Synthesis and Biological Evaluation of Novel Urea- and Guanidine-Based Derivatives for the Treatment of Obesity-Related Hepatic Steatosis
Abstract
:1. Introduction

2. Results and Discussion
2.1. Chemistry

| No. | n | R1 | R2 | Inhibition of Leptin expression in 3T3-L1 (%) a |
|---|---|---|---|---|
| 7a | 0 |  |  | 84.3 |
| 7b | 0 |  | 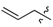 | 67.4 |
| 7c | 0 | 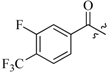 |  | 67.5 |
| 7d | 0 |  |  | 54.3 |
| 7e | 0 |  |  | 73.9 |
| 7f | 0 |  |  | 68.1 |
| 7g | 0 |  |  | 77.1 |
| 7h | 0 |  | 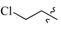 | 74.5 |
| 7i | 0 |  |  | 89.0 |
| 7j | 2 |  | H | 54.1 |
| 7k | 2 |  | H | 69.2 |
| 7l | 2 |  | H | 78.4 |
| 8a | 0 |  | 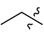 | 61.4 |
| 8b | 0 |  |  | 48.1 |
| 8c | 0 |  |  | 45.0 |
| Metformin | 39.3 |

| No. | n | R1 | Inhibition of Leptin expression in 3T3-L1(%) a |
|---|---|---|---|
| 17a | 0 | 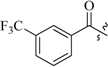 | 53.0 |
| 17b | 0 |  | 42.5 |
| 17c | 0 | 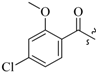 | 42.3 |
| 17d | 0 | 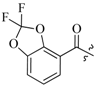 | 22.8 |
| 17e | 0 |  | 15.1 |
| 17f | 0 |  | 27.5 |
| 17g | 0 |  | 34.5 |
| 17h | 2 |  | 33.6 |
| 17i | 2 |  | 18.7 |
| Metformin | 39.3 |

| No. | R1 | R2 | Inhibition of Leptin expression in 3T3-L1 (%) a |
|---|---|---|---|
| 14a | 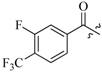 |  | 42.5 |
| 14b |  |  | 45.0 |
| 14c | 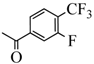 | 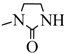 | 50.3 |
| 14d |  | 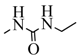 | 24.8 |
| Metformin | 39.3 |
2.2. Expression of Leptin in 3T3-L1 Adipocytes
2.3. Activity in Diet-Induced Obesity (DIO) Mice


| Parameter | Normal | HFD | HFD + Met | HFD + 7i |
|---|---|---|---|---|
| LDL-c (mM) | 0.2 | 0.3 | 0. 2 ± 0. 1 | 0.1 ** |
| HDL-c (mM) | 1.6 ± 0.4 | 2.8 ± 0.6 | 3.0 ± 0.9 | 2.2 * |
| ALT (U/L) | 35.0 ± 12.7 | 44.0 ± 9.6 | 34.0 ± 17.8 | 36.0 ± 9.6 |
| AST (U/L) | 96.0 ± 30.1 | 105.0 ± 10.0 | 121.0 ± 30.9 | 126.0 ± 23.6 |
| TP (g/L) | 51.9 ± 3.3 | 53.1 ± 3.4 | 57.1 ± 6.3 | 55.8 ± 3.7 |
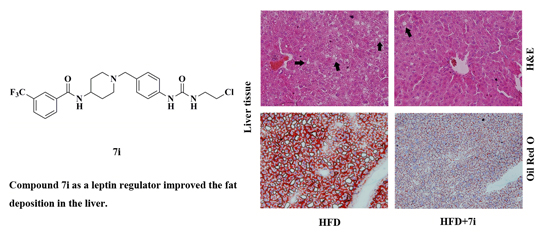
2.4. Histopathological Evaluation

3. Experimental
3.1. General Information
3.2. Chemistry
3.2.1. Synthesis of tert-Butyl (1-(4-nitrobenzyl)piperidin-4-yl)carbamate (3)
3.2.2. General Procedure for the Synthesis of Compounds 4
3.2.3. General Procedure for the Synthesis of Compounds 5 and 12
3.2.4. General Procedure for the Synthesis of Compounds 6 and 13
3.2.5. General Procedure for the Synthesis of Compounds 7a–7i, 8, 14a, 14b, 14d
3.2.6. General Procedure for the Synthesis of Compounds 7j, 7k, 7l and 14c
3.2.7. General Procedure for the Synthesis of 9
3.2.8. General Procedure for the Synthesis of 4-(4-Nitrophenoxy)piperidine (11)
3.2.9. General Procedure for the Synthesis of 16
3.2.10. General Procedure for the Synthesis of 17
3.3. Biological Assay Methods
3.3.2. Expression Levels of Leptin in 3T3-L1 Adipocytes
3.3.3. Animal Model and Treatment of Diet-Induced Obesity.
3.3.4. Assay for Serum Biochemical Markers
3.3.5. Histopathological Examination
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bray, G.A. Obesity: The disease. J. Med. Chem. 2006, 49, 4001–4007. [Google Scholar] [CrossRef]
- Kelly, T.; Yang, W.; Chen, C.-S.; Reynolds, K.; He, J. Global burden of obesity in 2005 and projections to 2030. Int. J. Obesity. 2008, 32, 1431–1437. [Google Scholar] [CrossRef]
- O’Brien, P.E.; Dixon, J.B. The extent of the problem of obesity. Am. J. Surg. 2002, 184, S4–S8. [Google Scholar] [CrossRef]
- Pi-Sunyer, F.X. Medical hazards of obesity. Ann. Intern. Med. 1993, 119, 655–660. [Google Scholar] [CrossRef]
- Feneberg, A.; Malfertheiner, P. Epidemic trends of obesity with impact on metabolism and digestive diseases. Digest. Dis. 2012, 30, 143–147. [Google Scholar] [CrossRef]
- Schindhelm, R.K.; Diamant, M.; Dekker, J.M.; Tushuizen, M.E.; Teerlink, T.; Heine, R.J. Alanine aminotransferase as a marker of non-alcoholic fatty liver disease in relation to type 2 diabetes mellitus and cardiovascular disease. Diabetes. Metab. Res. Rev. 2006, 22, 437–443. [Google Scholar] [CrossRef]
- Bacon, B.R.; Farahvash, M.J.; Janney, C.G.; Neuschwander-Tetri, B.A. Nonalcoholic steatohepatitis: An expanded clinical entity. Gastroenterology 1994, 107, 1103–1109. [Google Scholar]
- Henao-Mejia, J.; Elinav, E.; Jin, C.; Hao, L.; Mehal, W.Z.; Strowig, T.; Thaiss, C.A.; Kau, A.L.; Eisenbarth, S.C.; Jurczak, M.J.; et al. Inflammasome-mediated dysbiosis regulates progression of NAFLD and obesity. Nature 2012, 482, 179–186. [Google Scholar]
- Angulo, P. Nonalcoholic fatty liver disease. N. Engl. J. Med. 2002, 346, 1221–1231. [Google Scholar] [CrossRef]
- Browning, J.D.; Horton, J.D. Molecular mediators of hepatic steatosis and liver injury. J. Clin. Invest. 2004, 114, 147–152. [Google Scholar] [CrossRef]
- Jéquier, E. Leptin signaling, adiposity, and energy balance. Ann. N. Y. Acad. Sci. 2002, 967, 379–388. [Google Scholar] [CrossRef]
- Børbaek, C.; Kahn, B.B. Leptin signaling in the central nervous system and the periphery. Prog. Horm. Res. 2004, 59, 305–331. [Google Scholar] [CrossRef]
- Dellas, C.; Lankeit, M.; Reiner, C.; Schäfer, K.; Hasenfuß, G.; Konstantinides, S. BMI-independent inverse relationship of plasma leptin levels with outcome in patients with acute pulmonary embolism. Int. J. Obes. (Lond.) 2013, 37, 204–210. [Google Scholar] [CrossRef]
- Considine, R.V.; Sinha, M.K.; Heiman, M.L.; Kriauciunas, A.I.; Stephens, T.W.; Nyce, M.R.; Ohannesian, J.P.; Marco, C.C.; Mckee, L.J.; Bauer, T.L.; et al. Serum immunoreactive-leptin concentrations in normal-weight and obese humans. N. Engl. J. Med. 1996, 334, 292–295. [Google Scholar] [CrossRef]
- Roberts, E.A. Pediatric nonalcoholic fatty liver disease (NAFLD): A “growing” problem? J. Hepatol. 2007, 46, 1133–1142. [Google Scholar] [CrossRef]
- Pérez, C.; Fernández-Galaz, C.; Fernández-Agulló, T.; Arribas, C.; Andrés, A.; Ros, M.; Carrascosa, J.M. Leptin impairs insulin signaling in rat adipocytes. Diabetes 2004, 53, 347–353. [Google Scholar] [CrossRef]
- Cohen, B.; Novick, D.; Rubinstein, M. Modulation of insulin activities by leptin. Science 1996, 274, 1185–1188. [Google Scholar] [CrossRef]
- Muller, G.; Ertl, J.; Gerl, M.; Preibish, G. Leptin impairs metabolic actions of insulin in isolated rat adipocytes. J. Biol. Chem. 1997, 272, 10585–10593. [Google Scholar] [CrossRef]
- Valerio, N.; Melania, M.; Paolo, C.; Vincenzo, D.; Rita, D.; Fiorella, P.; Roberto, C.; Donatella, G.; Matilde, M. Leptin, free leptin index, insulin resistance and liver fibrosis in children with non-alcoholic fatty liver disease. Eur. J. Endocrinol. 2006, 155, 735–743. [Google Scholar] [CrossRef]
- Lewis, G.F.; Carpentier, A.; Adeli, K.; Giacca, A. Disordered fat storage and mobilization in the pathogenesis of insulin resistance and type 2 diabetes. Endocr. Rev. 2002, 23, 201–229. [Google Scholar] [CrossRef]
- Leclercq, I.A.; Farrell, G.C.; Schriemer, R.; Robertson, G.R. Leptin is essential for the hepatic fibrogenic response to chronic liver injury. J. Hepatol. 2002, 37, 206–213. [Google Scholar] [CrossRef]
- Imajo, K.; Fujita, K.; Yoneda, M.; Nozaki, Y.; Ogawa, Y.; Shinohara, Y.; Kato, S.; Mawatari, H.; Shibata, W.; Kitani, H.; et al. Hyperresponsivity to low-dose endotoxin during progression to nonalcoholic steatohepatitis is regulated by leptin-mediated signaling. Cell. MeTable 2012, 16, 44–54. [Google Scholar] [CrossRef]
- Patel, K.; Muir, A.; McHutchison, J.G.; Patton, H.M. A link between leptin and steatosis in chronic hepatitis C? Time to weigh up the fats. Am. J. Gastroenterol. 2003, 98, 952–955. [Google Scholar] [CrossRef]
- Freemark, M.; Bursey, D. The effects of metformin on body mass index and glucose tolerance in obese adolescents with fasting hyperinsulinemia and a family history of type 2 diabetes. Pediatrics 2001, 107, E55. [Google Scholar] [CrossRef]
- Duseja, A.; Das, A.; Dhiman, R.K.; Chawla, Y.K.; Thumburu, K.T.; Bhadada, S.; Bhansali, A. Metformin is effective in achieving biochemical response in patients with nonalcoholic fatty liver disease (NAFLD) not responding to lifestyle interventions. Ann. Hepatol. 2007, 6, 222–226. [Google Scholar]
- Stein, L.L.; Dong, M.H.; Loomba, R. Insulin Sensitizers in Nonalcoholic Fatty Liver Disease and Steatohepatitis: Current Status. Adv. Ther. 2009, 26, 893–907. [Google Scholar] [CrossRef]
- Plosker, GL.; Faulds, D. Troglitazone: A review of its use in themanagement of type 2 diabetes mellitus. Drugs 1999, 57, 409–438. [Google Scholar] [CrossRef]
- Pereira, C.V.; Moreira, A.C.; Pereira, S.P.; Machado, N.G.; Carvalho, F.S.; Sardao, V.A.; Oliveira, P.J. Investigating Drug-induced Mitochondrial Toxicity: A Biosensor to Increase Drug Safety? Curr. Drug Saf. 2009, 4, 34–54. [Google Scholar] [CrossRef]
- Venier, O.; Pascal, C.; Braun, A.; Namane, C.; Mougenot, P.; Crespin, O.; Pacquet, F.; Mougenot, C.; Monseau, C.; Onofri, B.; et al. Pyrrolidine-pyrazole ureas as potent and selective inhibitors of 11β-hydroxysteroid-dehydrogenase type 1. Bioorg. Med. Chem. Lett. 2011, 21, 2244–2251. [Google Scholar] [CrossRef]
- Sophie, P.; Peter, R.M.; Steve, S.; Veronique, G. Fluorine in medicinal chemistry. Chem. Soc. Rev. 2008, 37, 320–330. [Google Scholar] [CrossRef]
- Hoffler, U.; Hobbie, K.; Wilson, R.; Bai, R.; Rahman, A.; Malarkey, D.; Travlos, G.; Ghanayem, B.I. Diet-induced obesity is associated with hyperleptinemia, hyperinsulinemia, hepatic steatosis, and glomerulopathy in C57Bl/6J mice. Endocrine 2009, 36, 311–325. [Google Scholar] [CrossRef]
- Kang, H.S.; Okamoto, K.; Takeda, Y.; Beak, J.Y.; Gerrish, K.; Bortner, C.D.; DeGraff, L.M.; Wada, T.; Xie, W.; Jetten, A.M. Transcriptional profiling reveals a role for RORα in regulating gene expression in obesity-associated inflammation and hepatic steatosis. Physiol. Genomics 2011, 43, 818–828. [Google Scholar] [CrossRef]
- Adachi, M.; Brenner, D.A. High molecular weight adiponectin inhibits proliferation of hepatic stellate cells via activation of adenosine monophosphate-activated protein kinase. Hepatology 2008, 47, 677–685. [Google Scholar] [CrossRef]
- Polyzos, S.A.; Kountouras, J.; Zavos, C.; Tsiaousi, E. The role of adiponectin in the pathogenesis and treatment of non-alcoholic fatty liver disease. Diabetes Obes. Metab. 2010, 12, 365–383. [Google Scholar] [CrossRef]
- Wang, Y.; Zhou, M.; Lam, K.S.; Xu, A. Protective roles of adiponectin in obesity-related fatty liver diseases: Mechanisms and therapeutic implications. Arq. Bras. Endocrinol. Metabol. 2009, 53, 201–212. [Google Scholar]
- Hench, J.; Hench, I.B.; Pujol, C.; Ipsen, S.; Brodesser, S.; Mourier, A.; Tolnay, M.; Frank, S.; Trifunović, A. A tissue-specific approach to the analysis of metabolic changes in Caenorhabditis elegans. PLoS One 2011, 6, e28417. [Google Scholar] [CrossRef]
- Cook, H.C. Origins of Tinctorial methods in histology. J. Lin. Pathol. 1997, 50, 716–720. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 7a–l, 8a–c, 14a–d, 17a–i are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Liang, X.; Pei, H.; Ma, L.; Ran, Y.; Chen, J.; Wang, G.; Chen, L. Synthesis and Biological Evaluation of Novel Urea- and Guanidine-Based Derivatives for the Treatment of Obesity-Related Hepatic Steatosis. Molecules 2014, 19, 6163-6183. https://doi.org/10.3390/molecules19056163
Liang X, Pei H, Ma L, Ran Y, Chen J, Wang G, Chen L. Synthesis and Biological Evaluation of Novel Urea- and Guanidine-Based Derivatives for the Treatment of Obesity-Related Hepatic Steatosis. Molecules. 2014; 19(5):6163-6183. https://doi.org/10.3390/molecules19056163
Chicago/Turabian StyleLiang, Xiaolin, Heying Pei, Liang Ma, Yan Ran, Jinying Chen, Guangcheng Wang, and Lijuan Chen. 2014. "Synthesis and Biological Evaluation of Novel Urea- and Guanidine-Based Derivatives for the Treatment of Obesity-Related Hepatic Steatosis" Molecules 19, no. 5: 6163-6183. https://doi.org/10.3390/molecules19056163