1. Introduction
Carbon tetrachloride (CCl
4) is an industrial solvent and a xenobiotic used to induce chemical hepatic injuries in laboratory animals. The CCl
4-induced hepatic lesions are a common experimental model for the screening of the hepatoprotective activity of certain drugs, CCl
4 being a strong hepatotoxic agent and a single exposure to it rapidly leading to severe hepatic necrosis and steatosis [
1,
2,
3]. CCl
4 is metabolized by the hepatic microsomal P
450 isoenzymes through the reductive dehalogenation of the toxic in the endoplasmic reticulum of hepatic cells resulting in the unstable free radicals of trichloromethyl (CCl
3·) and trichloromethyl peroxyl (CCl
3O
2·).
It has been demonstrated that propolis has a hepatoprotective potential, as well as anti-inflammatory, immunostimulating, antiviral, and antibacterial effects [
4,
5,
6,
7]. Lin
et al. revealed that ethanolic propolis extract significantly prevents the increase of the levels of microsomal enzymes and the lipidic peroxidation in rats that were administered alcohol [
8]. The hepatoprotective effect of royal jelly (RJ) in CCl
4-induced liver damage has been reported by Cemek
et al. [
9]. Laboratory animals that were administered this toxic substance presented histopathological modifications in the liver and biochemical alterations. Bee pollen is a very important source of saccharides, lipids, proteins and amino acids, vitamins and polyphenolic compounds, mainly flavonoids, thus being able to exert hepatoprotective effects [
10,
11]. The hepatoprotective effects of honey have also been studied over time [
12]. Honey has a long history of use as a natural source of sugars and it is also an important ingredient in traditional medicine due to its antimicrobial and anti-inflammatory properties [
13,
14].
Many people suffer nowadays from hepatic disorders induced by alcohol consumption, exposure to chemical substances and infections. As chronic and acute hepatic affections continue to cause severe health problems all over the world, numerous experimental studies have taken considerable interest in the role of natural products such as extract of
Coriandrum sativum [
15], root of
Taraxacum officinale [
16], aqueous extract of
Coptidis rhizoma [
17], extract of
Pracparatum mungo (
Phaseolus mungo L.) [
18], sea buckthorn seed oil (
Hippophae rhamnoides L.,
Elaeagnaceae) [
19] and the alga
Laminaria japonica [
20] in treating hepatic disorders.
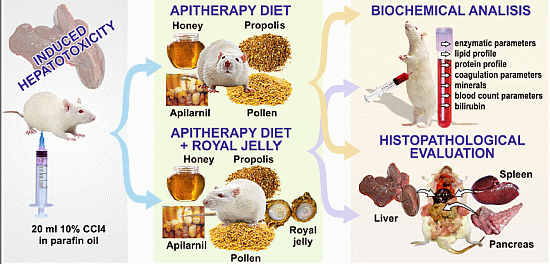
The present experiment follows the same direction of research, by investigating the protective effects of two apitherapy diet formulations containing a mixture of propolis, pollen, Apilarnil, and honey, with/without royal jelly, against carbon tetrachloride-induced toxicity in Wistar rats. The novelty value is given by the correlation established between the pathological changes in the enzymatic, lipid and protein profiles, the coagulation and blood count parameters, minerals, bilirubin, and the histopathological changes at the level of the liver, pancreas and spleen. The pancreatic and spleen tissues were included in our analysis because we intended to extend our assessment of the effect of CCl4 on these tissues, with the understanding that the toxic effects may also occur at other levels than the liver.
3. Discussion
The results of the present experiment are in agreement with results obtained in other experimental studies [
21,
22,
23,
24]. For example, the study of Cemek
et al.demonstrates that the administration of CCl
4 leads to severe acute liver affection in rats, revealed by significant increase of AST and ALT serum levels. The same study reveals that the treatment with RJ (included in the present experimental study in formulation II) remarkably counteracts the severe acute hepatic affection induced by CCl
4, which can be seen in the decrease of the serum activity of AST and ALT. Furthermore, the histopathological evaluation shows that the hepatic lesions induced by CCl
4 improve after treatment with RJ [
9].
Propolis, an ingredient in the two formulations administered in the present experiment, is known to improve the enzymatic profile. This result may be explained by the rapid regeneration of the parenchyma cells produced by the presence of the bioactive substances in propolis, such as flavonoids and their esters, mainly CAPE (caffeic acid phenethyl ester), which prevent the membranous fragility and subsequently decrease the levels of enzymatic markers in blood circulation [
25,
26].
The most important result obtained in the present experiment is the increase of HDL, following both the administration of formulation I and formulation II to laboratory animals with CCl4 induced toxicity. The positive effect of the administered apitherapy diet upon the lipid profile is based on the pharmacological action exerted by the ingredients in these preparations: honey, propolis, pollen, and RJ.
Thus, an experimental study regarding honey has demonstrated that the ingestion of this bee product has a protective effect upon the cardiovascular system by reducing blood cholesterol levels, LDL and triglycerides and by producing a slight increase of HDL levels [
27].
Propolis therapy has been demonstrated to reduce the high levels of triglycerides, total cholesterol and esters of cholesterol, probably through the antioxidant mechanism exerted by the flavones in propolis [
25]. The intensification of the oxidative stress enhances the influence of non-essential fatty acids which, in turn, increase the serum and tissue levels of cholesterol and triglycerides. It has been shown that antioxidants and flavonoids can act as inhibitors of lipid peroxidation by neutralizing the radicals of polyunsaturated fatty acids and by interrupting the chain reactions [
28]. Lipid peroxidation is an important biological consequence of cellular oxidative stress and is one of the main causes of hepatic lesions produced by CCl
4 mediated by the free radicals derived from this toxic substance [
3,
21,
29,
30,
31].
Regarding the association of RJ in modulating the lipid profile, it has been shown that RJ significantly reduces the values of cholesterol and triglycerides in rabbits [
27]. RJ has also demonstrated its physiological and biochemical cardioprotective effect in experiments on mice. It is not known, for the moment, the exact mechanism by which RJ exerts its strong anti-lipid peroxidation activity, but it is considered to be due to its antioxidant potential [
9].
The hepatic affection is characterized by the imbalance of the serum levels of total proteins, expressed by the deterioration of the albumin/globulin ratio. The decrease of the albumins level is accompanied by the increase of the globulins level, mainly of γ-globulins (less frequently of α-globulins or β-globulins). The improvement of the albumin/globulin ratio involves the increase of serum albumins, accompanied by a proportional decrease of the globulin values.
The deterioration of the albumin/globulin ratio is constantly accompanied by electrolytic damages, the most important of them being the imbalance of the phosphocalcium metabolism at the level of ionized calcium (hipocalcemia at the level of serum calcium is rarely met). The deterioration of the albumin/globulin ratio in favour of globulins is expressed by the decrease of albumin serum levels with the same amounts as the decrease of ionized calcium. The reduced albumin levels of are associated with reduced ionized calcium levels, even if the total serum calcium levels are normal. During the monitorization of the results after the apitherapy treatment, another recurring situation could be observed: the increase of the albumin levels and rebalance of the albumin/globulin ratio is clearly dependent on and accompanied by the reestablishment of the normal values of the ionized calcium. From the albumin-ionized calcium correlation, the following can be noted: the decrease or increase of the albumin levels are constantly correlated with the decrease or increase of the ionized calcium; the reduced values of albumin and ionized calcium concentrations are found at the beginning of liver damage.
Honey increases the
in vitro and
in vivo absorption of calcium in studies on laboratory animals [
27]. Propolis apparently increases the absorption and use of different minerals, due to the presence of the derivatives of the organic acids that improve physiological functions by regulating the enzyme-dependent ionic activity. Haro
et al. demonstrate the benefic effects of pollen and/or propolis on the metabolism of iron, calcium, phosphorus and magnesium in nutritional iron-deficiency anemia in rats [
32]. The optimal use of calcium by the organism cannot take place in the following conditions: avitaminosis (including some water-soluble vitamins), demineralization, excess of proteins and sugars, unbalanced diets through excessive intake of sodium, phosphorus, caffeine, foods containing too many vegetal fibers, oxalic acid, phytic acid, as well as the uncontrolled action of free radicals.
In the present experiment, it can be noticed that the administration of apiterapy diet (formulations I and II) leads to normal values of the serum iron and potassium levels, and also of the ionized calcium for the groups that have been given CCl
4. The biochemical mechanisms involved in acquiring CCl
4 hepatotoxicity are generated by the peroxidation of lipids produced by trichloromethyl radical (CCl
3). CCl
4 is metabolized by cytochrome P
450 to CCl
3 radical that induces the peroxidation of membrane lipids and disturbs Ca
2+ homeostasis thus inducing liver tissue damage [
33,
34]. Because the two apitherapy formulations have an effect on the normalization of calcium, it can be assumed that they also act on cytochrome P450. Further studies on lipid peroxidation and antioxidant systems will bring additional data into this matter. For now, we may safely assume that the two apitherapy formulations affect lipid peroxidation due to the antioxidant factors present in the composition of bee products and, not lastly, to the vitamin E in honey and pollen, since vitamin E is known to have an effect on lipid peroxidation in intoxication by CCl4 [
35].
As for the blood count parameters, the administration of the two apitherapy formulations to the groups with experimentally induced hepatopathy leads to the increase of WBC, the improvement of RBC, Hgb, and RDW values, the increase of HEM to normal levels, the increase of CHEM, the increase of thrombocytres, keeps MPV and PDW within normal range, brings neutrophils to normal values (expressed as % and 103/mcL), and decreases the eosinophils (expressed as % and 103/mcL).
The high values of total bilirubin for the group with CCl
4 intoxication is correlated with the decrease of RBC in the blood samples of the same group, as the increase of the RBC death is a possible source of bilirubin formation. In bilirubin levels, the administration of the two apitherapy formulations to the groups with experimental induced hepatopathy improves the bilirubin levels, that are albumin dependent. It is known that bilirubin, a hydrophobic and potentially toxic substance, circulates through the plasma bound to albumin [
36].Our results show the decrease in the total, direct and indirect bilirubin values in groups with CCl
4 induced hepatopathy that received the apitherapy treatment when compared to the animals with the same induced affection that received standard food only.
In our experiment, it has been shown that administration of CCl4 to Wistar rats produces important histological modifications at the level of the hepatic tissue, but less relevant histopathological changes at the level of spleen and pancreas.
4. Experimental Section
4.1. Animal Environment, Housing and Management
The experiment was performed on sixty adult male Wistar rats with a body weight of 220–250 g. The animals were kept in a diffusely lit and temperature-controlled room with a diurnal 12 h light cycle, where the temperature (22 ± 0.5 °C) and relative humidity (65%–70%) were kept constant. The animals were given free access to standard laboratory diet and water before the experiments.
All the experimental proceedings achieved on laboratory animals (Wistar rats) in this study were in agreement with the guidelines of animal bioethics from the Act on Animal Experimentation and Animal Health and Welfare Act from Romania and were in compliance with the European Council Directive of 24 November 1986 (86/609/EEC). The experiment was approved by the Ethics Commission of Grigore T. Popa University of Medicine and Pharmacy of Iasi.
4.2. Bee Products
Honey, propolis, Apilarnil, and pollen were provided by Stupina LLC, Balanesti, Gorj, while royal jelly was commercially acquired from the market (lyophilized royal jelly produced by The Beekeeping Research and Development Institute, Bucharest, Romania). Chemical composition of propolis: 45%–55% resins and balsams; 7.5% to 35% vegetable waxes and beeswax; 10%–15% volatile essential oils; 5% pollen; 5% phenolic acids and their esters, polyphenols, lignans, sesquiterpene, quinones, steroids and amino acids (pyroglutamic acid, arginine, proline). It also contains hydrosoluble vitamins: B1, B2, B3, B5 and C; liposoluble vitamins: A and E; and minerals Na, K, Mg, Ca, Ba, B, Cr, Zn, Se, Fe, Mn.
Chemical composition of pollen: 3.27%–34.62% reducing sugars (glucose, fructose, lactose, raffinose, stachiloza); 0.5%–20% non-reducing sugars; 1%–20% of fat to fatty acid esters, oleic, linoleic, linolinic acid, palmitic acid, stearic acid and arachidonic; amino acids (5.70% arginine, 2.4% histidine, 4.5% isoleucine, leucine 6.7%, 5.7% lysine, 1.8% methionine, phenylalanine 3.9%, 4% threonine, tryptophan, 1.3%, 5.7% valine); liposoluble vitamins (A, D, E, K); vitamins of group B and vitamin C, and minerals: K, Zn, Mg, Mn, C, Fe.
Chemical composition of Apilarnil: 65%–75% water; 25%–35% dry matter content; 12.9% protein; amino acids; carbohydrates: 6%–10% glucose, 3.16% fructose, 0.03% sucrose; 5%–8% lipids; liposoluble vitamins (A, D, E, K) and water-soluble vitamins (B group C); minerals, hormones, particularly somatotropic hormones and steroid hormones; antivirals; xantophylin; choline.
Chemical composition of honey: 0.4%–8% amino acids; 81.3% sugars of which: 7.5% sucrose; 38.19% fructose; 31.28% glucose; 6.89% maltase; other sugars; enzymes; traces of pollen and royal jelly; organic acids; lipids; antibiotic substances grouped under the heading “inhibin”; antigerminative factors; pigments; aromatic compounds, amino acids, minerals, vitamins-B1, B2, B3, B5, B6, B9, B12; vitamin C and liposoluble vitamins: provitamin A; vitamin D; vitamin E; vitamin K; honey enzymes: diastase, invertase, sucrase, zaharaza, catalase, α and β amylase, acid phosphatase, peroxidase, superoxide dismutase (SOD, superoxide oxidoreductase); α and β glucosidase, acetyltransferase, cytochrome oxidase, ascorbic acid oxidase, tyrosinase, monofenol monooxygenase, glycosyl transferases.
4.3. Study Design
The experimental model of hepatic lesion was induced by intraperitoneal (i.p.) injection of CCl4 (dissolved in paraffin oil, 10% solution). Two mL per 100 g were administered, every 2 days, for 2 weeks. Treatment was provided in the form of two formulations of the apitherapy diet:
formulation I, consisting of honey, propolis, Apilarnil, and pollen granules
formulation II that contains, besides the ingredients from formulation I, RJ
The two formulations were prepared daily. Formulation I was administered to groups II and V, in doses of 3.535 g/100 g body weight (bw), while formulation II was administered to groups III and VI, in doses of 3.635 g/100 g body weight (
Table 9).
Table 9.
The daily intake of the two apitherapy diet formulations calculated for 100 g bw.
Table 9.
The daily intake of the two apitherapy diet formulations calculated for 100 g bw.
| | Bee Products | Formulation I | Formulation II |
|---|
| 1. | Honey | 2.5 g | 2.5 g |
| 2. | Propolis | 0.01 g | 0.01 g |
| 3. | Apilarnil | 0.025 g | 0.025 g |
| 4. | Pollen | 1 g | 1 g |
| 5. | Royal jelly | - | 0.1 g |
| Total amount | 3.535 g | 3.635 g |
The Wistar rats were randomly divided into six groups of 10 animals each, as follows:
Group I (control group-standard food)-served as control, and was fed with standard food;
Group II (control group-apitherapy diet)-fed with apitherapy diet (formulation I–3.535 g/100 g bw/day, for 3 weeks);
Group III (control group-apitherapy diet + RJ)-fed with apitherapy diet and RJ (formulation II, 3.635 g/100 g bw/day, for 3 weeks);
Group IV (CCl4 group)-i.p. administration of 2 mL of 10% paraffin oil solution of CCl4 per 100 g, every 2 days, for 2 weeks, and fed with standard food;
Group V (group CCl4-apitherapy diet)-i.p. administration of 2 mL of 10% paraffin oil solution of CCl4 per 100 g, every 2 days, for 2 weeks, and fed with apitherapy diet (formulation I–3.535 g/100 g bw/day, for 3 weeks);
Group VI (group CCl4-apitherapy diet + RJ)-i.p. administration of 2 mL of 10% paraffin oil solution of CCl4 per 100 g, every 2 days, for 2 weeks, and fed with apitherapy diet and RJ (formulation II–3.635 g/100 g bw/day, for 3 weeks).
The amounts of the two apitherapy diet formulations calculated for each rat were added to a mixture of cereals (oat, barley, rye, and wheat). The diet was administered separately to each rat (housed in separate individual cages), twice a day: in the morning and at noon. In the end, after three weeks of apitherapy treatment, the animals were sacrificed.
4.4. Total Polyphenol and Flavonoids Content in the Studied Apitherapy Formulation
4.4.1. Determination of Total Polyphenol Content in the Studied Apitherapy Formulations
TPC was determined by spectrophotometry, with gallic acid as reference, according to the method described by the International Organization for Standardization (ISO) 14502-1 (ISO 14502-1: 2005). Briefly, 1.0 mL of hydroalcoholic/oil propolis extract was transferred to separate tubes containing 5.0 mL of 1/10 dilution of Folin-Ciocalteu reagent in water. Then, 4.0 mL sodium carbonate solution (7.5% w/v) was added. The tubes were then allowed to stand at room temperature for 60 min before absorbance at 765 nm was measured against water. The mean of three readings was used and the total phenolic content was expressed in mg of gallic acid (mg/100 g). The concentration of polyphenols in the samples was derived from a standard curve of gallic acid ranging from 10 to 200 μg/mL (Pearson’s correlation coefficient: r2 = 0.9998).
4.5. Biochemical Analysis
Anesthesia was achieved with thiopental (dose of 1 mL/100 g from a 0.01% thiopental solution), and blood samples were collected by the punction of the cord (i.e., the ventricular region) with a Vacuette® system (three types of vacutainers were used for collection, i.e., vacutainers without anticoagulant for general biochemical tests, vacutainers with Na citrate 0,105M for the samples in which the protein profile parameters were determined and vacutainers with EDTA anticoagulant for determining the blood count. Collected samples were processed by Synevo Laboratory (Iassy, România). Specific references of the procedure technology used 2010 Ref Type: Catalog; Laboratory Corporation of America, San Francisco, CA. Directory of Services and Interpretive Guide. Ref Type: Internet Communication).
Biochemical analysis and hematological tests:
enzymatic parameters (aspartate aminotransferase-AST, alanine aminotransferase-ALT, alkaline phosphatase-ALP, gamma-glutamyl transpeptidase-GGT),
lipid profile (total cholesterol-TC, triglyceride-TG, very low density lipoproteins–VLDL, high-density lipoproteins-HDL),
protein profile (total proteins-TP, albumin-ALB, globulins-GLO, alpha-1 globulins–ALPHA 1, alpha-2 globulins–ALPHA 2, beta globulins-BETA, gamma globulins–GAMMA, and albumin/globulin ratio–A/B),
coagulation parameters (Quick’s time-PT, thrombin time-TT, fibrinogen-F, International Normalized Ratio-INR),
minerals (iron-Fe, potassium-K, serum and ionized calcium–sCa and iCa),
blood count parameters (number of leukocytes–WBC, number of erythrocytes–RBC, hemoglobin–Hgb, hematocrit–HCT, mean corpuscular volume–MCV, mean corpuscular hemoglobin–MCH, mean corpuscular hemoglobin concentration–MCHC, red blood cell distribution width–RDW, number of neutrophils–Neu 103/mcL, percent of neutrophils–Neu%,number of eosinophils–Eo 103/mcL, percent of eosinophils–Eo%, number of lymphocytes–Ly 103/μL, percent of lymphocytes–Ly%, number of monocytes–Mo 103/mcL, percent of monocytes–Mo%)
bilirubin (total bilirubin-TB, direct bilirubin-DB, indirect bilirubin-IB).
The determination of the values of the investigated parameters was achieved with an automated analyzer (Aeroset, Abbott, Chicago, IL, USA) and commercial kits (Abbott).
4.6. Histopathological Examination
When the absence of vital signs (respiratory rate, heart rate, reflexes) was ascertained, the animals were dissected in order to collect the liver, spleen, and pancreas samples for the evaluation of the histopathological modifications.
The collected samples were fixed in 10% buffered formalin for at least 24 h, progressively dehydrated in solutions containing an increasing percentage of ethanol (60%, 80%, 90%, and 98%, v/v), clarified with amylic alcohol, embedded in paraffin under vacuum, sectioned at 5 μm thickness, deparaffinized, and stained with hematoxylin-eosin (HE).
4.7. Presentation of Results in Tables and Statistical Data
The statistical interpretation of the results was performed with One-Way ANOVA test and Tukey’s post-hoc test. The results were given as mean ± standard deviation. The value of p < 0.05 was considered significant.