Abscisic Acid Induced Changes in Production of Primary and Secondary Metabolites, Photosynthetic Capacity, Antioxidant Capability, Antioxidant Enzymes and Lipoxygenase Inhibitory Activity of Orthosiphon stamineus Benth.
Abstract
:1. Introduction
2. Results and Discussion
2.1. Total Phenolics and Flavonoids Profiling
| ABA (µM) | Parts | Total Phenolics (mg g−1 gallic acid dry weight) | Total Flavonoids (mg g−1 rutin dry weight) | Soluble sugar (mg g−1 sucrose dry weight) |
|---|---|---|---|---|
| Leaves | 3.11 ± 0.27 c | 1.47 ± 0.21 c | 79.12 ± 11.21 d | |
| 0 | Stems | 1.32 ± 0.02 l | 0.52 ± 0.02 g | 40.23 ± 8.98 l |
| Roots | 2.71 ± 1.24 e | 1.21 ± 0.34 k | 62.18 ± 12.12 h | |
| Leaves | 3.98 ± 0.34 b | 1.72 ± 0.56 b | 88.21 ± 9.76 c | |
| 2 | Stems | 1.50 ± 0.04 h | 0.76 ± 0.34 f | 47.21 ± 11.21 k |
| Roots | 2.87 ± 0.45 d | 1.18 ± 0.12 j | 68.21 ± 12.12 g | |
| Leaves | 4.10 ± 0.21 ab | 1.98 ± 0.32 ab | 90.17 ± 10.76 bc | |
| 4 | Stems | 1.57 ± 0.05 g | 0.86 ± 0.12 e | 50.11 ± 5.67 j |
| Roots | 2.92 ± 0.03 d | 1.27 ± 0.32 i | 70.82 ± 5.88 f | |
| Leaves | 4.21 ± 0.02 a | 2.12 ± 0.04 a | 98.12 ± 7.98 a | |
| 6 | Stems | 1.92 ± 0.21 f | 0.97 ± 0.08 d | 57.12 ± 12.12 l |
| Roots | 2.97 ± 0.11 de | 1.46 ± 0.12 h | 76.21 ± 10.12 e |
| Parameters | Pearson’s correlation coefficient (R2) | |
|---|---|---|
| Total phenolics | Total flavonoids | |
| 1. Soluble sugar | 0.971 * | 0.973 * |
| 2. H2O2 | 0.877 * | 0.812 * |
| 3. O2 | 0.778 * | 0.912 * |
| 4. PAL activity | 0.923 * | 0.901 * |
| 5. APX | 0.781 * | 0.822 ** |
| 6. SOD | 0.845 * | 0.816 * |
| 7. CAT | 0.912 * | 0.832 * |
| 8. ORAC | 0.904 * | 0.956 * |
| 9. DPPH | 0.781 * | 0.889 * |
| 10. Net Photosynthesis | −0.871 * | −0.921 * |
| 11. Stomata conductance | −0.881 ** | −0.824 * |
| 12. LOX | 0.951 * | 0.923 * |
2.2. Soluble Sugar Profiling
2.3. Antioxidant Content of H2O2 and O2−
| ABA (µM) | Parts | H2O2 (µmol g−1 fresh weight) | O2− (µmol g−1 dry weight min−1) | PAL Activity (nm transcinnamic mg−1 protein−1 h−1) |
|---|---|---|---|---|
| Leaves | 2.19 ± 0.34 d | 1.09 ± 0.02 d | 9.21 ± 0.62 d | |
| 0 | Stems | 0.67 ± 0.21 k | 0.32 ± 0.23 j | 2.08 ± 0.23 l |
| Roots | 1.32 ± 0.23 h | 0.66 ± 0.45 g | 5.02 ± 0.15 h | |
| Leaves | 2.43 ± 0.31 c | 1.17 ± 0.32 c | 10.16 ± 0.82 c | |
| 2 | Stems | 0.78 ± 0.03 k | 0.44 ± 0.12 i | 2.17 ± 0.12 k |
| Roots | 1.52 ± 0.34 g | 0.72 ± 0.02 f | 6.23 ± 0.32 g | |
| Leaves | 2.60 ± 0.06 b | 1.23 ± 0.12 b | 13.11 ± 2.13 b | |
| 4 | Stems | 0.90 ± 0.02 j | 0.56 ± 0.13 h | 3.21 ± 0.19 j |
| Roots | 1.66 ± 0.21 f | 0.87 ± 0.01 e | 7.11 ± 0.34 f | |
| Leaves | 2.81 ± 0.06 a | 1.57 ± 0.21 a | 17.21 ± 1.21 a | |
| 6 | Stems | 0.91 ± 0.12 i | 0.57 ± 0.11 h | 4.11 ± 0.6l i |
| Roots | 1.78 ± 0.02 e | 0.91 ± 0.04 e | 8.81 ± 0.54 e |
2.4. Phenyl Alanine Ammonia Lyase (PAL) Activity
2.5. Antioxidant Enzyme Activities
| ABA (µM) | Parts | Ascorbate peroxidase activity (APX; mg protein−1 min−1) | Superoxide dismutase activity (SOD mg protein−1 min−1) | Catalase activity (CAT; µmol mg protein−1 min−1) |
|---|---|---|---|---|
| Leaves | 15.23 ± 2.34 d | 4.62 ± 0.11 d | 19.21 ± 1.27 d | |
| 0 | Stems | 6.12 ± 0.81 k | 1.34 ± 0.01 l | 6.66 ± 2.11 l |
| Roots | 10.11 ± 0.03 h | 2.98 ± 0.41 h | 12.17 ± 0.97 h | |
| Leaves | 17.11 ± 0.51 c | 4.82 ± 0.21 c | 20.12 ± 0.82 c | |
| 2 | Stems | 6.11 ± 0.53 k | 1.52 ± 0.36 k | 8.27 ± 0.78 k |
| Roots | 11.27 ± 0.14 g | 3.62 ± 0.15 g | 13.24 ± 0.11 g | |
| Leaves | 19.71 ± 0.16 b | 5.01 ± 0.17 b | 23.17 ± 0.78 b | |
| 4 | Stems | 7.23 ± 0.42 j | 1.71 ± 2.11 j | 9.23 ± 1.19 j |
| Roots | 13.22 ± 0.31 f | 3.89 ± 1.02 f | 16.59 ± 0.89 f | |
| Leaves | 21.62 ± 0.26 a | 5.27 ± 0.81 a | 25.12 ± 1.21 a | |
| 6 | Stems | 9.12 ± 0.98 i | 1.76 ± 0.92 i | 10.24 ± 2.17 i |
| Roots | 14.21 ± 1.32 e | 4.02 ± 1.24 e | 17.21 ± 0.98 e |
2.6. 1,1-Diphenyl-2-picryl-hydrazyl (DPPH) and Oxygen Radical Absorbance Capacity (ORAC) Assay
| ABA (µM) | Parts | ORAC (µmol Trolox equivalent g−1) | DPPH (µmol Trolox equivalent g−1) |
|---|---|---|---|
| Leaves | 65.21 ± 2.41 d | 20.19 ± 9.02 d | |
| 0 | Stems | 35.67 ± 0.21 i | 7.72 ± 2.32 j |
| Roots | 51.07 ± 2.23 g | 13.66 ± 6.45 h | |
| Leaves | 69.31 ± 10.31 c | 25.17 ± 8.32 c | |
| 2 | Stems | 37.78 ± 9.03 k | 9.44 ± 7.12 k |
| Roots | 55.02 ± 0.34 l | 15.72 ± 3.02 g | |
| Leaves | 70.60 ± 8.96 b | 27.84 ± 9.12 b | |
| 4 | Stems | 40.90 ± 11.02 h | 10.23 ± 8.13 i |
| Roots | 57.21 ± 10.21 f | 16.21 ± 2.21 f | |
| Leaves | 77.81 ± 0.06 a | 31.57 ± 9.21 a | |
| 6 | Stems | 45.21 ± 0.12l g | 11.01 ± 2.11 h |
| Roots | 60.23 ± 8.02 e | 17.81 ± 2.04 e |
2.7. Leaf Gas Exchange

2.8. Lipoxygenase (LOX) Inhibitory Activity
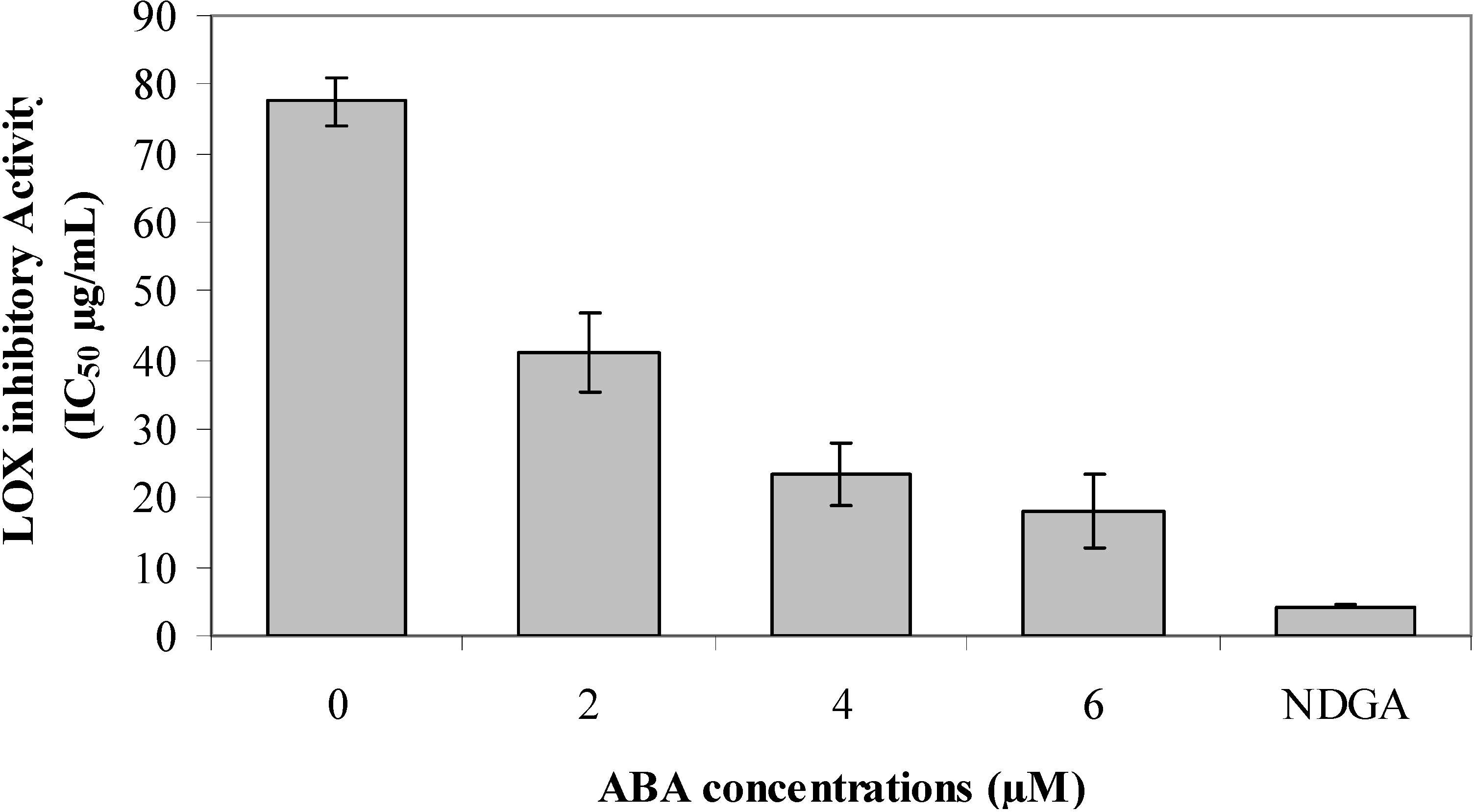
3. Experimental
3.1. Plant Materials and Maintenance
| Microclimate parameters | Quantification |
|---|---|
| Relative Humidity | 56.14–65.32% |
| Light intensity | 225–1450 µmol m−2s−1 |
| Day temperature | 27–31 °C |
| Night temperature | 18–22 °C |
| Ambient CO2 | 372.81 µmol mol−1 |
3.2. Determination of Total Phenolics and Flavonoids
3.3. Determination of Soluble Sugar
3.4. Superoxide Radical (O2−) Assay
3.5. H2O2 Assay
3.6. Oxygen Radical Absorbance Capacity (ORAC) Assay
3.7. DPPH Assay
3.8. Phenylalanine Ammonia-Lyase (PAL) Activity
3.9. Leaf Gas Exchange Measurement
3.10. Antioxidant Enzyme Activity
3.10.1. Preparation of Enzyme Extracts
3.10.2. Ascorbate Peroxidase (APX) Activity Assay
3.10.3. Catalase (CAT) Activity Assay
3.10.4. Superoxide Dismutase (SOD) Activity Assay
3.11. LOX Inhibitory Assay
3.12. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Akuwoah, A.; Zhari, G.; Norhayati, I.; Sadikun, A. The effects of different extraction solvents of varying polarities on polyphenols of Orthosiphon stamineus and evaluation of the free radical-scavenging activity. Food Chem. 2005, 93, 311–317. [Google Scholar]
- Akuwoah, A.; Zhari, G.; Norhayati, I.; Sadikun, A.; Khamsah, S.M. Sinensitin, eupatorin, 30-hydroxy-5,6,7, 40-tetramethoxyflavone and rosmarinic acid contents and antioxidative effect of Orthosiphon stamineus from Malaysia. Food Chem. 2004, 87, 559–566. [Google Scholar] [CrossRef]
- Sumaryono, W.; Proksch, P.; Wray, V.; Witte, L.; Hartmann, T. Qualitative and quantitative analysis of phenolic constituents from Orthosiphon aristatus. Planta Med. 1991, 57, 176–180. [Google Scholar]
- Tezuka, Y.; Stampoulis, P.; Banskota, A.; Awale, H.; Tran, H.; Saiki, K.Q. Constituents of the Vietnamese medicinal plant Orthosiphon stamineus. Chem. Pharma. Bull 2000, 48, 1711–1719. [Google Scholar]
- Mamoudou, H.D. Endogenous phenolics and starch modifying enzymes as determinats of sorghum for food use in Burkina Faso. Ph.D. Thesis, Wageningen University, The Netherlands, 2005. [Google Scholar]
- Djousse, L.; Arnett, D.K.; Coon, H.; Province, M.A.; Moore, L.L.; Ellison, R.C. Fruit and vegetable consumption and LDL cholesterol: The National Heart, Lung, and Blood Institute Family Heart Study. Am. J. Clinical Nut. 2004, 79, 213–217. [Google Scholar]
- Ector, B.J.; Magee, J.B.; Hegwood, C.B.; Coign, M.J. Resveratrol concentration in muscadine berries, juice, pomace, purees, seeds, and wines. Am. J. Enol. Vitic. 1996, 47, 57–62. [Google Scholar]
- Treutter, D. Managing phenol contents in crop plants by phytochemical farming and breeding-visions and constraints. Int. J. Mol. Sci. 2010, 11, 807–857. [Google Scholar] [CrossRef]
- Cantín, C.M.; Fidelibus, M.W.; Crisosto, C.H. Application of abscisic acid (ABA) at veraison advanced red colour development and maintained postharvest quality of ‘Crimson Seedless’ grapes. Postharvest Biol. Tech. 2007, 46, 237–241. [Google Scholar]
- Jiang, M.; Zhang, J. Effect of Abscisic acid on active oxygen species, antioxidative defence system, and oxidative damage in leaves of maize seedlings. Plant Cell Physiol. 2001, 42, 1265–1273. [Google Scholar]
- Cuello, J.; Quiles, M.J.; Rosauro, J.; Sabate, B. Effects of growth regulators and light on chloroplasts NAD(P)H dehydrogenase activities of senescent barley leaves. Plant Growth Regul. 1995, 17, 225–232. [Google Scholar] [CrossRef]
- Li, Z.; Zhao, X.; Sandhu, A.K.; Gu, L. Effects of exogenous abscisic acid on yield, antioxidant capacities, and phytochemical contents of greenhouse grown lettuces. J. Agric. Food Chem. 2010, 58, 6503–6509. [Google Scholar]
- Sandhu, A.K.; Gray, D.J.; Lu, J.; Gu, L. Effects of exogenous abscisic acid on antioxidant capacities, anthocyanins, and flavonol contents of muscadine grape (Vitis rotundifolia) skins. Food Chem. 2010, 126, 982–988. [Google Scholar]
- Pei, Z.M.; Murata, N.; Benning, G.; Thomine, S.; Klusener, B.; Allen, G.J.; Grill, E.; Schroeder, J.I. Calcium channels activated by hydrogen peroxide mediate abscisic acid signaling in guard cells. Nature 2000, 406, 731–734. [Google Scholar] [CrossRef]
- Lin, C.C.; Kao, C.H. Abscisic acid induced changes in cell wall peroxidase activity and hydrogen peroxide level in roots of rice seedlings. Plant Sci. 2001, 160, 323–329. [Google Scholar] [CrossRef]
- Lu, S.; Su, W.; Li, H.; Guo, Z. Abscisic acid improves drought tolerance of triploid bermuda grass and involves H2O2 and NO induced antioxidant enzyme activities. Plant Physiol. Biochem. 2009, 47, 132–138. [Google Scholar] [CrossRef]
- Bueno, P.; Piqueras, A.; Kurepa, J.; Savoure, A.; Verbruggen, N.; Van Montagu, M.; Inze, D. Expression of antioxidant enzymes in response to abscisic and high osmoticum in tobacco BY- cell cultures. Plant Sci. 1998, 138, 27–34. [Google Scholar] [CrossRef]
- Hung, K.T.; Kao, C.H. Hydrogen peroxide is necessary for abscisic acid-induced senescence of rice leaves. J. Plant Physiol. 2004, 161, 1347–1357. [Google Scholar] [CrossRef]
- Jannat, R.; Uraji, M.; Morofuji, M.; Islam, M.M.; Bloom, R.E.; Nakmura, Y.; McClung, C.R.; Schroeder, J.I.; Mori, I.C.; Murata, Y. Roles of intracellular hydrogen peroxide accumulation in abscisic acid signaling in Arabidopsis guard cells. J. Plant Physiol. 2011, 14, 112–123. [Google Scholar]
- Quan, L.J.; Zhang, B.; Shi, W.W.; Li, H.Y. Hydrogen peroxide in plants: A versatile molecule of the reactive oxygen species network. J. Integr. Plant Biol. 2008, 50, 2–18. [Google Scholar] [CrossRef]
- Tsai, Y.C.; Kao, C.H. The involvement of hydrogen peroxide in abscisic acid-induced activities of ascorbate peroxidase and glutathione reductase in rice roots. Plant Growth Regul. 2004, 43, 207–212. [Google Scholar]
- Hu, X.; Zhang, A.; Zheng, J.; Jiang, M. Abscisic acid is a key inducer of hydrogen peroxide production in leaves of maize plants exposed to water stress. Plant Cell Physiol. 2006, 47, 1484–1495. [Google Scholar]
- Cakmak, I.; Marschner, H. Magnesium deficiency and high light intensity enhance activities of superoxide dismutase, ascorbate peroxidase, and glutathione reductase in bean leaves. Plant Physiol. 1992, 98, 1222–1227. [Google Scholar]
- Kataoka, L.; Sugiura, A.; Utsunomiya, N.; Tomana, T. Effects of abscisic acid and defoliation on anthocyanin accumulation in kyoto grapes (Vitis vinefera x lab ruscana BAILEY). Vitis 2011, 21, 325–332. [Google Scholar]
- Wang, J.; Chen, J.; Pan, K. Effects of exogenous abscisic acid on the level of antioxidants in Atractylodes macrocephala Koids under lead stress. Env. Sci. Poll. Res. 1999, 12, 112–119. [Google Scholar]
- Shui, Y.C.; Feng, X.; Yan, W. Advances in the study of flavonoids in Gingko bilobaleaves. J. Med. Plant Res. 2009, 3, 1248–1252. [Google Scholar]
- Guo, R.; Yuan, G.; Wang, Q. Effect of sucrose and mannitol on the accumulation of health-promoting compounds and the activity of metabolic enzymes in broccoli sprouts. Sci. Hortic. 2011, 128, 159–165. [Google Scholar] [CrossRef]
- Bryant, J.P.; Chapin, F.S.; Klein, D.R. Carbon nutrient balance ofboreal plants in relation to vertebrate herbivory. Oikos 1983, 40, 357–368. [Google Scholar] [CrossRef]
- Herms, D.A.; Matson, W.J. The dilemma of plants: To grow or defend. Quar. Rev. Biol. 1992, 67, 283–335. [Google Scholar]
- Panuelas, J.; Estiarte, M. Can elevated CO2 affect secondary metabolism and ecosystem function? Trends Ecol. Evol. 1998, 13, 20–24. [Google Scholar] [CrossRef]
- Yang, D.; Sheng, D.; Duan, Q.; Liang, X.; Liang, Z.; Liu, Y. PEG and ABA trigger the burst of reactive oxygen species to increase Tanshinone production in Salvia miltiorrhiza Hairy roots. J. Plant Growth Regul. 2012, 31, 579–587. [Google Scholar]
- Ding, W.; Song, L.; Wang, X.; Bi, Y. Effects of abscisic acid on heat stress tolerance in the calli from the two ecotypes of Phragmites communis. Biol. Plant 2010, 54, 607–613. [Google Scholar] [CrossRef]
- Mansouri, H.; Asrar, Z.; Szopa, J. Effects of ABA on primary terpenoids and tetrahydrocannabitol in Cannabis sativa L. at flowering stage. Plant Growth Regul. 2009, 58, 269–277. [Google Scholar] [CrossRef]
- Jaafar, H.Z.E. Impact of Environmental Stress on Reproductive Development in Sweet pepper (Capsicum anuum). PhD Thesis, University of Nottingham, Nottingham, UK, 1995. [Google Scholar]
- Travaglia, C.; Balboa, G.; Esposito, G.; Reinoso, H. ABA action on the production and redistribution of filed grown maize carbohydrates in semiarid regions. Plant Growth Regul. 2012, 67, 27–34. [Google Scholar]
- Wang, S.; Hu, L.; Sun, J.; Sui, X.; Wei, Y.; Zhang, Z. Effects of exogenous abscisic acid on leaf carbohydrate metabolism during cucumber seedling dehyration. Plant Growth Regul. 2012, 66, 87–93. [Google Scholar] [CrossRef]
- Szabó, B.; Lakatos, Á.; Koszegi, T.; Botz, L. Investigation of abiogenic stress-induced alterations in the level of secondary metabolites in poppy plants (Papaver somniferum L.). Acta Biol. Hung. 2008, 59, 425–438. [Google Scholar]
- Ibrahim, M.H.; Jaafar, H.Z.E. The influence of carbohydrate, protein and phenylanine ammonia lyase on up-regulation of production of secondary metabolites (total phenolics and flavonoid) in Labisia pumila (Blume) Fern-Vill (Kacip Fatimah) under high CO2 and different nitrogen levels. Molecules 2011, 16, 4172–4190. [Google Scholar] [CrossRef]
- Jones, C.G.; Hartley, S.E. A protein competition model of phenolic allocation. Oikos 1999, 86, 27–44. [Google Scholar] [CrossRef]
- Fatima, Z.; Bano, A. The effects of plant growth regulators and temperate shock on IAA-oxidase activity of roots and endogenous ABA level of leaves. Pak. J. Biol. 1999, 2, 1601–1603. [Google Scholar] [CrossRef]
- Nakano, Y.; Asada, K. Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplast. Plant Cell Physiol. 1981, 22, 867–880. [Google Scholar]
- Guan, L.M.; Zhao, J.; Scandalice, J.G. Cis-element and trans-factors that regulate expression of the maize Cat 1 antioxidant gene in response to ABA and osmotic stress: H2O2 is likely intermediary signaling molecule for the response. Plant J. 2000, 22, 87–95. [Google Scholar] [CrossRef]
- Saleh, A.A.H. Amelioration of chilling injuries in mung bean (Vigna radiata L) seedlings by paclobutrazol, abscisic acid and hydrogen peroxide. Am. J. Plant Physiol. 2007, 2, 318–332. [Google Scholar] [CrossRef]
- Hu, X.; Jiang, M.; Zhang, A.; Lu, J. Abscisic acid induced apoplastic H2O2 accumulation up-regulates the activities of chloroplastic and cytosolic antioxidant enzymes in maize leaves. Planta 2005, 223, 57–68. [Google Scholar] [CrossRef]
- Cui, B.; Liang, Z.; Liu, Y.; Liu, F.; Zhu, J. Effects of ABA and its biosynthetic inhibitor fluridone on accumulation of phenolic acids and activity of PAL and TAT in hairy root of Salvia miltiorrhiza. Zhongguo Zhongyao Zazhi 2012, 37, 754–759. [Google Scholar]
- Duan, Y.Y.; Yue, X.; Chen, G.L. Effect of abscisic acid on wound healing and antioxidant enzyme activities of Cynomorium songaricum stem. Plant Physiol. J. 2012, 48, 298–302. [Google Scholar]
- Pociecha, E.; Płazek, A.; Janowiak, F.; Zwierzykowski, Z. ABA level, proline and phenolic concentration, and PAL activity induced during cold acclimation in androgenic Festulolium forms with contrasting resistance to frost and pink snow mould (Microdochium nivale). Physiol. Mol. Plant Pathol. 2009, 76, 126–132. [Google Scholar]
- Huang, D.; Ou, B.; Prior, R.L. The chemistry behind antioxidant capacity assays. J. Agric. Food Chem. 2006, 53, 1841–1856. [Google Scholar]
- Kalt, W.; Ryan, D.A.; Duy, J.; Prior, R.L.; Ehlenfeldt, M.K.; Vander, K.S.P. Inter-specific variation in anthocyanin, phenolics and antioxidant capacity among genotypes o high bush and lowbush blueberries (Vaccinium section Cyanococcus spp). J. Agri. Food Chem. 2001, 49, 4761–4767. [Google Scholar] [CrossRef]
- Mahmoodzadeh, H.; Esparham, E. Changes in hydrogen peroxide content and antioxidant enzymes in Abscisic acid induced antioxidant defence in leaves of bean seedlings. Int. J. Bot. 2011, 7, 195–199. [Google Scholar] [CrossRef]
- Percival, D.M.; Joanna, L. Use of plant growth regulators to increase polyphenolic compounds in the wild blueberry. Agric. Inst. Can. 2007, 87, 333–336. [Google Scholar]
- Prior, R.L.; Hoang, H.; Gu, L.; Wu, X.; Bacchiocca, M.; Howard, L. Assays for hydrophylic and lipophilic antioxidant capacity (ORAC) of plasma and other biological and food samples. J. Agric. Food Chem. 2003, 51, 3273–3279. [Google Scholar]
- Chaitanya, K.; Sundar, D.; Masilamani, S.; Ramachandra, R.A. Variation in heat stress induced antioxidant enzyme activities among three mulberry cultivars. Plant Growth Regul. 2002, 36, 175–180. [Google Scholar]
- Cakmak, I.; Horst, W.J. Effects of aluminium and lipid peroxidation, superoxide dismutase, catalase and peroxidase activities in root tips of soybean (Glycine max). Physiol. Plant 1991, 83, 463–468. [Google Scholar]
- Khorsidi, M.; Nijavan, A.M. The effects of abscisic acid and CaCl2 on the activities of antioxidant enzymes under cold stress in maize seedlings in the dark. Pak. J. Biol. Sci. 2006, 9, 54–59. [Google Scholar]
- Bowler, C.; Montagu, M.V.; Inze, D. Superoxide dismutase and stress tolerance. Ann. Rev. Plant Physiol. Mol. Bio. 1992, 43, 83–116. [Google Scholar]
- Moreno, J.J. New aspects of the role of hydroxyeicosatetraenoic acids in cell growth and cancer development. Biochem. Pharmacol. 2009, 77, 1–10. [Google Scholar] [CrossRef]
- Dobrian, A.D.; Lieb, D.C.; Cole, B.K.; Taylor-Fishwick, D.A.; Chakrabarti, S.K.; Nadler, J.L. Functional and pathological roles of the 12- and 15-lipoxygenases. Prog. Lipid. Res. 2011, 50, 115–131. [Google Scholar]
- Kumaraswamy, M.V.; Satish, S. Antioxidant and anti-lipoxygenase activity of Thespesia lampas Dalz and Gibs. Advan. Biol. Res. 2008, 2, 56–59. [Google Scholar]
- Woese, K.; Lange, D.; Boess, C; Bögl, K.W. A comparison of organically and conventionally grown foods—Results of a review of the relevant literature. J. Sci. Food Agric. 1997, 74, 281–293. [Google Scholar]
- Buran, T.J.; Sandhu, A.K.; Azeredo, A.M.; Bent, A.H.; Wiliamson, J.G.; Gu, L. Effect of exogenous abscisic acid on fruit quality, antioxidant capacities and phytochemical contents of southern high bush blueberries. Food Chem. 2012, 132, 1375–1381. [Google Scholar] [CrossRef]
- Ibrahim, M.H.; Jaafar, H.Z.E. Primary, secondary metabolites, H2O2, Malondialdehyde and photosynthetic responses of Orthosiphon stimaneus Benth. to different irradiance levels. Molecules 2012, 17, 1159–1176. [Google Scholar] [CrossRef]
- Ibrahim, M.H.; Jaafar, H.Z.E. Impact of elevated carbon dioxide on primary, secondary metabolites and antioxidant responses of Eleais guineensis Jacq. (Oil Palm) seedlings. Molecules 2012, 17, 5195–5211. [Google Scholar]
- Wang, Y.S.H.; Bunce, A.J.; Maas, L.J. Elevated carbon dioxide increases contents of antioxidant compounds in field-grown strawberries. J. Agric. Food Chem. 2003, 51, 4315–4320. [Google Scholar] [CrossRef]
- Patterson, B.D.; MacRae, E.A.; Ferguson, I.B. Estimation of hydrogen peroxide in plant extracts using titanium (IV). Anal. Biochem. 1984, 139, 487–492. [Google Scholar] [CrossRef]
- Brands, W.W.; Cuvelier, M.E.; Berset, C. Use of free radical method to evaluate antioxidant activity. Food Sci. Tech. 1995, 28, 25–80. [Google Scholar]
- Martínez-Téllez, M.A.; Lafuénte, M.T. Effects of high temperature conditioning on ethylene, phenylalanine ammonia lyase, peroxidase and polyphenol oxidase in flavedo of chilled “Fortune” mandarin fruit. J. Plant Physiol. 1997, 150, 674–678. [Google Scholar] [CrossRef]
- Ibrahim, M.H.; Jaafar, H.Z.E. Photosynthetic capacity, photochemical efficiency and chlorophyll content of three varieties of Labisia pumila benth. exposed to open field and greenhouse growing conditions. Acta Physiol. Plant. 2011, 33, 2179–2185. [Google Scholar] [CrossRef]
- Aebi, H. Catalase. In Methods of Enzymatic Analysis, 3rd ed.; Bergmeyer, H., Verlag, C., Weinheim, A., Eds.; Scientific Pulication: VA, USA, 1983. [Google Scholar]
- Giannopolitis, C.N.; Ries, S.K. Superoxide dismutase: Occurance in higher plants. Plant Physiol. 1977, 59, 309–314. [Google Scholar]
- Wu, H. Affecting the activity of soybean lipoxygenase-1. J. Mol. Graph. 1996, 14, 331–337. [Google Scholar] [CrossRef]
- Ibrahim, M.H.; Jaafar, H.Z.E. Reduced Photoinhibition under Low irradiance enhanced Kacip Fatimah (Labisia pumila Benth) secondary metabolites, Phenyl Alanine Lyase and antioxidant activity. Int. J. Mol. Sci. 2012, 13, 5290–5306. [Google Scholar] [CrossRef]
- Ibrahim, M.H.; Jaafar, H.Z.E.; Rahmat, A.; Rahman, Z.A. Involvement of nitrogen on flavonoids, glutathione, anthocyanin, ascorbic acid and antioxidant activities of Malaysian medicinal plant Labisia pumila Blume (Kacip Fatimah). Int. J. Mol. Sci. 2012, 13, 393–408. [Google Scholar]
- Sample Availability: Not available.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ibrahim, M.H.; Jaafar, H.Z.E. Abscisic Acid Induced Changes in Production of Primary and Secondary Metabolites, Photosynthetic Capacity, Antioxidant Capability, Antioxidant Enzymes and Lipoxygenase Inhibitory Activity of Orthosiphon stamineus Benth. Molecules 2013, 18, 7957-7976. https://doi.org/10.3390/molecules18077957
Ibrahim MH, Jaafar HZE. Abscisic Acid Induced Changes in Production of Primary and Secondary Metabolites, Photosynthetic Capacity, Antioxidant Capability, Antioxidant Enzymes and Lipoxygenase Inhibitory Activity of Orthosiphon stamineus Benth. Molecules. 2013; 18(7):7957-7976. https://doi.org/10.3390/molecules18077957
Chicago/Turabian StyleIbrahim, Mohd Hafiz, and Hawa Z. E. Jaafar. 2013. "Abscisic Acid Induced Changes in Production of Primary and Secondary Metabolites, Photosynthetic Capacity, Antioxidant Capability, Antioxidant Enzymes and Lipoxygenase Inhibitory Activity of Orthosiphon stamineus Benth." Molecules 18, no. 7: 7957-7976. https://doi.org/10.3390/molecules18077957




