Advances in Click Chemistry for Single-Chain Nanoparticle Construction
Abstract
:1. Introduction

1.1. Controlled Polymerization

| Polymerization Technique | Abbreviation | Nature of the SCNP Polymeric Precursors * |
|---|---|---|
| Reversible addition fragmentation chain transfer polymerization | RAFT | Poly(alkyl methacrylates) [5,6,7,10,15,21,22,23,24,25,26,27], poly(alkyl acrylates) [7,22,28,29], poly(styrene) [3,22,23,30,31,32], poly(haloalkyl styrene) [3,33], poly(4-N-Boc-vinylaniline) [33], poly(sodium 4-styrenesulfonate) [22], poly(N-alkyl acrylamide) [22,34,35] |
| Atom transfer radical polymerization | ATRP | Poly(alkyl methacrylates) [14], poly(alkyl acrylates) [11,12], poly(styrene) [36], poly(N-hydroxyethyl acrylamide) [37] |
| Nitroxide mediated radical polymerization | NMP | Poly(alkyl methacrylates) [9,38,39,40], poly(alkyl acrylates) [40], poly(styrene) [2,38,40], poly(haloalkyl styrene) [40], poly(fluorene) [41] |
| Ring opening metathesis polymerization | ROMP | Poly(ε-caprolactone) [38], poly(carbonates) [42] poly(norbornenes) [13,43,44] |
1.2. Polymer Functionalization
| Polymer Functionalization Technique | Functional Groups Involved | Functionalizable Polymers |
|---|---|---|
| Thiol-ene / thiol-yne additions * | Thiol/alkene, alkyne | Polymers bearing alkene-, alkyne- or thiol-groups |
| Modification of epoxides, anhydrides, oxazolines and isocyanates by reaction with amines / alcohols / thiols * | Epoxide, anhydride, oxazoline, isocyanate/ amine, alcohol, thiol | Polymers containing epoxide-, anhydride-, oxazoline-, isocyanate-, amine-, alcohol- or thiol-groups [23] |
| Modification of active esters by reaction with amines | N-Hydroxysuccinimide, pentafluorophenyl ester/ amine | Polymers bearing N-hydroxy-succinimide-, pentafluorophenyl ester- or amine-groups |
| Thiol-disulfide exchange | Pyridyl disulfide/thiol | Polymers containing pyridyl disulfide- or thiol-groups [6] |
| Diels-Alder reaction * | Diene/alkene | Diene- or alkene-bearing polymers [28,40,53] |
| Michael-type addition | Acrylate, N-substituted-maleimide, vinyl sulfone/ thiols | Polymers bearing acrylate-, N-substituted-maleimide-, vinyl sulfone- or thiol-groups |
| Copper-catalyzed azide alkyne cycloaddition (CuAAC) * | Azide / alkyne | Azide- or alkyne-bearing polymers [3,10,21,22,30,34] |
| Modification of ketones and aldehydes with amines/ alkoxyamines/hydrazines | Ketone, aldehyde / amine, alkoxyamine, hydrazine | Polymers containing ketone-, aldehyde-, amine-, alkoxyamine- or hydrazine- groups |
1.3. Intrachain Folding / Collapse
| Reactive functional groups | Covalent bonding interactions |
|---|---|
| Vinyl [33,38,39,42] | Radical coupling & Cross-Metathesis |
| Benzocyclobutene [40,53] | Diels-Alder reaction * |
| Benzosulfone [9,28,41] | Diels-Alder reaction * |
| Azide + Protected alkyne [3,10,21,22,30,34] | Copper-catalyzed [3+2] cycloaddition ** |
| Carboxilic acid [54] | Amide formation |
| Isocyanate [23] | Urea formation ** |
| Enediyne [11,12,24,29] | Bergman & Photo-Bergman cyclization |
| Sulfonyl azide [31] | Nitrene-mediated cross-linking |
| Benzoxazine [36] | Ring opening polymerization |
| Alkyne [25] | Glaser-Hay coupling * |
| Reactive functional groups | NC/DC bonding interactions |
|---|---|
| Benzamide [26] | Benzamide hydrogen bonding * |
| 2-Ureido-Pyrimidone (UPy) [43] | UPy dimerization * |
| Coumarin [35] | Coumarin photo-dimerization ** |
| Benzaldehyde [32] | Acylhydrazone formation ** |
| β-Ketoester [27] | Enamine formation ** |
| Methyl viologen + Naphtyl [37] | Cucurbit[n]uril complexation * |
| L-Phenylalanine (Phe) [55] | Hydrophobic Phe-Phe interactions * |
| Aminophenyl disulfide [44] | Disulfide formation ** |
2. Single-Chain Nanoparticle Construction via Click Chemistry
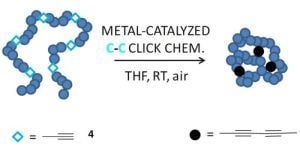
2.1. Intrachain Homocoupling via Click Chemistry



2.2. Intrachain Heterocoupling via Click Chemistry

2.3. Crosslinker-Induced Collapse via Click Chemistry


3. Applications of Single-Chain Nanoparticles
3.1. Nanomedicine
3.1.1. Peptide/Drug/siRNA Controlled Delivery
3.1.2. Image Contrast Agents
3.2. Catalysis
3.3. Other Uses
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Altintas, O.; Barner-Kowollik, C. Single chain folding of synthetic polymers by covalent and non-covalent interactions: Current status and future perspectives. Macromol. Rapid Commun. 2012, 33, 958–971. [Google Scholar] [CrossRef]
- Mackay, M.E.; Dao, T.T.; Tuteja, A.; Ho, D.L.; Horn, B.V.; Kim, H.-C.; Hawker, C.J. Nanoscale effects leading to non-Einstein-like decrease in viscosity. Nature Mater. 2003, 2, 762–766. [Google Scholar] [CrossRef]
- Oria, L.; Aguado, R.; Pomposo, J.A.; Colmenero, J. A versatile “click” chemistry precursor of functional polystyrene nanoparticles. Adv. Mater. 2010, 22, 3038–3041. [Google Scholar] [CrossRef] [Green Version]
- Wulff, G.; Chong, B.-O.; Kolb, U. Soluble single-molecule nanogels of controlled structure as a matrix for efficient artificial enzymes. Angew. Chem. Int. Ed. 2006, 45, 2955–2958. [Google Scholar] [CrossRef]
- Huerta, E.; Stals, P.J.M.; Meijer, E.W.; Palmans, A.R.A. Consequences of folding a water-soluble polymer around an organocatalyst. Angew. Chem. Int. Ed. 2013, 52, 2906–2910. [Google Scholar] [CrossRef]
- Ryu, J.-H.; Chacko, R.T.; Jiwpanich, S.; Bickerton, S.; Babu, R.P.; Thayumanavan, S. Self-cross-linked polymer nanogels: A versatile nanoscopic drug delivery platform. J. Am. Chem. Soc. 2010, 132, 17227–17235. [Google Scholar]
- Njiang, G.; Liu, G.; Hong, L. Chiral imprinting of diblock copolymer single-chain particles. Langmuir 2012, 27, 7176–7184. [Google Scholar]
- Tamura, A.; Oishi, M.; Nagaski, Y. Enhanced cytoplasmatic delivery of siRNA using a stabilized polyion complex based on PEGylated nanogels with a cross-linked polyamine structure. Biomacromolecules 2009, 10, 1818–1827. [Google Scholar] [CrossRef]
- Hamilton, S.K.; Harth, E. Molecular dendritic transporter nanoparticle vectors provide efficient intracellular delivery of peptides. ACS Nano 2009, 3, 402–410. [Google Scholar] [CrossRef]
- Perez-Baena, I.; Loinaz, I.; Padro, D.; Garcia, I.; Grande, H.J.; Odriozola, I. Single-chain polyacrylic nanoparticles with multiple Gd(III) centres as potential MRI constrast agents. J. Mater. Res. 2010, 20, 6916–6922. [Google Scholar]
- Qian, G.; Zhu, B.; Wang, Y.; Deng, S.; Hu, A. Size-tunable polymeric nanoreactors for one-pot synthesis and encapsulation of quantum dots. Macromol. Rapid Commun. 2012, 33, 1393–1398. [Google Scholar] [CrossRef]
- Zhu, B.; Sun, S.; Wang, Y.; Deng, S.; Qian, G.; Wang, M.; Hu, A. Preparation of carbon nanodots from single chain polymeric nanoparticles and theoretical investigation of the photoluminescence mechanism. J. Mater. Chem. C 2013, 1, 580–586. [Google Scholar] [CrossRef]
- Gillissen, M.A.J.; Voets, I.K.; Meijer, E.W.; Palmans, A.R.A. Single chain polymeric nanoparticles as compartmentalised sensors for metal ions. Polym. Chem. 2012, 3, 3166–3174. [Google Scholar] [CrossRef]
- Terashima, T.; Mes, T.; De Greef, T.F.A.; Gillissen, M.A.J.; Besenius, P.; Palmans, A.R.A.; Meijer, E.W. Single-chain folding of polymers for catalytic systems in water. J. Am. Chem. Soc. 2011, 133, 4742–4745. [Google Scholar] [CrossRef]
- Whitaker, D.E.; Mahon, C.S.; Fulton, D.A. Thermoresponsive dynamic covalent single-chain polymer nanoparticles reversibly transform into a hydrogel. Angew. Chem. Int. Ed. 2013, 52, 956–959. [Google Scholar] [CrossRef]
- Destarac, M. Controlled radical polymerization: industrial stakes, obstacles and achievements. Macromol. React. Eng. 2010, 4, 165–179. [Google Scholar] [CrossRef]
- Chiefari, J.; Chong, Y.K.; Ercole, F.; Krstina, J.; Jeffery, J.; Le, T.P.T.; Mayadunne, R.T.A.; Meijs, G.F.; Moad, C.L.; Moad, G.; et al. Living free-radical polymerization by reversible addition–fragmentation chain transfer: The RAFT process. Macromolecules 1998, 31, 5559–5562. [Google Scholar] [CrossRef]
- Wang, J.-S.; Matyjaszewski, K. Controlled/l“iving” radical polymerization: Atom transfer radical polymerization in the presence of transition-metal complexes. J. Am. Chem. Soc. 1995, 117, 5614–5615. [Google Scholar] [CrossRef]
- Hawker, C.J.; Bosman, A.W.; Harth, E. New polymer synthesis by nitroxide mediated living radical polymerizations. Chem. Rev. 2001, 101, 3661–3688. [Google Scholar] [CrossRef]
- Lynn, D.M.; Kanaoka, S.; Grubbs, R.H. Living ring-opening metathesis polymerization in aqueous media catalyzed by well-defined Ruthenium carbene complexes. J. Am. Chem. Soc. 1996, 118, 784–790. [Google Scholar] [CrossRef]
- Ruiz de Luzuriaga, A.; Ormategui, N.; Grande, H.J.; Odriozola, I.; Pomposo, J.A.; Loinaz, I. Intramolecular click cycloaddition: an efficient room-temperature route towards bioconjugable polymeric nanoparticles. Macromol. Rapid Commun. 2008, 29, 1156–1160. [Google Scholar] [CrossRef]
- Ruiz de Luzuriaga, A.; Perez-Baena, I.; Montes, S.; Loinaz, I.; Odriozola, I.; García, I.; Pomposo, J.A. New route to polymeric nanoparticles by click chemistry using bifunctional cross-linkers. Macromol. Symp. 2010, 296, 303–310. [Google Scholar] [CrossRef]
- Beck, J.B.; Killops, K.L.; Kang, T.; Sivanandan, K.; Bayles, A.; Mackay, M.E.; Wooley, K.; Hawker, C.J. Facile preparation of nanoparticles by intramolecular cross-linking of isocyanate functionalized copolymers. Macromolecules 2009, 42, 5629–5635. [Google Scholar] [CrossRef]
- Zhu, B.; Ma, J.; Li, Z.; Hou, J.; Cheng, X.; Qian, G.; Liu, P.; Hu, A. Formation of polymeric nanoparticles via Bergman cyclization mediated intramolecular chain collapse. J. Mater. Chem. 2011, 21, 2679–2683. [Google Scholar] [CrossRef]
- Sanchez-Sanchez, A.; Asenjo-Sanz, I.; Buruaga, L.; Pomposo, J.A. Naked and self-clickable propargylic-decorated single-chain nanoparticle precursors via Redox-initiated RAFT polymerization. Macromol. Rapid Commun. 2012, 33, 1262–1267. [Google Scholar] [CrossRef]
- Seo, M.; Beck, B.J.; Paulusse, J.M.; Hawker, C.J.; Kim, S.Y. Polymeric nanoparticles via noncovalent cross-linking of linear chains. Macromolecules 2008, 41, 6413–6418. [Google Scholar] [CrossRef]
- Buruaga, L.; Pomposo, J.A. Metal-free polymethyl methacrylate (PMMA) nanoparticles by enamine “click” chemistry at room temperature. Polymers 2011, 3, 1673–1683. [Google Scholar] [CrossRef] [Green Version]
- Croce, T.A.; Hamilton, S.K.; Chen, M.L.; Muchalski, H.; Harth, E. Alternative o-quinodimethane cross-linking precursors for intramolecular chain collapse nanoparticles. Macromolecules 2007, 40, 6028–6031. [Google Scholar] [CrossRef]
- Zhu, B.; Qian, G.; Xiao, Y.; Deng, S.; Wang, M.; Hu, A. A convergence of photo-bergman cyclization and intramolecular chain collapse towards polymeric nanoparticles. J. Polym. Sci. Pol. Chem. 2011, 49, 5330–5338. [Google Scholar] [CrossRef]
- Cengiz, H.; Aydogan, B.; Ates, S.; Acikalin, E.; Yagci, Y. Intramolecular cross-linking of polymers using difunctional acetylenes via click chemistry. Des. Monomers Polym. 2011, 14, 68–78. [Google Scholar]
- Jiang, X.; Pu, H.; Wang, P. Polymer nanoparticles via intramolecular crosslinking of sulfonyl azide functionalized polymers. Polymer 2011, 52, 3597–3602. [Google Scholar] [CrossRef]
- Murray, B.S.; Fulton, D.A. Dynamic covalent single-chain polymer nanoparticles. Macromolecules 2011, 44, 7242–7252. [Google Scholar] [CrossRef]
- Jiang, J.; Thayumanavan, S. Synthesis and characterization of amine-functionalized polystyrene nanoparticles. Macromolecules 2005, 38, 5886–5891. [Google Scholar] [CrossRef]
- Ormategui, N.; García, I.; Padro, D.; Cabanero, G.; Grande, H.J.; Loinaz, I. Synthesis of single chain thermoresponsive polymer nanoparticles. Soft Matter 2012, 8, 734–740. [Google Scholar] [CrossRef]
- He, J.; Tremblay, L.; Lacelle, S.; Zhao, Y. Preparation of polymer single chain nanoparticles using intramolecular photodimerization of coumarin. Soft Matter 2011, 7, 2380–2386. [Google Scholar] [CrossRef]
- Wang, P.; Pu, H.; Jin, M. Single-chain nanoparticles with well-defined structure via intramolecular crosslinking of linear polymers with pendant benzoxazine groups. J. Polym. Sci. Pol. Chem. 2011, 49, 5133–5141. [Google Scholar] [CrossRef]
- Appel, E.A.; del Barrio, J.; Dyson, J.; Isaacs, L.; Sherman, O.A. Metastable single-chain polymer nanoparticles prepared by dynamic cross-linking with nor-seco-cucurbit[10]uril. Chem. Sci. 2012, 3, 2278–2281. [Google Scholar] [CrossRef]
- Mecerreyes, D.; Lee, V.; Hawker, C.J.; Hedrick, J.L.; Wursh, A.; Volksen, W.; Magbitang, T.; Huang, E.; Miller, R.D. A novel approach to functionalized nanoparticles: Self-crosslinking of macromolecules in ultradilute solution. Adv. Mater. 2001, 13, 204–208. [Google Scholar] [CrossRef]
- Park, K.S.; Kim, D.Y.; Choi, S.K.; Suh, D.H. Novel approach to chemically amplified resist materials for next generation of lithography. Jpn. J. Appl. Phys. 2003, 42, 3877–3880. [Google Scholar] [CrossRef]
- Harth, E.; Horn, B.V.; Lee, V.Y.; Germack, D.S.; Gonzales, C.P.; Miller, R.D.; Hawker, C.J. A facile approach to architecturally defined nanoparticles via intramolecular chain collapse. J. Am. Chem. Soc. 2002, 124, 8653–8660. [Google Scholar]
- Adkins, C.T.; Muchalski, H.; Harth, E. Nanoparticles with individual site-isolated semiconducting polymers from intramolecular chain collapse processes. Macromolecules 2009, 42, 5786–5792. [Google Scholar] [CrossRef]
- Cherian, A.E.; Sun, F.C.; Sheiko, S.S.; Coates, G.W. Formation of nanoparticles by intramolecular cross-linking: following the reaction progress of single polymer chains by atomic force microscopy. J. Am. Chem. Soc. 2007, 129, 11350–11351. [Google Scholar] [CrossRef]
- Foster, E.J.; Berda, E.B.; Meijer, E.W. Metastable supramolecular polymer nanoparticles via intramolecular collapse of single polymer chains. J. Am. Chem. Soc. 2009, 131, 6964–6966. [Google Scholar] [CrossRef]
- Tuten, B.T.; Chao, D.; Lyon, C.K.; Berda, E.B. Single-chain polymer nanoparticles via reversible disulfide bridges. Polym. Chem. 2012, 3, 3068–3071. [Google Scholar] [CrossRef]
- Günay, K.A.; Theato, P.; Klok, H.-A. History of Post-Polymerization Modification. In Functional Polymers by Post-Polymerization Modification: Concepts, Guidelines, and applications, 1st ed.; Theato, P., Klok, H.-A., Eds.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2013; pp. 1–44. [Google Scholar]
- Cunningham, W.A. Sulfur. III. J. Chem. Educ. 1935, 12, 120–124. [Google Scholar] [CrossRef]
- Serniuk, G.E.; Banes, F.W.; Swaney, M.W. Study of the reaction of buna rubbers with aliphatic mercaptans. J. Am. Chem. Soc. 1948, 70, 1804–1808. [Google Scholar] [CrossRef]
- Pepper, K.W.; Paisley, H.M.; Young, M.A. Properties of ion-exchange resins in relation to their structure. Part VI. Anion-exchange resins derived from styrene-divinyl-benzene copolymers. J. Chem. Soc. 1953, 4097–4105. [Google Scholar] [CrossRef]
- Merrifield, R.B. Solid phase peptide synthesis. I. The synthesis of a tetrapeptide. J. Am. Chem. Soc. 1963, 85, 2149–2154. [Google Scholar] [CrossRef]
- Iwakura, Y.; Kurosaki, T.; Nakabayashi, N. Reactive fiber. Part I. Copolymerization and copolymer of acrylonitrile with glycidyl methacrylate and with glycidyl acrylate. Makromol. Chem. 1961, 44, 570–590. [Google Scholar] [CrossRef]
- Iwakura, Y.; Kurosaki, T.; Imai, Y. Reaction between amines and epoxy groups of acrylonitrile-glycidyl acrylate copolymers. Makromol. Chem. 1965, 86, 73–79. [Google Scholar] [CrossRef]
- Iwakura, Y.; Kurosaki, T.; Ariga, N.; Ito, T. Copolymerization of methyl methacrylate with glycidyl methacrylate and the reaction of the copolymer with amines. Makromol. Chem. 1966, 97, 128–138. [Google Scholar] [CrossRef]
- Dobish, J.N.; Hamilton, S.K.; Harth, E. Synthesis of low-temperature benzocyclobutene cross-linker and utilization. Polym. Chem. 2012, 3, 857–860. [Google Scholar] [CrossRef]
- Radu, J.E.F.; Novak, L.; Hartmann, J.F.; Beheshti, N.; Kjoniksen, A.-L.; Nyström, B.; Borbély, J. Structural and dynamical characterization of poly-gamma-glutamic acid-based cross-linked nanoparticles. Colloid. Polym. Sci. 2008, 286, 365–376. [Google Scholar] [CrossRef]
- Akagi, T.; Piyapakorn, P.; Akashi, M. Formation of unimer nanoparticles by controlling the self-association of hydrophobically modified poly(amino acid)s. Langmuir 2012, 28, 5249–5256. [Google Scholar] [CrossRef]
- Rostovtsev, V.V.; Green, L.G.; Fokin, V.V.; Sharpless, K.B. A stepwise Huisgen cycloaddition process: copper(I)-catalyzed regioselective “ligation” of azides and terminal alkynes. Angew. Chem. Int. Ed. 2002, 41, 2596–2599. [Google Scholar] [CrossRef]
- Tornøe, C.W.; Christensen, C.; Meldal, M. Peptidotriazoles on solid phase: [1,2,3]-triazoles by regiospecific copper(I)-catalyzed 1,3-dipolar cycloadditions of terminal alkynes to azides. J. Org. Chem. 2002, 67, 3057–3064. [Google Scholar] [CrossRef]
- Kolb, H.C.; Finn, M.G.; Sharpless, K.B. Click chemistry: Diverse chemical function from a few good reactions. Angew. Chem. Int. Ed. 2001, 40, 2004–2021. [Google Scholar] [CrossRef]
- Barner-Kowollik, C.; Du Prez, F.E.; Espeel, P.; Hawker, C.J.; Junkers, T.; Schlaad, H.; Van Camp, W. “Clicking” polymers or just efficient linking: What is the difference? Angew. Chem. Int. Ed. 2010, 49, 2–5. [Google Scholar]
- Tuteja, A.; Duxbury, P.M.; Mackay, M.E. Multifunctional nanocomposites with reduced viscosity. Macromolecules 2007, 40, 9427–9434. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sanchez-Sanchez, A.; Pérez-Baena, I.; Pomposo, J.A. Advances in Click Chemistry for Single-Chain Nanoparticle Construction. Molecules 2013, 18, 3339-3355. https://doi.org/10.3390/molecules18033339
Sanchez-Sanchez A, Pérez-Baena I, Pomposo JA. Advances in Click Chemistry for Single-Chain Nanoparticle Construction. Molecules. 2013; 18(3):3339-3355. https://doi.org/10.3390/molecules18033339
Chicago/Turabian StyleSanchez-Sanchez, Ana, Irma Pérez-Baena, and José A. Pomposo. 2013. "Advances in Click Chemistry for Single-Chain Nanoparticle Construction" Molecules 18, no. 3: 3339-3355. https://doi.org/10.3390/molecules18033339