Abstract
A series of novel IMB-070593 derivatives containing a substituted benzyloxime moiety and displaying a remarkable improvement in lipophilicity were synthesized and evaluated for their in vitro antimycobacterial and antibacterial activity. Our results reveal that the target compounds 19a–m have considerable Gram-positive activity (MIC: <0.008–32 µg/mL), although they are generally less active than the reference drugs against the Gram-negative strains. In particular, compounds 19h, 19j, 19k and 19m show good activity (MICs: <0.008–4 µg/mL) against all of the tested Gram-positive strains, including ciprofloxacin (CPFX)- and/or levofloxacin (LVFX)-resistant MSSA, MRSA and MSSE. Moreover, compound 19l (MIC: 0.125 µg/mL) is found to be 2–4 fold more active than the parent IMB070593, CPFX and LVFX against M. tuberculosis H37Rv ATCC 27294.
1. Introduction
As one of the largest classes of antimicrobial agents quinolones have been known for 50 years. These antibiotics, which inhibit the type II bacterial topoisomerases DNA gyrase and topoisomerase IV, are used mainly to fight both community-acquired and serious hospital-acquired infections [1]. On the other hand, DNA gyrase is considered to be the sole topoisomerase drug target of fluoroquinolones in Mycobacterium tuberculosis (MTB) [2]. Ciprofloxacin (CPFX), ofloxacin and sparfloxacin were recommended by the World Health Organization in 1996 as second-line agents for the treatment of tuberculosis (TB), mainly in cases involving resistance or intolerance to first-line anti-TB therapy [3]. Moreover, two newer C-8 methoxyfluoroquinolones, moxifloxacin (MXFX) and gatifloxacin, possessing a particularly strong in vitro and in vivo activity against MTB [4,5] are currently being further evaluated as anti-TB drugs.
However, since the mid-1990s, quinolone resistance started to increase in almost all Gram-positive and Gram-negative species as well as MTB [6,7]. The continued increase in resistance has put enormous pressure on public health systems worldwide [8], predominantly due to the high level of use and to some degree of abuse. Although considerable results have been achieved recently, there is an urgent need for the discovery and development of effective novel fluoroquinolones to confer desirable biological and pharmacological properties. Clearly, a more practical strategy is to modify the structures of existing fluoroquinolones to increase potency and overcome resistance.
Recently, a great number of syntheses of fluoroquinolone derivatives have been reported, together with the corresponding structure-activity relationship (SAR) studies [9,10,11]. As a result of these SAR studies, it appears evident that the substituent at C-7 position, the only area that substitution of bulky functional group is permitted, plays an important role in the antibacterial potency, antibacterial spectrum and toxicity of fluoroquinolones [12]. Moreover, it is generally believed that simply increasing the lipophilicity could also improve the anti-MTB and antibacterial activity of fluoroquinolones by introduction of an additional functional moiety on the primary or second amino group of the C-7 side chain [13,14,15,16,17,18]. Therefore, reasonable modification at C-7 position is likely to produce more effective anti-TB and antibacterial agents.
In our previous paper, we reported a series of gemifloxacin (GMFX) derivatives with remarkable improvement in lipophilicity and several target compounds featuring a substituted benzyloxime-incorporated pyrrolidino-substitution at C-7 position have superior Gram-positive activity to the corresponding methyloxime analog (GMFX) [19]. Similarly, some fluoroquinolone derivatives bearing a 3-(substituted benzyloximido)-2-(aminomethyl)azetidin-1-yl group at the C-7 position were found to be far more active than the corresponding methyloxime analog against Gram-positive strains in our study [1].
Inspired by the above research results with azetidinyl- and pyrrolidinyl-based fluoroquinolones, we planned to make structural modifications on IMB-070593 (Figure 1), a piperidinyl-based fluoroquinolone candidate discovered in our lab. In late pre-clinical stage of development currently, IMB-070593 possesses potent in vitro and in vivo antibacterial activity [20] and in vitro anti-MTB activity [21] as well as extremely low phototoxicity, hepatotoxicity and cardiac toxicity (unpublished data). Given that replacement of methyloxime of IMB-070593 by aliphatic moieties such as ethyl group with slightly increased lipophilicity, has almost no impact on the antibacterial activity [20], a series of novel IMB-070593 derivatives were designed, synthesized by introduction of diversified more lipophilic benzyloximes instead of methyloxime of the piperidine ring (Figure 1) in this study. Our primary object was to optimize the potency of IMB-070593 against MTB and clinically important pathogens including methicillin-resistant S. aureus (MRSA).
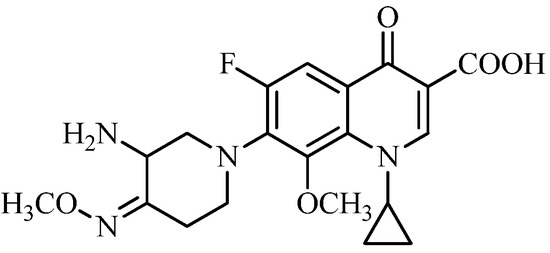
Figure 1.
Structure of IMB-070593.
Figure 1.
Structure of IMB-070593.
2. Results and Discussion
2.1. Chemistry
Commercially unavailable O-benzylhydroxylamines 4a–m were first prepared according to Scheme 1. Reduction of various benzaldehydes 1a–d with sodium borohydride in methanol gave the phenylmethanols 2a–d, and then 2a–d and commercially available compounds 2e–m were coupled with 2-hydroxyisoindoline-1,3-dione in the presence of diethylazodicarboxylate (DEAD) and triphenylphosphine (PPh3) in tetrahydrofuran to produce condensates 3a–m. The desired compounds 4a–m were obtained by treatment of 3a–m with hydrazine hydrate in dichloromethane according to well established procedures [22].
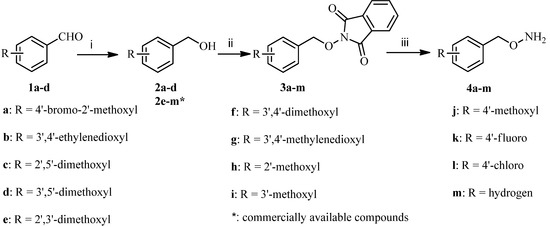
Scheme 1.
Synthesis of O-benzylhydroxylamines 4a–m.
Scheme 1.
Synthesis of O-benzylhydroxylamines 4a–m.
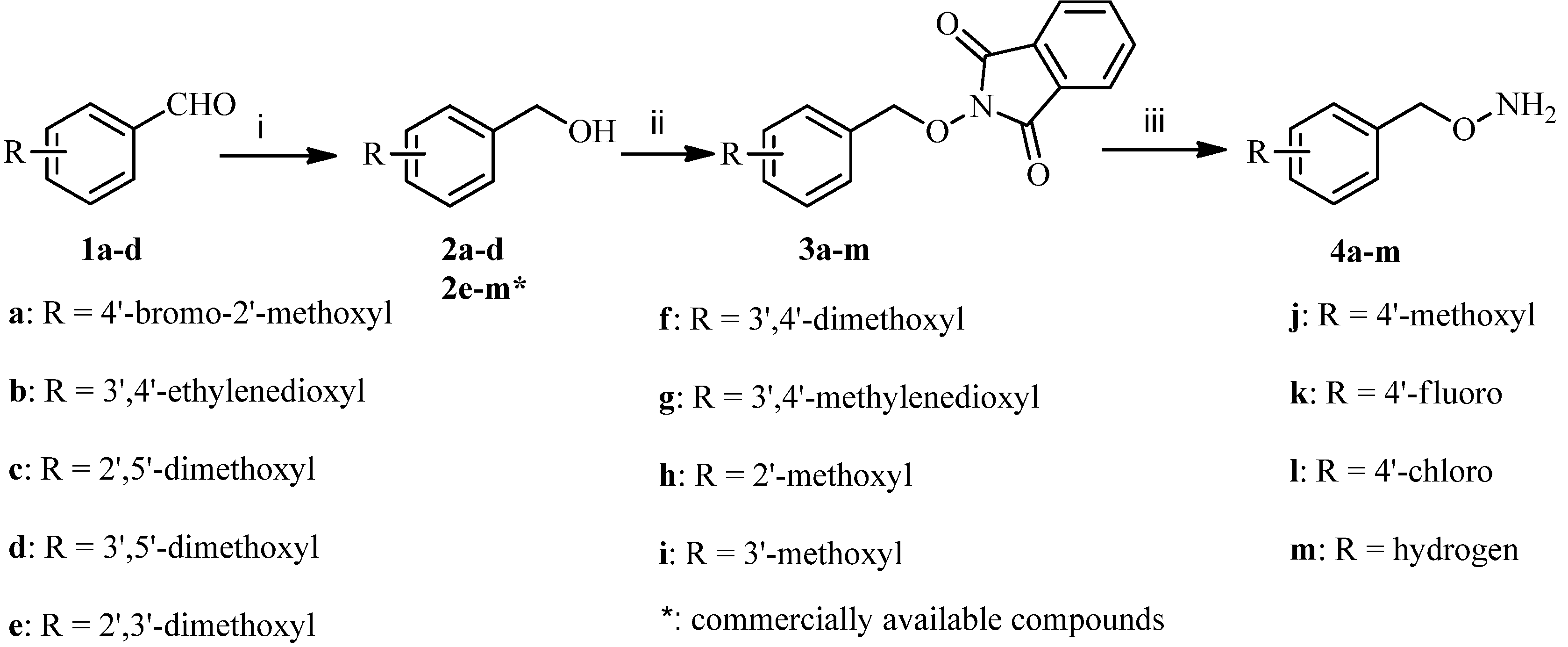
Reagents and conditions: (i) NaBH4, CH3OH, rt, 2 h, 84%–90%; (ii) 2-hydroxyisoindoline-1,3-dione, PPh3, DEAD, THF, 0 °C, 1 h, 79%–85%; (iii) N2H4.H2O, CH2Cl2, rt, 2 h, 89%–92%.
Detailed synthetic pathways to novel 3-amino-4-benzyloxyimino-piperidines 13a–m and IMB-070593 derivatives 19a–m are depicted in Scheme 2, Scheme 3, Scheme 4, respectively. The synthesis of 13m is illustrated in Scheme 2. We previously reported a low total yield (<5%) synthetic route to the key intermediate 9 from ethyl 1-benzyl-4-oxopiperidine-3-carboxylate via a 9-step procedure [20]. In order to overcome its disadvantages, a simpler route for synthesis of 9 was developed in this work. Oximation of readily available tert-butyl 3-cyano-4-oxopiperidine-1-carboxylate (5) [23] followed by treatment with DMSO-H2O2-K2CO3 system gave amide 7. Hoffmann degradation of the amide 7 was conducted successfully using freshly prepared sodium hypobromite instead of sodium hypochlorite to yield primary amine 8. The N-tert-butoxycarbonyl (Boc) protecting group on amine 8 was removed with hydrogen chloride gas in methylene chloride to afford the side chain compound 9 of IMB-070593 in good total yield (>20%, from 5).
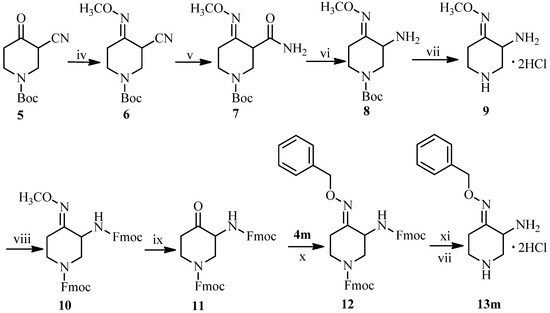
Scheme 2.
Synthesis of piperidine derivative 13m.
Scheme 2.
Synthesis of piperidine derivative 13m.
Reagents and conditions: (iv) CH3ONH2, HCl, NaOH, CH3OH, 50 °C, 3 h, 93%; (v) H2O2, K2CO3, DMSO, rt, 2 h, 71%; (vi) NaBrO, CH3CN, rt, overnight, 63%; (vii) HCl gas, CH2Cl2, rt, 3h, 52%; (viii) FmocCl, Et3N, CH2Cl2, rt, 57%; (ix) Ethyl acetoacetate, 36% HCl, CH3OH, 60 °C, 4 h, 52%; (x) C2H5OH, rt, 5 h, 75%; (xi) 10%NaOH, THF, reflux, 10 h, 45%.
However, the conversion of the methyl oxime 8 or 9 into the corresponding ketone by acidic hydrolysis turned out to be rather complicated and the products difficult to purify. Even though various acids (HCl, HBr, HI, H2SO4, CH3SO3H) in different solvents (H2O, CH3OH, C2H5OH) were used for the hydrolysis, we were not able to obtain the desired product in acceptable yield. Therefore, the primary and second amino groups of 9 had to be protected by treatment with 9-fluorenylmethyl chloroformate (FmocCl) to form the bis-Fmoc protected methyloxime 10, which upon hydrolysis afforded the desired ketone 11 successfully albeit in poor yield (<30% for the two steps). Oximation of the ketone 11 with O-benzylhydroxylamine 4m in ethanol followed by deprotection of the bis-Fmoc groups on the amine 12 in NaOH-THF system provided 3-amino-4-(benzyloxyimino)piperidine dihydrochloride (13m).
The synthesis of the other side chain compounds 13a–l is illustrated in Scheme 3. Reaction of ketone 5 with various substitueted O-benzylhydroxylamines 4a–l followed by treatment with DMSO-H2O2-K2CO3 system afforded amides 15a–l. Hoffmann degradation of 15a–l and subsequently deprotection of the resulting amines 16a–l gave 3-amino-4-(substituted benzyloxyimino)piperidine dihydrochlorides 13a–l.
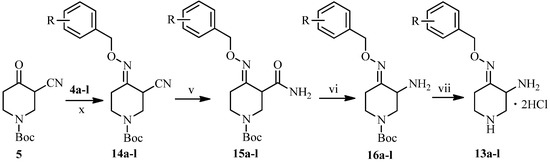
Scheme 3.
Synthesis of piperidine derivatives 13a–l.
Scheme 3.
Synthesis of piperidine derivatives 13a–l.
Reagents and conditions: (x) C2H5OH, rt, 5 h, 90%–95%; (v) H2O2, K2CO3, DMSO, rt, 2 h, 71%–75%; (vi) NaBrO, CH3CN, rt, overnight, 59%–65%; (vii) HCl gas, CH2Cl2, rt, 3 h, 50%–56%. See Scheme 1 for structures of the groups a–l.
Finally, as illustrated in Scheme 4, the novel IMB-070593 derivatives 19a–m were obtained by coupling the side chain compounds 13a–m with the boric chelate 18 of the fluoroquinolone nucleus 17 according to well-established literature procedures [12,24].
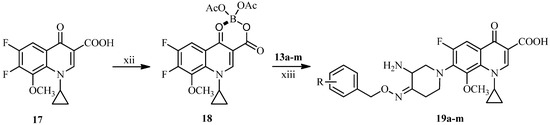
Scheme 4.
Synthesis of target compounds 19a–m.
Scheme 4.
Synthesis of target compounds 19a–m.
Reagents and conditions: (xii) H3BO3, (CH3CO)2O, CH3CO2H, 100 °C, 2 h, 70%; (xiii) (1) Et3N, CH3CN, 50 °C, overnight; (2) 5% NaOH, 50 °C, 1 h, 18%–34% (for two steps). See Table 1 for structures.
Because the oxime group is present in the E- or Z- configuration, it was necessary to determine the geometries of all the oxime target compounds 19a–m. Although we were unable to prepare X-ray-quality single crystals of any oxime intermediate or product in this study, we had previously obtained single crystals of 4-(methoxyimino)-3-methylaminopiperidinone dihydrochloride, an N-methylated 9 derivative, in which the piperidine ring adopts a chair conformation and the methyloxime geometry exists in an E-configuration [20]. Accordingly, we can speculate that the oxime group of the target compounds in this study should have the same E-configuration due to single signals of the piperidine ring observed in the 1H-NMR spectra of the compounds.
2.2. Lipophilicity
Lipophilicity of the target compounds 19a–m and the parent IMB-070593 is expressed in the term of their ClogP values which were calculated by Chemoffice 2010 software. As shown in Table 1, there is a remarkable improvement in the lipophilicity of the derivatives 19a–m as evidenced by their ClogP values (0.60–1.86) which are much more than that of IMB-070593 (−0.72) (statistically significant at p < 0.001 using t-test) (Table 1).

Table 1.
Structures, lipophilicity and antimycobacterial activity of compounds 19a–m. 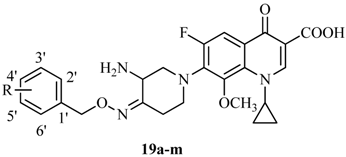

| Compd. | R | Clog p a | MIC (µg/mL) | |
|---|---|---|---|---|
| MTB b | MDR-MTB c | |||
| 19a | 4′-bromo-2′-methoxyl | 1.86 | 16 | 16 |
| 19b | 3′,4′-ethylenedioxyl | 0.87 | 8 | 4 |
| 19c | 2′,5′-dimethoxyl | 0.95 | 8 | 8 |
| 19d | 3′,5′-dimethoxyl | 0.95 | 16 | 4 |
| 19e | 2′,3′-dimethoxyl | 0.60 | 8 | 8 |
| 19f | 3′,4′-dimethoxyl | 0.60 | 8 | 8 |
| 19g | 3′,4′-methylenedioxyl | 0.91 | 4 | 1 |
| 19h | 2′-methoxyl | 0.86 | 8 | 4 |
| 19i | 3′-methoxyl | 0.86 | 4 | 1 |
| 19j | 4′-methoxyl | 0.86 | 4 | 2 |
| 19k | 4′-fluoro | 1.08 | 4 | 4 |
| 19l | 4′-chloro | 1.65 | 0.125 | 4 |
| 19m | hydrogen | 0.94 | 4 | 2 |
| IMB | −0.72 | 0.25 | 0.125 | |
| CPFX | 0.5 | 0.25 | ||
| LVFX | 0.25 | 0.125 | ||
| MXFX | 0.125 | 0.125 | ||
| INH | 0.06 | 2 | ||
| RIP | 0.06 | >40 | ||
IMB, IMB070593; CPFX, ciprofloxacin; LVFX, levofloxacin; MXFX, moxifloxacin; RIP, rifampicin; INH, isoniazid; a The Clog p is calculated by Chemoffice 2010 software; b MTB: MTB H37Rv ATCC 27294; c MDR-MTB: MDR-MTB 20161 is resistant to RIP and INH.
2.3. Anti-MTB Activity
The target compounds 19a–m were initially evaluated for their in vitro activity against MTB H37Rv ATCC 27294 and MDR-MTB 20161 clinical isolate using the Microplate Alamar Blue Assay (MABA) [25,26]. The minimum inhibitory concentration (MIC) is defined as the lowest concentration effecting a reduction in fluorescence of ≥90% relative to the mean of replicate bacterium-only controls and MICs of the compounds 19a–m along with CPFX, levofloxacin (LVFX), MXFX, isoniazid (INH) and rifampicin (RIP) for comparison are presented in Table 1.
The data reveal that the target compounds 19a–m have considerable activity against both of the tested MTB strains (MICs: 0.125–16 µg/mL). As for MTB H37Rv ATCC 27294, the most active compound 19l (MIC: 0.125 µg/mL) was found to be 2–4 fold more potent than the parent IMB070593, CPFX and LVFX (MICs: 0.25–0.5 µg/mL), and comparable to MXFX (MIC: 0.125 µg/mL), the fluoroquinolone with the strongest anti-MTB activity.
In the case of MDR-MTB 20161 clinical isolate resistant to RIP and INH, the target compounds 19a–m (MICs: 1–16 µg/mL) show less active than the parent IMB070593 and the three other reference fluoroquinolones, but compounds 19g and 19i have useful activity (MICs: 1 µg/mL) against this strain.
2.4. Antibacterial Activity
The target compounds 19a–m were evaluated for their in vitro antibacterial activity against representative strains using standard techniques [27]. Minimum inhibitory concentration (MIC) is defined as the concentration of the compound required to give complete inhibition of bacterial growth, and MIC values of 19a–m against Gram-positive and Gram-negative strains along with the parent IMB-070593, CPFX and LVFX for comparison, are listed in Table 2 and Table 3, respectively.
Generally, the target compounds 19a–m have potent in vitro antibacterial activity against the tested Gram-positive strains (MICs: <0.008–32 µg/mL). Among of them, compounds 19h, 19j, 19k and 19m show good potency in inhibiting the growth of Methicillin-susceptible Staphylococcus aureus (MSSA), MRSA, methicillin-susceptible Staphylococcus epidermidis (MSSE), methicillin-resistant Staphylococcus epidermidis (MRSE) and Streptococcus pneumonia (MICs: <0.008–4 µg/mL). Notably, the four target compounds 19h, 19j, 19k and 19m have also useful activity (MICs: <0.008– 4 µg/mL) against both CPFX- and LVFX-resistant MRSA12-1/12-2 (MICs: 32->128 µg/mL) and MRSE12-3 (MICs: 16–32 µg/mL), CPFX-resistant MRSA12-3 (MIC: 64 µg/mL), as well as LVFX-resistant MSSA12-3 (MIC: 16 µg/mL) and MRSA12-4 (MIC: 32 µg/mL).
On the other hand, the target compounds 19a–m are generally less active than the parent IMB-070593, CPFX and LVFX against the tested Gram-negative strains with few exceptions. It is noted that compounds 19e and 19h possess good potency against all of the four clinical strains of Klebsiella pneumonia (MICs: <0008–0.5 µg/mL). However, all of 19a–m, like the three reference drugs, have virtually no activity against the extended-spectrum β-lactamase (ESBLs)-producing Escherichia coli and Klebsiella pneumonia, due partly to resistance of these ESBLs-producing strains inherent to fluoroquinolones.
Variations (R) on the benzene ring of the benzyl moiety in this study include mono-/bi-methoxyl, 3′,4′-methylenedioxyl/ethylenedioxyl, fluorine, chlorine and bromine substitution (Table 1). The activity imparted to IMB-070593 derivatives by R groups against Gram-positive strains was in the order: 4′-fluoro > 4′-chloro (cf. 19k vs. 19l) or 4′-methoxyl ≥ 2′-methoxyl > 3′-methoxyl (19j vs. 19h vs. 19i) for mono-substitution. However, introduction of an additional bromine atom at the 4′ position of 2′-methoxybenzyl moiety goes against the activity (19h vs. 19a). In addition, compound 19c is found to have antibacterial activity comparable to 19d–f, and the same result is obtained in comparisons of the activity of 19b and 19g, therefore, analogs containing a disubstituted benzyl moiety seem to possess similar antibacterial activity.

Table 2.
In vitro antibacterial activity of compounds 19a–m against Gram-positive strains.
| Strains | Compd. MIC (µg/mL) | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 19a | 19b | 19c | 19d | 19e | 19f | 19g | 19h | 19i | 19j | 19k | 19l | 19m | IMB-070593 | CPFX | LVFX | |
| S.a. ATCC | 0.06 | <0.008 | 0.5 | 0.015 | <0.008 | 0.125 | 1 | 0.125 | <0.008 | <0.008 | <0.008 | 0.06 | 0.03 | 0.015 | 1 | 0.25 |
| MSSA12-1 | 0.25 | 0.125 | 0.06 | 0.06 | 0.125 | 0.125 | 0.125 | 0.06 | 0.125 | 0.06 | 0.015 | 0.25 | 0.06 | <0.008 | 0.5 | 0.125 |
| MSSA12-2 | 0.06 | 0.015 | 0.06 | 0.125 | 0.25 | 0.125 | 0.125 | 0.03 | 0.125 | 0.125 | 0.03 | 0.06 | 0.06 | 0.015 | 0.5 | 0.03 |
| MSSA12-3 | 4 | 2 | 2 | 0.25 | 4 | 2 | 2 | 2 | 0.125 | 2 | 0.5 | 4 | 1 | 1 | 0.25 | 16 |
| MSSA12-4 | 0.125 | 0.03 | 0.03 | 0.06 | <0.008 | 0.06 | 0.06 | 0.03 | 0.06 | 0.03 | <0.008 | 0.06 | 0.03 | <0.008 | <0.008 | 0.015 |
| MRSA12-1 | 8 | 4 | 4 | 8 | 8 | 8 | 4 | 4 | 4 | 4 | 4 | 8 | 4 | 2 | >128 | 64 |
| MRSA12-2 | 8 | 4 | 4 | 4 | 8 | 4 | 4 | 4 | 4 | 4 | 4 | 8 | 2 | 2 | 64 | 32 |
| MRSA12-3 | 0.25 | 0.03 | 0.125 | 0.125 | 0.25 | 0.125 | 0.06 | 0.03 | 0.125 | 0.125 | <0.008 | 0.015 | 0.03 | 0.015 | 64 | 0.03 |
| MRSA12-4 | 8 | 4 | 4 | 4 | 8 | 4 | 4 | 4 | 4 | 4 | 4 | 8 | 4 | 2 | 0.5 | 32 |
| MSSE12-1 | 0.125 | 0.25 | 0.125 | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 | 0.125 | 0.125 | 0.03 | 0.25 | 0.125 | 0.015 | 0.5 | 0.25 |
| MSSE12-2 | 0.5 | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 | 0.125 | 0.03 | 0.5 | 0.125 | 0.03 | 2 | 0.25 |
| MSSE12-3 | 0.5 | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 | 0.125 | 0.25 | 0.125 | 0.03 | 0.5 | 0.25 | 0.06 | 2 | 0.03 |
| MSSE12-4 | 0.125 | 0.06 | 0.015 | 0.03 | <0.008 | 0.03 | 0.06 | <0.008 | 0.03 | 0.125 | <0.008 | 0.015 | 0.015 | 0.015 | <0.008 | 0.03 |
| MRSE12-1 | 8 | 16 | 16 | 32 | 8 | 16 | 16 | 4 | 16 | 4 | 4 | 32 | 4 | 1 | 0.015 | 0.03 |
| MRSE12-2 | 2 | 2 | 2 | 8 | 2 | 0.5 | 1 | 2 | 4 | 1 | 4 | 8 | 2 | 1 | 1 | <0.008 |
| MRSE12-3 | 8 | 2 | 4 | 16 | 8 | 4 | 1 | 2 | 8 | 4 | 4 | 16 | 2 | 1 | 32 | 16 |
| MRSE12-4 | 0.5 | 0.125 | 0.125 | 0.125 | 0.25 | 0.25 | 0.25 | 0.125 | 4 | 0.125 | 0.03 | 0.25 | 2 | 0.015 | 0.015 | 0.03 |
| S.p. 12-7 | 0.5 | 0.125 | 0.06 | 0.125 | 0.25 | 0.25 | 0.25 | 0.125 | 0.03 | 0.25 | 0.03 | 0.125 | 0.125 | 0.03 | 0.015 | 0.03 |
| S.p. 12-23 | 0.5 | 0.125 | 0.015 | 0.25 | <0.008 | 0.5 | 0.25 | 0.25 | 0.125 | 0.06 | 0.015 | 0.25 | 0.06 | 0.015 | <0.008 | 0.015 |
LVFX, levofloxacin; CPFX, ciprofloxacin; S.a. ATCC, S.a. ATCC25923; S.a., Staphylococcus aureus; MSSA, Methicillin-susceptible Staphylococcus aureus; MRSA, Methicillin-resistant Staphylococcus aureus; MSSE, Methicillin-susceptible Staphylococcus epidermidis; MRSE, Methicillin-resistant Staphylococcus epidermidis; S.p., Streptococcus pneumonia.

Table 3.
In vitro antibacterial activity of compounds 19a–m against Gram-negative strains.
| Strains | Compd. MIC (µg/mL) | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 19a | 19b | 19c | 19d | 19e | 19f | 19g | 19h | 19i | 19j | 19k | 19l | 19m | IMB-070593 | CPFX | LVFX | |
| E. co. ATCC | 8 | 1 | 0.5 | 4 | <0.008 | 0.25 | <0.008 | <0.008 | <0.008 | 2 | 1 | 0.25 | 2 | 0.06 | <0.008 | <0.008 |
| E. co. 12-1 | 64 | 8 | 16 | 32 | 4 | 16 | 8 | 2 | 1 | 8 | 8 | 16 | 8 | 0.5 | 0.125 | 0.5 |
| E. co. 12-2 | >64 | >128 | >64 | >128 | >128 | >128 | >128 | 16 | >128 | 64 | >128 | 128 | 16 | 16 | 8 | 4 |
| E. co. 12-3 | >64 | >128 | >64 | >128 | >128 | >128 | >128 | >64 | >128 | >64 | >128 | >128 | >128 | 64 | 64 | 32 |
| E. co. 12-4 | >64 | >128 | >64 | >128 | >128 | >128 | >128 | >64 | >128 | >64 | >128 | >128 | >128 | 32 | 16 | 8 |
| E. co. a12-1 | >64 | >128 | >64 | >128 | >128 | >128 | >128 | >64 | >128 | >64 | >128 | >128 | >128 | 16 | 32 | 16 |
| E. co. a 12-2 | >64 | >128 | >64 | >128 | >128 | >128 | >128 | >64 | >128 | >64 | >128 | >128 | >128 | 32 | 64 | 16 |
| E. co. a 12-3 | >64 | >128 | >64 | >128 | >128 | >128 | >128 | >64 | >128 | >64 | >128 | >128 | >128 | 64 | 64 | 32 |
| E. co. a 12-4 | 64 | 8 | 16 | 16 | 4 | 16 | 2 | 1 | 8 | 8 | 8 | 16 | 4 | 0.5 | 0.125 | 0.25 |
| K. p. 12-1 | 16 | 1 | 1 | 16 | <0.008 | 4 | 8 | 0.125 | 2 | 2 | 1 | 1 | 2 | 0.25 | 0.125 | 0.03 |
| K. p. 12-2 | 16 | 4 | 2 | 16 | 0.5 | 16 | 8 | 0.125 | 4 | 4 | 4 | 1 | 4 | 0.5 | 0.015 | 0.06 |
| K. p. 12-3 | 16 | 4 | 2 | 8 | <0.008 | 8 | 8 | 0.5 | 2 | 2 | 4 | 1 | 4 | 0.25 | 0.06 | 0.06 |
| K. p. 12-4 | 32 | 4 | 2 | 16 | <0.008 | 8 | 8 | 0.5 | 2 | 4 | 4 | 1 | 4 | 0.5 | 0.015 | 0.06 |
| K. p. a12-1 | >64 | 64 | >64 | 128 | 128 | 128 | 64 | 64 | 64 | 64 | 64 | 64 | 32 | 4 | 0.5 | 1 |
| K. p. a 12-2 | >64 | 64 | >64 | 128 | 128 | 128 | 64 | 64 | 32 | 64 | 64 | 64 | 32 | 8 | 4 | 4 |
| K. p. a 12-3 | >64 | >128 | >64 | >128 | >128 | >128 | >128 | >64 | >128 | >64 | >128 | >128 | >128 | 32 | 64 | 16 |
| K. p. a 12-4 | >64 | 64 | >64 | 128 | >128 | 128 | 64 | 64 | 128 | 64 | 128 | 128 | 32 | 16 | 4 | 8 |
| K. p. a 12-5 | >64 | >128 | >64 | >128 | >128 | 128 | >128 | >64 | >128 | >64 | >128 | >128 | >128 | 32 | 32 | 16 |
| P. a. ATCC | 16 | 2 | 2 | 8 | 4 | 16 | 8 | 1 | 4 | 2 | 2 | 1 | 2 | 2 | 1 | 8 |
| P. a. 12-12 | 64 | 16 | 32 | 64 | 8 | 64 | 16 | 32 | 32 | 8 | 16 | 32 | 16 | 2 | 0.125 | 0.25 |
| P. a. 12-14 | 8 | 4 | 16 | 8 | 8 | 8 | 4 | 8 | 4 | 2 | 4 | 8 | 2 | 2 | 32 | 32 |
LVFX, levofloxacin; CPFX, ciprofloxacin; E. co. ATCC, E. co. ATCC25922; E. co., Escherichia coli; K.p., Klebsiella pneumoniae; P. a. ATCC, P. a. ATCC27853; P.a., Pseudomonas aeruginosa; a Extended-spectrum β-lactamase-producing.
3. Experimental
3.1. Chemistry
Melting points were determined in open capillaries and are uncorrected. 1H-NMR (400, 500 or 600 MHz) and 13C-NMR (100 or 150 MHz) spectra were recorded at 25 °C on Varian Mercury spectrometers. Chemical shifts (δ) are given in ppm relative to tetramethylsilane or the respective NMR solvent. Electrospray ionization (ESI) mass spectra and high resolution mass spectra (HRMS) were obtained on a MDSSCIEX Q-Tap mass spectrometer. The reagents were all of analytical grade or chemically pure. TLC was performed on silica gel plates (Merck, ART5554 60F254).
3.2. Synthesis
3.2.1. General Procedure for the Synthesis of Substituted O-Benzylhydroxylamines 4a–m
To a solution of benzaldehydes 1a-d (100 mmol) dissolved in methanol (500 mL) was added sodium borohydride (200 mmol) at room temperature, and the mixture was stirred at the same temperature for 2 h and concentrated under reduced pressure. The residue was diluted with dichloromethane (500 mL) and washed with water, dried over anhydrous sodium sulfate, and concentrated under reduced pressure to give the corresponding phenylmethanols 2a–d as colorless oils.
To a solution of the above obtained phanylmethanols 2a–d or commercially available 2e–m (60 mmol), 2-hydroxyisoindoline-1,3-dione (60 mmol) and triphenylphosphine (75 mmol) in tetrahydrofuran (300 mL) was added dropwise a solution of diethyl azodicarboxylate (75 mmol) in tetrahydrofuran (15 mL) at 0 °C over 0.5 h. The mixture was stirred at the same temperature for 1 h and concentrated under reduced pressure. The residue was dissolved in methanol (300 mL) and stirred at room temperature for 1 h and filtered to give 3a–m as white solids. Next, to a stirred solution of 3a–m (60 mmol) in dichloromethane (200 mL) was added dropwise hydrazine hydrate (120 mmol). The reaction mixture was stirred at room temperature for 3 h, and then filtered. The filtrate was washed successively with 2 mol/L sodium hydroxide solution (200 mL) and brine (200 mL), dried over anhydrous sodium sulfate and concentrated under reduced pressure to give the corresponding benzyloxyamines 4a–m as colorless oils (56%–70%, from 2a–d; 66%–78%, from 2e–m).
O-(4-Bromo-2-methoxybenzyl) hydroxylamine (4a). The title compound was obtained from 3a as a colorless oil (87%), 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 7.53 (1H, d, J = 8.4 Hz, Ar-H), 7.21 (1H, d, J = 2.4 Hz, Ar-H), 6.98 (1H, q, J1 = 8.4 Hz, J2 = 2.4 Hz, Ar-H), 5.21 (2H, s, OCH2Ar), 3.79 (3H, s, O-CH3). MS-ESI (m/z): 233 (M+H)+.
O-((2,3-Dihydrobenzo[b][1,4]dioxin-6-yl)methyl) hydroxylamine (4b). The title compound was obtained from 3b as a colorless oil (85%), 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 6.81–6.79 (2H, m, Ar-H), 6.77–6.75 (1H, m, Ar-H), 5.95 (2H, s, OCH2Ar), 4.21 (4H, s, 2 × OCH2). MS-ESI (m/z): 182 (M+H)+.
O-(2,5-Dimethoxybenzyl) hydroxylamine (4c). The title compound was obtained from 3c as a colorless oil (90%), 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 6.89 (2H, m, Ar-H), 6.81 (1H, m, Ar-H), 4.57 (2H, s, OCH2Ar), 3.71 (6H, s, O-CH3). MS-ESI (m/z): 184 (M+H)+.
O-(3,5-Dimethoxybenzyl) hydroxylamine (4d). The title compound was obtained from 3d as a colorless oil (88%), 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 6.46 (2H, s), 6.39 (1H, s), 4.50 (2H, s), 3.73 (6H, s). MS-ESI (m/z): 184 (M+H)+.
O-(2,3-Dimethoxybenzyl) hydroxylamine (4e). The title compound was obtained from 3e as a colorless oil (91%), 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 7.03 (1H, t, J = 7.8 Hz, Ar-H), 6.68 (1H, d, J = 7.8 Hz, Ar-H), 6.90 (1H, d, J = 7.8 Hz, Ar-H), 4.56 (2H, s, OCH2Ar), 3.78 (3H, s, O-CH3), 3.69 (3H, s, O-CH3). MS-ESI (m/z): 184 (M+H)+.
O-(3,4-Dimethoxybenzyl) hydroxylamine (4f). The title compound was obtained from 3f as a colorless oil (89%), 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 7.20 (1H, m, Ar-H), 6.93 (2H, m, Ar-H), 5.05 (2H, s, OCH2Ar), 3.78 (6H, s, O-CH3). MS-ESI (m/z): 184 (M+H)+.
O-(Benzo[d][1,3]dioxol-5-ylmethyl) hydroxylamine (4g). The title compound was obtained from 3g as a colorless oil (84%), 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 6.98 (1H, d, J = 1.2 Hz, Ar-H), 6.94 (1H, d, J = 8 Hz, Ar-H), 6.91 (1H, q, J1 = 8 Hz, J2 = 1.2 Hz, Ar-H), 6.05 (2H, s, OCH2Ar), 4.91 (2H, s, OCH2O). MS-ESI (m/z): 168 (M+H)+.
O-(2-Methoxybenzyl) hydroxylamine (4h). The title compound was obtained from 3h as a colorless oil (85%), 1H-NMR (600 MHz, DMSO-d6) δ (ppm): 7.26 (2H, m, Ar-H), 6.95 (1H, d, J = 7.8 Hz, Ar-H), 6.91 (1H, J = 7.8 Hz, Ar-H), 4.56 (2H, s, Ar-H), 3.76 (3H, s, O-CH3). MS-ESI (m/z): 154 (M+H)+.
O-(3-Methoxybenzyl) hydroxylamine (4i). The title compound was obtained from 3i as a colorless oil (86%), 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 7.33 (1H, m, Ar-H), 6.97 (3H, m, Ar-H) 5.00 (2H, s, OCH2Ar), 3.77 (3H, s, O-CH3). MS-ESI (m/z): 154 (M+H)+.
O-(4-Methoxybenzyl) hydroxylamine (4j). The title compound was obtained from 3j as a colorless oil (88%), 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 7.34 (2H, d, J = 8.4 Hz, Ar-H), 6.97 (2H, d, J = 8.4 Hz, Ar-H), 4.90 (2H, s, OCH2Ar), 3.77 (3H, s, O-CH3). MS-ESI (m/z): 154 (M+H)+.
O-(4-Fluorobenzyl) hydroxylamine (4k). The title compound was obtained from 3k as a colorless oil (84%), 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 7.35 (2H, m, Ar-H), 7.16 (2H, m, Ar-H), 4.54 (2H, s, OCH2Ar). MS-ESI (m/z): 142 (M+H)+.
O-(4-Chlorobenzyl) hydroxylamine (4l). The title compound was obtained from 3l as a colorless oil (90%), 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 7.38 (2H, d, J = 8.4 Hz, Ar-H), 7.34 (2H, d, J = 8.4 Hz, Ar-H), 4.56 (2H, s, OCH2Ar). MS-ESI (m/z): 158 (M+H)+.
O-Benzyl hydroxylamine (4m). The title compound was obtained from 3m as a colorless oil (92%), 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 7.39 (4H, m, Ar-H), 7.33 (1H, m, Ar-H), 5.12 (2H, s, OCH2Ar). MS-ESI (m/z): 124 (M+H)+.
tert-Butyl 3-cyano-4-(methoxyimino)piperidine-1-carboxylate (6). To a solution of hydroxylamine hydrochloride (5.01 g, 60 mmol) in methanol (150 mL) was added sodium hydroxide (2.40 g, 60 mmol), and the mixture stirred at room temperature for 30 minutes. The piperidinone 5 (11.18 g, 50 mmol) was added to the mixture, stirred at 50 °C for 3 h and then filtered. The filtrate was concentrated under reduced pressure. The residue was diluted with ethyl acetate (200 mL), washed with water, dried over anhydrous sodium sulfate, and concentrated under reduced pressure to afford the title compound 6 (14.12 g, 93%) as a yellow oil. 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 4.48 (1H, s, piperidine-H), 4.25 (1H, m, piperidine-H), 4.04 (1H, m, piperidine-H), 3.84 (3H, s, NOCH3), 3.14 (1H, brs, piperidine-H), 2.93 (1H, brs, piperidine-H), 2.41 (2H, m, piperidine-H), 1.42 (9H, s, Boc-9H). MS-ESI (m/z): 254 (M+H)+.
tert-Butyl 3-carbamoyl-4-(methoxyimino)piperidine-1-carboxylate (7). To a solution of 6 (10.12 g, 40 mmol) and potassium carbonate (11.06 g, 80 mmol) in dimethyl sulfoxide (100 mL) was added hydrogen peroxide (36.27 mL, 320 mmol) at 15 °C for 1 h, and stirred at room temperature for 3 h. The mixture was diluted with water (200 mL), and extracted by ethyl acetate (200 mL). The combined extracts were washed with brine, dried over anhydrous sodium sulfate, and then concentrated under reduced pressure to give the title compound 7 (7.71 g, 71%) as a light yellow oil. 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 7.37 (1H, s, CONH2), 7.00 (1H, s, CONH2), 4.19–4.10 (1H, brs, piperidine-H), 4.00–3.92 (1H, m, piperidine-H), 3.84 (1H, s, piperidine-H), 3.72 (3H, s, NOCH3), 3.11 (1H, m, piperidine-H), 2.61–2.53 (1H, m, piperidine-H), 2.24 (1H, brs, piperidine-H), 1.42 (1H, s, piperidine-H), 1.38 (9H, s, Boc-9H). MS-ESI (m/z): 294 (M+Na)+.
tert-Butyl 3-amino-4-(methoxyimino)piperidine-1-carboxylate (8). To a stirred solution of 7 (13.55 g, 50 mmol) in acetonitrile (350 mL) was added dropwise 10% sodium hypobromite solution (106.20 mL, 90 mmol) at 5 °C for 1 h. The reaction mixture was stirred at room temperature for 10 h, and adjusted to pH 6.5–7 with 20% acetic acid, and then concentrated under reduced pressure. The residue was dissolved in water (200 mL), adjusted to pH 4–3 with 10% hydrochloride and washed with ethyl acetate. The water layer was adjusted to pH 9 with 15% sodium hydroxide and extracted with ethyl acetate. The combined extracts were dried over anhydrous sodium sulfate and concentrated under reduced pressure to yield the title compound 8 (7.65 g, 63%) as a yellow oil. 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 4.09–4.02 (1H, m, piperidine-H), 3.74 (3H, s, NOCH3), 3.57 (1H, m, piperidine-H), 3.31–3.25 (1H, m, piperidine-H), 3.11 (1H, s, piperidine-H), 2.99–2.92 (1H, brs, piperidine-H), 2.71–2.66 (1H, m, piperidine-H), 1.98 (1H, brs, piperidine-H), 1.38 (9H, s, Boc-9H). MS-ESI (m/z): 244 (M+H)+.
3-Amino-4-methoxyiminopiperidine dihydrocholoride (9). To a solution of 8 (7.29 g, 30 mmol) in dichloromethane was pumped dried hydrochloride gas at room temperature for 30 minutes. The mixture was stirred for another 1 h at room temperature, and concentrated under reduced pressure. The residue was treated with ethyl acetate. The precipitate was collected by suction, and dried under vacuum to afford the title compound 9 (2.15 g, 52%) as a white solid, m.p.: 224–226 °C. 1H-NMR (300 MHz, D2O) δ (ppm): 4.45–4.50 (1H, m, piperidine-H), 4.01–3.97 (4H, m, piperidine-H, NOCH3), 3.70–3.65 (1H, m, piperidine-H), 3.59–3.53 (1H, m, piperidine-H), 3.34–3.28 (1H, m, piperidine-H), 3.23–3.15 (1H, m, piperidine-H), 2.46–2.37 (1H, m, piperidine-H). M.p.: 178–181 °C. MS-FAB (m/z): 144 (M+H)+.
(9H-Fluoren-9-yl)methyl 3-[(9H-fluoren-9-yl)methoxycarbonylamino]-4-(methoxyimino)piperidine-1-carboxylate (10). To a stirring solution of 9 (3.24g, 15 mmol) and triethylamine (6.24 mL,45 mmol) in dichloromethane (300 mL) was added in portions 9-fluorenylmethyl chloroformate (7.76 g, 30 mmol) at 0 °C, and the mixture stirred at room temperature for 5 h, then washed with water, dried over anhydrous sodium sulfate, and concentrated under reduced pressure. The crude product was purified by column chromatography (silica gel), eluting with petroleum ether and ethyl acetate (v:v = 3:1) to produce the title compound 10 (5.02 g, 57%) as an off-white solid, m.p.: 152–154 °C. 1H-NMR (400 MHz, CDCl3) δ (ppm): 7.76 (4H, m, Fmoc-ArH), 7.61 (4H, m, Fmoc-ArH), 7.39 (4H, m, Fmoc-ArH), 7.32 (4H, m, Fmoc-ArH), 4.44 (4H, d, J = 6.8 Hz, 2 × Fmoc-CH2), 4.27 (2H, m, 2 × Fmoc-CH), 4.12 (1H, m, piperidine-H), 3.92 (3H, s, NOCH3), 3.20 (1H, brs, piperidine-H), 3.04 (1H, t, J = 8.8 Hz, piperidine-H), 2.89 (1H, t, J = 11 Hz, piperidine-H), 2.10 (2H, brs, piperidine-H), 1.25 (1H, m, piperidine-H). MS-ESI (m/z): 610 (M+Na)+.
(9H-Fluoren-9-yl)methyl 3-[(9H-fluoren-9-yl)methoxycarbonylamino]-4-oxopiperidine-1-carboxylate (11). To a solution of 10 (11.75 g, 20 mmol) and ethyl acetoacetate (26.02 g, 200 mmol) in methanol (200 mL) was added concentrated hydrochloride (0.4 mmol, 33.33 mL) and stirred at 60 °C for 6 h. The mixture was concentrated under reduced pressure. The residue was dissolved in water (200 mL), adjusted to pH 6.5–7 with saturated sodium bicarbonate solution, and extracted with dichloromethane. The combined extracts were dried over anhydrous sodium sulfate and concentrated under reduced pressure. The crude product was purified by column chromatography (silica gel), eluting with petroleum ether and ethyl acetate (v:v = 2:1) to afford the title compound 11 (5.81 g, 52%) as a white solid, m.p.: 141–142 °C. 1H-NMR (600 MHz, CDCl3) δ (ppm): 7.77 (4H, m, Fmoc-ArH), 7.61 (4H, m, Fmoc-ArH), 7.41 (4H, m, Fmoc-ArH), 7.31 (4H, m, Fmoc-ArH), 4.63 (4H, s, 2 × Fmoc-CH2), 4.28 (2H, m, 2 × Fmoc-CH), 4.12 (1H, m, piperidine-H), 3.72 (1H, brs, piperidine-H), 3.08 (1H, brs, piperidine-H), 2.84 (1H, brs, piperidine-H), 2.66–2.59 (2H, m, piperidine-H), 1.26 (1H, m, piperidine-H). MS-ESI (m/z): 581 (M+Na)+.
(9H-Fluoren-9-yl)methyl 3-[(9H-fluoren-9-yl)methoxycarbonylamino]-4-benzyloxyiminopiperidine-1-carboxylate (12). To a solution of 11 (5.58 g, 10 mmol) in ethanol (100 mL) was added 4m (2.46 g, 20 mmol) and the mixture stirred at room temperature for 10 h and concentrated under reduced pressure. The residue was dissolved in ethyl acetate (100 mL) and washed with brine (200 mL). The combined extracts were dried over anhydrous sodium sulfate and concentrated under reduced pressure. The crude product was purified by column chromatography (silica gel), eluting with petroleum ether acetate (v:v = 2:1) to yield the title compound 12 (4.95 g, 75%) as a white solid, m.p.: 169–171 °C. 1H-NMR (600 MHz, CDCl3) δ (ppm): 7.75 (4H, m, Fmoc-ArH), 7.61 (4H, m, Fmoc-ArH), 7.42–7.37 (9H, m, Fmoc-ArH, Ar-H), 7.31 (4H, m, Fmoc-ArH), 5.15 (2H, s, OCH2Ar), 4.43 (4H, s, 2 × Fmoc-CH2), 4.26 (2H, m, 2 × Fmoc-CH), 4.18 (1H, m, piperidine-H), 3.28 (1H, brs, piperidine-H), 3.00 (1H, m, piperidine-H), 2.85 (1H, m, piperidine-H), 2.15 (1H, brs, piperidine-H), 1.53 (2H, m, piperidine-H). MS-ESI (m/z): 664 (M+H)+, 686 (M+Na)+, 702 (M+K)+.
3-Amino-4-benzyloxyiminopiperidine dihydrocholoride (13m). To a solution of 12 (2 g, 3 mmol) in tetrahydrofuran (50 mL) was added 10% sodium hydroxide (6 mL, 15 mmol) and stirred at 60 °C for 20 h. The mixture was concentrated under reduced pressure. The residue was treated with dichloromethane (100 mL) and washed with water. The organic layer was dried over anhydrous sodium sulfate and filtered. To the filtrate was pumped dried hydrochloride gas at room temperature for 30 minutes and concentrated under reduced pressure to afford the title compound 13m (0.3 g, 45%) as a white solid, m.p.: 153–155 °C. 1H-NMR (600 MHz, DMSO-d6) δ (ppm): 7.39 (4H, m), 7.33 (1H, m, Ar-H), 5.12 (2H, s, OCH2Ar), 3.95 (1H, m, piperidine-H), 3.64 (1H, m, piperidine-H), 3.07 (1H, m, piperidine-H), 2.97 (1H, m, piperidine-H), 2.88 (1H, m, piperidine-H), 2.78 (1H, m, piperidine-H), 2.38 (1H, m, piperidine-H). MS-ESI (m/z): 220 (M+H)+, 242 (M+Na)+. M.p.: 153–155 °C.
3.2.2. General Procedure for the Synthesis of 3-Amino-4-(substituted benzyloxyimino)piperidine Dihydrocholorides 13a–l
To a solution of 5 (70 mmol) in ethanol (100 mL) was added 4a–l (140 mmol) and stirred at room temperature for 10 h and concentrated under reduced pressure. The residue was dissolved in ethyl acetate (200 mL) and washed with brine (200 mL). The organic layer was dried over anhydrous sodium sulfate and concentrated under reduced pressure to afford compounds 14a–l as yellow oils.
To a solution of 14a–l (50 mmol) and potassium carbonate (100 mmol) in dimethyl sulfoxide (150 mL) was added hydrogen peroxide (400 mmol) at 15 °C for 1 h, and stirred at room temperature for 3 h. The mixture was diluted with water (300 mL) and extracted by ethyl acetate (400 mL). The combined extracts were washed with brine (400 mL), dried over anhydrous sodium sulfate, and then concentrated under reduced pressure to give compounds 15a–l as colorless or light yellow oils.
To a stirred solution of 15a–l (50 mmol) in acetonitrile (350 mL) was added dropwise 10% sodium hypobromite solution (90 mmol) at 5 °C for 1 h. The reaction mixture was stirred at room temperature for 10 h, adjusted to pH 6.5–7 by 20% acetic acid and then concentrated under reduced pressure. The residue was dissolved in water (200 mL), adjusted to pH 4 with 10% hydrochloride and washed by ethyl acetate. The water layer was adjusted to pH 9 with 10% sodium hydroxide and extracted by ethyl acetate. The combined extracts were dried over anhydrous sodium sulfate, and concentrated under reduced pressure to afford compounds 16a–l as yellow oils.
To a solution of 16a–l (7.29 g, 30 mmol) in dichloromethane was pumped dried hydrochloride gas at room temperature for 30 minutes. The mixture was stirred for another 1 h at room temperature, and concentrated under reduced pressure. The residue was treated with ethyl acetate. The precipitate was collected by suction, and dried under vacuum to afford compounds 13a–l (19%–26%, from 5) as white or yellow solids.
3-Amino-4-(4′-bromo-2′-methoxybenzyloxyimino)piperidine dihydrocholoride (13a). The title compound was obtained from 16a as an off-white solid easily absorbing moisture (50%).
3-Amino-4-(3′,4′-ethylenedioxylbenzyloxyimino)piperidine dihydrocholoride (13b). The title compound was obtained from 16b as a white solid (51%), m.p.: 190–192 °C. 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 6.87 (3H, m, Ar-H), 5.02 (2H, s, OCH2Ar), 4.41 (1H, m, piperidine-H), 4.23 (4H, s, 2 × OCH2), 3.71 (1H, m, piperidine-H), 3.53 (1H, m, piperidine-H), 3.37 (1H, m, piperidine-H), 3.23–3.11 (2H, m, piperidine-H), 2.98 (1H, m, piperidine-H). MS-ESI (m/z): 278 (M+H)+.
3-Amino-4-(2′,5′-dimethoxylbenzyloxyimino)piperidine dihydrocholoride (13c). The title compound was obtained from 16c as a light yellow solid (53%), m.p.: 189–192 °C. 1H-NMR (400 MHz, D2O) δ (ppm): 7.07 (1H, m, Ar-H), 7.02 (2H, m, Ar-H), 5.20 (2H, s, OCH2Ar), 4.50 (1H, J1 = 12.4 Hz, J2 = 5.6 Hz, piperidine-H), 4.00 (1H, m, piperidine-H), 3.84 (3H, s, OCH3), 3.82 (3H, s, OCH3), 3.67 (1H, m, piperidine-H), 3.56 (1H, m, piperidine-H), 3.31 (1H, t, J = 12.4 Hz, piperidine-H), 3.18 (1H, m, piperidine-H), 2.44 (1H, m, piperidine-H). MS-ESI (m/z): 280 (M+H)+.
3-Amino-4-(3′,5′-dimethoxylbenzyloxyimino)piperidine dihydrocholoride (13d). The title compound was obtained from 16d as a white solid (50%), m.p.: 212–213 °C. 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 6.55 (2H, m, Ar-H), 6.45 (1H, m, Ar-H), 5.09 (2H, s, OCH2Ar), 4.45 (1H, m, piperidine-H), 3.75 (6H, s, OCH3), 3.72 (1H, m, piperidine-H), 3.38 (1H, m, piperidine-H), 3.26 (1H, m, piperidine-H), 3.17 (1H, m, piperidine-H), 2.98 (1H, m, piperidine-H), 2.58 (1H, m, piperidine-H). MS-ESI (m/z): 280 (M+H)+, 302 (M+Na)+.
3-Amino-4-(2′,3′-dimethoxylbenzyloxyimino)piperidine dihydrocholoride (13e). The title compound was obtained from 16e as a light yellow solid (54%), m.p.: 194–196 °C. 1H-NMR (600 MHz, DMSO-d6) δ (ppm): 7.07 (2H, m, Ar-H), 6.98 (1H, m, Ar-H), 5.15 (2H, s, OCH2Ar), 4.44 (1H, m, piperidine-H), 3.81 (3H, s, OCH3), 3.74 (3H, s, OCH3), 3.70 (1H, m, piperidine-H), 3.36 (1H, m, piperidine-H), 3.23 (1H, m, piperidine-H), 3.16 (1H, m, piperidine-H), 2.98 (1H, m, piperidine-H), 2.58 (1H, m, piperidine-H). MS-ESI (m/z): 280 (M+H)+.
3-Amino-4-(3′,4′-dimethoxylbenzyloxyimino)piperidine dihydrocholoride (13f). The title compound was obtained from 16f as a light yellow solid (52%), m.p.: 183–184 °C. 1H-NMR (600 MHz, DMSO-d6) δ (ppm): 6.95 (3H, m, Ar-H), 5.05 (2H, s, OCH2Ar), 4.45 (1H, m, piperidine-H), 3.75 (6H, s, OCH3), 3.37 (1H, m, piperidine-H), 3.28 (1H, m, piperidine-H), 3.13 (1H, m, piperidine-H), 2.96 (1H, m, piperidine-H), 2.55 (1H, m, piperidine-H), 1.21 (1H, m, piperidine-H). MS-ESI (m/z): 280 (M+H)+.
3-Amino-4-(3′,4′-methylenedioxybenzyloxyimino)piperidine dihydrocholoride (13g). The title compound was obtained from 16e as a white solid (50%), m.p.: 199–201 °C. 1H-NMR (600 MHz, DMSO-d6) δ (ppm): 7.01 (1H, s, Ar-H), 6.89 (2H, m, Ar-H), 6.02 (2H, s, OCH2Ar), 5.04 (2H, s, 2 × OCH2), 4.36 (1H, m, piperidine-H), 3.68 (1H, brs, piperidine-H), 3.35 (1H, m, piperidine-H), 3.22 (1H, m, piperidine-H), 3.12 (1H, m, piperidine-H), 2.98 (1H, m, piperidine-H), 2.48 (1H, m, piperidine-H). MS-ESI (m/z): 264 (M+H)+, 527 (2M+H)+.
3-Amino-4-(2′-methoxylbenzyloxyimino)piperidine dihydrocholoride (13h). The title compound was obtained from 16e as a light yellow solid (55%), m.p.: 199–200 °C. 1H-NMR (400 MHz, D2O) δ (ppm): 7.44 (2H, m, Ar-H), 7.13 (1H, d, J = 8.4 Hz, Ar-H), 7.07 (1H, t, J = 7.6 Hz, Ar-H), 5.24 (2H, s, OCH2Ar), 4.44 (1H, q, J1 = 12 Hz, J2 = 5.2 Hz, piperidine-H), 3.96 (1H, q, J1 = 12 Hz, J2 = 5.2 Hz, piperidine-H), 3.88 (3H, s, OCH3), 3.63 (1H, q, J1 = 12.8 Hz, J2 = 5.6 Hz, piperidine-H), 3.53 (1H, m, piperidine-H), 3.25 (1H, t, J = 12 Hz, piperidine-H), 3.13 (1H, m, piperidine-H), 2.41 (1H, m, piperidine-H). MS-ESI (m/z): 250 (M+H)+.
3-Amino-4-(3′-methoxylbenzyloxyimino)piperidine dihydrocholoride (13i). The title compound was obtained from 16e as a light yellow solid (51%), m.p.: 215–217 °C. 1H-NMR (400 MHz, D2O) δ (ppm): 7.43 (1H, m, Ar-H), 7.04 (3H, m, Ar-H), 5.15 (2H, s, OCH2Ar), 4.38 (1H, m, piperidine-H), 3.91 (1H, m, piperidine-H), 3.85 (3H, s, OCH3), 3.62–3.52 (2H, m, piperidine-H), 3.24–3.17 (1H, m, piperidine-H), 3.14–3.07 (1H, m, piperidine-H), 2.42–2.34 (1H, m, piperidine-H).MS-ESI (m/z): 250 (M+H)+.
3-Amino-4-(4′-methoxylbenzyloxyimino)piperidine dihydrocholoride (13j). The title compound was obtained from 16e as an off-white solid (53%), m.p.: 221–222 °C. 1H-NMR (600 MHz, DMSO-d6) δ (ppm): 7.35 (2H, d, J = 9 Hz, Ar-H), 6.93 (2H, J = 9 Hz, Ar-H), 5.07 (2H, s, OCH2Ar), 4.41 (1H, m, piperidine-H), 3.76 (3H, s, OCH3), 3.71 (1H, m, piperidine-H), 3.38 (1H, m, piperidine-H), 3.26 (1H, m, piperidine-H), 3.20 (1H, m, piperidine-H), 3.12 (1H, m, piperidine-H), 2.95 (1H, m, piperidine-H). MS-ESI (m/z): 250 (M+H)+.
3-Amino-4-(4′-flourobenzyloxyimino)piperidine dihydrocholoride (13k). The title compound was obtained from 16e as a yellow solid as yellow solid easily absorbing moisture (50%).
3-Amino-4-(4′-chlorobenzyloxyimino)piperidine dihydrocholoride (13l). The title compound was obtained from 16e as a white solid (56%), m.p.: 200–202 °C. 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 7.44 (4H, m, Ar-H), 5.14 (2H, s, OCH2Ar), 4.43 (1H, m, piperidine-H), 3.72 (1H, m, piperidine-H), 3.38 (1H, m, piperidine-H), 3.24 (1H, m, piperidine-H), 3.16 (1H, m, piperidine-H), 2.98 (1H, m, piperidine-H), 2.57 (1H, m, piperidine-H). MS-ESI (m/z): 254 (M+H)+.
3.2.3. General Procedure for the Synthesis of 7-[3-Amino-4-(substituted benzyloxyimino)piperidin-1-yl]-1-cyclopropyl-6-fluoro-8-methoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic Acids 19a–m
A mixture of boric acid (9.27 g, 150 mmol) and acetic anhydride (54.06 g, 530 mmol) was stirred at 110 °C for 1.5 h, added acetic acid (50 mL) and then stirred for 1h at the same temperature. To the reaction mixture temperature was added 17 (32.3 g, 100 mmol) at 95 °C and stirred at the same temperature for 2 h. After cooling to room temperature, the mixture was poured into ice water (500 mL) slowly and stirred for 0.5 h. The resulting solid was collected by suction and washed successively with water (50 mL), chilled ethanol (50 mL) and ethyl ether (50 mL), and dried under vacuum to afford compound 18 (29.4 g, 69%) as a white solid, m.p.: 195–196 °C.
To a solution of 13a–m (1 mmol) and triethylamine (3 mmol) in acetonitrile (10 mL) was added 18 (0.8 mmol) at room temperature. The reaction mixture was stirred overnight at 50 °C, and concentrated under reduced pressure. The residue was dissolved in a solution of 5% sodium hydroxide solution (8 mL) and stirred for 1 h at 50 °C. After cooling to room temperature, the mixture was adjusted to pH 7.0–7.5 with 5% acetic acid, and extracted with dichloromethane. The combined extracts were concentrated under reduced pressure. The residue was dissolved in 20% acetic acid (10 mL), stirred for 0.5 h at 50 °C, and filtered. The filtrate was adjusted to pH 6.5–7.5 with 15% sodium hydroxide and extracted by dichloromethane. The combined extracts were dried over anhydrous sodium sulfate and concentrated under reduced pressure. The crude product was purified by column chromatography (silica gel), eluting with dichloromethane and methanol (v:v = 10:1) to afford the target compounds 19a–m as off-white or yellow solids.
7-[3-Amino-4-(4′-bromo-2′-methoxybenzyloxyimino)piperidin-1-yl]-1-cyclopropyl-6-fluoro-8-methoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic acid (19a). Prepared from 13a as an off-white solid (19%), m.p.: >230 °C. 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 8.74 (1H, s, C2-H), 7.65 (1H, d, J = 12 Hz, C5-H), 7.42 (1H, d, J = 8 Hz, Ar-H), 7.22 (1H, d, J = 2 Hz, Ar-H), 6.97 (1H, q, J1 = 8 Hz, J2 = 6 Hz, Ar-H), 5.10 (2H, s, O-CH2Ar), 4.86 (1H, m, cyclopropyl CH), 4.45 (1H, m, piperidine-H), 4.15 (1H, m, piperidine-H), 3.68 (6H, s, O-CH3), 2.96 (1H, m, piperidine-H), 2.85 (1H, m, piperidine-H), 2.77 (1H, m, piperidine-H), 2.68 (1H, m, piperidine-H), 2.40 (1H, m, piperidine-H), 0.93 (4H, m, 2 × cyclopropyl CH2). MS-ESI (m/z): 603 (M+H)+. HRMS-ESI (m/z): Calcd. for C27H29O6N4FBr (M+H)+: 603.1254; Found 603.1276.
7-[3-Amino-4-(3′,4′-ethylenedioxybenzyloxyimino)piperidin-1-yl]-1-cyclopropyl-6-fluoro-8-methoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic acid (19b). Prepared from 13b as an off-white solid (33%), m.p.: 78–81 °C. 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 8.70 (1H, s, C2-H), 7.77 (1H, d, J = 12 Hz, C5-H), 6.87 (1H, s, Ar-H), 6.83 (2H, m, Ar-H), 4.96 (2H, s, O-CH2Ar), 4.80 (1H, m, cyclopropyl CH), 4.42 (1H, m, piperidine-H), 4.23 (4H, s, 2 × OCH2), 3.74 (3H, s, O-CH3), 3.40 (1H, m, piperidine-H), 3.19 (1H, m, piperidine-H), 3.03 (1H, m, piperidine-H), 2.89 (1H, m, piperidine-H), 2.72 (1H, m, piperidine-H), 2.33 (1H, m, piperidine-H), 1.05 (4H, m, 2 × cyclopropyl CH2). MS-ESI (m/z): 553 (M+H)+, 555 (M+Na)+. HRMS-ESI (m/z): Calcd. for C28H30O7N4F (M+H)+: 553.2093; Found 553.2109. M.p.: 78–81 °C.
7-[3-Amino-4-(2′,5′-dimethoxybenzyloxyimino)piperidin-1-yl]-1-cyclopropyl-6-fluoro-8-methoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic acid (19c). Prepared from 13c as an off-white solid (21%), m.p.: 80–81 °C. 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 8.62 (1H, s, C2-H), 7.78 (1H, d, J = 12 Hz, C5-H), 6.94 (1H, d, J = 8 Hz, Ar-H), 6.87 (1H, q, J1 = 8H, J2 = 3 Hz, Ar-H), 6.84 (1H, d, J = 3 Hz, Ar-H), 5.08 (2H, s, O-CH2Ar), 4.93 (1H, m, cyclopropyl CH), 4.42 (1H, m, piperidine-H), 4.17 (1H, m, piperidine-H), 3.69 (9H, s, O-CH3), 3.17 (1H, m, piperidine-H), 3.11 (1H, m, piperidine-H), 2.91–2.86 (1H, m, piperidine-H), 2.75 (1H, m, piperidine-H), 2.39 (1H, m, piperidine-H), 0.98 (4H, m, 2 × cyclopropyl CH2). MS-ESI (m/z): 555 (M+H)+. HRMS-ESI (m/z): Calcd. for C28H32O7N4F (M+H)+: 555.2249; Found 555.2252. M.p.: 80–81 °C.
7-[3-Amino-4-(3′,5′-dimethoxybenzyloxyimino)piperidin-1-yl]-1-cyclopropyl-6-fluoro-8-methoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic acid (19d). Prepared from 13d as an off-white solid (23%), m.p.: 68–70 °C. 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 8.70 (1H, s, C2-H), 7.78 (1H, d, J = 12 Hz, C5-H), 6.52 (1H, s, Ar-H), 6.42 (1H, s, Ar-H), 6.25 (1H, s, Ar-H), 5.03 (2H, s, O-CH2Ar), 4.87 (1H, m, cyclopropyl CH), 4.41 (1H, m, piperidine-H), 4.16 (1H, m, piperidine-H), 3.67 (9H, s, O-CH3), 3.43 (1H, m, piperidine-H), 3.18 (1H, m, piperidine-H), 2.88 (1H, m, piperidine-H), 2.75 (1H, m, piperidine-H), 2.41 (1H, m, piperidine-H), 1.03 (4H, m, 2 × cyclopropyl CH2). MS-ESI (m/z): 555 (M+H)+. HRMS-ESI (m/z): Calcd. for C28H32O7N4F (M+H)+: 555.2249; Found 555.2264. M.p.: 68–70 °C.
7-[3-Amino-4-(2′,3′-dimethoxybenzyloxyimino)piperidin-1-yl]-1-cyclopropyl-6-fluoro-8-methoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic acid (19e). Prepared from 13e as an off-white solid (18%), m.p.: 73–74 °C. 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 8.70 (1H, s, C2-H), 7.78 (1H, d, J = 12 Hz, C5-H), 6.83 (1H,d, J = 8 Hz, Ar-H), 6.74 (1H, t, J = 8 Hz, Ar-H ), 6.45 (1H, d, J = 8 Hz, Ar-H), 5.11 (2H, s, O-CH2Ar), 4.41 (1H, m, cyclopropyl CH), 4.17–4.13 (2H, m, piperidine-H), 3.80 (3H, s, O-CH3), 3.74 (6H, s, O-CH3), 3.09–3.61 (1H, m, piperidine-H), 2.92–2.81 (1H, m, piperidine-H), 2.75–2.62 (2H, m, piperidine-H), 2.38–2.32 (1H, m, piperidine-H), 1.04–0.96 (4H, m, 2 × cyclopropyl CH2). MS-ESI (m/z): 555 (M+H)+. HRMS-ESI (m/z): Calcd. for C28H32O7N4F (M+H)+: 555.2249; Found 555.2254. M.p.: 73–74 °C.
7-[3-Amino-4-(3′,4′-dimethoxybenzyloxyimino)piperidin-1-yl]-1-cyclopropyl-6-fluoro-8-methoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic acid (19f). Prepared from 13f as an off-white solid (20%), m.p.: 88–91 °C. 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 8.69 (1H, s, C2-H), 7.83 (1H, d, J = 11 Hz, C5-H), 6.79 (1H, d, J = 1.6 Hz, Ar-H), 6.68 (1H, d, J = 8, Ar-H), 6.7 (1H, q, J1 = 8 Hz, J2 = 1.6 Hz, Ar-H), 5.08 (2H, s, O-CH2Ar), 4.87 (1H, m, cyclopropyl CH), 4.58 (1H, m, piperidine-H), 4.20–4.16 (1H, m, piperidine-H), 3.77 (9H, O-CH3s), 3.53–3.49 (1H, m, piperidine-H), 3.18–3.15 (1H, m, piperidine-H), 3.02–2.96 (1H, m, piperidine-H), 2.82–2.80 (1H, m, piperidine-H), 2.42–2.35 (1H, m, piperidine-H), 1.12–1.04 (4H, m, 2 × cyclopropyl CH2). MS-ESI (m/z): 555 (M+H)+. HRMS-ESI (m/z): Calcd. for C28H32O7N4F (M+H)+: 555.2249; Found 555.2265.
7-[3-Amino-4-(3′,4′-methylenedioxybenzyloxyimino)piperidin-1-yl]-1-cyclopropyl-6-fluoro-8-methoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic acid (19g). Prepared from 13g as an off-white solid (25%), m.p.: 79–80 °C. 1H-NMR (600 MHz, DMSO-d6) δ (ppm): 8.68 (1H, s, C2-H), 7.78 (1H, d, J = 11 Hz, C5-H), 6.96 (1H, s, Ar-H), 6.90 (1H, d, J = 7.8 Hz, Ar-H), 6.87 (1H, d, J = 7.8 Hz, Ar-H), 6.01 (2H, s, OCH2O), 4.99 (2H, s, O-CH2Ar), 4.83 (1H, m, cyclopropyl CH), 4.17 (1H, m, piperidine-H), 3.74 (3H, s, O-CH3), 3.04–3.02 (1H, m, piperidine-H), 2.91–2.88 (1H, m, piperidine-H), 2.82–2.88 (1H, m, piperidine-H), 2.73–2.70 (1H, m, piperidine-H), 2.63–2.60 (1H, m, piperidine-H), 2.38–2.33 (1H, m, piperidine-H), 0.92 (4H, m, 2 × cyclopropyl CH2). 13C-NMR (150 MHz, DMSO-d6) δ (ppm): 176.22, 165.60, 157.19 (d, J = 268 Hz), 150.57, 150.05, 147.19, 146.92, 146.79, 139.05, 134.09, 133.03, 131.73, 131.55, 121.80, 121.16, 108.71, 106.78 (d, J = 24 Hz), 74.75, 63.10, 58.17, 51.31, 49.60, 40.73, 24.39, 9.02. MS-ESI (m/z): 539 (M+H)+, 561 (M+Na)+. HRMS-ESI (m/z): Calcd. for C27H28O7N4F (M+H)+: 539.1941; Found 539.1946.
7-[3-Amino-4-(2′-methoxybenzyloxyimino)piperidin-1-yl]-1-cyclopropyl-6-fluoro-8-methoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic acid (19h). Prepared from 13h as an off-white solid (19%), m.p.: 208–211 °C. 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 8.71 (1H, s, C2-H), 7.88 (1H, d, J = 12 Hz, C5-H), 7.34–7.29 (2H, m, Ar-H), 7.03–6.89 (2H, m, Ar-H), 5.12 (2H, s, O-CH2Ar), 4.16 (1H, brs, cyclopropyl CH), 3.80–3.68 (5H, m, piperidine-H), 3.50–3.41 (1H, brs, piperidine-H), 3.32 (6H, s, O-CH3), 2.05–1.90 (1H, m, piperidine-H), 1.12–0.84 (4H, m, 2 × cyclopropyl CH2). MS-ESI (m/z): 525 (M+H)+, 547 (M+Na)+. HRMS-ESI (m/z): Calcd. for C27H30O6N4F (M+H)+: 525.2143; Found 525.2147.
7-[3-Amino-4-(3′-methoxybenzyloxyimino)piperidin-1-yl]-1-cyclopropyl-6-fluoro-8-methoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic acid (19i). Prepared from 13i as an off-white solid (23%), m.p.: 94–95 °C. 1H-NMR (600 MHz, DMSO-d6) δ (ppm): 8.65 (1H, s, C2-H), 7.75 (1H, d, J = 11 Hz, C5-H), 7.14 (1H, s, Ar-H), 6.86–6.83 (3H, m, Ar-H), 5.03 (2H, s, O-CH2Ar), 4.43–4.41 (1H, brs, cyclopropyl CH), 4.17–4.14 (1H, brs, piperidine-H), 3.75 (6H, s, O-CH3), 3.45–3.43 (1H, m, piperidine-H), 3.23–3.18 (1H, m, piperidine-H), 3.06–3.04 (1H, m, piperidine-H), 2.87–2.84 (1H, m, piperidine-H), 2.70–2.66 (1H, m, piperidine-H), 2.34–2.29 (1H, m, piperidine-H), 1.17–1.12 (4H, m, 2 × cyclopropyl CH2). 13C-NMR (150 MHz, DMSO-d6) δ (ppm): 176.23, 165.83, 165.60, 158.29 (d, J = 235 Hz), 156.98, 154.63, 150.63, 150.12, 129.88, 129.76, 129.24, 113.68, 113.29, 106.92, 106.76, 106.63, 106.25 (d, J = 25 Hz), 74.36, 63.18, 55.06, 54.87, 49.57, 45.27, 40.73, 24.50, 8.59. MS-ESI (m/z): 525 (M+H)+, 547 (M+Na)+. HRMS-ESI (m/z): Calcd. for C27H30O6N4F (M+H)+: 525.2143; Found 525.2149.
7-[3-Amino-4-(4′-methoxybenzyloxyimino)piperidin-1-yl]-1-cyclopropyl-6-fluoro-8-methoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic acid (19j). Prepared from 13j as an off-white solid (27%), m.p.: 80–82 °C. 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 8.66 (1H, s, C2-H), 7.76–7.68 (1H, m, C5-H), 7.31 (2H, d, J = 8 Hz, Ar-H), 6.91 (2H, d, J = 8 Hz, Ar-H), 4.99 (2H, s, O-CH2Ar), 4.14 (1H, brs, cyclopropyl CH), 3.74 (6H, s, O-CH3), 3.65 (1H, m, piperidine-H), 3.37 (1H, brs, piperidine-H), 3.27 (1H, brs, piperidine-H), 3.16 (1H, brs, piperidine-H), 2.99 (1H, brs, piperidine-H), 2.30 (1H, m, piperidine-H), 1.96 (1H, m, piperidine-H), 1.09–1.02 (4H, m, 2 × cyclopropyl CH2). 13C-NMR (150 MHz, DMSO-d6) δ (ppm): 176.32, 165.71, 158.91, 153.05 (d, J = 250 Hz), 150.58, 134.09, 129.82, 129.65, 129.23, 122.12, 113.65, 107.00, 106.48 (d, J = 22 Hz), 74.63, 63.08, 55.04, 51.44, 49.64, 45.40, 25.09, 8.90. MS-ESI (m/z): 525 (M+H)+. HRMS-ESI (m/z): Calcd. for C27H30O6N4F (M+H)+: 525.2143; Found 525.2146.
7-[3-Amino-4-(4′-flourobenzyloxyimino)piperidin-1-yl]-1-cyclopropyl-6-fluoro-8-methoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic acid (19k). Prepared from 13k as an off-white solid (30%), m.p.: 74–77 °C. 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 8.63 (1H, s, C2-H), 7.77 (1H, d, J =12 Hz, C5-H), 7.45 (2H, m, Ar-H), 7.11 (2H, m, Ar-H), 5.09 (2H, s, O-CH2Ar), 4.91(1H, m, cyclopropyl CH), 4.11 (1H, m, piperidine-H), 3.73 (3H, s, O-CH3), 3.47 (1H, m, piperidine-H), 3.25 (1H, m, piperidine-H), 3.12 (1H, m, piperidine-H), 2.91 (1H, m, piperidine-H), 2.72 (1H, m, piperidine-H), 2.35 (1H, m, piperidine-H), 0.96 (4H, m, 2 × cyclopropyl CH2). 13C-NMR (150 MHz, DMSO-d6) δ (ppm): 176.32, 165.56, 163.78 (d, J = 243 Hz), 155.42 (d, J = 247 Hz), 150.68, 149.61, 145.21, 133.75, 133.04, 130.24, 129.62, 129.54, 115.22, 114.62, 106.31 (d, J = 24 Hz), 74.43, 63.25, 55.98, 50.23, 49.52, 40.94, 24.71, 8.67. MS-ESI (m/z): 513 (M+H)+, 535 (M+Na)+. HRMS-ESI (m/z): Calcd. for C26H27O5N4F2 (M+H)+: 513.1949; Found 513.1952.
7-[3-Amino-4-(4′-chlorobenzyloxyimino)piperidin-1-yl]-1-cyclopropyl-6-fluoro-8-methoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic acid (19l). Prepared from 13l as an off-white solid (34%), m.p.: 67–69 °C. 1H-NMR (400 MHz, DMSO-d6) δ (ppm): 8.67 (1H, s, C2-H), 7.79 (1H, d, J = 12 Hz, C5-H), 7.14 (2H, m, Ar-H), 7.07 (2H, m, Ar-H), 5.11 (2H, s, O-CH2Ar), 4.94 (1H, m, cyclopropyl CH), 4.40 (1H, m, piperidine-H), 4.01 (1H, m, piperidine-H), 3.75 (3H, s, O-CH3), 3.18–3.14 (1H, m, piperidine-H), 3.07–3.05 (1H, m, piperidine-H), 2.92–2.90 (1H, m, piperidine-H), 2.79–2.76 (1H, m, piperidine-H), 2.43–2.38 (1H, m, piperidine-H), 1.07–1.04 (4H, m, 2 × cyclopropyl CH2). 13C-NMR (150 MHz, DMSO-d6) δ (ppm): 176.18, 165.59, 157.75, 155.47 (d, J = 248 Hz), 150.59, 146.11, 137.12, 134.08, 132.25, 129.79, 129.00, 121.19, 117.10, 106.67 (d, J = 23 Hz), 106.61, 73.98, 63.13, 57.79, 51.14, 45.26, 40.72, 24.91, 8.86. MS-ESI (m/z): 529 (M+H)+. HRMS-ESI (m/z): Calcd. for C26H27O5N4FCl (M+H)+: 529.1648; Found 529.1637.
7-(3-Amino-4-benzyloxyiminopiperidin-1-yl)-1-cyclopropyl-6-fluoro-8-methoxy-4-oxo-1,4-dihydro-quinoline-3-carboxylic acid (19m). Prepared from 13m as an off-white solid (32%), m.p.: 84–85 °C. 1H-NMR (600 MHz, DMSO-d6) δ (ppm): 8.68 (1H, s, C2-H), 8.74 (1H, d, J = 12 Hz, C5-H), 7.29–7.21 (5H, m, Ar-H), 5.10 (2H, s, O-CH2Ar), 4.97–4.92 (1H, m, cyclopropyl CH), 4.43–4.39 (1H, m, piperidine-H), 4.16–4.13 (1H, m, piperidine-H), 3.72 (3H, s, O-CH3), 3.65–3.63 (1H, m, piperidine-H), 3.2–3.15 (1H, m, piperidine-H), 2.86–2.83 (1H, m, piperidine-H), 2.71–2.67 (1H, m, piperidine-H), 2.37–2.34 (1H, m, piperidine-H), 1.08–0.95 (4H, m, 2 × cyclopropyl CH2). 13C-NMR (150 MHz, DMSO-d6) δ (ppm): 176.26, 165.73, 155.65 (d, J = 255 Hz), 150.60, 150.19, 146.05, 137.89, 133.09, 128.14, 127.98, 127.68, 127.73, 127.29, 106.86 (d, J = 21 Hz), 106.35, 74.62, 61.29, 53.11, 51.35, 45.39, 40.75, 24.78, 8.98. MS-ESI (m/z): 495 (M+H)+, 517 (M+Na)+. HRMS-ESI (m/z): Calcd. for C26H28O5N4F (M+H)+: 495.2038; Found 495.2048. M.p.: 84–85 °C.
3.3. MIC Determination
Compounds 19a–m were evaluated for their in vitro antibacterial activity using standard techniques in comparison to the reference drugs IMB-070593, CPFX and LVFX. Drugs (10.0 mg) were dissolved in 0.1 N NaOH solution and water (10 mL). Further progressive two fold serial dilution with melted Mueller-Hinton agar was performed to obtain the required concentrations of 128, 64, 32, 16, 8, 4, 2, 1, 0.5, 0.25, 0.125, 0.06, 0.03, 0.015 and 0.008 mg/mL. Petri dishes were incubated with 104 colony-forming units (cfu) and incubated at 35 °C for 18 h. MIC was the lowest concentration of the test compound, which resulted in no visible growth on the plate.
4. Conclusions
In summary, a series of novel IMB-070593 derivatives with remarkable improvement in lipophilicity, as compared to the parent IMB-070593, were designed, synthesized and evaluated for their in vitro anti-MTB and antibacterial activity. Our results reveal that compound 19l has good in vitro activity against MTB H37Rv ATCC 27294 (MIC: 0.125 µg/mL) which is 2–4 fold more potent than the parent IMB070593, CPFX and LVFX (MICs: 0.25–0.5 µg/mL). Whereas, compounds 19h, 19j, 19k and 19m show interesting antibacterial activity (MICs: <0.008–4 µg/mL) against all of the tested Gram-positive strains including CPFX- and/or LVFX-resistant MSSA, MRSA and MSSE. However, the target compounds 19a–m are generally less active than IMB-070593 against MDR-MTB 20161 and Gram-negative strains. It suggests that merely an increase in lipophilicity of the tested compounds does not result in enhanced antimycobacterial and antibacterial activities.
Acknowledgments
This work was supported by the National S&T Major Special Project on Major New Drug Innovation (Number 2012ZX09301002-001-017).
References and Notes
- Lv, K.; Sun, Y.X.; Sun, L.Y.; Guo, H.Y.; Wu, J.W.; Liu, M.L. Design, synthesis and in vitro antibacterial activity of fluoroquinolone derivatives containing a chiral 3-(alkoxyimino)-2-(aminomethyl)azetidine moiety. ChemMedChem 2012, 7, 1230–1236. [Google Scholar] [CrossRef]
- Aubry, A.; Pan, X.S.; Aubry, L.M.; Jarlier, V.; Emmanuelle, C. Mycobacterium tuberculosis DNA Gyrase: Interaction with quinolones and correlation with antimycobacterial drug activity. Antimicrob. Agents Chemother. 2004, 48, 1281–1288. [Google Scholar] [CrossRef]
- Crofton, J.; Choculet, P.; Maher, D. Guidelines for the Management of Drug-Resistant Tuberculosis. WHO/TB/96–210 (Rev.1); World Health Organization: Geneva, Switzerland, 1997. [Google Scholar]
- Bradbury, B.J.; Pucci, M.J. Recent advances in bacterial topoisomerase inhibitors. Curr. Opin. Pharmacol. 2008, 8, 574–581. [Google Scholar] [CrossRef]
- Ginsburg, A.S.; Grosset, J.H.; Bishai, W.R. Fluoroquinolones, tuberculosis, and resistance. Lancet Infect. Dis. 2003, 3, 432–442. [Google Scholar] [CrossRef]
- Dalhoff, A. Resistance surveillance studies: A multifaceted problem—The fluoroquinolone example. Infection 2012, 40, 239–262. [Google Scholar] [CrossRef]
- Grimaldo, E.R.; Tupasi, T.E.; Rivera, A.B.; Quelapio, M.I. D.; Cardano, R.C.; Derilo, J.O.; BelenIncreased, V.A. Increased resistance to ciprofloxacin and ofloxacin in multidrug-resistant Mycobacterium tuberculosis isolates from patients seen at a tertiary hospital in the Philippines. Int. J. Tuberc. Lung. Dis. 2001, 5, 546–550. [Google Scholar]
- Lv, K.; Wu, J.W.; Wang, J.; Liu, M.L.; Wei, Z.Q.; Cao, J.; Sun, Y.X.; Guo, H.Y. Synthesis and in vitro antibacterial activity of quinolone/naphthyridone derivatives containing 3-alkoxyimino-4-(methyl)aminopiperidine scaffolds. Bioorg. Med. Chem. Lett. 2013, 23, 1754–1759. [Google Scholar] [CrossRef]
- Zhang, Y.B.; Liu, M.L.; Guo, H.Y. Structure characteristics of quinolones and structure-activity relationships against Gram-positive bacteria. World Notes Antibiot. 2008, 29, 12–19. [Google Scholar]
- Liu, M.L.; Guo, H.Y. Non-fluoroquinolones: discovery and structure-activity relationship studies. World Notes Antibiot. 2006, 27, 16–21. [Google Scholar]
- Chai, Y.; Liu, M.L.; Lv, K.; Feng, L.S.; Li, S.J.; Sun, L.Y.; Wang, S.; Guo, H.Y. Synthesis and in vitro antibacterial activity of a series of novel gatifloxacin derivatives. Eur. J. Med. Chem. 2011, 46, 4267–4273. [Google Scholar] [CrossRef]
- Feng, L.S.; Liu, M.L.; Wang, S.; Chai, Y.; Li, S.J.; Guo, H.Y. Synthesis and in vitro antimycobacterial activity of moxifloxacin methylene and ethylene isatin derivatives. Chem. Res. Chin. Univ. 2012, 28, 61–66. [Google Scholar]
- Sharma, P.C.; Jain, A.; Jain, S.; Pahwa, R.; Yar, M.S. Ciprofloxacin: Review on developments in synthetic, analytical, and medicinal aspects. J. Enzyme Inhib. Med. Chem. 2010, 25, 557–589. [Google Scholar] [CrossRef]
- Sriram, D.; Yogeeswari, P.; Basha, J.S.; Radha, D.R.; Nagaraja, V.J. Synthesis and antimycobacterial evaluation of various 7-substituted ciprofloxacin derivatives. Bioorg. Med. Chem. 2005, 13, 5774–5778. [Google Scholar] [CrossRef]
- Sriram, D.; Aubry, A.; Yogeeswaria, P.; Fisher, L.M. Gatifloxacin derivatives: Synthesis, antimycobacterial activities, and inhibition of Mycobacterium tuberculosis DNA gyras. Bioorg. Med. Chem. Lett. 2006, 16, 2982–2985. [Google Scholar] [CrossRef]
- Feng, L.-S.; Liu, M.-L.; Zhang, S.; Chai, Y.; Wang, B.; Zhang, Y.-B.; Lv, K.; Guan, Y.; Guo, H.-Y.; Xiao, C.-L. Synthesis and in vitro antimycobacterial activity of 8-OCH3 ciprofloxacin methylene and ethylene isatin derivatives. Eur. J. Med. Chem. 2011, 46, 341–348. [Google Scholar] [CrossRef]
- Guo, Q.; Liu, M.-L.; Feng, L.-S.; Lv, K.; Guan, Y.; Guo, H.-Y.; Xiao, C.-L. Synthesis and in-vitro antimycobacterial activity of fluoroquinolone derivatives containing a coumarin moiety. Arch. Pharm. 2011, 344, 802–809. [Google Scholar] [CrossRef]
- Feng, L.S.; Liu, M.L.; Wang, B.; Chai, Y.; Hao, X.Q.; Meng, S.; Guo, H.Y. Synthesis and in vitro antimycobacterial activity of balofloxacin ethylene isatin derivatives. Eur. J. Med. Chem. 2010, 45, 3407–3412. [Google Scholar] [CrossRef]
- Feng, L.S.; Lv, K.; Liu, M.L.; Wang, S.; Zhao, J.; You, X.F.; Li, S.J.; Cao, J.; Guo, H.Y. Synthesis and in vitro antibacterial activity of gemifloxacin derivatives containing a substituted benzyloxime moiety. Eur. J. Med. Chem. 2012, 55, 125–136. [Google Scholar] [CrossRef]
- Wang, X.Y.; Guo, Q.; Wang, Y.C.; Liu, B.Q.; Liu, M.L.; Sun, L.Y.; Guo, H.Y. Synthesis and antibacterial activity of 7-(4-alkoxyimino-3-aminopiperidin-1-yl)fluoroquinolone derivatives. Acta Pharm. Sin. 2008, 43, 819–827. [Google Scholar]
- Gui, H.Y.; Liu, M.L.; Liu, B.Q.; Hu, J.S.; Wu, J.W.; Wang, Z. Application of 7-(4-alkoxyimino-3-aminopiperidin-1-yl)fluoroquinolones and their combinations. CN Patent 101863876, 20 October 2010. [Google Scholar]
- Brown, P.; Calvert, S.H.; Chapman, P.C.A.; Cosham, S.C.; Eglington, A.J.; Elliot, R.L.; Harris, M.A.; Hinks, J.D.; Lowther, J.; Merrikin, D.J.; et al. β-Lactamase-stable penicillins. Synthesis and structure-activity relationships of (Z)-alkyloxyimino penicillins; selection of BRL 44154. J. Chem. Soc. Perkin Trans. 1 1991, 4, 881–891. [Google Scholar]
- Chai, Y.; Liu, M.L.; Wang, B.; You, X.F.; Feng, L.S.; Zhang, Y.B.; Cao, J.; Guo, H.Y. Synthesis and in vitro antibacterial activity of novel fluoroquinolone derivatives containing substituted piperidines. Bioorg. Med. Chem. Lett. 2010, 20, 5195–5198. [Google Scholar] [CrossRef]
- Chai, Y.; Wan, Z.L.; Wang, B.; Guo, H.Y.; Liu, M.L. Synthesis and in vitro antibacterial activity of 7-(4-alkoxyimino-3-amino-3-methylpiperidin-1-yl)fluoroquinolone derivatives. Eur. J. Med. Chem. 2009, 44, 4063–4069. [Google Scholar] [CrossRef]
- Collins, L.; Franzblau, S.G. Microplate alamar blue assay versus BACTEC 460 system for high-throughput screening of compounds against Mycobacterium tuberculosis and Mycobacterium avium. Antimicrob. Agents Chemother. 1997, 41, 1004–1009. [Google Scholar]
- Lu, Y.; Zheng, M.Q.; Wang, B.; Fu, L.; Zhao, W.J.; Li, P.; Xu, J.; Zhu, H.; Jin, H.X.; Yin, D.L.; et al. Clofazimine analogs with efficacy against experimental tuberculosis and reduced potential for accumulation. Antimicrob. Agents Chemother. 2011, 55, 5185–5193. [Google Scholar] [CrossRef]
- MIC values were determined as described by the NCCLS (see: National Committee for Clinical Laboratory Standards, Performance Standards for Antimicrobial Susceptibility Testing: 11th Informational Supplement, Volume 21; NCCLS: Wayne, PA, USA, 2001; pp. M100–S11.). MIC was defined as the lowest concentration of each compound that inhibits visible growth of bacteria after incubation at 35 °C for 18–24 h.
- Sample Availability: Contact the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
