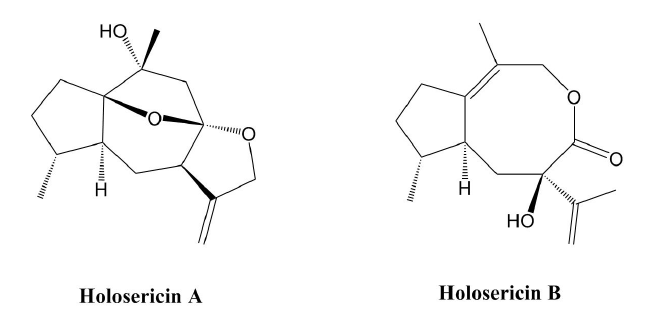
Two New Guaiane Sesquiterpenoids from Daphne holosericea (Diels) Hamaya
Abstract
:1. Introduction

2. Results and Discussion
| No. | 1 | 2 | ||
|---|---|---|---|---|
| δC | δH, J (Hz) | δC | δH, J (Hz) | |
| 1 | 93.4 | 157.0 | ||
| 2 | 32.1 | 1.74 m, 1.61 m (α-H) | 39.7 | 1.86 m (α-H), 1.69 m |
| 3 | 31.8 | 1.60 m, 1.45 m (α-H) | 32.4 | 1.85 m, 1.55 m (α-H) |
| 4 | 38.3 | 1.96 m | 36.0 | 2.25 m |
| 5 | 39.1 | 2.38 ddd (5.0, 8.5, 13.5) | 45.5 | 2.46 m |
| 6 | 24.9 | 1.82 m (α-H), 1.21 m | 19.5 | 2.40 dd (14.0, 9.5, α-H), 2.28 dd (14.0, 6.0) |
| 7 | 49.8 | 2.50 dd (10.5, 8.0) | 84.7 | |
| 8 | 110.2 | 176.5 | ||
| 9 | 50.6 | 2.22 d (13.6), 2.03 d (13.6, α-H) | 72.8 | 4.56 s |
| 10 | 76.1 | 126.9 | ||
| 11 | 152.1 | 149.9 | ||
| 12 | 72.2 | 4.48 d (13.0, α-H), 4.56 d (13.0) | 19.6 | 1.67 s |
| 13 | 104.6 | 4.95 d (2.4), 4.93 d (2.4) | 109.3 | 4.90 s, 4.72 s |
| 14 | 14.6 | 0.96 d (6.9) | 17.5 | 1.06 d (7.5) |
| 15 | 26.5 | 1.38 s | 12.5 | 2.02 s |


3. Experimental Section
3.1. General Information
3.2. Plant Material
3.3. Extraction and the Isolation
 + 6.7 (c 0.5, MeOH); UV (MeOH) λmax (logε) 203 (3.26); IR (KBr) νmax 3417, 2924, 2852, 1597, 1420, 1083, 1032; 1H- and 13C-NMR data see Table 1; positive ESI-MS m/z [M+Na]+ 273 (100); HR-EI-MS m/z [M]+ 250.1567 (calcd. for C15H22O3, 150.1569).
+ 6.7 (c 0.5, MeOH); UV (MeOH) λmax (logε) 203 (3.26); IR (KBr) νmax 3417, 2924, 2852, 1597, 1420, 1083, 1032; 1H- and 13C-NMR data see Table 1; positive ESI-MS m/z [M+Na]+ 273 (100); HR-EI-MS m/z [M]+ 250.1567 (calcd. for C15H22O3, 150.1569). + 5.2 (c 0.5, MeOH); UV (MeOH) λmax (logε) 217 (3.76), 204 (3.73); IR (KBr) νmax 3398, 2922, 2852, 1643, 1597, 1419, 1042; 1H- and 13C-NMR data see Table 1; positive ESI-MS m/z [M+Na]+ 273 (60); HR-EI-MS m/z [M]+ 250.1575 (calcd. for C15H22O3, 150.1569).
+ 5.2 (c 0.5, MeOH); UV (MeOH) λmax (logε) 217 (3.76), 204 (3.73); IR (KBr) νmax 3398, 2922, 2852, 1643, 1597, 1419, 1042; 1H- and 13C-NMR data see Table 1; positive ESI-MS m/z [M+Na]+ 273 (60); HR-EI-MS m/z [M]+ 250.1575 (calcd. for C15H22O3, 150.1569).3.4. Bioassay of AChE Inhibitory Activity
4. Conclusions
Supplementary Materials
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kunming Institute of Botany, Chinese Academy of Sciences. Flora of Yunnan; Science Press: Kunming, China, 1997; p. 219. [Google Scholar]
- Huang, S.Z.; Li, X.N.; Ma, Q.Y.; Dai, H.F.; Li, L.C.; Cai, X.H.; Liu, Y.Q.; Zhou, J.; Zhao, Y.X. Daphnauranols A-C, new antifeedant sesquiterpenoids with a 5/6/7 ring system from Daphne aurantiaca. Tetrahedron Lett. 2014, 55, 3693–3696. [Google Scholar] [CrossRef]
- Li, L.Z.; Gao, P.Y.; Peng, Y.; Wang, L.H.; Yang, J.Y.; Wu, C.F.; Zhang, Y.; Song, S.J. Daphnane-type diterpenoids from the flower buds of Daphne genkwa. Helv. Chim. Acta 2010, 93, 1172–1179. [Google Scholar] [CrossRef]
- Bang, K.K.; Yun, C.Y.; Lee, C.; Jin, Q.H.; Lee, J.W.; Jung, S.H.; Lee, D.; Lee, M.K.; Hong, J.T.; Kim, Y.; et al. Melanogenesis inhibitory daphnane diterpenoids from the flower buds of Daphne genkwa. Bioorg. Med. Chem. Lett. 2013, 23, 3334–3337. [Google Scholar] [CrossRef]
- Zhao, Y.X.; Huang, S.Z.; Ma, Q.Y.; Mei, W.L.; Dai, H.F. Two new daucane sesquiterpenoids from Daphne aurantiaca. Molecules 2012, 17, 10046–10051. [Google Scholar] [CrossRef]
- Liang, S.; Shen, Y.H.; Feng, Y.; Tian, J.M.; Liu, X.H.; Xiong, Z.; Zhang, W.D. Terpenoids from Daphne aurantiaca and their potential anti-inflammatory activity. J. Nat. Prod. 2010, 73, 532–535. [Google Scholar] [CrossRef]
- Huang, S.Z.; Zhang, X.J.; Li, X.Y.; Kong, L.M.; Jiang, H.Z.; Ma, Q.Y.; Liu, Y.Q.; Hu, J.M.; Zheng, Y.T.; Li, Y.; et al. Daphnane-type diterpene esters with cytotoxic and anti-HIV-1 activities from Daphne acutiloba Rehd. Phytochemistry 2012, 75, 99–107. [Google Scholar] [CrossRef]
- Xu, W.C.; Shen, J.G.; Jiang, J.Q. Phytochemical and biological studies of the plants from the genus Daphne. Chem. Biodivers. 2011, 8, 1215–1233. [Google Scholar] [CrossRef]
- Huang, S.Z.; Zhang, X.J.; Li, X.Y.; Jiang, H.Z.; Ma, Q.Y.; Wang, P.C.; Liu, Y.Q.; Hu, J.M.; Zheng, Y.T.; Zhou, J.; et al. Phenols with anti-HIV activity from Daphne acutiloba. Planta Med. 2012, 78, 182–185. [Google Scholar] [CrossRef]
- Editorial committee of flora of China, Chinese Academy of Sciences. Flora of China; Science Press: Beijing, China, 1999; p. 344. [Google Scholar]
- Chen, Y.Q.; Su, J.; Shen, Y.H.; Zhang, W.; Liang, S.; Zhang, W.D.; Kong, L.Y. Flavonoids from Daphne holosericea. Chem. Nat. Comp. 2009, 45, 542–545. [Google Scholar] [CrossRef]
- Chen, Y.Q.; Su, J.; Shen, Y.H.; Zhang, W.; Hu, X.J.; Xu, W.Z.; Zhang, W.D.; Kong, L.Y. Studies on chemical constituents of Daphne holosericea (Diels) Hamaya. Chin. Pharm. J. 2008, 19, 1453–1456. [Google Scholar]
- Li, X.W.; Weng, L.; Gao, X.; Zhao, Y.; Pang, F.; Liu, J.H.; Zhang, H.F.; Hu, J.F. Antiproliferative and apoptotic sesquiterpene lactones from Carpesium faberi. Bioorg. Med. Chem. Lett. 2011, 21, 366–372. [Google Scholar] [CrossRef]
- Zhang, S.S.; Ma, Q.Y.; Zou, X.S.; Dai, H.F.; Huang, S.Z.; Luo, Y.; Yu, Z.F.; Luo, H.R.; Zhao, Y.X. Chemical constituents and their in vitro acetylcholinesterase inhibitory activities from the fungus Amauroderma amoiensis. Planta Med. 2013, 79, 87–91. [Google Scholar]
- Adewusi, E.A.; Fouche, G.; Steenkamp, V. Cytotoxicity and acetylcholinesterase inhibitory activity of an isolated crinine alkaloid from Boophane disticha (Amaryllidaceae). J. Ethnopharmacol. 2012, 143, 572–578. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ma, Q.-Y.; Chen, Y.-C.; Huang, S.-Z.; Guo, Z.-K.; Dai, H.-F.; Hua, Y.; Zhao, Y.-X. Two New Guaiane Sesquiterpenoids from Daphne holosericea (Diels) Hamaya. Molecules 2014, 19, 14266-14272. https://doi.org/10.3390/molecules190914266
Ma Q-Y, Chen Y-C, Huang S-Z, Guo Z-K, Dai H-F, Hua Y, Zhao Y-X. Two New Guaiane Sesquiterpenoids from Daphne holosericea (Diels) Hamaya. Molecules. 2014; 19(9):14266-14272. https://doi.org/10.3390/molecules190914266
Chicago/Turabian StyleMa, Qing-Yun, Yi-Chun Chen, Sheng-Zhuo Huang, Zhi-Kai Guo, Hao-Fu Dai, Yan Hua, and You-Xing Zhao. 2014. "Two New Guaiane Sesquiterpenoids from Daphne holosericea (Diels) Hamaya" Molecules 19, no. 9: 14266-14272. https://doi.org/10.3390/molecules190914266