Biological Activity and Molecular Structures of Bis(benzimidazole) and Trithiocyanurate Complexes
Abstract
:1. Introduction

2. Results and Discussion
2.1. Synthesis and Characterization
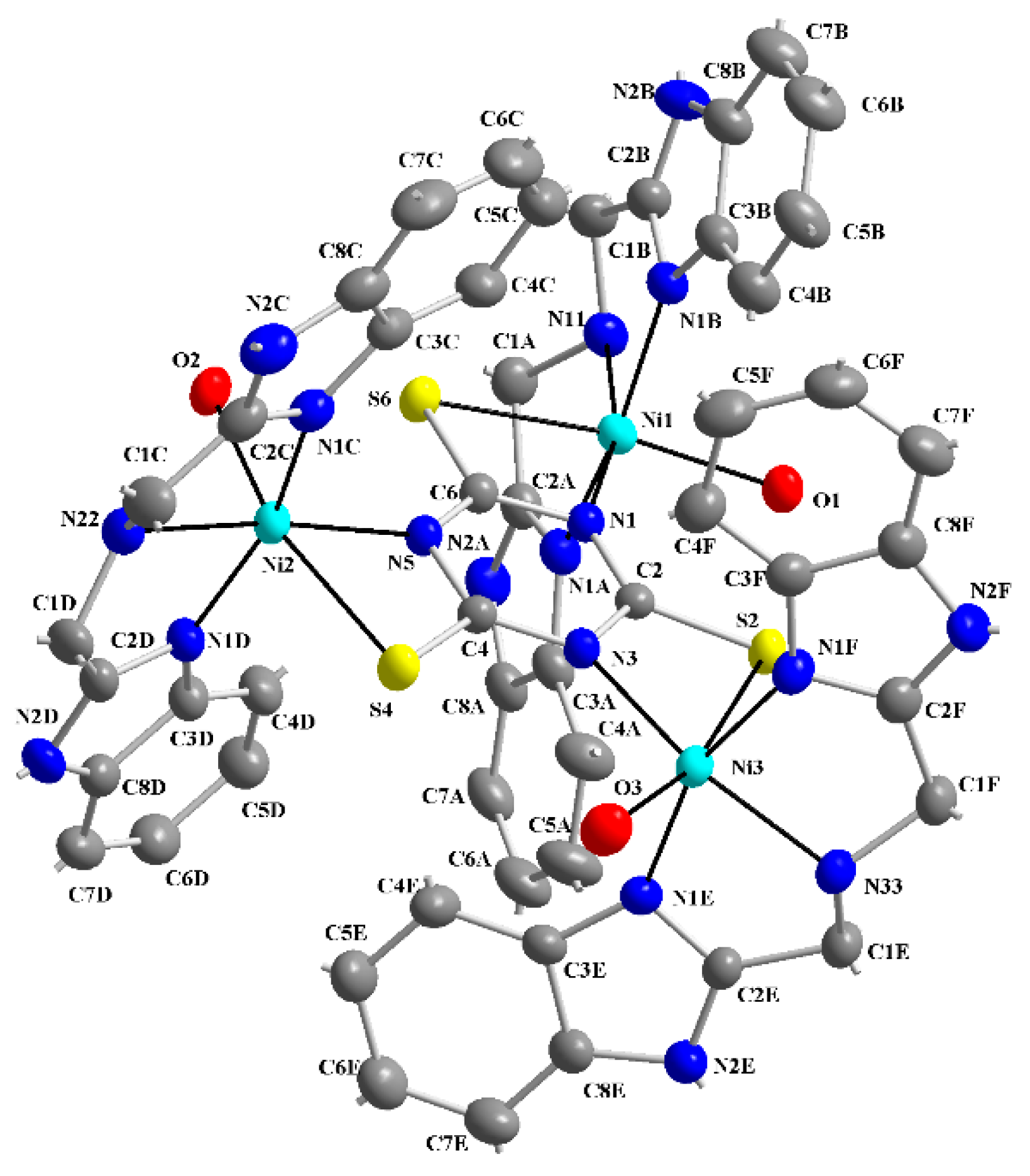
2.2. X-ray Structures of the Complexes [Ni3(abb)3(H2O)3(μ-ttc)](ClO4)3·3H2O·EtOH (1) and [(tbbH2)(ttcH2)2(ttcH3)(H2O)] (2)
| Ni1-N1B | 2.041(3) | Ni2-N5 | 2.030(3) | Ni3-N1F | 2.044(3) |
| Ni1-N1 | 2.047(3) | Ni2-N1C | 2.056(4) | Ni3-N3 | 2.043(3) |
| Ni1-N1A | 2.057(3) | Ni2-N1D | 2.062(4) | Ni3-N1E | 2.057(3) |
| Ni1-O1 | 2.102(3) | Ni2-N22 | 2.101(4) | Ni3-O3 | 2.100(3) |
| Ni1-N11 | 2.121(3) | Ni2-O2 | 2.109(3) | Ni3-N33 | 2.107(3) |
| N1B-Ni1-N1 | 101.50(14) | N5-Ni2-N1C | 99.59(14) | N1F-Ni3-N3 | 100.24(13) |
| N1B-Ni1-N1A | 159.97(15) | N5-Ni2-N1D | 98.94(13) | N1F-Ni3-N1E | 160.83(14) |
| N1-Ni1-N1A | 97.76(13) | N1C-Ni2-N1D | 161.06(14) | N3-Ni3-N1E | 97.92(13) |
| N1B-Ni1-O1 | 92.65(13) | N5-Ni2-N22 | 170.79(14) | N1F-Ni3-O3 | 90.79(14) |
| N1-Ni1-O1 | 97.07(12) | N1C-Ni2-N22 | 80.76(15) | N3-Ni3-O3 | 98.86(13) |
| N1A-Ni1-O1 | 90.41(13) | N1D-Ni2-N22 | 80.33(14) | N1E-Ni3-O3 | 92.42(14) |
| N1B-Ni1-N11 | 80.27(14) | N5-Ni2-O2 | 96.53(12) | N1F-Ni3-N33 | 79.92(14) |
| N1-Ni1-N11 | 165.69(14) | N1C-Ni2-O2 | 90.31(13) | N3-Ni3-N33 | 163.52(14) |
| N1A-Ni1-N11 | 79.71(14) | N1D-Ni2-O2 | 91.45(13) | N1E-Ni3-N33 | 80.92(14) |
| O1-Ni1-N11 | 97.03(13) | N22-Ni2-O2 | 92.67(14) | O3-Ni3-N33 | 97.62(13) |
| N1B-Ni1-S6 | 89.81(10) | N5-Ni2-S4 | 67.54(9) | N1F-Ni3-S2 | 88.42(11) |
| N1-Ni1-S6 | 67.36(9) | N1C-Ni2-S4 | 89.22(10) | N3-Ni3-S2 | 67.60(9) |
| N1A-Ni1-S6 | 92.54(10) | N1D-Ni2-S4 | 94.26(10) | N1E-Ni3-S2 | 92.90(10) |
| O1-Ni1-S6 | 164.40(9) | N22-Ni2-S4 | 103.30(11) | O3-Ni3-S2 | 166.03(10) |
| N11-Ni1-S6 | 98.56(10) | O2-Ni2-S4 | 163.73(9) | N33-Ni3-S2 | 95.98(10) |



| S1-C1A | 1.810(4) | S1C-C1C | 1.666(4) | C8A-C3A-N1A | 106.9(3) |
| S1-C1B | 1.828(4) | S2C-C2C | 1.635(4) | N2A-C8A-C3A | 105.9(3) |
| C1A-C2A | 1.490(5) | S3C-C3C | 1.648(4) | C2B-C1B-S1 | 111.4(3) |
| N1A-C2A | 1.332(5) | S1D-C1D | 1.656(4) | C2B-N1B-C3B | 109.4(3) |
| N1A-C3A | 1.405(5) | S2D-C2D | 1.647(4) | C2B-N2B-C8B | 109.8(3) |
| N2A-C2A | 1.335(5) | S3D-C3D | 1.681(4) | N2B-C2B-N1B | 109.0(3) |
| N2A-C8A | 1.389(5) | S1E-C1E | 1.675(4) | N1B-C3B-C8B | 105.9(3) |
| C3A-C8A | 1.404(5) | S2E-C2E | 1.692(4) | N1C-C1C-N3C | 115.0(3) |
| C1B-C2B | 1.485(5) | S3E-C3E | 1.645(4) | N2C-C2C-N1C | 113.5(3) |
| N1B-C2B | 1.331(5) | C1A-S1-C1B | 101.20(18) | N2C-C3C-N3C | 114.1(3) |
| N1B-C3B | 1.397(5) | C2A-C1A-S1 | 115.2(3) | N1D-C2D-N2D | 113.5(3) |
| N2B-C2B | 1.324(5) | C2A-N1A-C3A | 107.7(3) | N2E-C3E-N3E | 113.4(3) |
| N2B-C8B | 1.392(5) | C2A-N2A-C8A | 109.1(3) | ||
| C3B-C8B | 1.397(5) | N1A-C2A-N2A | 110.4(3) |
2.3. Biological Activity

| Compounds | IC50 (μg/mL) Measurement after 24 h | |||||
|---|---|---|---|---|---|---|
| S. aureus | E. coli | S. cerevisiae | S. agalactiae | C. striatum | S. marcescens | |
| Ligand abb | 5.6 | 19 | 21.4 | 22.4 | 30.8 | 26.7 |
| Ligand tbb | 485.2 | 182.3 | 204.7 | 126.1 | 112.5 | 122.6 |
| Ligand ttcNa3 | 625 | 636.8 | 86.4 | 119.5 | 115.8 | 123.9 |
| Complex 1 | 4.8 | 23.7 | 15.4 | 21.4 | 24.3 | 27.2 |
| Complex 2 | 562.3 | 257.6 | 64.3 | 102.1 | 119.5 | 61.5 |
| Complex 3 | 1036.7 | 12.6 | 18.2 | 113.4 | 124.9 | 110.4 |
3. Experimental Section
3.1. General Information
3.2. Preparation of the Compounds
3.2.1. 1-(1H-Benzimidazol-2-yl)-N-(1H-benzimidazol-2-ylmethyl)methanamine (abb)
3.2.2. 2-(1H-Benzimidazol-2-ylmethylsulfanylmethyl)-1H-benzimidazole (tbb)
3.2.3. [Ni3(abb)3(H2O)3(μ-ttc)](ClO4)3·3H2O·EtOH (1)
3.2.4. [(tbbH2)(ttcH2)2(ttcH3)(H2O)] (2)
3.2.5. [Ni3(pmdien)3(μ-ttc)](ClO4)3 (3)
3.3. X-ray Crystallography
3.4. Biological Activity Testing
3.4.1. Cultivation of Commercially Supplied Bacteria and Yeasts
3.4.2. Preparation of Hospital Samples and Their Cultivation
Cohort of Patients with Bacterial Infections
Collection of Wound Swabs from Patients with Bacterial Infections
Cultivation of Clinical Specimens
3.4.3. Determination of Growth Curves
3.4.4. Statistical Analyses
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Giovine, A.; Muraglia, M.; Florio, M.A.; Rosato, A.; Corbo, F.; Franchini, C.; Degennaro, L.; Musio, B.; Luisi, R. Synthesis of Functionalized Arylaziridines as Potential Antimicrobial Agents. Molecules 2014, 19, 11505–11519. [Google Scholar] [CrossRef] [PubMed]
- Ranjith, P.K.; Rajeesh, P.; Haridas, K.R.; Susanta, N.K.; Row, T.N.G.; Rishikesan, R.; Kumari, N.S. Design and synthesis of positional isomers of 5 and 6-bromo-1-(phenyl)sulfonyl-2-(4-nitrophenoxy)methyl-1H-benzimidazoles as possible antimicrobial and antitubercular agents. Bioorg. Med. Chem. Lett. 2013, 23, 5228–5234. [Google Scholar] [CrossRef] [PubMed]
- Toro, P.; Klahn, A.H.; Pradines, B.; Lahoz, F.; Pascual, A.; Biot, C.; Arancibia, R. Organometallic benzimidazoles: Synthesis, characterization and antimalarial activity. Inorg. Chem. Commun. 2013, 35, 126–129. [Google Scholar] [CrossRef]
- Sondhi, S.M.; Rajvanshi, S.; Johar, M.; Bharti, N.; Azam, A.; Singh, A.K. Anti-inflammatory, analgesic and antiamoebic activity evaluation of pyrimido 1,6-alpha benzimidazole derivatives synthesized by the reaction of ketoisothiocyanates with mono and diamines. Eur. J. Med. Chem. 2002, 37, 835–843. [Google Scholar] [CrossRef]
- Grassmann, S.; Sadek, B.; Ligneau, X.; Elz, S.; Ganellin, C.R.; Arrang, J.M.; Schwartz, J.C.; Stark, H.; Schunack, W. Progress in the proxifan class: Heterocyclic congeners as novel potent and selective histamine H3-receptor antagonists. Eur. J. Pharm. Sci. 2002, 15, 367–378. [Google Scholar] [CrossRef]
- Haque, R.A.; Iqbal, M.A.; Asekunowo, P.; Majid, A.; Ahamed, M.B.K.; Umar, M.I.; Al-Rawi, S.S.; Al-Suede, F.S.R. Synthesis, structure, anticancer, and antioxidant activity of para-xylyl linked bis-benzimidazolium salts and respective dinuclear Ag(I) N-heterocyclic carbene complexes (Part-II). Med. Chem. Res. 2013, 22, 4663–4676. [Google Scholar] [CrossRef]
- Nile, S.H.; Kumar, B.; Park, S.W. In Vitro Evaluation of Selected Benzimidazole Derivatives as an Antioxidant and Xanthine Oxidase Inhibitors. Chem. Biol. Drug Des. 2013, 82, 290–295. [Google Scholar] [CrossRef] [PubMed]
- Kralova, V.; Hanusova, V.; Stankova, P.; Knoppova, K.; Canova, K.; Skalova, L. Antiproliferative effect of benzimidazole anthelmintics albendazole, ricobendazole, and flubendazole in intestinal cancer cell lines. Anti-Cancer Drugs 2013, 24, 911–919. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.; Sharma, R.; Chaturvedi, S.C. A rational design, synthesis, characterization, and antihypertensive activities of some new substituted benzimidazoles. Med. Chem. Res. 2013, 22, 4622–4632. [Google Scholar] [CrossRef]
- Beaulieu, C.; Wang, Z.Y.; Denis, D.; Greig, G.; Lamontagne, S.; O’Neill, G.; Slipetz, D.; Wang, J. Benzimidazoles as new potent and selective DP antagonists for the treatment of allergic rhinitis. Bioorg. Med. Chem. Lett. 2004, 14, 3195–3199. [Google Scholar] [CrossRef] [PubMed]
- White, A.W.; Curtin, N.J.; Eastman, B.W.; Golding, B.T.; Hostomsky, Z.; Kyle, S.; Maegley, K.A.; Li, J.; Skalitzky, D.J.; Webber, S.E.; et al. Potentiation of cytotoxic drug activity in human tumour cell lines, by amine-substituted 2-arylbenzimidazole-4-carboxamide PARP-1 inhibitors. Bioorg. Med. Chem. Lett. 2004, 14, 2433–2437. [Google Scholar] [CrossRef] [PubMed]
- Kahveci, B.; Mentes, E.; Ozil, M.; Ulker, S.; Erturk, M. An efficient synthesis of benzimidazoles via a microwave technique and evaluation of their biological activities. Mon. Chem. 2013, 144, 993–1001. [Google Scholar] [CrossRef]
- Purushottamachar, P.; Godbole, A.M.; Gediya, L.K.; Martin, M.S.; Vasaitis, T.S.; Kwegyir-Afful, A.K.; Ramalingam, S.; Ates-Alagoz, Z.; Njar, V.C.O. Systematic Structure Modifications of Multitarget Prostate Cancer Drug Candidate Galeterone To Produce Novel Androgen Receptor Down-Regulating Agents as an Approach to Treatment of Advanced Prostate Cancer. J. Med. Chem. 2013, 56, 4880–4898. [Google Scholar] [CrossRef] [PubMed]
- Evans, T.M.; Gardiner, J.M.; Mahmood, N.; Smis, M. Structure-activity relationships of anti-HIV-1 N-alkoxy- and N-allyloxy-benzimidazoles. Bioorg. Med. Chem. Lett. 1997, 7, 409–412. [Google Scholar] [CrossRef]
- Yoon, Y.K.; Ali, M.A.; Wei, A.C.; Choon, T.S.; Khaw, K.Y.; Murugaiyah, V.; Masand, V.H.; Osman, H. Synthesis, characterization, and molecular docking analysis of novel benzimidazole derivatives as cholinesterase inhibitors. Bioorg. Chem. 2013, 49, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.M.; Wu, C.F.; Li, X.B.; Wu, G.S.; Xie, S.; Hu, Q.N.; Deng, Z.X.; Zhu, M.X.; Hong, X.C.; Luo, H.R. Synthesis, biological evaluation and molecular modeling of substituted 2-aminobenzimidazoles as novel inhibitors of acetylcholinesterase and butyrylcholinesterase. Bioorg. Med. Chem. 2013, 21, 4218–4224. [Google Scholar] [CrossRef] [PubMed]
- Ceballos, L.; Virkel, G.; Elissondo, C.; Canton, C.; Canevari, J.; Murno, G.; Denegri, G.; Lanusse, C.; Alvarez, L. A pharmacology-based comparison of the activity of albendazole and flubendazole against Echinococcus granulosus metacestode in sheep. Acta Trop. 2013, 127, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Perez-Villanueva, J.; Hernandez-Campos, A.; Yepez-Mulia, L.; Mendez-Cuesta, C.; Mendez-Lucio, O.; Hernandez-Luis, F.; Castillo, R. Synthesis and antiprotozoal activity of novel 2-[2-(1H-imidazol-1-yl)ethyl sulfanyl]-1H-benzimidazole derivatives. Bioorg. Med. Chem. Lett. 2013, 23, 4221–4224. [Google Scholar] [CrossRef] [PubMed]
- Henke, K.R.; Robertson, D.; Krepps, M.K.; Atwood, D.A. Chemistry and stability of precipitates from aqueous solutions of 2,4,6-trimercaptotriazine, trisodium salt, nonahydrate (TMT-55) and mercury (II) chloride. Water Res. 2000, 34, 3005–3013. [Google Scholar] [CrossRef]
- Matlock, M.M.; Henke, K.R.; Atwood, D.A.; Robertson, D. Aqueous leaching properties and environmental implications of cadmium, lead and zinc trimercaptotriazine (TMT) compounds. Water Res. 2001, 35, 3649–3655. [Google Scholar] [CrossRef]
- Rosso, V.W.; Lust, D.A.; Bernot, P.J.; Grosso, J.A.; Modi, S.P.; Rusowicz, A.; Sedergran, T.C.; Simpson, J.H.; Srivastava, S.K.; Humora, M.J.; et al. Removal of palladium from organic reaction mixtures by trimercaptotriazine. Org. Process Res. Dev. 1997, 1, 311–314. [Google Scholar] [CrossRef]
- Garrett, C.E.; Prasad, K. The art of meeting palladium specifications in active pharmaceutical ingredients produced by Pd-catalyzed reactions. Adv. Synth. Catal. 2004, 346, 889–900. [Google Scholar] [CrossRef]
- Chen, W.; Hong, S.; Xiang, B.; Luo, H.Q.; Li, M.; Li, N.B. Corrosion inhibition of copper in hydrochloric acid by coverage with trithiocyanuric acid self-assembled monolayers. Corros. Eng. Sci. Technol. 2013, 48, 98–107. [Google Scholar] [CrossRef]
- Chen, W.; Hong, S.; Luo, H.Q.; Li, N.B. Inhibition Effect of 2,4,6-Trimercapto-1,3,5-triazine Self-Assembled Monolayers on Copper Corrosion in NaCl Solution. J. Mater. Eng. Perform. 2014, 23, 527–537. [Google Scholar] [CrossRef]
- Iltzsch, M.H.; Tankersley, K.O. Structure-activity relationship of ligands of uracil phosphoribosyltransferase from toxoplasma-gondii. Biochem. Pharmacol. 1994, 48, 781–791. [Google Scholar] [CrossRef]
- Kar, S.; Miller, T.A.; Chakraborty, S.; Sarkar, B.; Pradhan, B.; Sinha, R.K.; Kundu, T.; Ward, M.D.; Lahiri, G.K. Synthesis, mixed valence aspects and non-linear optical properties of the triruthenium complexes [{(bpy)2RuII}3(L)]3+ and [{(phen)2RuII}3L3+ (bpy = 2,2′-bipyridine, phen = 1,10-phenanthroline and L3− = 1,3,5-triazine-2,4,6-trithiol). Dalton Trans. 2003, 2591–2596. [Google Scholar] [CrossRef]
- Kar, S.; Pradhan, B.; Sinha, K.; Kundu, T.; Kodgire, P.; Rao, K.K.; Puranik, V.G.; Lahiri, G.K. Synthesis, structure, redox, NLO and DNA interaction aspects of [{(L′–′′′)2RuII}3(mu3-L)]3+ and [(L′)2RuII(NC5H4S−)]+, L3− = 1,3,5-triazine-2,4,6-trithiolato, L′–′′′ = arylazopyridine. Dalton Trans. 2004, 11, 1752–1760. [Google Scholar] [CrossRef] [PubMed]
- Aoki, S.; Zulkefeli, M.; Shiro, M.; Kimura, E. New supramolecular trigonal prisms from zinc(II)-1,4,7,10-tetraazacyclododecane (cyclen) complexes and trithiocyanurate in aqueous solution. Proc. Natl. Acad. Sci. USA 2002, 99, 4894–4899. [Google Scholar] [CrossRef] [PubMed]
- Aoki, S.; Shiro, M.; Kimura, E. A cuboctahedral supramolecular capsule by 4:4 self-assembly of tris(ZnII-cyclen) and trianionic trithiocyanurate in aqueous solution at neutral pH (cyclen = 1,4,7,10-tetraazacyclododecane). Chem. Eur. J. 2002, 8, 929–939. [Google Scholar] [CrossRef]
- Zulkefeli, M.; Sogon, T.; Takeda, K.; Kimura, E.; Aoki, S. Design and Synthesis of a Stable Supramolecular Trigonal Prism Formed by the Self-Assembly of a Linear Tetrakis(Zn2+-cyclen) Complex and Trianionic Trithiocyanuric Acid in Aqueous Solution and Its Complexation with DNA (Cyclen = 1,4,7,10-Tetraazacyclododecane). Inorg. Chem. 2009, 48, 9567–9578. [Google Scholar] [PubMed]
- Kopel, P.; Dolezal, K.; Machala, L.; Langer, V. Synthesis, characterization and screening of biological activity of Zn(II), Fe(II) and Mn(II) complexes with trithiocyanuric acid. Polyhedron 2007, 26, 1583–1589. [Google Scholar] [CrossRef]
- Kopel, P.; Dolezal, K.; Langer, V.; Jun, D.; Adam, V.; Kuca, K.; Kizek, R. Trithiocyanurate Complexes of Iron, Manganese and Nickel and Their Anticholinesterase Activity. Molecules 2014, 19, 4338–4354. [Google Scholar] [CrossRef] [PubMed]
- Kopel, P.; Mrozinski, J.; Dolezal, K.; Langer, V.; Boca, R.; Bienko, A.; Pochaba, A. Ferromagnetic Properties of a Trinuclear Nickel(II) Complex with a Trithiocyanurate Bridge. Eur. J. Inorg. Chem. 2009, 36, 5475–5482. [Google Scholar] [CrossRef]
- Ranganathan, A.; Pedireddi, V.R.; Rao, C.N.R. Hydrothermal synthesis of organic channel structures: 1:1 Hydrogen-bonded adducts of melamine with cyanuric and trithiocyanuric acids. J. Am. Chem. Soc. 1999, 121, 1752–1753. [Google Scholar] [CrossRef]
- Nagarajan, V.; Pedireddi, V.R. Preparation of Multiple Cocrystals of Trithiocyanuric Acid with Some N-Donor Compounds. Cryst. Growth Des. 2014, 14, 4803–4810. [Google Scholar] [CrossRef]
- Kopel, P.; Travnicek, Z.; Kvitek, L.; Biler, M.; Pavlicek, M.; Sindelar, Z.; Marek, J. Coordination compounds of nickel with trithiocyanuric acid. Part IV. Structure of Ni(pmdien)(ttcH) (pmdien = N,N,N′,N′,N′′-pentamethyldiethylenetriamine, ttcH3 = trithiocyanuric acid). Transit. Met. Chem. 2001, 26, 282–286. [Google Scholar] [CrossRef]
- Bienko, A.; Kopel, P.; Kizek, R.; Kruszynski, R.; Bienko, D.; Titis, J.; Boca, R. Synthesis, crystal structure and magnetic properties of trithiocyanurate or thiodiacetate polynuclear Ni(II) and Co(II) complexes. Inorg. Chim. Acta 2014, 416, 147–156. [Google Scholar] [CrossRef]
- Kopel, P.; Travnicek, Z.; Panchartkova, R.; Sindelar, Z.; Marek, J. Coordination compounds of nickel with trithiocyanuric acid. J. Coord. Chem. 1998, 44, 205–215. [Google Scholar] [CrossRef]
- Kopel, P.; Travnicek, Z.; Panchartkova, R.; Biler, M.; Marek, J. Coordination compounds of nickel with trithiocyanuric acid. Part II. Crystal and molecular structure of Ni(taa)(ttcH) (taa = tris-(2-aminoethyl)amine, ttcH3 = trithiocyanuric acid). Transit. Met. Chem. 1999, 24, 239–243. [Google Scholar] [CrossRef]
- Marek, J.; Kopel, P.; Travnicek, Z. Tris(1,10-phenanthroline)sodium 2,4,6-trimercapto-1,3,5-triazin-1-ide. Acta Crystallogr. Sect. C Cryst. Struct. Commun. 2003, 59, M399–M401. [Google Scholar] [CrossRef]
- Kopel, P.; Travnicek, Z.; Zboril, R.; Marek, J. Synthesis, X-ray and Mossbauer study of iron(II) complexes with trithiocyanuric acid (ttcH3): The X-ray structures of [Fe(bpy)3) (ttcH) 2bpy 7H2O and [Fe(phen)3](ttcH2)(ClO4) 2CH3OH 2H2O. Polyhedron 2004, 23, 2193–2202. [Google Scholar] [CrossRef]
- Li, W.R.; Xie, X.B.; Shi, Q.S.; Duan, S.S.; Ouyang, Y.S.; Chen, Y.B. Antibacterial effect of silver nanoparticles on Staphylococcus aureus. Biometals 2011, 24, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Park, H.J.; Kim, J.Y.; Kim, J.; Lee, J.H.; Hahn, J.S.; Gu, M.B.; Yoon, J. Silver-ion-mediated reactive oxygen species generation affecting bactericidal activity. Water Res. 2009, 43, 1027–1032. [Google Scholar] [CrossRef] [PubMed]
- Kwakye-Awuah, B.; Williams, C.; Kenward, M.A.; Radecka, I. Antimicrobial action and efficiency of silver-loaded zeolite X. J. Appl. Microbiol. 2008, 104, 1516–1524. [Google Scholar] [CrossRef] [PubMed]
- Matthews, C.J.; Leese, T.A.; Clegg, W.; Elsegood, M.R.J.; Horsburgh, L.; Lockhart, J.C. A route to bis(benzimidazole) ligands with built-in asymmetry: Potential models of protein binding sites having histidines of different basicity. Inorg. Chem. 1996, 35, 7563–7571. [Google Scholar] [CrossRef]
- SMART (Version 5.63) and SAINT (Version 6.45), Area Detector Control and Integration Software, Bruker AXS Inc.: Madison, WI, USA, 2003.
- Sheldrick, G.M. SADABS Program for Empirical Absorption Correction for Area Detectors, Version 2.10; University of Gottingen: Gottingen, Germany, 2003. [Google Scholar]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. Sect. A 2008, 64, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Bautista-Trujillo, G.U.; Solorio-Rivera, J.L.; Renteria-Solorzano, I.; Carranza-German, S.I.; Bustos-Martinez, J.A.; Arteaga-Garibay, R.I.; Baizabal-Aguirre, V.M.; Cajero-Juarez, M.; Bravo-Patino, A.; Valdez-Alarcon, J.J. Performance of culture media for the isolation and identification of Staphylococcus aureus from bovine mastitis. J. Med. Microbiol. 2013, 62, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Predrag, S.; Branislava, K.; Miodrag, S.; Biljana, M.S.; Suzana, T.; Natasa, M.T.; Tatjana, B. Clinical importance and representation of toxigenic and non-toxigenic Clostridium difficile cultivated from stool samples of hospitalized patients. Braz. J. Microbiol. 2012, 43, 215–223. [Google Scholar] [PubMed]
- Bosch-Mestres, J.; Martin-Fernandez, R.M.; de Anta-Losada, M.T.J. Comparative study of three culture media for detecting group B Streptococcus colonization in pregnant women. Enferm. Infec. Microbiol. Clin. 2003, 21, 346–349. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the prepared compounds are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kopel, P.; Wawrzak, D.; Langer, V.; Cihalova, K.; Chudobova, D.; Vesely, R.; Adam, V.; Kizek, R. Biological Activity and Molecular Structures of Bis(benzimidazole) and Trithiocyanurate Complexes. Molecules 2015, 20, 10360-10376. https://doi.org/10.3390/molecules200610360
Kopel P, Wawrzak D, Langer V, Cihalova K, Chudobova D, Vesely R, Adam V, Kizek R. Biological Activity and Molecular Structures of Bis(benzimidazole) and Trithiocyanurate Complexes. Molecules. 2015; 20(6):10360-10376. https://doi.org/10.3390/molecules200610360
Chicago/Turabian StyleKopel, Pavel, Dorota Wawrzak, Vratislav Langer, Kristyna Cihalova, Dagmar Chudobova, Radek Vesely, Vojtech Adam, and Rene Kizek. 2015. "Biological Activity and Molecular Structures of Bis(benzimidazole) and Trithiocyanurate Complexes" Molecules 20, no. 6: 10360-10376. https://doi.org/10.3390/molecules200610360










