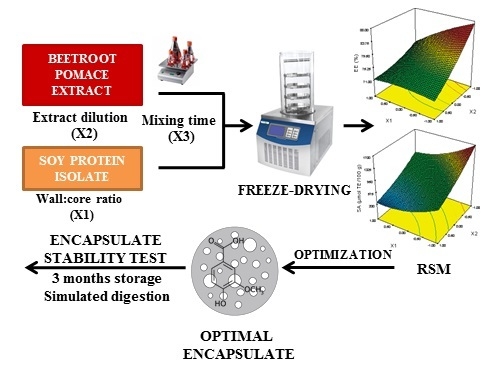
Encapsulation of Beetroot Pomace Extract: RSM Optimization, Storage and Gastrointestinal Stability
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characterization of Beetroot Pomace Extract
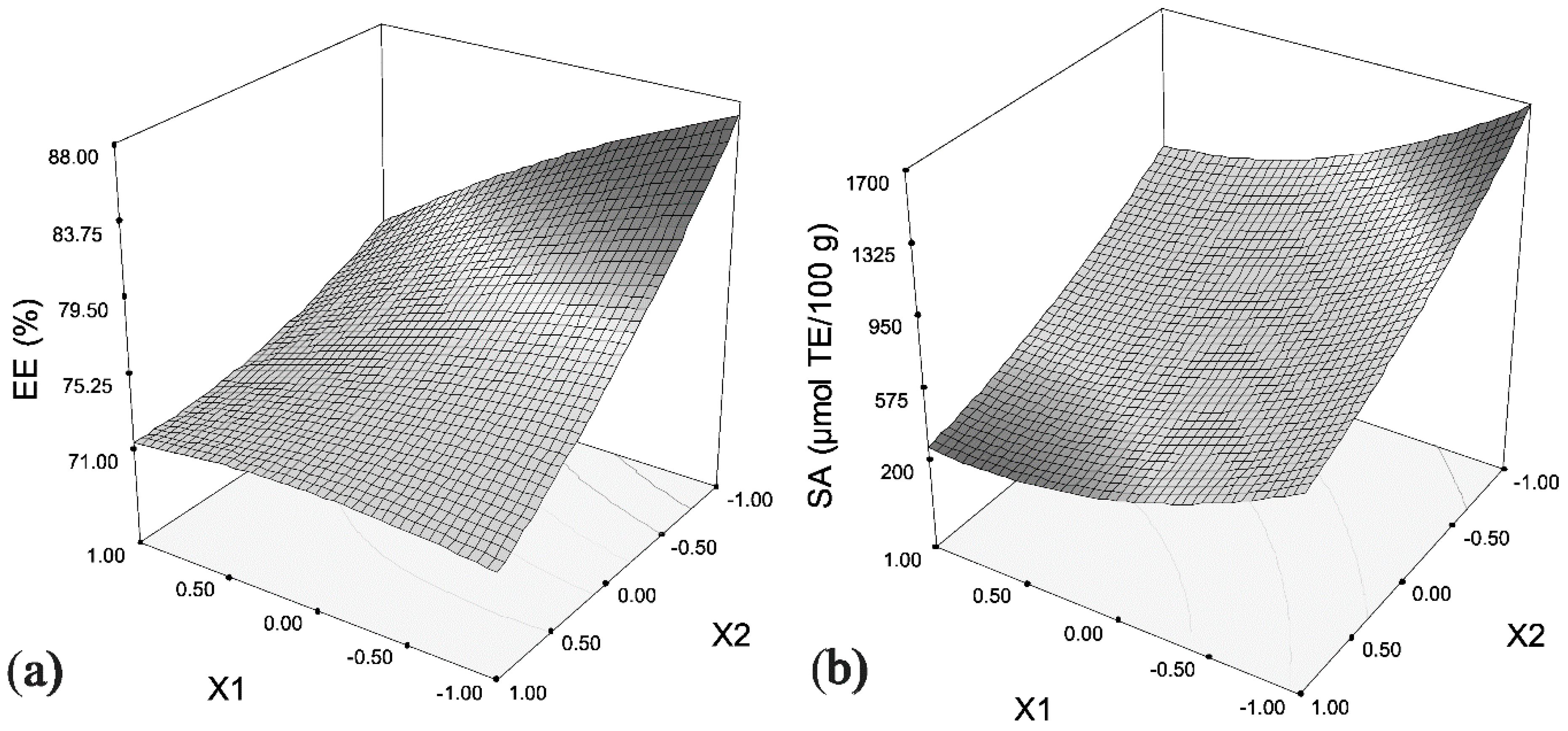
2.2. Optimization of Beetroot Pomace Extract Encapsulation
2.3. Characterization of the OE
2.3.1. Bioactive Compounds and Biological Activity
2.3.2. Storage Stability Studies
2.3.3. In Vitro Digestion of OE in the Intestinal Fluids
3. Material and Methods
3.1. Chemicals and Instruments
3.2. Plant Material
3.3. Pomace Extraction Procedure
3.4. Encapsulation Optimization
3.5. Total Phenolic Content
Phenolic Content in the Core (CPC) and Surface (SPC) of Encapsulate and Encapsulating Efficiency (EE)
3.6. Total Flavonoids Content
3.7. Determination of Betalain Content
3.8. HPLC Analysis
3.9. Radical Scavenging Activity (SA) by DPPH● Assay
3.10. Reducing Power (RP)
3.11. Storage Stability Test
3.12. In Vitro Simulated Gastric and Intestinal Digestion Release Study
3.13. Statistical Analysis
4. Conclusion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kris-Etherton, P.M.; Etherton, T.D.; Carlson, J.; Gardner, C. Recent discoveries in inclusive food-based approaches and dietary patterns for reduction in risk for cardiovascular disease. Curr. Opin. Lipidol. 2002, 13, 397–407. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J.P.E.; Tognolini, M.; Borges, G.; Crozier, A. Dietary (poly)phenolics in human health: Structures, bioavailability and evidence of protective effects against chronic diseases. Antiox. Redox Signal. 2013, 18, 1818–1888. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Mateos, A.; Vauzour, D.; Krueger, C.G.; Shanmuganayagam, D.; Reed, J.; Calani, L.; Mena, P.; Del Rio, D.; Crozier, A. Bioavailability, bioactivity and impact on health of dietary flavonoids and related compounds: An update. Arch. Toxicol. 2014, 88, 1803–1853. [Google Scholar] [CrossRef] [PubMed]
- Jackson, C.-J.C.; Paliyath, G. Functional foods, Nutraceuticals, and Degenerative Disease Prevention; Paliyath, G., Bakovic, M., Shetty, K., Eds.; Wiley-Blackwell: Oxford, UK, 2011; Chapter 2; pp. 11–43. [Google Scholar]
- Vulić, J.; Čanadanović-Brunet, J.; Ćetković, G.; Tumbas, V.; Djilas, S.; Četojević-Simin, D.; Čanadanović, V. Antioxidant and cell growth activities of beet root pomace extracts. J. Functional Foods 2012, 4, 670–678. [Google Scholar] [CrossRef]
- Tumbas Šaponjac, V.; Gironés-Vilaplana, A.; Djilas, S.; Mena, P.; Ćetković, G.; Moreno, D.A.; Čanadanović-Brunet, J.; Vulić, J.; Stajčić, S.; Krunić, M. Anthocyanin profiles and biological properties of caneberry (Rubus spp.) press residues. J. Sci. Food Agr. 2014, 94, 2393–2400. [Google Scholar] [CrossRef] [PubMed]
- Tumbas Šaponjac, V.; Gironés-Vilaplana, A.; Djilas, S.; Mena, P.; Ćetković, G.; Moreno, D.A.; Čanadanović-Brunet, J.; Vulić, J.; Stajčić, S.; Vinčić, M. Chemical composition and potential bioactivity of strawberry pomace. RSC Adv. 2015, 5, 5397–5405. [Google Scholar] [CrossRef]
- Mena, P.; Ascacio-Valdés, J.A.; Gironés-Vilaplana, A.; del Rio, D.; Moreno, D.A.; García-Viguera, C. Assessment of pomegranate wine lees as a valuable source for the recovery of (poly)phenolic compounds. Food Chem. 2014, 145, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Čanadanović-Brunet, J.M.; Savatović, S.M.; Ćetković, G.S.; Vulić, J.J.; Djilas, S.M.; Markov, S.L.; Cvetković, D.D. Antioxidant and Antimicrobial Activities of Beet Root Pomace Extracts. Czech J. Food Sci. 2011, 29, 575–585. [Google Scholar]
- Kujala, T.S.; Loponen, J.M.; Klika, K.D.; Pihlaja, K. Phenolics and betacyanins in red beetroot (Beta vulgaris) root: Distribution and effect of cold storage on the content of total phenolics and three individual compounds. J. Agric. Food Chem. 2000, 48, 5338–5342. [Google Scholar] [CrossRef] [PubMed]
- Vulić, J.J.; Ćebović, T.N.; Čanadanović-Brunet, J.M.; Ćetković, G.S.; Čanadanović, V.M.; Djilas, S.M.; Tumbas Šaponjac, V.T. In vivo and in vitro antioxidant effects of beetroot pomace extracts. J. Functional Foods 2014, 6, 168–175. [Google Scholar] [CrossRef]
- Vulić, J.J.; Ćebović, T.N.; Čanadanović, V.M.; Ćetković, G.S.; Djilas, S.M.; Čanadanović-Brunet, J.M.; Velićanski, A.S.; Cvetković, D.D.; Tumbas Šaponjac, V.T. Antiradical, antimicrobial and cytotoxic activities of commercial beetroot pomace. Food Funct. 2013, 4, 713–721. [Google Scholar] [CrossRef] [PubMed]
- Pedreno, M.A.; Escribano, J. Correlation between antiradical activity and stability of betanine from Beta vulgaris L roots under different pH, temperature and light conditions. J. Sci. Food Agric. 2001, 81, 627–631. [Google Scholar]
- Champagne, C.P.; Fustier, P. Microencapsulation for the improved delivery of bioactive compounds into foods. Curr. Opin. Biotechnol. 2007, 18, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Thies, C. A survey of microencapsulation processes. In Microencapsulation: Methods and Industrial Applications; Benita, S., Ed.; Marcel Dekker Inc: New York, NY, USA, 1996; pp. 133–154. [Google Scholar]
- Shahidi, F.; Han, X.Q. Encapsulation of food ingredients. Crit. Rev. Food Sci. Nutr. 1993, 33, 501–547. [Google Scholar] [CrossRef] [PubMed]
- Franzen, K.L.; Kinsella, J.E. Functional properties of succinylated and acetylated soy protein. J. Agric. Food Chem. 1976, 24, 788–795. [Google Scholar] [CrossRef]
- Roopchand, D.E.; Krueger, C.G.; Moskal, K.; Fridlender, B.; Lila, M.A.; Raskin, I. Food-compatible method for the efficient extraction and stabilization of cranberry pomace polyphenols. Food Chem. 2013, 141, 3664–3669. [Google Scholar] [CrossRef] [PubMed]
- Orhan, D.D.; Hartevioğlu, A.; Küpeli, E.; Yesilada, E. In vivo anti-inflammatory and antinociceptive activity of the crude extract and fractions from Rosa canina L. fruits. J. Ethnopharmacol. 2007, 112, 394–400. [Google Scholar] [CrossRef] [PubMed]
- Caldwell, C.R.; Britz, S.J.; Mirecki, R.M. Effect of temperature, elevated carbon dioxide, and drought during seed development on the isoflavone content of dwarf soybean (Glycine max (L.) Merrill) grown in controlled environments. J. Agric. Food Chem. 2005, 53, 1125–1129. [Google Scholar] [CrossRef] [PubMed]
- Marmion, D.M. Handbook of US Colorants: Foods, Drugs, Cosmetics, and Medicinal Devices; Wiley-Interscience: New York, NY, USA, 1991; pp. 154–196. [Google Scholar]
- Georgiev, V.G.; Weber, J.; Kneschke, E.; Denev, P.N.; Bley, T.; Pavlov, A.I. Antioxidant activity and phenolic content of betalain extracts from intact plants and hairy root cultures of the beetroot Beta vulgaris cv. Detroit dark red. Plant Foods Hum. Nutr. 2010, 65, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Eder, B. Pigments. In Handbook of Food Analysis; Nollet, L.M.L., Ed.; CRC: Boca Raton, FL, USA, 2004; Volume 1, pp. 805–878. [Google Scholar]
- Kugler, F.; Stintzing, C.F.; Carle, R. Evaluation of the antioxidant capacity of betalanic fruits and vegetables. J. Appl. Bot. Food Qual. 2007, 81, 69–76. [Google Scholar]
- Ramírez, M.J.; Giraldo, G.I.; Orrego, C.E. Modeling and stability of polyphenol in spray-dried and freeze-dried fruit encapsulates. Powder Technol. 2015, 277, 89–96. [Google Scholar] [CrossRef]
- Ezhilarasi, P.N.; Indrani, D.; Jena, B.S.; Anandharamakrishnan, C. Freeze drying technique for microencapsulation of Garcinia fruit extract and its effect on bread quality. J. Food Eng. 2013, 117, 513–520. [Google Scholar] [CrossRef]
- Saikia, S.L.; Mahnot, N.K.; Mahanta, C.L. Optimisation of phenolic extraction from Averrhoa carambola pomace by response surface methodology and its microencapsulation by spray and freeze drying. Food Chem. 2015, 171, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Šaponjac, V.T.; Ćetković, G.; Čanadanović-Brunet, J.; Pajin, B.; Djilas, S.; Petrović, J.; Lončarević, I.; Stajčić, S.; Vulić, J. Sour cherry pomace extract encapsulated in whey and soy proteins: Incorporation in cookies. Food Chem. 2016, 207, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Cheng, D.M.; Kuhn, P.; Poulev, A.; Rojo, L.E.; Lila, M.A.; Raskin, I. In vivo and in vitro antidiabetic effects of aqueous cinnamon extract and cinnamon polyphenol-enhanced food matrix. Food Chem. 2012, 135, 2994–3002. [Google Scholar] [CrossRef] [PubMed]
- Robert, P.; Gorena, T.; Romero, N.; Sepulveda, E.; Chavez, J.; Sáenz, C. Encapsulation of polyphenols and anthocyanins from pomegranate (Punica granatum) by spray drying. Int. J. Food Sci. Tech. 2010, 45, 1386–1394. [Google Scholar] [CrossRef]
- Robert, P.; Torres, V.; García, P.; Vergara, C.; Sáenz, C. The encapsulation of purple cactus pear (Opuntia ficus-indica) pulp by using polysaccharide-proteins as encapsulating agents. LWT-Food Sci. Technol. 2015, 60, 1039–1045. [Google Scholar] [CrossRef]
- Changchub, L.; Maisuthisakul, P. Thermal Stability of Phenolic Extract and Encapsulation from Mango Seed Kernel. Agricultural Sci. J. 2011, 42, 397–400. [Google Scholar]
- Janiszewska, E. Microencapsulated beetroot juice as a potential source of betalain. Powder Tech. 2014, 264, 190–196. [Google Scholar] [CrossRef]
- Gandía-Herrero, F.; Jiménez-Atiénzar, M.; Cabanes, J.; García-Carmona, F.; Escribano, J. Stabilization of the Bioactive Pigment of Opuntia Fruits through Maltodextrin Encapsulation. J. Agric. Food Chem. 2010, 58, 10646–10652. [Google Scholar] [CrossRef] [PubMed]
- Meir, S.; Kanner, J.; Akiri, B.; Hadas, S.P. Determination and involvement of aqueous reducing compounds in oxidative defense systems of various senescing leaves. J. Agric. Food Chem. 1995, 43, 1813–1819. [Google Scholar] [CrossRef]
- Sáenz, C.; Tapia, S.; Chávez, J.; Robert, P. Microencapsulation by spray drying of bioactive compounds from cactus pear (Opuntia ficus-indica). Food Chem. 2009, 114, 616–622. [Google Scholar] [CrossRef]
- Kaimainen, M.; Laaksonen, O.; Järvenpää, E.; Sandell, M.; Huopalahti, R. Consumer acceptance and stability of spray dried betanin in model juices. Food Chem. 2015, 187, 398–406. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Corke, H. Amaranthus betacyanin pigments applied in model food systems. J. Food Sci. 1999, 64, 869–873. [Google Scholar] [CrossRef]
- Vergara, C.; Saavedra, J.; Sáenz, C.; García, P.; Robert, P. Microencapsulation of pulp and ultrafiltrated cactus pear (Opuntia ficus-indica) extracts and betanin stability during storage. Food Chem. 2014, 157, 246–251. [Google Scholar] [CrossRef] [PubMed]
- Flores, F.P.; Singh, R.K.; Kong, F. Physical and storage properties of spray-dried blueberry pomace extract with whey protein isolate as wall material. J. Food Eng. 2014, 137, 1–6. [Google Scholar] [CrossRef]
- Işık, N.; Alteheld, B.; Kühn, S.; Schulze-Kaysers, N.; Kunz, B.; Wollseifen, H.R.; Stehle, P.; Lesser, S. Polyphenol release from protein and polysaccharide embedded plant extracts during in vitro digestion. Food Res. Int. 2014, 65, 109–114. [Google Scholar] [CrossRef]
- Saura-Calixto, F.; Serrano, J.; Goñi, I. Intake and bioaccessibility of total polyphenols in a whole diet. Food Chem. 2007, 101, 492–501. [Google Scholar] [CrossRef]
- Siracusa, L.; Kulisic-Bilusic, T.; Politeo, O.; Krause, I.; Dejanovic, B.; Ruberto, G. Phenolic composition and antioxidant activity of aqueous infusions from Capparis spinosa L. and Crithmum maritimum L. before and after submission to a two-step in vitro digestion model. J. Agric. Food Chem. 2011, 59, 12453–12459. [Google Scholar] [CrossRef] [PubMed]
- Ravichandran, K.; Palaniraj, R.; Saw, N.M.M.T.; Gabr, A.M.M.; Ahmed, A.R.; Knorr, D.; Smetanska, I. Effects of different encapsulation agents and drying process on stability of betalains extract. J. Food Sci. Technol. 2014, 51, 2216–2221. [Google Scholar] [CrossRef] [PubMed]
- González-Molina, E.; Moreno, D.A.; García-Viguera, C. Genotype and harvest time influence the phytochemical quality of Fino lemon juice (Citrus limon (L.) Burm. F.) for industrial use. J. Agric. Food Chem. 2008, 56, 1669–1675. [Google Scholar] [CrossRef] [PubMed]
- Markham, K.R. Flavones, flavonols and their glycosides. In Methods in Plant Biochemistry; Harborne, J.B., Dey, P.M., Eds.; Academic Press: London, UK, 1989; pp. 193–237. [Google Scholar]
- Von Elbe, J.H. Betalains. In Handbook of Food Analytical Chemistry. Pigments, Colorants, Flavors, Texture, and Bioactive Food Components; Wrolstad, R.E., Acree, T.E., Decker, E.A., Penner, M.H., Reid, D.S., Schwartz, S.J., Shoemaker, C.F., Smith, D., Sporns, P., Eds.; John Wiley and Sons, Inc: New York, NY, USA, 2003; F.3.1.1.-F.3.1.7.; pp. 123–131. [Google Scholar]
- Francis, G.W.; Andersen, Ø.M. Natural pigments. In Handbook of Thin-Layer Chromatography, Third Edition, Revised and Expanded; Sherman, J., Fried, B., Eds.; Marcel Dekker Inc: New York, NY, USA, 2003; pp. 911–961. [Google Scholar]
- Girones-Vilaplana, A.; Mena, P.; Moreno, D.A.; Garcia-Viguera, C. Evaluation of sensorial, phytochemical and biological properties of new isotonic beverages enriched with lemon and berries during shelf life. J. Sci. Food Agric. 2014, 94, 1090–1100. [Google Scholar] [CrossRef] [PubMed]
- Oyaizu, M. Studies on product of browning reaction from glucose amine. Jpn. J. Nutr. 1986, 44, 307–315. [Google Scholar] [CrossRef]
- Bakowska-Barczak, A.M.; Kolodziejczyk, P.P. Black currant polyphenols: Their storage stability and microencapsulation. Ind. Crops Prod. 2011, 34, 1301–1309. [Google Scholar] [CrossRef]
- Ji, Y.; Zhu, K.; Chen, Z.-C.; Zhou, H.; Ma, J.; Qian, H. Effects of different additives on rice cake texture and cake staling. J. Texture Stud. 2010, 41, 703–713. [Google Scholar]
- González-Molina, E.; Moreno, D.A.; García-Viguera, C. Aronia-Enriched Lemon Juice: A New Highly Antioxidant Beverage. J. Agric. Food Chem. 2008, 56, 11327–11333. [Google Scholar] [CrossRef] [PubMed]
- U.S. Pharmacopeia. Test Solutions, 2012. Available online: http://www.pharmacopeia.cn (accessed on 30 April 2016).
- Sample Availability: Not available.


| Bioactive Compounds/Bioactivity | Content (per 100 g Pomace Dry Weight) |
|---|---|
| Polyphenols a | 89.06 ± 4.62 |
| Flavonoids b | 4.74 ± 0.16 |
| Betacyanins c | 79.22 ± 2.04 |
| Betaxanthins d | 80.06 ± 2.06 |
| Gallic acid e | 7.45 ± 0.24 |
| Protocatechuic acid e | 11.53 ± 0.40 |
| p-Hydroxybenzoic acid e | 0.78 ± 0.02 |
| Catechin e | 53.42 ± 1.53 |
| Epicatechin e | 2.09 ± 0.08 |
| Chlorogenic acid e | 0.30 ± 0.01 |
| Caffeic acid e | 0.18 ± 0.00 |
| Ferulic acid e | 0.48 ± 0.02 |
| Sinapic acid e | 0.52 ± 0.03 |
| Coumaric acid e | 0.34 ± 0.01 |
| Myricetin e | 3.52 ± 0.14 |
| Luteolin e | 0.03 ± 0.00 |
| Quercetin e | 0.22 ± 0.10 |
| Apigenin e | 0.10 ± 0.00 |
| Isorhamnetin e | 0.35 ± 0.01 |
| Betanin e | 88.94 ± 3.57 |
| Vulgaxanthin-I e | 71.08 ± 1.33 |
| SA f | 59.75 ± 2.09 |
| RP f | 206.84 ± 9.81 |
| Experiment | Wall–Core Ratio (X1, g/L) | Extract Dilution (X2) | Mixing Time (X3, min) | EE (%) a | SA (µmol TE/100 g) a |
|---|---|---|---|---|---|
| 1 | 50 (−1) | 0 (−1) | 15 (0) | 84.41 ± 1.56 | 1619.22 ± 58.64 |
| 2 | 150 (+1) | 0 (−1) | 15 (0) | 77.54 ± 1.49 | 947.13 ± 29.74 |
| 3 | 50 (−1) | 4 (+1) | 15 (0) | 69.69 ± 4.64 | 772.19 ± 11.40 |
| 4 | 150 (+1) | 4 (+1) | 15 (0) | 74.18 ± 3.09 | 339.76 ± 3.96 |
| 5 | 50 (−1) | 2 (0) | 5 (−1) | 82.72 ± 5.50 | 959.41 ± 18.98 |
| 6 | 150 (+1) | 2 (0) | 5 (−1) | 77.69 ± 5.00 | 316.30 ± 10.47 |
| 7 | 50 (−1) | 2 (0) | 25 (+1) | 75.40 ± 4.18 | 998.77 ± 47.09 |
| 8 | 150 (+1) | 2 (0) | 25 (+1) | 60.26 ± 0.32 | 254.67 ± 24.62 |
| 9 | 100 (0) | 0 (−1) | 5 (−1) | 85.03 ± 2.00 | 921.69 ± 14.62 |
| 10 | 100 (0) | 4 (+1) | 5 (−1) | 76.36 ± 3.95 | 223.61 ± 14.17 |
| 11 | 100 (0) | 0 (−1) | 25 (+1) | 78.16 ± 4.17 | 1077.27 ± 29.22 |
| 12 | 100 (0) | 4 (+1) | 25 (+1) | 64.23 ± 0.15 | 166.96 ± 19.77 |
| 13 | 100 (0) | 2 (0) | 15 (0) | 75.87 ± 4.75 | 630.29 ± 24.48 |
| 14 | 100 (0) | 2 (0) | 15 (0) | 75.69 ± 3.46 | 604.68 ± 32.94 |
| 15 | 100 (0) | 2 (0) | 15 (0) | 76.48 ± 0.90 | 776.99 ± 0.28 |
| Optimization | Variable Codes | Variable Values | Optimal Responses | |||||
|---|---|---|---|---|---|---|---|---|
| X1 | X2 | X3 | X1 | X2 | X3 | EE (%) | SA (µmol/100 g) | |
| Single response (EE) | −0.99 | −0.89 | −0.09 | 50.50 | 0.22 | 14.1 | 86.16 | - |
| Single response (SA) | −0.98 | −0.98 | 0.10 | 51.00 | 0.04 | 16.00 | - | 1676.17 |
| Multi response (EE + SA) | −0.98 | −0.98 | 0.36 | 51.00 | 0.04 | 18.60 | 86.14 | 1668.37 |
| Bioactive Compounds/Bioactivity | Content (per 100 g OE) |
|---|---|
| Polyphenols a | 326.51 ± 3.00 |
| Flavonoids b | 10.23 ± 1.22 |
| Betacyanins c | 60.52 ± 0.92 |
| Betaxanthins d | 61.33 ± 0.92 |
| Gallic acid e | 26.54 ± 0.89 |
| Protocatechuic acid e | 65.58 ± 2.65 |
| p-Hydroxybenzoic acide | 30.18 ± 0.02 |
| Catechin e | 156.04 ± 1.03 |
| Epicatechin e | 3.88 ± 0.05 |
| Chlorogenic acid e | 0.55 ± 0.01 |
| Caffeic acid e | 0.34 ± 0.00 |
| Ferulic acid e | 0.89 ± 0.02 |
| Sinapic acid e | 0.97 ± 0.01 |
| Coumaric acid e | 0.64 ± 0.01 |
| Myricetin e | 6.55 ± 0.09 |
| Luteolin e | 0.06 ± 0.00 |
| Quercetin e | 0.41 ± 0.08 |
| Apigenin e | 0.18 ± 0.00 |
| Isorhamnetin e | 0.65 ± 0.01 |
| Betanin e | 65.36 ± 2.18 |
| Vulgaxanthin-I e | 59.29 ± 2.05 |
| SA f | 1655.36 ± 80.98 |
| RP f | 394.95 ± 13.05 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tumbas Šaponjac, V.; Čanadanović-Brunet, J.; Ćetković, G.; Jakišić, M.; Djilas, S.; Vulić, J.; Stajčić, S. Encapsulation of Beetroot Pomace Extract: RSM Optimization, Storage and Gastrointestinal Stability. Molecules 2016, 21, 584. https://doi.org/10.3390/molecules21050584
Tumbas Šaponjac V, Čanadanović-Brunet J, Ćetković G, Jakišić M, Djilas S, Vulić J, Stajčić S. Encapsulation of Beetroot Pomace Extract: RSM Optimization, Storage and Gastrointestinal Stability. Molecules. 2016; 21(5):584. https://doi.org/10.3390/molecules21050584
Chicago/Turabian StyleTumbas Šaponjac, Vesna, Jasna Čanadanović-Brunet, Gordana Ćetković, Mirjana Jakišić, Sonja Djilas, Jelena Vulić, and Slađana Stajčić. 2016. "Encapsulation of Beetroot Pomace Extract: RSM Optimization, Storage and Gastrointestinal Stability" Molecules 21, no. 5: 584. https://doi.org/10.3390/molecules21050584