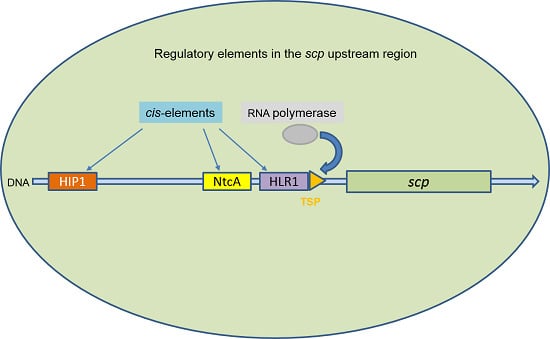
Regulation of the scp Genes in the Cyanobacterium Synechocystis sp. PCC 6803—What is New?
Abstract
:1. Introduction
2. Results and Discussion
2.1. NtcA Binds to the Promoter Regions of scpB and scpE

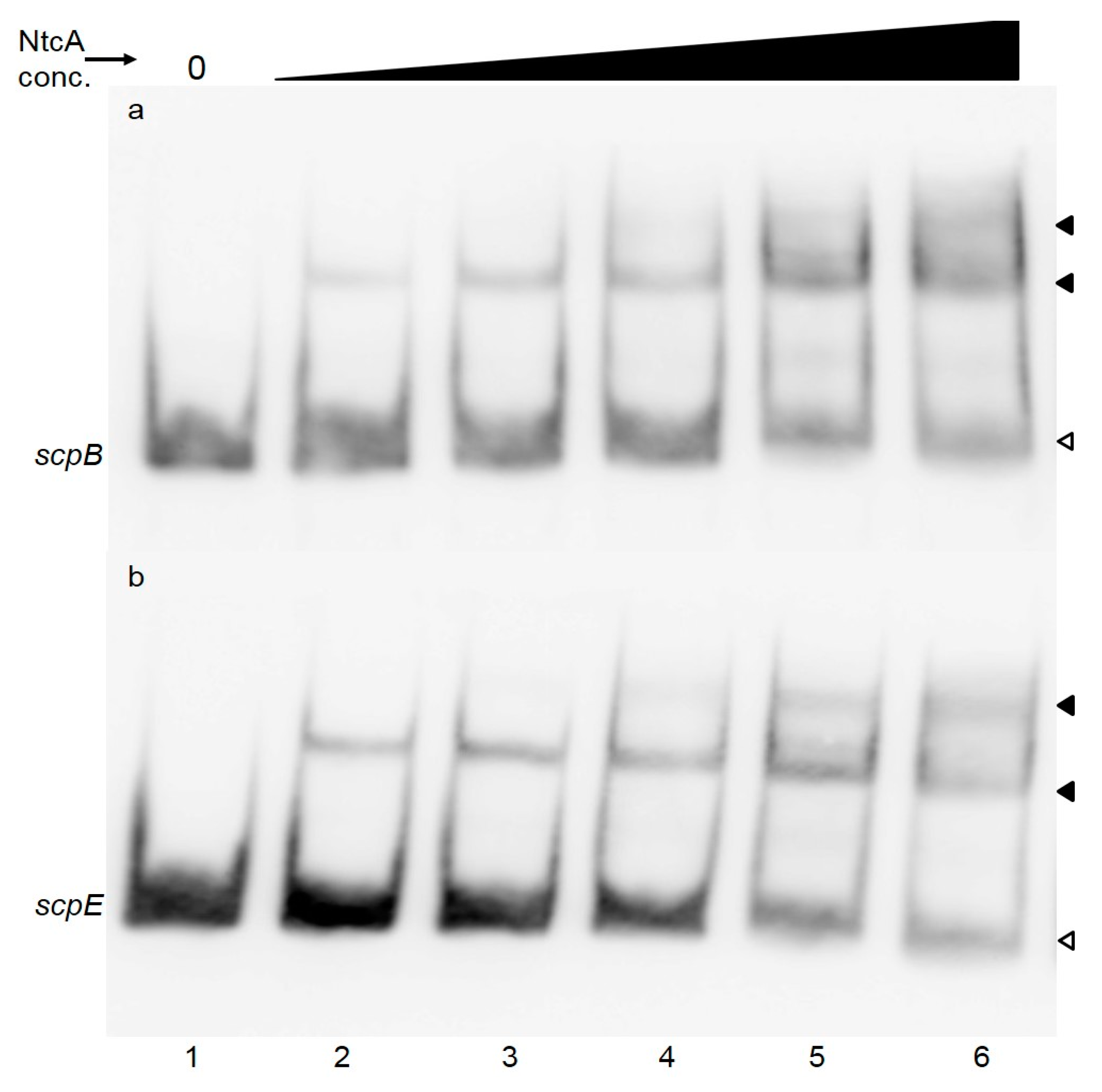
2.2. 2-Oxoglutarate does not Stimulate NtcA Binding

2.3. scpB and scpC Contain a HIP1 Motif


2.4. HLR1 Motif
| Gene ID | Gene Product | HLR1 | p-Value | Position Relative to the Translational Start Site | Strand |
|---|---|---|---|---|---|
| ssl2542 * | ScpB | TTACAATAGTTAACAT | 1.16 × 10−8 | −42 | + |
| sll1483 | Hypothetical protein | TTCACAAAAATTTATA | 1.41 × 10−8 | −63 | + |
| sll0157 | Hypothetical protein | TTACAATCGTTTACAA | 1.54 × 10−8 | −50 | + |
| sll2012 | SigD | TGAGAAAACTTTACAA | 6.38 × 10−8 | −44 | − |
| slr1894 | MrgA | TAACAAAATTTAACAC | 1.97 × 10−8 | −32 | − |
| slr0320 | Hypothetical protein | TTAAGATATATTACAA | 6.57 × 10−8 | −78 | + |
| slr1687 | Hypothetical protein | TAACAAAACTTTATAA | 1.07 × 10−8 | −47 | − |
| ssl3044 | Probable ferredoxin | TAACAAAACTTTATAA | 1.07 × 10−8 | −142 | + |
| ssl2162 | Unknown protein | TGTAAACCTTTGTTAA | 1.13 × 10−8 | −75 | − |
3. Experimental Section
3.1. Strains and Growth Conditions
3.2. Protein Expression and Purification
3.3. Electrophoretic Mobility Shift Assay (EMSA)
| NtcA-Fw | 5ʹ-AACGTCCATGGATCAGTCCCTA-3ʹ |
| NtcA-R | 5ʹ-CATAGAGGTACCTTAGGTAAACTG-3ʹ |
| Sll1130-Fw | 5ʹ-GCGCCATGGATACAATTTACGA ACAATTTG-3ʹ |
| Sll1130-R | 5ʹ-GCGAAGCTTACCGAGTTTAAAAACATGGGG-3′ |
| scpB-Fw | 5ʹ-TTTGAGCCTAACATTATCCTCC-3ʹ |
| scpB-R | 5ʹ-ATGTAATTACGAGCTGGGAGG-3ʹ |
| scpE-Fw | 5ʹ-ACTCTGACGTACCCATTGAC-3ʹ |
| scpE-R | 5ʹ-CTCATGGGATTTCCTGGGATA-3ʹ |
| scpB-CR-Fw | 5ʹ-TAAATTTGGATTCACTGCTTTCG-3ʹ |
| scpB-CR-R | 5ʹ-AGTGAAGTACACCTTGCCCAGAG-3ʹ |
| Fw-HIP1 | 5ʹ-ATCCTGGGACTGGAGAATCA-3ʹ |
| R-HIP1 | 5ʹ-CGGGTGGTCATAATTGGATTA-3ʹ |
3.4. Bioinformatic Analysis
3.5. DNA Pull down Assay
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Funk, C.; Vermaas, W. A cyanobacterial gene family coding for single-helix proteins resembling part of the light-harvesting proteins from higher plants. Biochemistry 1999, 38, 9397–9404. [Google Scholar] [CrossRef] [PubMed]
- Kufryk, G.; Hernandez-Prieto, M.; Kieselbach, T.; Miranda, H.; Vermaas, W.; Funk, C. Association of small Cab-like proteins (Scps) of Synechocystis sp. PCC 6803 with photosystem II. Photosynth. Res. 2008, 95, 135–145. [Google Scholar] [CrossRef] [PubMed]
- Storm, P.; Hernandez-Prieto, M.; Eggink, L.; Hoober, J.; Funk, C. The small Cab-like proteins of Synechocystis sp. PCC 6803 bind chlorophyll. Photosynth. Res. 2008, 98, 479–488. [Google Scholar] [CrossRef] [PubMed]
- Staleva, H.; Komenda, J.; Shukla, M.K.; Šlouf, V.; Kaňa, R.; Polívka, T.; Sobotka, R. Mechanism of photoprotection in the cyanobacterial ancestor of plant antenna proteins. Nat. Chem. Biol. 2015, 11, 287–291. [Google Scholar] [CrossRef] [PubMed]
- Promnares, K.; Komenda, J.; Bumba, L.; Nebesarova, J.; Vacha, F.; Tichy, M. Cyanobacterial small chlorophyll-binding protein ScpD (HliB) is located on the periphery of photosystem II in the vicinity of PsbH and CP47 subunits. J. Biol. Chem. 2006, 281, 32705–32713. [Google Scholar] [CrossRef] [PubMed]
- Yao, D.; Kieselbach, T.; Komenda, J.; Promnares, K.; Prieto, M.A.H.; Tichy, M.; Vermaas, W.; Funk, C. Localization of the small Cab-like proteins in photosystem II. J. Biol. Chem. 2007, 282, 267–276. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.X.; Hall, M.; Funk, C.; Schröder, W.P. Photosystem II, a growing complex: Updates on newly discovered components and low molecular mass proteins. Biochim. Biophys. Acta 2012, 1817, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Knoppová, J.; Sobotka, R.; Tichý, M.; Yu, J.; Konik, P.; Halada, P.; Nixon, P.J.; Komenda, J. Discovery of a chlorophyll binding protein complex involved in the early steps of photosystem II assembly in Synechocystis. Plant Cell 2014, 26, 1200–1212. [Google Scholar] [CrossRef] [PubMed]
- Yao, D.C.I.; Brune, D.C.; Vavilin, D.; Vermaas, W.F.J. Photosystem II component lifetimes in the cyanobacterium Synechocystis sp. PCC 6803: Small Cab-like proteins stabilize biosynthesis intermediates and affect early steps in chlorophyll synthesis. J. Biol. Chem. 2012, 287, 682–692. [Google Scholar] [CrossRef] [PubMed]
- Daddy, S.; Zhan, J.; Jantaro, S.; He, C.; He, Q.; Wang, Q. A novel high light-inducible carotenoid-binding protein complex in the thylakoid membranes of Synechocystis PCC 6803. Sci. Rep. 2015, 5. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Vavilin, D.; Funk, C.; Vermaas, W. Small cab-like proteins regulating tetrapyrrole biosynthesis in the cyanobacterium Synechocystis sp. PCC 6803. Plant Mol. Biol. 2002, 49, 149–160. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Vavilin, D.; Funk, C.; Vermaas, W. Multiple deletions of small Cab-like proteins in the cyanobacterium Synechocystis sp. PCC 6803. J. Biol. Chem. 2004, 279, 27971–27979. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Prieto, M.A.; Tibiletti, T.; Abasova, L.; Kirilovsky, D.; Vass, I.; Funk, C. The small Cab-like proteins of the cyanobacterium Synechocystis sp. PCC 6803: Their involvement in chlorophyll biogenesis for photosystem II. Biochim. Biophys. Acta 2011, 1807, 1143–1151. [Google Scholar] [CrossRef] [PubMed]
- Sinha, R.K.; Komenda, J.; Knoppová, J.; Sedlářová, M.; Pospíśil, P. Small Cab-like proteins prevent formation of singlet oxygen in the damaged photosystem II complex of the cyanobacterium Synechocystis sp. PCC 6803. Plant Cell Environ. 2012, 35, 806–818. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Prieto, M.A.; Futschik, M.E. Cyanoexpress: A web database for exploration and visualisation of the integrated transcriptome of cyanobacterium Synechocystis sp. PCC 6803. Bioinformation 2012, 8, 634–638. [Google Scholar] [CrossRef] [PubMed]
- Mähler, N.; Cheregi, O.; Funk, C.; Netotea, S.; Hvidsten, T.R. Synergy: A web resource for exploring gene regulation in Synechocystis sp. PCC 6803. PLoS ONE 2014, 9, e113496. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Herrero, A.; Muro-Pastor, A.M.; Flores, E. Nitrogen control in cyanobacteria. J. Bacteriol. 2001, 183, 411–425. [Google Scholar] [CrossRef] [PubMed]
- López-Gomollón, S.; Hernández, J.A.; Pellicer, S.; Angarica, V.E.; Peleato, M.L.; Fillat, M.F. Cross-talk between iron and nitrogen regulatory networks in Anabaena (Nostoc) sp. PCC 7120: Identification of overlapping genes in FurA and NtcA regulons. J. Mol. Biol. 2007, 374, 267–281. [Google Scholar] [CrossRef] [PubMed]
- Frías, J.E.; Flores, E.; Herrero, A. Requirement of the regulatory protein NtcA for the expression of nitrogen assimilation and heterocyst development genes in the cyanobacterium Anabaena sp. PCC 7120. Mol. Microbiol. 1994, 14, 823–832. [Google Scholar] [CrossRef] [PubMed]
- Osanai, T.; Oikawa, A.; IIjima, H.; Kuwahara, A.; Asayama, M.; Tanaka, K.; Ikeuchi, M.; Saito, K.; Hirai, M.Y. Metabolomic analysis reveals rewiring of Synechocystis sp. PCC 6803 primary metabolism by NtcA overexpression. Environ. Microbiol. 2014, 16, 3304–3317. [Google Scholar] [CrossRef] [PubMed]
- Tanigawa, R.; Shirokane, M.; Maeda, S.-I.; Omata, T.; Tanaka, K.; Takahashi, H. Transcriptional activation of NtcA-dependent promoters of Synechococcus sp. PCC 7942 by 2-oxoglutarate in vitro. Proc. Natl. Acad. Sci. USA 2002, 99, 4251–4255. [Google Scholar] [CrossRef] [PubMed]
- Vázquez-Bermúdez, M.A.F.; Herrero, A.; Flores, E. 2-oxoglutarate increases the binding affinity of the NtcA (nitrogen control) transcription factor for the Synechococcus glnA promoter. FEBS Lett. 2002, 512, 71–74. [Google Scholar] [CrossRef]
- Zhao, M.X.; Jiang, Y.L.; He, Y.X.; Chen, Y.F.; Teng, Y.B.; Chen, Y.; Zhang, C.C.; Zhou, C.Z. Structural basis for the allosteric control of the global transcription factor NtcA by the nitrogen starvation signal 2-oxoglutarate. Proc. Natl. Acad. Sci. USA 2010, 107, 12487–12492. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Morby, A.P.; Turner, J.S.; Whitton, B.A.; Robinson, N.J. Deletion within the metallothionein locus of cadmium-tolerant Synechococcus PCC 6301 involving a Highly Iterated Palindrome (HIP1). Mol. Microbiol. 1993, 7, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Robinson, P.J.; Cranenburgh, R.M.; Head, I.M.; Robinson, N.J. HIP1 propagates in cyanobacterial DNA via nucleotide substitutions but promotes excision at similar frequencies in Escherichia coli and Synechococcus PCC 7942. Mol. Microbiol. 1997, 24, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Delaye, L.; Moya, A. Abundance and distribution of the Highly Iterated Palindrome 1 (HIP1) among prokaryotes. Mob. Genet. Elem. 2011, 1, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Elhai, J. Highly Iterated Palindromic sequences (HIPs) and their relationship to DNA methyltransferases. Life 2015, 5, 921–948. [Google Scholar] [CrossRef] [PubMed]
- Krishna, P.S.; Rani, B.R.; Mohan, M.K.; Suzuki, I.; Shivaji, S.; Prakash, J.S.S. A novel transcriptional regulator, Sll1130, negatively regulates heat-responsive genes in Synechocystis sp. PCC 6803. Biochem. J. 2013, 449, 751–760. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, J.; Salih, G.F.; Ghebramedhin, H.; Jansson, C. Deletion mutagenesis of the 5′ psbA2 region in Synechocystis 6803: Identification of a putative cis element involved in photoregulation. Mol. Cell Biol. Res. Commun. 2000, 3, 292–298. [Google Scholar] [CrossRef] [PubMed]
- Kappell, A.D.; Bhaya, D.; van Waasbergen, L.G. Negative control of the high light-inducible hlia gene and implications for the activities of the NblS sensor kinase in the cyanobacterium Synechococcus elongatus strain PCC 7942. Arch. Microbiol. 2006, 186, 403–413. [Google Scholar] [CrossRef] [PubMed]
- Kappell, A.; van Waasbergen, L. The response regulator RpaB binds the High Light Regulatory 1 sequence upstream of the high-light-inducible hlib gene from the cyanobacterium Synechocystis PCC 6803. Arch. Microbiol. 2007, 187, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Seino, Y.; Takahashi, T.; Hihara, Y. The response regulator RpaB binds to the upstream element of photosystem I genes to work for positive regulation under low-light conditions in Synechocystis sp. strain PCC 6803. J. Bacteriol. 2009, 191, 1581–1586. [Google Scholar] [CrossRef] [PubMed]
- Muramatsu, M.; Sonoike, K.; Hihara, Y. Mechanism of downregulation of photosystem I content under high-light conditions in the cyanobacterium Synechocystis sp. PCC 6803. Microbiology 2009, 155, 989–996. [Google Scholar] [CrossRef] [PubMed]
- García-Domínguez, M.; Reyes, J.C.; Florencio, F.J. NtcA represses transcription of gifA and gifB, genes that encode inhibitors of glutamine synthetase type I from Synechocystis sp. PCC 6803. Mol. Microbiol. 2000, 35, 1192–1201. [Google Scholar] [CrossRef] [PubMed]
- Ramasubramanian, T.S.; Wei, T.F.; Golden, J.W. Two Anabaena sp. strain PCC 7120 DNA-binding factors interact with vegetative cell- and heterocyst-specific genes. J. Bacteriol. 1994, 176, 1214–1223. [Google Scholar] [PubMed]
- Kuniyoshi, T.M.; Gonzalez, A.; Lopez-Gomollon, S.; Valladares, A.; Bes, M.T.; Fillat, M.F.; Peleato, M.L. 2-oxoglutarate enhances NtcA binding activity to promoter regions of the microcystin synthesis gene cluster. FEBS Lett. 2011, 585, 3921–3926. [Google Scholar] [CrossRef] [PubMed]
- Krasikov, V.; Aguirre von Wobeser, E.; Dekker, H.L.; Huisman, J.; Matthijs, H.C.P. Time-series resolution of gradual nitrogen starvation and its impact on photosynthesis in the cyanobacterium Synechocystis PCC 6803. Physiol. Plant. 2012, 145, 426–439. [Google Scholar] [CrossRef] [PubMed]
- Osanai, T.; Imamura, S.; Asayama, M.; Shirai, M.; Suzuki, I.; Murata, N.; Tanaka, K. Nitrogen induction of sugar catabolic gene expression in Synechocystis sp. PCC 6803. DNA Res. 2006, 13, 185–195. [Google Scholar] [CrossRef] [PubMed]
- Von Wobeser, A.E.; Ibelings, B.W.; Bok, J.; Krasikov, V.; Huisman, J.; Matthijs, H.C.P. Concerted changes in gene expression and cell physiology of the cyanobacterium Synechocystis sp. strain PCC 6803 during transitions between nitrogen and light-limited growth. Plant Physiol. 2011, 155, 1445–1457. [Google Scholar] [CrossRef] [PubMed]
- He, Q.; Dolganov, N.; Björkman, O.; Grossman, A.R. The high light-inducible polypeptides in Synechocystis PCC 6803. J. Biol. Chem. 2001, 276, 306–314. [Google Scholar] [CrossRef] [PubMed]
- Picossi, S.; Flores, E.; Herrero, A. ChIP analysis unravels an exceptionally wide distribution of DNA binding sites for the NtcA transcription factor in a heterocyst-forming cyanobacterium. BMC Genom. 2014, 15, 22. [Google Scholar] [CrossRef] [PubMed]
- Tolonen, A.C.; Aach, J.; Lindell, D.; Johnson, Z.I.; Rector, T.; Steen, R.; Church, G.M.; Chisholm, S.W. Global gene expression of Prochlorococcus ecotypes in response to changes in nitrogen availability. Mol. Syst. Biol. 2006, 2. [Google Scholar] [CrossRef] [PubMed]
- Forcada-Nadal, A.; Forchhammer, K.; Rubio, V. SPR analysis of promoter binding of Synechocystis PCC6803 transcription factors NtcA and CRP suggests cross-talk and sheds light on regulation by effector molecules. FEBS Lett. 2014, 588, 2270–2276. [Google Scholar] [CrossRef] [PubMed]
- Muro-Pastor, M.I.; Reyes, J.C.; Florencio, F.J. Cyanobacteria perceive nitrogen status by sensing intracellular 2-oxoglutarate levels. J. Biol. Chem. 2001, 276, 38320–38328. [Google Scholar] [PubMed]
- Robinson, N.J.; Robinson, P.J.; Gupta, A.; Bleasby, A.J.; Whitton, B.A.; Morby, A.P. Singular over-representation of an octameric palindrome, HIP1, in DNA from many cyanobacteria. Nucleic Acids Res. 1995, 23, 729–735. [Google Scholar] [CrossRef] [PubMed]
- Drewett, V.; Molina, H.; Millar, A.; Muller, S.; Hesler, F.V.; Shaw, P.E. DNA-bound transcription factor complexes analysed by mass-spectrometry: Binding of novel proteins to the human c-fos SRE and related sequences. Nucleic Acids Res. 2001, 29, 479–487. [Google Scholar] [CrossRef] [PubMed]
- López-Redondo, M.L.; Moronta, F.; Salinas, P.; Espinosa, J.; Cantos, R.; Dixon, R.; Marina, A.; Contreras, A. Environmental control of phosphorylation pathways in a branched two-component system. Mol. Microbiol. 2010, 78, 475–489. [Google Scholar] [CrossRef] [PubMed]
- Hanaoka, M.; Tanaka, K. Dynamics of RpaB–promoter interaction during high light stress, revealed by chromatin immunoprecipitation (ChIP) analysis in Synechococcus elongatus PCC 7942. Plant J. 2008, 56, 327–335. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, T.; Nakai, N.; Muramatsu, M.; Hihara, Y. Role of multiple HLR1 sequences in the regulation of the dual promoters of the psaAB genes in Synechocystis sp. PCC 6803. J. Bacteriol. 2010, 192, 4031–4036. [Google Scholar] [CrossRef] [PubMed]
- Muramatsu, M.; Hihara, Y. Acclimation to high-light conditions in cyanobacteria: From gene expression to physiological responses. J. Plant Res. 2012, 125, 11–39. [Google Scholar] [CrossRef] [PubMed]
- Satoh, A.; Kurano, N.; Senger, H.; Miyachi, S. Regulation of energy balance in photosystems in response to changes in CO2 concentrations and light intensities during growth in extremely-high-CO2-tolerant green microalgae. Plant Cell Physiol. 2002, 43, 440–451. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.K.; Elvitigala, T.; Bhattacharyya-Pakrasi, M.; Aurora, R.; Ghosh, B.; Pakrasi, H.B. Integration of carbon and nitrogen metabolism with energy production is crucial to light acclimation in the cyanobacterium Synechocystis. Plant Physiol. 2008, 148, 467–478. [Google Scholar] [CrossRef] [PubMed]
- Tu, C.J.; Shrager, J.; Burnap, R.L.; Postier, B.L.; Grossman, A.R. Consequences of a deletion in DspA on transcript accumulation in Synechocystis sp. strain PCC 6803. J. Bacteriol. 2004, 186, 3889–3902. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.L.; Postier, B.L.; Burnap, R.L. Alterations in global patterns of gene expression in Synechocystis sp. PCC 6803 in response to inorganic carbon limitation and the inactivation of NdhR, a LysR family regulator. J. Biol. Chem. 2004, 279, 5739–5751. [Google Scholar] [CrossRef] [PubMed]
- Hihara, Y.; Kamei, A.; Kanehisa, M.; Kaplan, A.; Ikeuchi, M. DNA microarray analysis of cyanobacterial gene expression during acclimation to high light. Plant Cell 2001, 13, 793–806. [Google Scholar] [CrossRef] [PubMed]
- Marin, K.; Kanesaki, Y.; Los, D.A.; Murata, N.; Suzuki, I.; Hagemann, M. Gene expression profiling reflects physiological processes in salt acclimation of Synechocystis sp. strain PCC 6803. Plant Physiol. 2004, 136, 3290–3300. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Singh, A.K.; McIntyre, L.M.; Sherman, L.A. Differential gene expression in response to hydrogen peroxide and the putative PerR regulon of Synechocystis sp. strain PCC 6803. J. Bacteriol. 2004, 186, 3331–3345. [Google Scholar] [CrossRef] [PubMed]
- Ohta, H.; Shibata, Y.; Haseyama, Y.; Yoshino, Y.; Suzuki, T.; Kagasawa, T.; Kamei, A.; Ikeuchi, M.; Enami, I. Identification of genes expressed in response to acid stress in Synechocystis sp. PCC 6803 using DNA microarrays. Photosynth. Res. 2005, 84, 225–230. [Google Scholar] [CrossRef] [PubMed]
- Shoumskaya, M.A.; Paithoonrangsarid, K.; Kanesaki, Y.; Los, D.A.; Zinchenko, V.V.; Tanticharoen, M.; Suzuki, I.; Murata, N. Identical Hik-Rre systems are involved in perception and transduction of salt signals and hyperosmotic signals but regulate the expression of individual genes to different extents in Synechocystis. J. Biol. Chem. 2005, 280, 21531–21538. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, I.; Kanesaki, Y.; Mikami, K.; Kanehisa, M.; Murata, N. Cold-regulated genes under control of the cold sensor Hik33 in Synechocystis. Mol. Microbiol. 2001, 40, 235–244. [Google Scholar] [CrossRef] [PubMed]
- Hsiao, H.Y.; He, Q.; van Waasbergen, L.G.; Grossman, A.R. Control of photosynthetic and high-light-responsive genes by the histidine kinase DspA: Negative and positive regulation and interactions between signal transduction pathways. J. Bacteriol. 2004, 186, 3882–3888. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.X.; Li, H.C.; Wei, Y.P.; Chu, W.Y.; Chong, Y.L.; Long, X.H.; Liu, Z.P.; Qin, S.; Shao, H.B. Signal transduction pathways in Synechocystis sp. PCC 6803 and biotechnological implications under abiotic stress. Crit. Rev. Biotechnol. 2015, 35, 269–280. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, T.; Sato, S.; Kotani, H.; Tanaka, A.; Asamizu, E.; Nakamura, Y.; Miyajima, N.; Hirosawa, M.; Sugiura, M.; Sasamoto, S.; et al. Sequence analysis of the genome of the unicellular cyanobacterium Synechocystis sp. strain PCC 6803. II. Sequence determination of the entire genome and assignment of potential protein-coding regions. DNA Res. 1996, 3, 109–136. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.; Kaneko, T.; Hirosawa, M.; Miyajima, N.; Tabata, S. Cyanobase, a www database containing the complete nucleotide sequence of the genome of Synechocystis sp. strain PCC 6803. Nucleic Acids Res. 1998, 26, 63–67. [Google Scholar] [CrossRef] [PubMed]
- Cheregi, O.; Vermaas, W.; Funk, C. The search for new chlorophyll-binding proteins in the cyanobacterium Synechocystis sp. PCC 6803. J. Biotechnol. 2012, 162, 124–133. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, P.; Lindblad, P. Novel insights into the regulation of LexA in the cyanobacterium Synechocystis sp. strain PCC 6803. J. Bacteriol. 2011, 193, 3804–3814. [Google Scholar] [CrossRef] [PubMed]
- Samples Availability: Not available.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheregi, O.; Funk, C. Regulation of the scp Genes in the Cyanobacterium Synechocystis sp. PCC 6803—What is New? Molecules 2015, 20, 14621-14637. https://doi.org/10.3390/molecules200814621
Cheregi O, Funk C. Regulation of the scp Genes in the Cyanobacterium Synechocystis sp. PCC 6803—What is New? Molecules. 2015; 20(8):14621-14637. https://doi.org/10.3390/molecules200814621
Chicago/Turabian StyleCheregi, Otilia, and Christiane Funk. 2015. "Regulation of the scp Genes in the Cyanobacterium Synechocystis sp. PCC 6803—What is New?" Molecules 20, no. 8: 14621-14637. https://doi.org/10.3390/molecules200814621