Synthesis of a Functionalized Benzofuran as a Synthon for Salvianolic Acid C Analogues as Potential LDL Antioxidants
Abstract
:1. Introduction




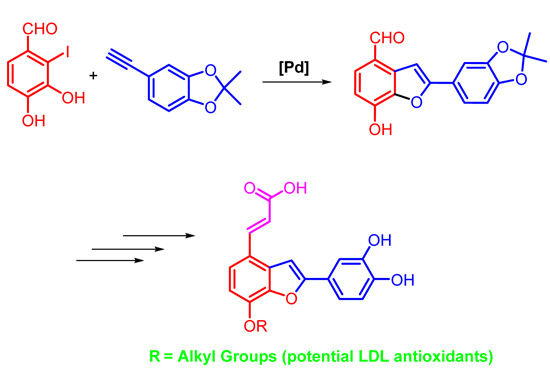
2. Results and Discussion




3. Experimental Section
3.1. General Methods
3.2. Synthesis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Li, H.; Horke, S.; Förstermann, U. Oxidative stress in vascular disease and its pharmacological prevention. Trends Pharmacol. Sci. 2013, 34, 313–319. [Google Scholar] [CrossRef] [PubMed]
- Pop-Busui, R.; Mehta, M.; Pennathur, S. Oxidative Stress and Cardiovascular Disease in Diabetes. In Studies in Diabetes. Oxidative Stress in Applied Basic Research and Clinical Practice; Obrosova, I., Stevens, M.J., Yorek, M.A., Eds.; Springer: New York, NY, USA, 2014; pp. 189–235. [Google Scholar]
- Dokken, B.B. The Pathophysiology of Cardiovascular Disease and Diabetes: Beyond Blood Pressure and Lipids. Diabetes Spectrum. 2008, 21, 160–165. [Google Scholar] [CrossRef]
- Garrett, A.R.; Gupta-Elera, G.; Keller, M.A.; O’Neill, K.L. Bioactive Foods in Aging: The Role in Cancer Prevention and Treatment. In Bioactive Food as Dietary Interventions for the Aging Population; Watson, R.R., Preedy, V.R., Eds.; Academic Press: San Diego, CA, USA, 2013; pp. 33–45. [Google Scholar]
- Zamora, R.; Delgado, R.M.; Hidalgo, F.J. Effects of Oxidation on the Nutritive and Health-Promoting Value of Food Components. In Food Oxidants and Antioxidants: Chemical, Biological, and Functional Properties; CRC Press: Boca Raton, FL, USA, 2013; pp. 197–234. [Google Scholar]
- González-Vallinas, M.; González-Castejón, M.; Rodríguez-Casado, A.; Ramírez de Molina, A. Dietary phytochemicals in cancer prevention and therapy: A complementary approach with promising perspectives. Nutr. Rev. 2013, 71, 585–599. [Google Scholar] [CrossRef] [PubMed]
- Rubió, L.; Motilva, M.J.; Romero, M.P. Recent Advances in Biologically Active Compounds in Herbs and Spices: A Review of the Most Effective Antioxidant and Anti-Inflammatory Active Principles. Crit. Rev. Food Sci. Nutr. 2013, 53, 943–953. [Google Scholar] [CrossRef] [PubMed]
- Martin, C.; Zhang, Y.; Tonelli, C.; Petroni, K. Plants, Diet, and Health. Annu. Rev. Plant Biol. 2013, 64, 19–46. [Google Scholar] [CrossRef] [PubMed]
- Murphy, M.P. Antioxidants as therapies: can we improve on nature? Free Radic. Biol. Med. 2014, 66, 20–23. [Google Scholar] [CrossRef] [PubMed]
- Hao, H.; Zheng, X.; Wang, G. Insights into drug discovery from natural medicines using reverse pharmacokinetics. Trends Pharmacol. Sci. 2014, 35, 168–177. [Google Scholar] [CrossRef] [PubMed]
- Velderrain-Rodríguez, G.R.; Palafox-Carlos, H.; Wall-Medrano, A.; Ayala-Zavala, J.F.; Chen, C.Y.O.; Robles-Sánchez, M.; Astiazaran-García, H.; Alvarez-Parrillab, E.; González-Aguilar, G.A. Phenolic compounds: Their journey after intake. Food Funct. 2014, 5, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Ding, W.X.; Gu, J.Y.; Cao, L.; Li, N.; Ding, G.; Wang, Z.; Chen, L.; Xu, X.J.; Xiao, W. Traditional Chinese herbs as chemical resource library for drug discovery of anti-infective and anti-inflammatory. J. Ethnopharm. 2014, 155, 589–598. [Google Scholar] [CrossRef]
- Sucher, N.J. The application of Chinese medicine to novel drug discovery. Expert Opin. Drug Discov. 2013, 8, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Yeap Foo, L. Polyphenolics of Salvia—A review. Phytochemistry 2002, 59, 117–140. [Google Scholar] [CrossRef] [PubMed]
- Kintzios, S.E. Sage: The Genus Salvia; Harwood Academic Publishers: Amsterdam, NY, USA, 2002. [Google Scholar]
- Wang, M.; Li, J.; Rangarajan, M.; Shao, Y.; La Voie, E.J.; Huang, T.C.; Ho, C.T. Antioxidative phenolic compounds from sage (Salvia officinalis). J. Agric. Food Chem. 1998, 46, 4869–4873. [Google Scholar] [CrossRef]
- Li, L.N.; Tan, R.; Chen, W.M. Salvianolic Acid A, a New Depside from Roots of Salvia miltiorrhiza. Planta Med. 1984, 50, 227–228. [Google Scholar] [CrossRef] [PubMed]
- Ai, C.B.; Li, L.N. Stereostructure of salvianolic acid B and isolation of salvianolic acid C from Salvia miltiorrhiza. J. Nat. Prod. 1988, 51, 145–149. [Google Scholar] [CrossRef]
- Lin, Y.L.; Chang, Y.Y.; Kuo, Y.H.; Liu, Y.H.; Shiao, M.S. Anti-Lipid Peroxidative Principles from Tournefortia sarmentosa. J. Nat. Prod. 2002, 65, 745–747. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, J.T. Hydroxyl radical scavenging effect of salvianolic acids. J. Chin. Pharm. Sci. 1994, 3, 43–50. [Google Scholar]
- Li, D.Y.; Xu, L.N.; Liu, X.G. Effects of water-soluble components isolated from Salvia miltiorrhiza on oxygen free radical generation and lipid peroxidation. J. Chin. Pharm. Sci. 1995, 4, 107–112. [Google Scholar]
- Zhang, Y.; Akao, T.; Nakamura, N.; Duan, C.L.; Hattori, M.; Yang, X.W.; Liu, J.X. Extremely Low Bioavailability of Magnesium Lithospermate B, An Active Component from Salvia miltiorrhiza, in Rat. Planta Med. 2004, 70, 138–142. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Akao, T.; Nakamura, N.; Hattori, M.; Yang, X.W.; Duan, C.L.; Liu, J.X. Magnesium lithospermate B is excreted rapidly into rat bile mostly as methylated metabolites, which are potent antioxidants. Drug Metab. Dispos. 2004, 32, 52–57. [Google Scholar]
- Zhang, X.; Song, Z.; Xu, J.; Ma, Z. Improving the NQO1-Inducing Activities of Phenolic Acids from Radix Salvia miltiorrhiza: A Methylation Strategy. Chem. Biol. Drug Des. 2011, 78, 558–566. [Google Scholar] [CrossRef] [PubMed]
- Alford, B.L.; Hügel, H.M. Total synthesis of (+)-pentamethylsalvianolic acid C. Org. Biomol. Chem. 2013, 11, 2724–2727. [Google Scholar] [CrossRef] [PubMed]
- Visioli, F.; Poli, A.; Gall, C. Antioxidant and Other Biological Activities of Phenols from Olives and Olive Oil. Med. Res. Rev. 2002, 22, 65–75. [Google Scholar] [CrossRef] [PubMed]
- Foti, M.C. Antioxidant Properies of Phenols. J. Pharm. Pharmacol. 2007, 59, 1673–1685. [Google Scholar] [CrossRef] [PubMed]
- Roy, K.; Mitra, I. Advances in quantitative structure–activity relationship models of antioxidants. Exp. Opin. Drug Discov. 2009, 4, 1157–1175. [Google Scholar] [CrossRef]
- Razzaghi-Asl, N.; Garrido, J.; Khazraei, H.; Borges, F.; Firuzi, O. Antioxidant Properties of Hydroxycinnamic Acids: A Review of Structure-Activity Relationships. Curr. Med. Chem. 2013, 20, 4436–4450. [Google Scholar] [CrossRef] [PubMed]
- Menezes, J.C.; Kamat, S.P.; Cavaleiro, J.A.; Gaspar, A.; Garrido, J.; Borges, F. Synthesis and antioxidant activity of long chain alkyl hydroxycinnamates. Eur. J. Med. Chem. 2011, 46, 773–777. [Google Scholar] [CrossRef] [PubMed]
- Roleira, F.M.; Siquet, C.; Orru, E.; Garrido, E.M.; Garrido, J.; Milhazes, N.; Podda, G.; Paiva-Martins, F.; Reis, S.; Carvalho, R.A.; et al. Lipophilic phenolic antioxidants: Correlation between antioxidant profile, partition coefficients and redox properties. Bioorg. Med. Chem. 2010, 18, 5816–5825. [Google Scholar] [CrossRef] [PubMed]
- Garrido, J.; Gaspar, A.; Garrido, E.M.; Miri, R.; Tavakkoli, M.; Pourali, S.; Saso, L.; Borges, F.; Firuzi, O. Alkyl esters of hydroxycinnamic acids with improved antioxidant activity and lipophilicity protect PC12 cells against oxidative stress. Biochimie 2012, 94, 961–967. [Google Scholar] [CrossRef] [PubMed]
- Dawood, K.M. Benzofuran derivatives: A patent Review. Exp. Opin. Ther. Pat. 2013, 23, 133–156. [Google Scholar] [CrossRef]
- Shen, S.D.; Zhang, G.P.; Leib, M.; Hu, LH. First total synthesis of salvianolic acid C, tournefolic acid A, and tournefolal. ARKIVOC 2011, 6, 204–213. [Google Scholar]
- Motoki, I.; Ryosuke, R.; Janagiraman, N.; Hitomi, M.; Koji, T.; Hisanori, N.; Shunichi, H. Asymmetric Total Synthesis of (−)-trans-Blechnic Acid via Rhodium(II)-Catalyzed C–H Insertion and Palladium(II)-Catalyzed C–H Olefination Reactions. Synlett 2014, 25, 288–292. [Google Scholar]
- Kwiecien, H.; Smist, M.; Kowalewska, M. Recent Development on the Synthesis of Benzo[b]- and Naphtho[b]furans: A Review. Curr. Org. Synth. 2012, 9, 529–560. [Google Scholar] [CrossRef]
- De Luca, L.; Nieddu, G.; Porcheddu, A.; Giacomelli, G. Some Recent Approaches to the Synthesis of 2-Substituted Benzofurans. Curr. Med. Chem. 2009, 16, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, A.V.; Svinareva, P.A.; Tomilova, L.G.; Zefirov, N.S. Synthesis of 4,5-dihydroxyphthalonitrile. Russ. Chem. Bull. 2001, 50, 919–920. [Google Scholar] [CrossRef]
- Mitchell, R.H.; Lai, Y.H.; Williams, R.V. N-Bromosuccinimide-dimethylformamide: A mild, selective nuclear monobromination reagent for reactive aromatic compounds. J. Org. Chem. 1979, 44, 4733–4735. [Google Scholar] [CrossRef]
- Zysman-Colman, E.; Arias, K.; Siegel, J.S. Synthesis of arylbromides from arenes and N-bromosuccinimide (NBS) in acetonitrile-A convenient method for aromatic bromination. Can. J. Chem. 2009, 87, 440–447. [Google Scholar] [CrossRef]
- Sonogashira, K. Palladium-Catalyzed Alkynylation. In Handbook of Organopalladium Chemistry for Organic Synthesis; Negishi, E.I., Ed.; Wiley: New York, NY, USA, 2002; pp. 493–529. [Google Scholar]
- Bernini, R.; Cacchi, S.; Fabrizi, G.; Filisti, E. 2-Arylhydroxytyrosol Derivatives via Suzuki-Miyaura Cross-Coupling. Org. Lett. 2008, 10, 3457–3460. [Google Scholar] [CrossRef] [PubMed]
- Thorand, S.; Krause, N. Improved Procedures for the Palladium-Catalyzed Coupling of terminal Alkynes with Aryl Bromides (Sonogashira Coupling). J. Org. Chem. 1998, 63, 8551–8553. [Google Scholar] [CrossRef]
- Jung, K.Y.; Vanommeslaeghe, K.; Lanning, M.E.; Yap, J.L.; Gordon, C.; Wilder, P.T.; MacKerrell, A.D.; Fletcher, S. Amphipathic α-Helix Mimetics Based on a 1,2-Diphenylacetylene Scaffold. Org. Lett. 2013, 15, 3234–3237. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Not available.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
López-Frías, G.; Camacho-Dávila, A.A.; Chávez-Flores, D.; Zaragoza-Galán, G.; Ramos-Sánchez, V.H. Synthesis of a Functionalized Benzofuran as a Synthon for Salvianolic Acid C Analogues as Potential LDL Antioxidants. Molecules 2015, 20, 8654-8665. https://doi.org/10.3390/molecules20058654
López-Frías G, Camacho-Dávila AA, Chávez-Flores D, Zaragoza-Galán G, Ramos-Sánchez VH. Synthesis of a Functionalized Benzofuran as a Synthon for Salvianolic Acid C Analogues as Potential LDL Antioxidants. Molecules. 2015; 20(5):8654-8665. https://doi.org/10.3390/molecules20058654
Chicago/Turabian StyleLópez-Frías, Gabriela, Alejandro A. Camacho-Dávila, David Chávez-Flores, Gerardo Zaragoza-Galán, and Víctor H. Ramos-Sánchez. 2015. "Synthesis of a Functionalized Benzofuran as a Synthon for Salvianolic Acid C Analogues as Potential LDL Antioxidants" Molecules 20, no. 5: 8654-8665. https://doi.org/10.3390/molecules20058654