USA, 1982; p. 14. [Google Scholar]
- Konoshima, T.; Kokumai, M.; Kozuka, M.; Tokuda, H.; Nishino, H.; Iwahima, I. Anti-tumor-promoting activities of afromosin and soyasaponin I isolated from Wistaria brachybotrys. J. Nat. Prod. 1992, 55, 1776–1778. [Google Scholar] [CrossRef]
- Konoshima, T.; Kozuka, M.; Haruna, M.; Ito K.; Kimura, T. Studies on the constituents of leguminous. XI: The structures of new triterpenoids from Wistaria brachybotrys Sieb. XI: The structures of new triterpenoid from Wistaria brachybotrys Sieb. et Zucc. Chem. Pharm. Bull. 1989, 37, 1550–1553. [Google Scholar] [CrossRef]
- Konoshima, T.; Kozuka, M.; Haruna, M.; Ito, K. constituents of leguminous plants, XIII. New triterpenoid saponins from Wistaria brachybotrys. J. Nat. Prod. 1991, 54, 830–836. [Google Scholar] [CrossRef]
- Konoshima, T.; Tokuda, H.; Kozuka, M.; Okamoto, E.; Tanabe, M. Promoting effects of crude drugs. Shoyakugaku Zasshi 1987, 41, 344–348. [Google Scholar]
- Konoshima, T.; Okamoto, E.; Kozuka, M.; Nishino, H.; Tokuda, H. Studies on Inhibitors of Skin Tumor Promotion, III. Inhibitory effects of isoflavonoids from Wisteria brachybotrys on Epstein - Barr virus activation. J. Nat. Prod. 1988, 51, 1266–1270. [Google Scholar] [CrossRef]
- Konoshima, T.; Takasaki, M.; Kozuka, M.; Tokuda, H.; Nishino, H.; Matsuda, E.; Nagai, M. Anti-tumor promoting activities of isoflavonoids from Wistaria brachybotrys. Biol. Pharm. Bull. 1997, 20, 865–868. [Google Scholar] [CrossRef]
- Kaneko, M.; Nakata, H.; Takada, F.; Matsumura, M.; Kitagawa, C.; Sakashita, S.; Nuno, M.; Saitoh, T. Isoflavones from the gall and wood of Wisteria brachybotry. Phytochemistry 1988, 27, 267–269. [Google Scholar] [CrossRef]
- Konoshima, T.; Takasaki, M.; Kozuka, M.; Tokuda, H.; Nishino, H.; Matsuda, E.; Nagai, M. Anti-tumor promoting activities of isoflavonoids from Wistaria brachybotrys. Biol. Pharm. Bull. 1997, 20, 865–868. [Google Scholar] [CrossRef]
- Oh, W.G.; Jang, I.C.; Jeon, G.I.; Park, E.J.; Park, H.R.; Lee, S.C. Antioxidative activity of extracts from Wisteria floribunda flower. J. Korean Soc. Food Sci. Nutr. 2008, 37, 677–683. [Google Scholar] [CrossRef]
- Yan-haul, J.; Xiao-huil, F.; Hua, J. Extraction of Wisteria sinensis active constituent and its inhibitory effect on fungi and bacteria. J. Henan Agric. Sci. 2009, 3, 60. [Google Scholar]
- Joulain, D.; Tabacchi, R. Two volatile β-chromenes from Wisteria sinensis flowers. Phytochemistry 1994, 37, 1767–1770. [Google Scholar] [CrossRef]
- Kurokawa, T.; Tsude, M.; Sugino, Y. Purification and characterization of a lectin from Wisteria floribunda seeds. J. Biol. Chem. 1976, 18, 5686–5693. [Google Scholar]
- Vchida, T.; Yamaizumi, M.; Mekada, E.; Okada, Y.; Tsude, M.; Kurokawa, T.; Sugino, Y. Reconstitution of hubrid toxin from fragment A of diphtheria toxin and a subunit of Wisteria floribunda lectin. J. Biol. Chem. 1978, 18, 6307–6310. [Google Scholar]
- Kaladas, P.M.; Poretz, R.D. Purification and properties of a mitogenic lectin from Wisteria floribunda seeds. Biochemistry US 1979, 22, 4806–4812. [Google Scholar] [CrossRef]
- Agrawal, P.K. Flavonoid Glycosides. In Studies in Organic Chemistry 39,Carbon-13 NMR of Flavonoids; Agrawal, P.K., Bansal, M.C., Eds.; Elsevier: Amsterdam, The Netherlands, 1989; pp. 283–364. [Google Scholar]
- Harborne, J.B. Phytochemical Methods: A Guide to Modern Techniques of Plant Analysis, 2nd ed; Chapman & Hall Ltd: London, UK, 1984; pp. 49–50. [Google Scholar]
- Harborne, J.B., Mabry. The flavonoids: Advances in research. In C 13 NMR Spectroscopy of Flavonoids; Markham, K.R., Mohanchari, V., Eds.; Chapman & Hall Ltd: University Press: Cambridge, London, UK, 1982; pp. 119–132. [Google Scholar]
- Mabry, T.J.; Markham, K.R.; Thomas, M.B. The systematic identification of flavonoids. In The Ultraviolet Spectra of Flavones and Flavonols; Springer: Verlag, Berlin, Germany, 1970; pp. 41–164, Part II, Chapter V. [Google Scholar]
- Nie, R.L.; Morita, T.; Kasai, R.; Zhou, J.; Wu, C.Y.; Tanaka, O. Saponins from Chinese medicinal plants, (1). Isolation and structures of hemslosides. Planta Med. 1984, 50, 322–327. [Google Scholar] [CrossRef]
- Jung, M.J.; Kang, S.S.; Jung, Y.J.; Choi, J.S. Phenolic glycosides from the stem bark of Albizzia julibrissin. Chem. Pharm. Bull. 2004, 52, 1501. [Google Scholar] [CrossRef]
- Mathias, L.; Vieira, I.J.C.; Braz-Filho, R.; Rodrigues-Filho, E.A. Retamatrioside, a new flavonol triglycoside from Retama sphaerocarpa. J. Nat. Prod. 1998, 61, 1158–1161. [Google Scholar] [CrossRef]
- Bashir, A.; Hamburger, M.; Gupta, M.P.; Solis, P.N.; Hostettmann, K. Flavonol glycosides from Monnina sylvatica. Phytochemistry 1991, 30, 3781–3784. [Google Scholar] [CrossRef]
- Meng, Y.; Krzysiak, A.K.; Durako, M.J.; Kunzelman, J.I.; Wright, J.L.C. Flavones and flavones glycosides from Halophila johnsonii. Phytochemistry 2008, 69, 2603–2608. [Google Scholar] [CrossRef]
- Kamel, M.S. Flavone C-glycosides from Lupinus hartwegii. Phytochemistry 2003, 63, 449–452. [Google Scholar] [CrossRef]
- Wang, AX.; Zhong, Q.; Jia, Z.J. A new furobenzopyranone and other constituents from Anaphalis lacteal. Pharmazie 2004, 59, 807–811. [Google Scholar]
- Lu, C.M.; Yang, J.J.; Wang, P.Y.; Lin, C.C. A new acylated flavanol glycosides and antioxidant effects of Hedyotis diffusa. Planta Med. 2000, 66, 374–376. [Google Scholar] [CrossRef]
- Qbe, F.; Iwase, Y.; Yamauchi, T.; Yahara, S.; Nohara, T. Flavanol sinapoyl glycosides from leaves of Thevetia peruviana. Phytochemistry 1995, 40, 577–581. [Google Scholar] [CrossRef]
- Kanadaswami, C.; Lee, L.T.; Hlee, P.P.; Hwang, J.J.; Ke, F.C.; Huang, Y.T.; Lee, M.T. The antitumor activities of flavonoids. In Vivo 2005, 19, 895–909. [Google Scholar]
- Shi, R.; Ong, C.; Shen H. Luteolin sensitizes tumor necrosis factor-α-induced apoptosis in human tumor cells. Oncogene 2004, 23, 7712–7721. [Google Scholar] [CrossRef]
- Pietta, P.G. Flavonoids as antioxidants. J. Nat. Prod. 2000, 63, 1035–1042. [Google Scholar] [CrossRef]
- Rice-Evans, C. Flavonoid antioxidants. Curr. Med. Chem. 2001, 8, 797–807. [Google Scholar] [CrossRef]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radic. Biol. Med. 1996, 20, 933–956. [Google Scholar] [CrossRef]
- Mira, L.; Fernandez, M.T.; Santos, M.; Rocha, R.; Florencio, M.H.; Jennings, K.R. Interactions of flavonoids with iron and copper ions: A mechanism for their antioxidant activity. Free Radic. Res. 2002, 36, 1199–1208. [Google Scholar] [CrossRef]
- Braca, A.; De Tommasi, N.; Di Bari, L.; Pizza, C.; Politi, M.; Morelli, I. Antioxidant principles from Bauhinia tarapotensis. J. Nat. Prod. 2001, 64, 892–295. [Google Scholar] [CrossRef]
- Cai, Q.; Rahm, R.O.; Zhang, R. Dietary flavonoids, quercetin, luteolin and genistein, reduce oxidative DNA damage and lipid peroxidation and quench free radicals. Cancer Lett. 1997, 199, 99–107. [Google Scholar]
- Hu, C. and kitts, D.D. luteolin and luteolin-7-O-glucoside from dandelion flower suppress iNOS and COX-2 in RAW264.7 cells. Mol. Cell Biochem. 2004, 265, 107–113. [Google Scholar] [CrossRef]
- Sadik, C.D.; Sies, H.; Schewe, T. Inhibition of 15-lipoxygenases by flavonoids: structure-activity relations and mode of action. Biochem. Pharamcol. 2003, 65, 773–788. [Google Scholar] [CrossRef]
- Choi, B.N.; Lim, D.W.; Lee, J.A.; Gao, S.S.; Kwon, D.Y.; Kim, B.R. Luteolin supperesses Cisplatin-induced apoptosis in auditory cells: possible mediation through induction of heme oxygenase-l expression. J. Med. Food. 2008, 11, 230–236. [Google Scholar] [CrossRef]
- Lim, J.H.; Park, H.S.; Choi, J.k.; Lee, I.S.; Choi, H.J. Isoorientin induces Nrf2 pathway-driven antioxidant response through phosphatidylinositol 3-kinase signaling. Arch. Pharm. Res. 2007, 30, 1590–1598. [Google Scholar] [CrossRef]
- Wruck, C.J.; Claussen, M.; fuhrmann, G.; Romer, L.; Schulz, A.; Pufe, T.; Waetzig, V.; Peipp, M.; Herdegen, T.; Gotz, M.E. Lueteolin protects rat PCl2 and C6 cells against MPP+ induced toxicity via an ERK depedent Keap 1-Nrf2-ARE pathway. J. Neural Transm. Suppl. 2007, 72, 57–67. [Google Scholar] [CrossRef]
- Qiusheng, Z.; Yuntao, Z.; Rongliang, Z.; Dean, G.; Changling, L. Effects of verbascoside and luteolin on oxidative damage in brain of heroin treated mice. Pharmazie 2005, 60, 539–543. [Google Scholar]
- Skehan, P.; Storeng, R.; Scudiero, D.; Monks, A.; McMahon, J.; Vistica, D.; Warren, J.T.; Bokesch, H.; Kenney, S.; Boyd, M.R. New colorimetric cytotoxicity assay for anticancer drug screening. J. Nat. Cancer Inst. 1990, 82, 1107–1112. [Google Scholar] [CrossRef]
- Peiwu, L.; Hopia, A.; Jari, S.; Yrjonen, T.; Vuorela, H. TLC method for evaluation of free radical scavenging activity of rapeseed meal by video scanning technology 1999. In Proceedings of the 10th International Rapeseed Congress, Canberra, Australia, 26–29 September 1999; Available online: http: www.regional.org.au/ au/gcirc/1/551.htm accessed on 11 March 2003.
- Govindarajan, R.; Rastog, I.S.; Vijayakumar, M.; Shirwaikar, A.; Rawat, A.K.S.; Mehrotra, S.; Pushpangadan, P. Studies on the antioxidant activities of Desmodium gangeticum. Biol. Pharm. Bull. 2003, 26, 1424–1427. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 6–9 are available from the authors.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mohamed, M.A.; Hamed, M.M.; Abdou, A.M.; Ahmed, W.S.; Saad, A.M. Antioxidant and Cytotoxic Constituents from Wisteria sinensis. Molecules 2011, 16, 4020-4030. https://doi.org/10.3390/molecules16054020
Mohamed MA, Hamed MM, Abdou AM, Ahmed WS, Saad AM. Antioxidant and Cytotoxic Constituents from Wisteria sinensis. Molecules. 2011; 16(5):4020-4030. https://doi.org/10.3390/molecules16054020
Chicago/Turabian StyleMohamed, Mona A., Manal M. Hamed, Allia M. Abdou, Wafaa S. Ahmed, and Amal M. Saad. 2011. "Antioxidant and Cytotoxic Constituents from Wisteria sinensis" Molecules 16, no. 5: 4020-4030. https://doi.org/10.3390/molecules16054020
APA StyleMohamed, M. A., Hamed, M. M., Abdou, A. M., Ahmed, W. S., & Saad, A. M. (2011). Antioxidant and Cytotoxic Constituents from Wisteria sinensis. Molecules, 16(5), 4020-4030. https://doi.org/10.3390/molecules16054020




