Effects of Velvet Antler with Blood on Bone in Ovariectomized Rats
Abstract
:1. Introduction
2. Results
2.1. Combination Therapy of VAM and VA Blood is Effective in Inhibiting Osteoblast-Like Cells
| VA:VA Blood = 1:0.2 | Combination Index (CI) | ||
|---|---|---|---|
| ED50 | ED75 | ED90 | |
| VAU | 5.76 × 103 | 7.63 × 105 | 1.05 × 108 |
| VAM | 0.97 | 0.74 | 0.55 |
| VAB | 3.12 | 6.42 | 13.14 |
2.2. Chemical Constituents
2.3. Body Weight and Serum Markers
| Item | VAM-B |
|---|---|
| Protein (mg/g) | 0.5 |
| Glycoprotein (mg/g) | 3.3 |
| Testosterone (ng/g) | 4.4 |
| Estradiol (ng/g) | 3.1 |
| Insulin-like growth factor-1 (ng/g) | 33.8 |
| Cholesterol (mg/g) | 0.9 |
| Calcium (%) | 13.2 |
| Iron (ppm) | 434.0 |
| Amino acid (mg/g) | VAM-B |
|---|---|
| Alanine | 14.45 |
| Arginine | 15.95 |
| Aspartic acid | 18.21 |
| Cysteine | 1.73 |
| Glutamic acid | 21.00 |
| Glycine | 18.86 |
| Histidine | 6.51 |
| Isoleucine | 2.90 |
| Leucine | 15.66 |
| Lysine | 10.84 |
| Methionine | 2.11 |
| Phenylalanine | 8.72 |
| Proline | 12.78 |
| Serine | 8.39 |
| Threonine | 8.24 |
| Tyrosine | 4.00 |
| Valine | 10.01 |
| Total amounts | 180.39 |
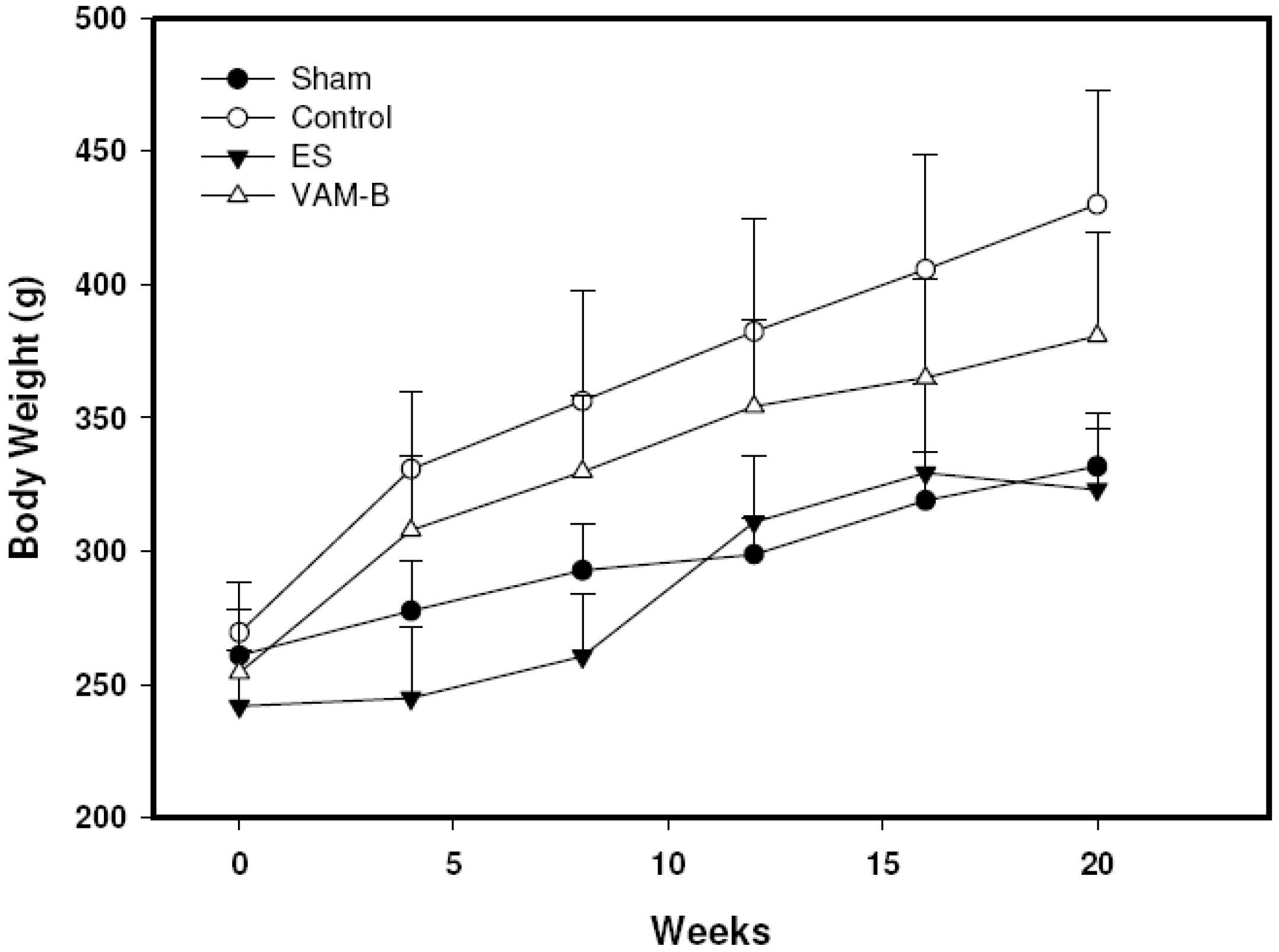

2.4. Bone Biomechanics and Histomorphometric Parameters


| Groups | BV/TV (%) | Tb.Th (μm) | Tb.Sp (μm) | Tb.N (μm) | SMI |
|---|---|---|---|---|---|
| Sham | 40.84 ± 10.73 | 101.66 ± 16.09 | 168.28 ± 29.25 | 0.0040 ± 0.0004 | 0.60 ± 0.38 |
| Control | 10.70 ± 3.32 # | 107.69 ± 9.77 | 696.22 ± 158.76 # | 0.0010 ± 0.0003 # | 2.01 ± 0.22 # |
| VAM-B | 16.15 ± 2.23 * | 105.60 ± 2.63 | 447.25 ± 65.28 * | 0.0015 ± 0.0001 * | 1.70 ± 0.10 # |
| ES | 25.69 ± 10.41 * | 90.29 ± 4.01 | 255.99 ± 92.24 * | 0.0028 ± 0.0010 * | 1.29 ± 0.52 # |
3. Discussion
4. Experimental
4.1. Preparations of VA with Blood Powder
4.2. Cell Proliferation Assay and Combination Effect between VA and VA Blood
4.3. Protein Content
4.4. Estradiol, Testosterone, and IGF-1 Analyses
4.5. Rat Osteoporosis Model
4.6. Bone Breaking Strength Test
4.7. Micro-Computed Tomographic (Micro-CT) Analysis
4.8. Statistical Analysis
5. Conclusions
Acknowledgments
References
- WHO Regional Office for the Western Pacific, Basic Theories. In WHO International Standard Terminologies on Traditional Medicine in the Western Pacific Region; World Health Organization: Geneva, Switzerland, 2007; p. 23.
- Zhou, J.; Qu, F. Treating gynaecological disorders with Traditional Chinese Medicine: A Review. Afr. J. Tradit. Complement. Altern. Med. 2009, 6, 494–517. [Google Scholar]
- Wong, R.W.; Rabie, B.; Bendeus, M.; Hägg, U. The effects of Rhizoma Curculiginis and Rhizoma Drynariae extracts on bones. Chin. Med. 2007, 2, 13. [Google Scholar] [CrossRef] [Green Version]
- Xie, F.; Wu, C.F.; Lai, W.P.; Yang, X.J.; Cheung, P.Y.; Yao, X.S.; Leung, P.C.; Wong, M.S. The osteoprotective effect of Herba epimedii (HEP) extract in vivo and in vitro. Evid. Based Complement. Alternat. Med. 2005, 2, 353–361. [Google Scholar] [CrossRef]
- Chen, C.C.; Liu, M.H.; Wang, M.F.; Chen, C.C. Effects of aging and dietary antler supplementation on the calcium-regulating hormones and bone status in ovariectomized SAMP8 mice. Chin. J. Physiol. 2007, 50, 308–314. [Google Scholar]
- Li, Y.; Zhao, Y.; Tang, R.; Qu, X. Preventive and therapeutic effects of antler collagen on osteoporosis in ovariectomized rats. Afr. J. Biotechnol. 2010, 9, 6437–6341. [Google Scholar]
- Shi, B.; Li, G.; Wang, P.; Yin, W.; Sun, G.; Wu, Q.; Yu, G. Effect of antler extract on corticosteroid-induced avascular necrosis of the femoral head in rats. J. Ethnopharmacol. 2010, 127, 124–129. [Google Scholar] [CrossRef]
- Yang, J.H.; Cao, Y.; Wang, R.L.; Fei, Y.R.; Zhang, H.; Feng, P.; Liu, J. Anti-resorptive effect of pilose antler blood (Cervus nippon Temminck) in ovariectomized rats. Indian J. Exp. Biol. 2010, 48, 554–558. [Google Scholar]
- Suttie, J.M.; Haines, S.R.; Brown-Smith, A.P.; Fennesy, P.F. The effect of removal technique and post-removal handling on velvet antler colour. NZ J. Agric. Res. 2000, 43, 207–225. [Google Scholar] [CrossRef]
- Ahlborg, H.G.; Rosengren, B.E.; Järvinen, T.L.N.; Rogmark, C.; Nilsson, J.Å.; Sernbo, I.; Karlsson, M.K. Prevalence of osteoporosis and incidence of hip fracture in women—Secular trends over 30 years. BMC Musculoskelet Disord 2010, 11, 48. [Google Scholar] [CrossRef]
- Davidson, A. Traditional Uses. In Velvet Antler: Powerful Tonic for Joints, Infertility, Blood Pressure, Athletic Performance and More…; Safe Goods: Sheffield, MA, USA, 1999; pp. 15–22. [Google Scholar]
- Wang, Y.M.; Chu, L.W.; Wang, Y.B.; Wang, S.L. Comparative analysis of contents of amino acid, total phospolipid, calcium and phosphor in Sika Deer velvet bone slices with blood and without blood. J. Econ. Anim. 2003, 7, 21–23. [Google Scholar]
- Oury, F. A crosstalk between bone and gonads. Ann. NY Acad. Sci. 2012, 1260. [Google Scholar] [CrossRef]
- Bubenik1, G.A.; Miller, K.; Lister, A.L.; Osborn, D.A.; Bartos, L.; van der Kraak, G.J. Testosterone and estradiol concentrations in serum, velvet skin, and growing antler bone of male white-tailed deer. J. Exp. Zool. A. Comp. Exp. Biol. 2005, 303A, 186–192. [Google Scholar] [CrossRef]
- Jeon, B.T.; Kang, S.K.; Lee, S.M.; Hong, S.K.; Moon, S.H. Serum biochemical values during antler growth in sika deer (Cervus nippon). Asian-Aust. Anim. Sci. 2007, 20, 748–753. [Google Scholar]
- Xian, L.; Wu, X.; Pang, L.; Lou, M.; Rosen, C.J.; Qiu, T.; Crane, J.; Frassica, F.; Zhang, L.; Rodriguez, J.P.; et al. Matrix IGF-1 maintains bone mass by activation of mTOR in mesenchymal stem cells. Nat. Med. 2012. [Google Scholar] [CrossRef]
- Armagan, O.; Sermagül, E.; Ekim, A.; Sirmagül, B.; Taçioglu, F. Levels of IGF-1 and their relationship with bone mineral density in the premenopausal women with fibromyalgia syndrome. Rheumatism 2008, 23, 118–123. [Google Scholar]
- Bartos, L.; Schams, D.; Bubenik, G.A. Testosterone, but not IGF-1, LH, prolactin or cortisol, may serve as antler-stimulating hormone in red deer stags (Cervus elaphus). Bone 2009, 44, 691–698. [Google Scholar] [CrossRef]
- Sadighi, M.; Li, C.; Littlejohn, R.P.; Suttie, J.M. Effects of testosterone either alone or with IGF-I on growth of cells derived from the proliferation zone of regenerating antlers in vitro. Growth Horm. IGF Res. 2001, 11, 240–246. [Google Scholar] [CrossRef]
- Gaspar-López, E.; Landete-Castillejos, T.; Estevez, J.A.; Ceacero, F.; Gallego, L.; García, A.J. Seasonal variations in red deer (Cervus elaphus) hematology related to antler growth and biometrics measurements. J. Exp. Zool. A. Ecol. Genet. Physiol. 2011, 315, 242–249. [Google Scholar]
- Papaioannou, A.; Kennedy, C.C.; Dolovich, L.; Lau, E.; Adachi, J.D. Patient adherence to osteoporosis medications: problems, consequences and management strategies. Drugs Aging 2007, 24, 37–55. [Google Scholar] [CrossRef]
- Li, C.W.; Jiang, I.G.; Zeng, Y.; Yan, C.E. Velvet tissue estradiol concentrations’ in Pere David’s deer, Sika deer, and fallow deer. Acta Zool. Sin. 2003, 49, 124–127. [Google Scholar]
- Ikeda, H.; Taira, N.; Nogami, T.; Shien, K.; Okada, M.; Shien, T.; Doihara, H.; Miyoshi, S. Combination treatment with fulvestrant and various cytotoxic agents (doxorubicin, paclitaxel, docetaxel, vinorelbine, and 5-fluorouracil) has a synergistic effect in estrogen receptor-positive breast cancer. Cancer Sci. 2011, 102, 2038–2042. [Google Scholar] [CrossRef]
- Wang, K.T.; Chen, L.G.; Tseng, S.H.; Huang, J.S.; Hsieh, M.S.; Wang, C.C. Anti-inflammatory effects of resveratrol and oligostilbenes from Vitis thunbergii var. taiwaniana against lipopolysaccharide-induced arthritis. J. Agric. Food Chem. 2011, 59, 3649–3656. [Google Scholar]
- Sample Availability: Not available.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tseng, S.-H.; Sung, H.-C.; Chen, L.-G.; Lai, Y.-J.; Wang, K.-T.; Sung, C.-H.; Wang, C.-C. Effects of Velvet Antler with Blood on Bone in Ovariectomized Rats. Molecules 2012, 17, 10574-10585. https://doi.org/10.3390/molecules170910574
Tseng S-H, Sung H-C, Chen L-G, Lai Y-J, Wang K-T, Sung C-H, Wang C-C. Effects of Velvet Antler with Blood on Bone in Ovariectomized Rats. Molecules. 2012; 17(9):10574-10585. https://doi.org/10.3390/molecules170910574
Chicago/Turabian StyleTseng, Sung-Hui, Hsin-Ching Sung, Lih-Geeng Chen, Ying-Jang Lai, Kun-Teng Wang, Chun-Hsien Sung, and Ching-Chiung Wang. 2012. "Effects of Velvet Antler with Blood on Bone in Ovariectomized Rats" Molecules 17, no. 9: 10574-10585. https://doi.org/10.3390/molecules170910574
APA StyleTseng, S.-H., Sung, H.-C., Chen, L.-G., Lai, Y.-J., Wang, K.-T., Sung, C.-H., & Wang, C.-C. (2012). Effects of Velvet Antler with Blood on Bone in Ovariectomized Rats. Molecules, 17(9), 10574-10585. https://doi.org/10.3390/molecules170910574





