Antiplasmodial and Leishmanicidal Activities of 2-Cyano-3-(4-phenylpiperazine-1-carboxamido) Quinoxaline 1,4-Dioxide Derivatives
Abstract
:1. Introduction

2. Results and Discussion
2.1. Chemistry

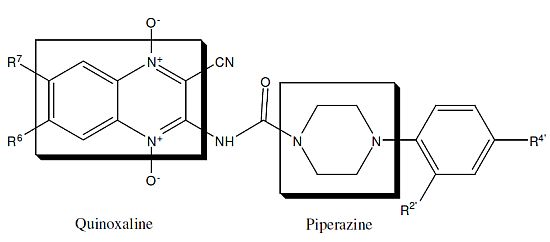
2.2. Pharmacology and Structure-Activity Relationship
| Compd. | MW | R 6 | R 7 | R 2' | R 4' | IC50 (µM) a | IC50 (µM) b | CC50 (µM) c | SI d |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 537.5 | H | Cl | NO2 | CF3 | 24.5 | 21.8 | 7.0 | 0.3 |
| 2 | 517 | H | CH3 | NO2 | CF3 | 44.7 | 36.3 | 17.7 | 0.5 |
| 3 | 521 | H | F | NO2 | CF3 | 14.6 | 41.1 | 11.0 | 0.3 |
| 4 | 572 | Cl | Cl | NO2 | CF3 | 13.9 | 22.7 | 1.6 | 0.1 |
| 5 | 492.5 | H | Cl | H | CF3 | 18.6 | 7.6 | 6.4 | 0.8 |
| 6 | 472 | H | CH3 | H | CF3 | 30.5 | 23.3 | 12.1 | 0.5 |
| 7 | 476 | H | F | H | CF3 | 30.9 | 28.8 | 12.2 | 0.4 |
| 8 | 527 | Cl | Cl | H | CF3 | 18.5 | 5.7 | 2.2 | 0.4 |
| 9 | 422 | H | CH3 | H | F | 36.3 | 23.0 | 24.1 | 1.1 |
| 10 | 426 | H | F | H | F | 34.3 | 31.3 | 24.3 | 0.8 |
| 11 | 454.5 | H | Cl | H | CH3O | 12.8 | 18.8 | 47.5 | 2.5 |
| 12 | 434 | H | CH3 | H | CH3O | 30.4 | 30.0 | 183.5 | 6.1 |
| 13 | 489 | Cl | Cl | H | CH3O | 26.1 | 10.9 | 14.0 | 1.3 |
| CQ | 320 | 0.1 | |||||||
| DOX | 543.5 | 6.4 | 0.4 | >10  |
3. Experimental
3.1. Chemical Synthesis
3.1.1. General Remarks
3.1.2. General Procedure for the Synthesis of Cyanoamines II
3.1.3. General Procedure for the Synthesis of 2-Cyano-3-(4-phenylpiperazine-1-carboxamido) Quinoxaline 1,4-Dioxide (CPCQs)
3.2. Pharmacology
3.2.1. In Vitro Antiplasmodial Drug Assay
3.2.2. In Vitro Cytotoxicity
3.2.3.In Vitro Antileishmanial Drug Assay
4. Conclusions
Acknowledgments
References
- Vicente, E.; Charnaud, S.; Bongard, E.; Villar, R.; Burguete, A.; Solano, B.; Ancizu, S.; Perez-Silanes, S.; Aldana, I.; Vivas, L.; et al. Synthesis and Antiplasmodial Activity of 3-Furyl and 3-Thienylquinoxaline-2-carbonitrile 1,4-Di-N-oxide Derivatives. Molecules 2008, 13, 69–77. [Google Scholar] [CrossRef]
- WHO. World Malaria Report 2011. Available online: http://www.who.int/malaria/world_malaria_report_2011/en/index.html (accessed on 11 June 2011).
- WHO. Leishmaniasis. Available online: http://www.who.int/entity/tdr/diseases-topics/leishmaniasis/en/ (accessed on 11 June 2011).
- Palumbo, E. Oral miltefosine treatment in children with visceral leishmaniasis: A brief review. Braz. J. Infect. Dis. 2008, 12, 2–4. [Google Scholar]
- Lima, L.M.; Barreiro, E.J. Bioisosterism: A useful strategy for molecular modification and drug design. Curr. Med. Chem. 2005, 12, 23–49. [Google Scholar]
- Carta, A.; Corona, P.; Loriga, M. Quinoxaline 1,4-dioxide: A versatile scaffold endowed with manifold activities. Curr. Med. Chem. 2005, 12, 2259–2272. [Google Scholar] [CrossRef]
- Monge, A.; Palop, J.A.; Piñol, A.; Martínez-Crespo, F.J.; Narro, S.; González, M.; Sáinz, Y.; López de Ceráin, A.; Hamilton, E.; Barker, A.J. 3-Amino-2-Quinoxalinecarbonitrile. New Fused Quinoxaline with Potential Cytotoxic Activity. J. Heterocycl. Chem. 1994, 31, 1135–1139. [Google Scholar] [CrossRef]
- Burguete, A.; Estevez, Y.; Castillo, D.; González, G.; Villar, R.; Solano, B.; Vicente, E.; Pérez-Silanes, S.; Aldana, I.; Monge, A.; et al. Anti-leishmanial and structure-activity relationship of ring substituted 3-phenyl-1-(1,4-di-N-oxide quinoxalin-2-yl)-2-propen-1-one derivatives. Mem. Inst. Oswaldo Cruz 2008, 103, 778–780. [Google Scholar] [CrossRef]
- Estevez, Y.; Quiliano, M.; Burguete, A.; Zimic, M.; Málaga, E.; Verástegui, M.; Pérez-Silanes, S.; Aldana, I.; Monge, A.; Castillo, D.; et al. Trypanocidal properties, structure-activity relationship and computational studies of quinoxaline 1,4-di-N-oxide derivatives. Experiment. Parasitol. 2011, 127, 745–751. [Google Scholar] [CrossRef]
- Ancizu, S.; Moreno, E.; Torres, E.; Burguete, A.; Perez-Silanes, S.; Benitez, D.; Villar, R.; Solano, B.; Marin, A.; Aldana, I.; et al. Heterocyclic-2-carboxylic acid (3-cyano-1,4-di-N-oxidequinoxalin-2-yl)amide derivatives as hits for the development of neglected diseases drugs. Molecules 2009, 14, 2256–2272. [Google Scholar] [CrossRef]
- Marin, A.; Lima, L.M.; Solano, B.; Vicente, E.; Pérez-Silanes, S.; Maurel, S.; Sauvain, M.; Aldana, I.; Monge, A.; Deharo, E. Antiplasmodial structure-activity relationship of 3-trifluoromethyl-2-arylcarbonylquinoxaline 1,4-di-N-oxide derivatives. Experiment. Parasitol. 2008, 118, 25–31. [Google Scholar] [CrossRef]
- Vicente, E.; Lima, L.M.; Bongard, E.; Charnaud, S.; Villar, R.; Solano, B.; Burguete, A.; Pérez-Silanes, S.; Aldana, I.; Monge, A. Synthesis and structure-activity relationship of 3-phenylquinoxaline 1,4-di-N-oxide derivatives as antimalarial agents. Eur. J. Med. Chem. 2008, 43, 1903–1910. [Google Scholar] [CrossRef]
- Zarranz, B.; Jaso, A.; Lima, L.M.; Aldana, I.; Monge, A.; Maurel, S.; Sauvain, M. Antiplasmodial activity of 3-trifluoromethyl-2-carbonylquinoxaline di-N-oxide derivatives. Braz. J. Pharm. Sci. 2006, 42, 357–361. [Google Scholar]
- Zarranz, B.; Jaso, A.; Aldana, I.; Monge, A.; Maurel, S.; Deharo, E.; Jullian, V.; Sauvain, M. Synthesis and antimalarial activity of new 3-arylquinoxaline-2-carbonitrile derivatives. Arzneimittelforschung 2005, 55, 754–761. [Google Scholar]
- Aldana, I.; Ortega, M.A.; Jaso, A.; Zarranz, B.; Oporto, P; Gimenez, A.; Monge, A.; Deharo, E. Anti-malarial activity of some 7-chloro-2-quinoxalinecarbonitrile-1,4-di-N-oxide derivatives. Pharmazie 2002, 58, 68–69. [Google Scholar]
- Barea, C.; Pabón, A.; Castillo, D.; Zimic, M.; Quiliano, M.; Galiano, S.; Pérez-Silanes, S.; Monge, A.; Deharo, E.; Aldana, I. New salicylamide and sulfonamide derivatives of quinoxaline 1,4-di-N-oxide with antileishmanial and antimalarial activities. Bioorg. Med. Chem. Lett. 2011, 21, 4498–4502. [Google Scholar]
- Cunico, W.; Gomes, C.R.; Facchinetti, V.; Moreth, M.; Penido, C.; Henriques, M.G.; Varotti, F.P.; Krettli, L.G.; Krettli, A.U.; da Silva, F.S.; et al. Synthesis, antimalarial evaluation and molecular modeling studies of hydroxyethylpiperazines, potential aspartyl protease inhibitors, part 2. Eur. J. Med. Chem. 2009, 44, 3816–3820. [Google Scholar]
- Mendoza, A.; Pérez-Silanes, S.; Quiliano, M.; Pabón, A.; Galiano, S.; González, G.; Garavito, G.; Zimic, M.; Vaisberg, A.; Aldana, I.; et al. Aryl piperazine and pyrrolidine as antimalarial agents. Synthesis and investigation of structure-activity relationships. Exp. Parasitol. 2011, 128, 97–103. [Google Scholar]
- Ortega, M.A.; Sainz, Y.; Montoya, M.E.; Jaso, A.; Zarranz, B.; Aldana, I.; Monge, A. Anti-Mycobacterium tuberculosis agents derived from quinoxaline-2-carbonitrile and quinoxaline-2-carbonitrile 1,4-di-N-oxide. Arzneimittelforschung 2002, 52, 113–119. [Google Scholar]
- González, M.; Cerecetto, H. Benzofuroxan and Furoxan. Chemistry and Biology. In Topics in Heterocyclic Chemistry. Bioactive Heterocycles V; Khan, M.T.H., Ed.; Springer: Berlin, Germany, 2007; Volume 10, pp. 265–308. [Google Scholar]
- Ley, K.; Seng, F. Synthesis unter verwendung von benzofuroxan. Synthesis 1975, 7, 415–422. [Google Scholar]
- Keizer, H.M.; Sijbesma, R.P.; Meijder, E.W. The convenient synthesis of hydrogen-bonded ureidopyrimidiones. Eur. J. Org. Chem. 2004, 2553–2555. [Google Scholar]
- Desjardins, R.E.; Canfield, C.J.; Haynes, J.D.; Chulay, J.D. Quantitative assessment of antimalarial activity in vitro by a semiautomated microdilution technique. Antimicrob. Agents Chemother. 1979, 16, 710–718. [Google Scholar] [CrossRef]
- Sereno, D.; Lemesre, J.L. Use of an enzymatic micromethod to quantity amastigotes stage of Leishmania amazonensis in vitro. Parasitol. Res. 1997, 83, 401–403. [Google Scholar] [CrossRef]
- Cachet, N.; Hoakwie, F.; Bertani, S.; Bourdy, G.; Deharo, E.; Stien, D.; Houel, E.; Gornitzka, H.; Fillaux, J.; Chevalley, S.; et al. Antimalarial activity of simalikalactone E, a new quassinoid from Quassia amara L. (Simaroubaceae). Antimicrob. Agents Chemother. 2009, 53, 4393–4398. [Google Scholar]
- Muñoz, V.; Sauvain, M.; Mollinedo, P.; Callapa, J.; Rojas, I.; Gimenez, A.; Valentin, A.; Mallie, M. Antimalarial activity and cytotoxicity of (−)-roemrefidine isolated from the stem bark of Sparattanthelium amazonum. Planta Med. 1999, 65, 448–449. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Barea, C.; Pabón, A.; Galiano, S.; Pérez-Silanes, S.; Gonzalez, G.; Deyssard, C.; Monge, A.; Deharo, E.; Aldana, I. Antiplasmodial and Leishmanicidal Activities of 2-Cyano-3-(4-phenylpiperazine-1-carboxamido) Quinoxaline 1,4-Dioxide Derivatives. Molecules 2012, 17, 9451-9461. https://doi.org/10.3390/molecules17089451
Barea C, Pabón A, Galiano S, Pérez-Silanes S, Gonzalez G, Deyssard C, Monge A, Deharo E, Aldana I. Antiplasmodial and Leishmanicidal Activities of 2-Cyano-3-(4-phenylpiperazine-1-carboxamido) Quinoxaline 1,4-Dioxide Derivatives. Molecules. 2012; 17(8):9451-9461. https://doi.org/10.3390/molecules17089451
Chicago/Turabian StyleBarea, Carlos, Adriana Pabón, Silvia Galiano, Silvia Pérez-Silanes, German Gonzalez, Chloe Deyssard, Antonio Monge, Eric Deharo, and Ignacio Aldana. 2012. "Antiplasmodial and Leishmanicidal Activities of 2-Cyano-3-(4-phenylpiperazine-1-carboxamido) Quinoxaline 1,4-Dioxide Derivatives" Molecules 17, no. 8: 9451-9461. https://doi.org/10.3390/molecules17089451