Easy Access to Evans’ Oxazolidinones. Stereoselective Synthesis and Antibacterial Activity of a New 2-Oxazolidinone Derivative
Abstract
:1. Introduction
1.1. Evans’ Oxazolidinones

1.2. Oxazolidinone Derivatives

2. Results and Discussion
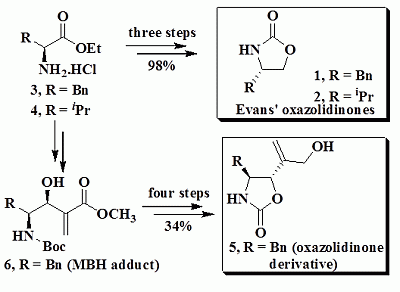
2.1. Preparation of Evans’ Oxazolidinones 1 and 2

2.2. Preparation of 2-Oxazolidinone Derivative 5


2.3. Antibacterial Activity
| S. aureus | Compound 5 | Compound 14 | Ciclopirox Olamine | DMSO |
|---|---|---|---|---|
| 680 | 19.0 ± 0.58 | 2.0 ± 0.48 | 17.0 ± 0.58 | 0.00 |
| 2221 | 18.0 ± 0.60 | NA | 18.0 ± 0.60 | 0.00 |
| 4006 | 21.0 ± 0.33 | 2.0 ± 0.58 | 17.0 ± 0.58 | 0.00 |
| 4075 | 15.0 ± 0.67 | NA | 17.0 ± 0.58 | 0.00 |
| 4127 | 16.0 ± 0.58 | 2.0 ± 0.50 | 17.0 ± 0.50 | 0.00 |
| MIC (μg mL−1) | |
|---|---|
| Compound 5 | 0.03 |
| Ciclopirox olamine | 0.05 |
3. Experimental
3.1. General Procedures
3.2. Synthesis
3.2.1. General Procedure for the Synthesis of Evans’ Oxazolidinones 1 and 2
3.2.2. (−)-(3S,4S)-4-Tert-butoxycarbonylamino-3-hydroxy-2-methylene-5-phenylpentanoic Acid Methyl Ester (6) [11]
3.2.3. (+)-(4S,5S)-4-Benzyl-5-(1-Methoxycarbonylvinyl)-2,2-dimethyloxazolidine-3-carboxylic Acid Tert-Butyl Ester (14)
3.2.4. (−)-(3S,4S)-4-Benzyl-5-(1-Hydroxymethylvinyl)-2-oxazolidinone (5)
3.3. Antibacterial Assays
3.3.1. Bacterial Strains and Culture Media
3.3.2. Antibacterial Screening Assay
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Evans, D.A.; Bartroli, J.; Shih, T.L. Enantioselective aldol condensations. 2. Erythro-selective chiral aldol condensations via boron enolates. J. Am. Chem. Soc. 1981, 103, 2127–2129. [Google Scholar] [CrossRef]
- Gaul, C.; Seebach, D. A valine-derived lithiated 3-methylthiomethyl-1,3-oxazolidin-2-one for enantioselective nucleophilic hydroxymethylation, Formylation, and alkoxycarbonylation of aldehydes. Org. Lett. 2000, 2, 1501–1504. [Google Scholar] [CrossRef]
- Evans, D.A.; Wu, L.D.; Wiener, J.J.M.; Johnson, J.S.; Ripin, D.H.B.; Tedrow, J.S. A general method for the synthesis of enantiomerically pure β-substituted β-amino acids through α-substituted succinic acid derivatives. J. Org. Chem. 1999, 64, 6411–6417. [Google Scholar]
- Sibi, M.P. Are two phenyls better than one? Synthesis and applications of (R)-4-diphenylmethyl-2-oxazolidinone. Aldrichim. Acta 1999, 32, 93–103. [Google Scholar]
- Ager, D.J.; Prakash, I.; Schaad, D.R. 1,2-Amino alcohols and their heterocyclic derivatives as chiral auxiliaries in asymmetric synthesis. Chem. Rev. 1996, 96, 835–875. [Google Scholar] [CrossRef]
- Newman, M.S.; Kutner, A. New reactions involving alkaline nitroso-2-oxazolidones. J. Am. Chem. Soc. 1951, 73, 4199–4204. [Google Scholar] [CrossRef]
- Slee, A.M.; Wuonola, M.A.; McRipley, R.J.; Zajac, L.; Zawada, M.J.; Bartholomew, P.T.; Gregory, W.A.; Forbes, M. Oxazolidinones, a new class of synthetic antibacterial agents: In vitro and in vivo. Antimicrob. Agents Chemother. 1987, 31, 1791–1797. [Google Scholar] [CrossRef]
- Ford, C.W.; Zurenko, G.E.; Barbachyn, M.R. The discovery of linezolid, the first oxazolidinone antibacterial agent. Curr. Drug. Targets Infect. Disord. 2001, 1, 181–199. [Google Scholar] [CrossRef]
- Mishra, R.K.; Revell, K.D.; Coates, C.M.; Turos, E.; Dickey, S.; Lim, D.V. N-thiolated 2-oxazolidinones: A new family of antibacterial agents for methicillin-resistant Staphylococcus aureus and Bacillus anthracis. Bioorg. Med. Chem. Lett. 2006, 16, 2081–2083. [Google Scholar] [CrossRef]
- Heldreth, B.; Long, T.E.; Jang, S.; Reddy, G.S.K.; Turos, E.; Dickey, S.; Lim, D.V. N-Thiolated beta-lactam antibacterials: Effects of the N-organothio substituent on anti-MRSA activity. Bioorg. Med. Chem. Lett. 2006, 14, 3775–3784. [Google Scholar]
- Coelho, F.; Diaz, G.; Abella, C.A.; Almeida, W.P. The Baylis-Hillman reaction with chiral α-amino aldehydes under racemization-free conditions. Synlett 2006, 3, 435–439. [Google Scholar]
- Hoffman, R.V.; Maslouh, N.; Cervantes-Lee, F. Highly stereoselective syntheses of syn- and anti-1,2-amino alcohols. J. Org. Chem. 2002, 67, 1045–1056. [Google Scholar] [CrossRef]
- Kempf, D.J. Dipeptide analogs. Versatile synthesis of hydroxyethylene isostere. J. Org. Chem. 1986, 51, 3921–3926. [Google Scholar]
- Rich, D.H.; Sun, E.T.O.; Ulm, E. Synthesis of analogs of the carboxyl protease inhibitor pepstatin. Effect of structure on inhibition of pepsin and renin. J. Med. Chem. 1980, 23, 27–33. [Google Scholar] [CrossRef]
- Gupta, A.K.; Plott, T. Ciclopirox olamine: A broad-spectrum antifungal with antibacterial and anti-inflammatory properties. Int. J. Dermatol. 2004, 43, 3–8. [Google Scholar] [CrossRef]
- Jue, S.G.; Daeson, G.W.; Brodgen, R.N. Ciclopirox olamine 1% cream: A preliminary review of its antimicrobial activity and therapeutic use. Drugs 1985, 29, 330–341. [Google Scholar] [CrossRef]
- Caetano, N.; Saraiva, A.; Pereira, R.; Carvalho, D.; Pimentel, M.C.B.; Maia, M.B.S. Determinação de atividades antimicrobiana de extratos de plantas de uso popular como anti-inflamatório. Braz. J. Pharmacog. 2002, 12, 132–135. [Google Scholar]
- Sample Availability: Samples of the compounds are available from the authors.
© 2014 by the authors. licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Diaz, G.; De Freitas, M.A.A.; Ricci-Silva, M.E.; Diaz, M.A.N. Easy Access to Evans’ Oxazolidinones. Stereoselective Synthesis and Antibacterial Activity of a New 2-Oxazolidinone Derivative. Molecules 2014, 19, 7429-7439. https://doi.org/10.3390/molecules19067429
Diaz G, De Freitas MAA, Ricci-Silva ME, Diaz MAN. Easy Access to Evans’ Oxazolidinones. Stereoselective Synthesis and Antibacterial Activity of a New 2-Oxazolidinone Derivative. Molecules. 2014; 19(6):7429-7439. https://doi.org/10.3390/molecules19067429
Chicago/Turabian StyleDiaz, Gaspar, Michelle A. A. De Freitas, Maria E. Ricci-Silva, and Marisa A. N. Diaz. 2014. "Easy Access to Evans’ Oxazolidinones. Stereoselective Synthesis and Antibacterial Activity of a New 2-Oxazolidinone Derivative" Molecules 19, no. 6: 7429-7439. https://doi.org/10.3390/molecules19067429