Characterization and Antitumor Activity of a Polysaccharide from Sarcodia ceylonensis
Abstract
:1. Introduction
2. Results and Discussion
2.1. Polysaccharide Isolation and Purification
2.2. Infrared Spectrum of SCP-60
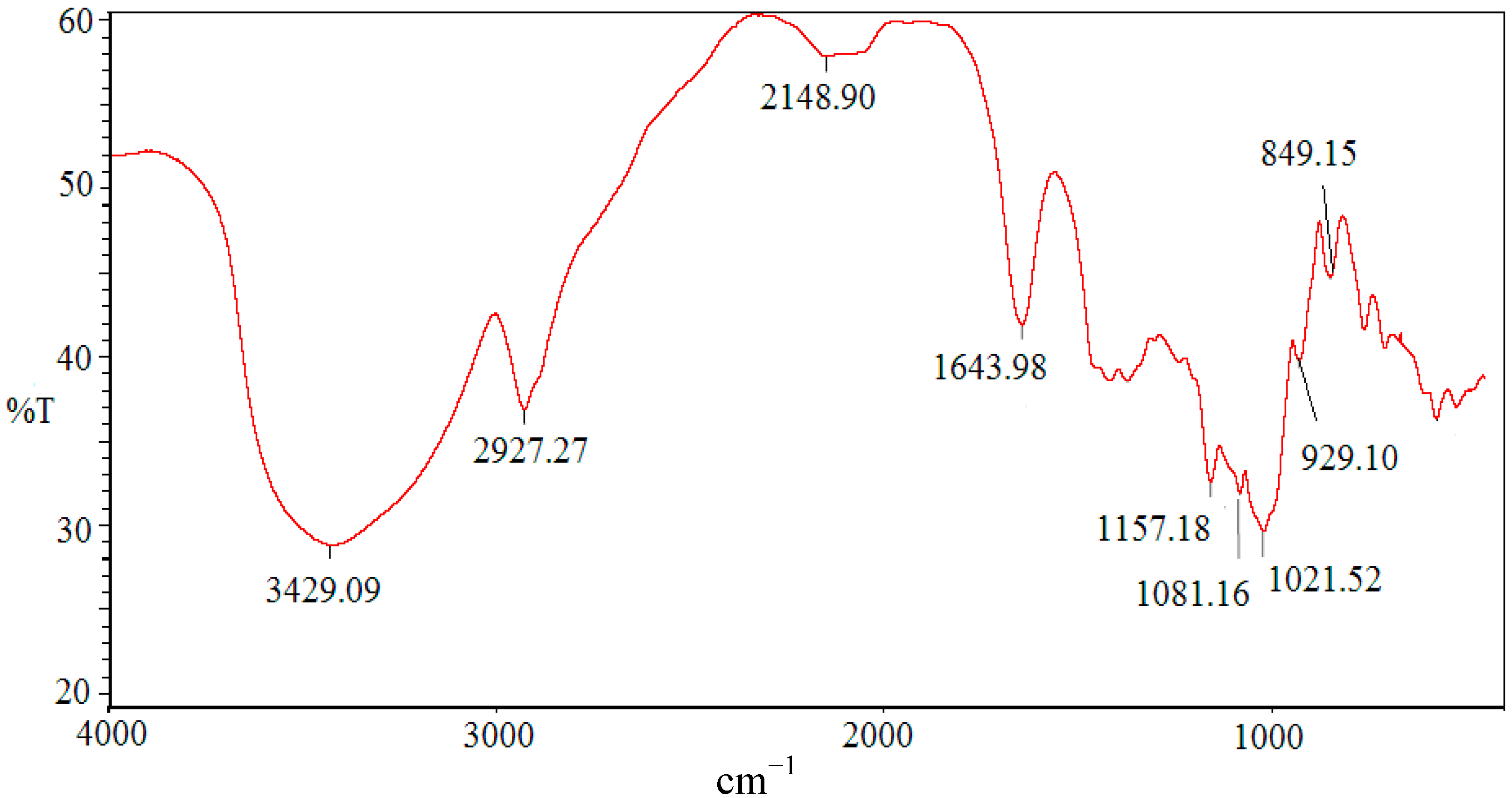
2.3. NMR Identification of SCP-60


| Sugar Residues | C-1 | C-2 | C-3 | C-4 | C-5 | C-6 |
|---|---|---|---|---|---|---|
| H-1 | H-2 | H-3 | H-4 | H-5 | H-6 | |
| α-d-Glcp-(1→ | 97.75 | 71.50 | 75.41 | 69.67 | 73.00 | 60.63 |
| 5.058 | 3.711 | 3.776 | 3.643 | 3.908 | 3.908 | |
| →6)-α- Manp-(1→ | 97.75 | 75.41 | 71.50 | 68.92 | 71.90 | 66.23 |
| 5.058 | 4.072 | 3.908 | 3.776 | 3.711 | 3.998 | |
| →6)-α-d-Glcp-(1→ | 97.75 | 73.00 | 75.41 | 69.68 | 71.90 | 68.92 |
| 5.058 | 3.643 | 3.776 | 3.422 | 3.908 | 3.998 | |
| →6)-α-Galp-(1→ | 97.75 | 68.92 | 69.68 | 68.92 | 69.60 | 66.23 |
| 5.058 | 3.711 | 3.776 | 3.908 | 3.998 | 3.691 |
2.4. Tumor Inhibition Effects
| Group | Dose (mg/kg) | Inhibition Rate of Tumor (%) | Thymus Index(mg/g) | Spleen Index(mg/g) |
|---|---|---|---|---|
| MC | ‒ | ‒ | 3.01 ± 0.66 | 12.99 ± 0.97 |
| CTX | 20 | 65.62 ± 3.29 | 1.13 ± 0.05 | 8.72 ± 0.55 |
| Polysaccharide | 100 | 69.01 ± 5.01 | 5.69 ± 0.37 bc | 16.79 ± 1.09 bc |
| 50 | 57.95 ± 3.75 | 4.85 ± 0.21 ac | 15.37 ± 1.16 bc | |
| 25 | 36.78 ± 2.22 | 3.78 ± 0.09 c | 13.06 ± 0.86 c |
2.5. Analysis of Immune Index
2.6. Effect of SCP-60 on Body Weight in Mice

2.7. Determination of IL-2, TNF-α, and IFN-γ using ELISA Kits

2.8. Analysis of SOD and MDA in S180 Mice
| Group | Dose (mg/kg) | SOD (U/mL) | MDA (nmol/mL) |
|---|---|---|---|
| MC | ‒ | 34.69 ± 3.17 | 30.51 ± 3.66 |
| CTX | 20 | 38.34 ± 2.77 | 41.69 ± 4.29 |
| Polysaccharide | 100 | 63.63 ± 4.82 bc | 7.62 ± 1.03 bc |
| 50 | 52.15 ± 3.62 bc | 10.12 ± 2.77 bc | |
| 25 | 41.71 ± 3.86 a | 26.57 ± 2.82 c |
3. Experimental Section
3.1. Materials and Chemicals
3.2. Extraction of Crude Polysaccharide from Sarcodia ceylonensis
3.3. Purification of the Crude Polysaccharide
3.4. Determination of the Molecular Weights of Purification Polysaccharide
3.5. Infrared Spectra Analysis
3.6. Analysis of Monosaccharide Compositions
3.7. NMR Identification
3.8. Antitumor Test in Vivo
3.8.1. Animals and Treatment
3.8.2. Tumor Inhibition Effect
3.8.3. Analysis of Immune Index
3.8.4. Determination of IL-2, TNF-α, and IFN-γ by ELISA Method
3.8.5. Detection of SOD Activity and MDA Level in S180-Bearing Mice
3.9. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Schepetkin, I.A.; Quinn, M.T. Botanical polysaccharides: Macrophage immunomodulation and therapeutic potential. Int. Immunopharmacol. 2006, 6, 317–333. [Google Scholar] [CrossRef]
- Fan, Y.J.; Luo, A.X. Evaluation of anti-tumor activity of water-soluble polysaccharides from Dendrobium denneanum. Afr. J. Pharm. Pharm. 2011, 5, 415–420. [Google Scholar] [CrossRef]
- Silva, R.O.; Santana, A.P.M.; Carvalho, N.S.; Bezerra, T.S.; Oliveira, C.B.; Damasceno, S.R.B.; Chaves, L.S.; Freitas, A.L.P.; Soares, P.M.G.; Souza, M.H.L.P.; et al. A sulfated-polysaccharide fraction from seaweed gracilaria birdiae prevents naproxen-induced gastrointestinal damage in rats. Mar. Drugs 2012, 10, 2618–2633. [Google Scholar] [CrossRef]
- Bouhlal, R.; Haslin, C.; Chermann, J.C.; Colliec-Jouault, S.; Sinquin, C.; Simon, G.; Cerantola, S.; Riadi, H.; Bourgougnon, N. Antiviral activities of sulfated polysaccharides isolated from Sphaerococcus coronopifolius (Rhodophytha, Gigartinales) and Boergeseniella thuyoides (Rhodophyta, Ceramiales). Mar. Drugs 2011, 9, 1187–1209. [Google Scholar] [CrossRef]
- Esteves, A.I.S.; Nicolai, M.; Humanes, M.; Goncalves, J. Sulfated polysaccharides in marine sponges: Extraction methods and anti-hiv activity. Mar. Drugs 2011, 9, 139–153. [Google Scholar] [CrossRef]
- Thinh, P.D.; Menshova, R.V.; Ermakova, S.P.; Anastyuk, S.D.; Ly, B.M.; Zvyagintseva, T.N. Structural characteristics and anticancer activity of fucoidan from the Brown Alga Sargassum mcclurei. Mar. Drugs 2013, 11, 1456–1476. [Google Scholar] [CrossRef]
- Maruyama, H.; Tamauchi, H.; Hashimoto, M.; Nakano, T. Antitumor activity and immune response of mekabu fucoidan extracted from sporophyll of Undaria pinnatifida. In Vivo 2003, 17, 245–249. [Google Scholar]
- Eldeen, A.M.G.; Ahmed, E.F.; Zeid, M.A.A. In vitro cancer chemopreventive properties of polysaccharide extract from the brown alga, Sagassum latifolium. Food Chem. Toxicol. 2009, 47, 1378–1384. [Google Scholar] [CrossRef]
- Chen, X.; Nie, W.; Yu, G.; Li, Y.; Hu, Y.; Lu, J.; Jin, L. Antitumor and immunomodulatory activity of polysaccharides from Sargassum fusiforme. Food Chem. Toxicol. 2012, 50, 695–700. [Google Scholar] [CrossRef]
- Han, X.Q.; Wu, X.M.; Chai, X.Y.; Chen, D.; Dai, H.; Dong, H.L.; Ma, Z.Z.; Gao, X.M.; Tu, P.F. Isolation, characterization and immunological activity of a polysaccharide from the fruit bodies of an edible mushroom, Sarcodon aspratus (Berk.) S. Ito. Food Res. Int. 2011, 44, 489–493. [Google Scholar] [CrossRef]
- Zhao, T.; Mao, G.H.; Mao, R.; Zou, Y.; Zheng, D.H.; Feng, W.W.; Ren, M.; Wang, W.; Zheng, W.; Song, J.; et al. Antitumor and immunomodulatory activity of a water-soluble low molecular weight polysaccharide from Schisandra chinensis (Turcz.) Baill. Food Chem. Toxicol. 2013, 55, 609–616. [Google Scholar] [CrossRef]
- Wu, X.; Mao, G.; Fan, Q.; Zhao, T.; Zhao, J.; Li, F.; Yang, L. Isolation, purification, immunological and anti-tumor activities of polysaccharides from Gymnema sylvestre. Food Res. Int. 2012, 48, 935–939. [Google Scholar] [CrossRef]
- Xia, B.M. Chinese Journal of Marine Algae Volume II Rhodophyta; Science Press: Beijing, China, 1999; p. 107. [Google Scholar]
- Luo, D.H. Identification of structure and antioxidant activity of a fraction of polysaccharide purified from Dioscorea nipponica Makino. Carbohydr. Polym. 2008, 71, 544–549. [Google Scholar] [CrossRef]
- Liu, Y.H.; Wang, F.S. Structural characterization of an active polysaccharide from Phellinus ribis. Carbohydr. Polym. 2007, 70, 386–392. [Google Scholar] [CrossRef]
- Zhao, M.M.; Yang, N.; Yang, B. Structural characterization of water-soluble olysaccharides from Opuntia monacanthap cladodes in relation to their anti-glycated activities. Food Chem. 2007, 105, 1480–1486. [Google Scholar] [CrossRef]
- Barker, S.A.; Bourne, E.J.; Stacey, M.; Whiffen, D.H. Infrared spectra of carbohydrates. Part I. Some derivatives of d-glucopyranose. J. Chem. Soc. 1954, 75, 171–176. [Google Scholar]
- Yoon, S.; Kim, M.K.; Lee, I.Y.; Yun, M.; Nam Shin, J.E. Production and structural features of a water-soluble polysaccharide from a mutant strain of Agrobacterium sp. J. Ind. Eng. Chem. 2008, 14, 759–764. [Google Scholar] [CrossRef]
- Kath, F.; Kulicke, W.M. Mild enzymatic isolation of mannan and glucan from yeast Saccharomyces cerevisiae. Angew. Makromol. Chem. 1999, 268, 59–68. [Google Scholar] [CrossRef]
- Byun, E.B.; Sung, N.Y.; Kim, J.H.; Choi, J.; Matsui, T.; Byun, M.W. Enhancement of anti-tumor activity of gamma-irradiated silk fibroin via immunomodulatory effects. Chem.-Biol. Interact. 2010, 186, 90–95. [Google Scholar] [CrossRef]
- Chen, J.R.; Hu, T.J.; Zheng, R.L. Antioxidant activities of Sophora subprosrate polysaccharide in immunosuppressed mice. Int. Immunopharmacol. 2007, 7, 547–553. [Google Scholar] [CrossRef]
- Valenzuela, A. The biological significance of determination in the assessment of tissue oxidative stress. Life Sci. 1990, 48, 301–309. [Google Scholar] [CrossRef]
- Neilsen, F.; Mikkelsen, B.B.; Neilsen, J.B.; Andersen, H.R.; Grandjean, P. Plasma malondialdehyde as biomar reference interval and effects of life-style factors. Clin. Chem. 1997, 47, 1209–1214. [Google Scholar]
- Yagi, K. Lipid peroxides and human diseases. Chem. Phys. Lipids 1987, 45, 337–351. [Google Scholar] [CrossRef]
- Jiau-Jian, L.; Larry, W.O. Over expression of manganese-containing superoxide dismutase confers resistance to the cyto-toxicity of tumor necrosis factor and/or hyperthermia. Cancer Res. 1977, 57, 1991–1998. [Google Scholar]
- Ming, L.; Jill, C.P.; Jingfang, J.N.; Edward, C.; Brash, E. Antioxidant action via p53 mediated apoptosis. Cancer Res. 1998, 58, 1723–1729. [Google Scholar]
- Yerra, R.; Malaya, G.; Upal, K.M. Antitumor activity and in vivo antioxidant status of Mucuna pruriens (Fabaceae) Seeds against Ehrlich Ascites Carcinoma in Swiss Albino Mice. Iran J. Pharmacol. Ther. 2005, 4, 46–53. [Google Scholar]
- Asakawa, T.; Matsuhita, S. Colouring conditions of Thiobarbituric acid test for detecting lipid hydroperoxides. Lipids 1980, 15, 137–140. [Google Scholar] [CrossRef]
- Navarini, L.; Gilli, R.; Gombac, V.; Abatangelo, A.; Bosco, M.; Toffanin, R. Polysaccharides from hot water extracts of roasted Coffea arabica beans: Isolation and characterization. Carbohydr. Polym. 1999, 40, 71–81. [Google Scholar] [CrossRef]
- Luo, A.X.; Ge, Z.F.; Fan, Y.J.; Luo, A.S.; Chun, Z.; He, X.J. In vitro and in vivo antioxidant activity of a water-soluble polysaccharide from Dendrobium denneanum. Molecules 2011, 16, 1579–1592. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Nunome, T.; Yamauchi, R.; Kato, K.; Sone, Y. Structure of an exocellular polysaccharide of Lactobacillus helveticus TN-4, a spontaneous mutant strain of Lactobacillus helveticus TY1–2. Carbohydr. Res. 1995, 275, 319–332. [Google Scholar] [CrossRef]
- Kumar, C.G.; Joo, H.S.; Choi, J.W.; Koo, Y.M.; Chang, C.S. Purification and characterization of extracellular polysaccharide from haloalkalophilic Bacillus sp. I-450. Enzyme Microb. Technol. 2004, 34, 673–681. [Google Scholar] [CrossRef]
- Fan, Y.J.; He, X.J.; Zhou, S.D.; Luo, A.X.; He, T.; Chun, Z. Composition analysis and antioxidant activity of polysaccharide from Dendrobium denneanum. Int. J. Biol. Macromol. 2009, 45, 169–173. [Google Scholar] [CrossRef]
- Pang, X.B.; Yao, W.B.; Yang, X.B.; Xie, C.; Liu, D.; Zhang, J.; Gao, X.D. Purification, characterization and biological activity on hepatocytes of a polysaccharide from Flammulina velutipes mycelium. Carbohydr. Res. 2007, 70, 291–297. [Google Scholar]
- Furukawa, T.; Kubota, T.; Tanino, H.; Oura, S.; Yuasa, S.; Murate, H.; Morita, K.; Kozakai, K.; Yano, T.; Hoffman, R.M. Chemosensitivity of breast cancer lymph node metastasis compared to the primary tumor from individual patients tested in the histoculture drug response assay. Anticancer Res. 2000, 20, 3657–3658. [Google Scholar]
- Sample availability: Not available.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Fan, Y.; Lin, M.; Luo, A.; Chun, Z.; Luo, A. Characterization and Antitumor Activity of a Polysaccharide from Sarcodia ceylonensis. Molecules 2014, 19, 10863-10876. https://doi.org/10.3390/molecules190810863
Fan Y, Lin M, Luo A, Chun Z, Luo A. Characterization and Antitumor Activity of a Polysaccharide from Sarcodia ceylonensis. Molecules. 2014; 19(8):10863-10876. https://doi.org/10.3390/molecules190810863
Chicago/Turabian StyleFan, Yijun, Mengchuan Lin, Aoshuang Luo, Ze Chun, and Aoxue Luo. 2014. "Characterization and Antitumor Activity of a Polysaccharide from Sarcodia ceylonensis" Molecules 19, no. 8: 10863-10876. https://doi.org/10.3390/molecules190810863





