Pyrrole and Fused Pyrrole Compounds with Bioactivity against Inflammatory Mediators
Abstract
:1. Introduction
2. Results
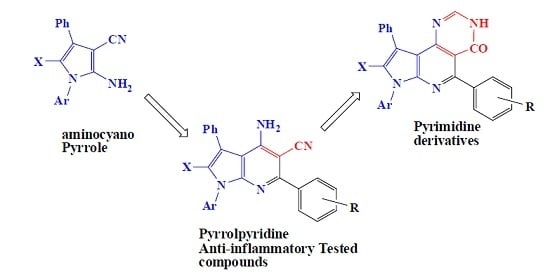
2.1. Chemistry
2.2. Anti-Inflammatory Assays
2.3. Docking and Modelling
2.4. Inflammatory Mediators Assay
3. Discussion
4. Materials and Methods
4.1. General Information
4.2. Synthesis
4.2.1. General Procedure for the Synthesis of Compounds 3a–l
4.2.2. General Procedure for the Synthesis of Compounds 4a–l
4.3. Anti-Inflammatory Activity
4.4. Docking and Modelling Methods
4.5. Determination of Inflammatory Mediators
4.5.1. Determination of Immunoglobulin E (IgE)
4.5.2. Determination of Interleukin 1β (IL-1β)
4.5.3. Determination of Tumor Necrosis Factor α (TNF-α)
4.5.4. Determination of C Reactive Protein (CRP)
4.5.5. Determination of Histamine
4.6. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gasparini, L.; Ongini, E.; Wenk, G. Non-steroidal anti-inflammatory drugs (NSAIDs) in Alzheimer’s disease: Old and new mechanisms of action. J. Neurochem. 2004, 91, 521–536. [Google Scholar] [CrossRef] [PubMed]
- Nagao, M.; Sato, Y.; Yamauchi, A. Meta-Analysis of Interleukin Polymorphisms and NSAID Usage Indicates Correlations to the Risk of Developing Cancer. Int. J. Genom. Med. 2014, 2, 1–7. [Google Scholar]
- Perković, I.; Džolić, Z.R.; Zorc, B. A convenient synthesis of new NSAID esters containing amino acid, urea and amide moieties. Acta Pharm. 2013, 63, 409–418. [Google Scholar] [CrossRef] [PubMed]
- Kotake, S.; Yago, T.; Kawamoto, M.; Nanke, Y. Effects of NSAIDs on Differentiation and Function of Human and Murine Osteoclasts—Crucial “Human Osteoclastology”. Pharmaceuticals 2010, 3, 1394–1410. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.; Walker, A.; Williams, J.; Hasty, K.A. Study of Osteoarthritis Treatment with Anti-Inflammatory Drugs: Cyclooxygenase-2 Inhibitor and Steroids. Biomed. Res. Int. 2015, 2015, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Bansal, Y.; Silakari, O. Multifunctional compounds: Smart molecules for multifactorial diseases. Eur. J. Med. Chem. 2014, 76, 31–42. [Google Scholar] [CrossRef] [PubMed]
- Ajmone-Cat, M.A.; Bernardo, A.; Greco, A.; Minghetti, L. Non-steroidal anti-inflammatory drugs and brain inflammation: Effects on microglial functions. Pharmaceuticals 2010, 3, 1949–1964. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, E.; Costa, D.; Toste, S.A.; Lima, J.L.F.C.; Reis, S. In vitro scavenging activity for reactive oxygen and nitrogen species by nonsteroidal anti-inflammatory indole, pyrrole, and oxazole derivative drugs. Free Radic. Biol. Med. 2004, 37, 1895–1905. [Google Scholar] [CrossRef] [PubMed]
- Ushiyama, S.; Yamada, T.; Murakami, Y.; Kumakura, S.I.; Inoue, S.I.; Suzuki, K.; Nakao, A.; Kawara, A.; Kimura, T. Preclinical pharmacology profile of CS-706, a novel cyclooxygenase-2 selective inhibitor, with potent antinociceptive and anti-inflammatory effects. Eur. J. Pharmacol. 2008, 578, 76–86. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.C.; Yadav, S.; Pahwa, R.; Kaushik, D.; Jain, S. Synthesis and evaluation of novel prodrugs of naproxen. Med. Chem. Res. 2011, 20, 648–655. [Google Scholar] [CrossRef]
- Praveen Rao, P.N.; Knaus, E.E. Evolution of nonsteroidal anti-inflammatory drugs (NSAIDs): Cyclooxygenase (COX) inhibition and beyond. J. Pharm. Pharm. Sci. 2008, 11, 81–110. [Google Scholar]
- Sánchez-Borges, M.; Caballero-Fonseca, F.; Capriles-Hulett, A.; González-Aveledo, L. Hypersensitivity Reactions to Nonsteroidal Anti-Inflammatory Drugs: An Update. Pharmaceuticals 2010, 3, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Joshi, S.D.; More, U.A.; Dixit, S.R.; Korat, H.H.; Aminabhavi, T.M.; Badiger, A.M. Synthesis, characterization, biological activity, and 3D-QSAR studies on some novel class of pyrrole derivatives as antitubercular agents. Med. Chem. Res. 2013, 23, 1123–1147. [Google Scholar] [CrossRef]
- Dannhardt, G.; Kiefera, W.; Krämera, G.; Maehrlein, S.; Nowe, U.; Fiebich, B. The pyrrole moiety as a template for COX-1/COX-2 inhibitors. Eur. J. Med. Chem. 2000, 35, 499–510. [Google Scholar] [CrossRef]
- Sarg, M.T.; Bayoumi, M.M.K.; Gilil, A.H.; El, S.M.A. Synthesis of Pyrroles and Condensed Pyrroles as Anti-Inflammatory Agents with Multiple Activities and Their Molecular Docking Study. Open J. Med. Chem. 2015, 49–96. [Google Scholar] [CrossRef]
- Said, S.A.; Amr, E.G.E.; Sabry, N.M.; Abdalla, M.M. Analgesic, anticonvulsant and anti-inflammatory activities of some synthesized benzodiazipine, triazolopyrimidine and bis-imide derivatives. Eur. J. Med. Chem. 2009, 44, 4787–4792. [Google Scholar] [CrossRef] [PubMed]
- Connolly, S.; Aberg, A.; Arvai, A.; Beaton, H.G.; Cheshire, D.R.; Cook, A.R.; Cooper, S.; Cox, D.; Hamley, P.; Mallinder, P.; et al. 2-Aminopyridines as Highly Selective Inducible Nitric Oxide Synthase Inhibitors. Differential Binding Modes Dependent on Nitrogen Substitution. J. Med. Chem. 2004, 47, 3320–3323. [Google Scholar] [CrossRef] [PubMed]
- Bukhari, S.N.A.; Jantan, I.; Jasamai, M. Anti-inflammatory trends of 1,3-diphenyl-2-propen-1-one derivatives. Mini Rev. Med. Chem. 2013, 13, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, M.S.; Fathallah, S.S. Pyrroles and Fused Pyrroles: Synthesis and Therapeutic Activities. Mini Rev. Org. Chem. 2014, 6, 477–507. [Google Scholar] [CrossRef]
- Han, W.B.; Zhang, A.H.; Deng, X.Z.; Lei, X.; Tan, R.X. Curindolizine, an Anti-Inflammatory Agent Assembled via Michael Addition of Pyrrole Alkaloids Inside Fungal Cells. Org. Lett. 2016, 18, 1816–1819. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.T.; Mou, X.Q.; Xi, Q.M.; Liu, W.T.; Liu, W.F.; Sheng, Z.J.; Zheng, X.; Zhang, K.; Du, Z.Y.; Zhao, S.Q.; et al. Anti-inflammatory activity effect of 2-substituted-1,4,5,6-tetrahydrocyclopenta[b]pyrrole on TPA-induced skin inflammation in mice. Bioorg. Med. Chem. Lett. 2016, 26, 5334–5339. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Kim, Y.; Jeong, D.; Kim, J.H.; Kim, S.; Son, Y.; Yoo, B.C.; Jeong, E.J.; Kim, T.W.; Lee, I.H.; et al. Pyrrole-Derivative of Chalcone, (E)-3-Phenyl-1-(2-Pyrrolyl)-2-Propenone, Inhibits Inflammatory Responses via Inhibition of Src, Syk, and TAK1 Kinase Activities. Biomol. Ther. 2016, 24, 595–603. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, P.; Kishore Kumar, M.; Narasu, L.; Gundla, R.; Samanta, S.; Cuthbertson, C.; Neamati, N. Designing novel MEK1 inhibitors as anticancer agents. Int. J. Life Sci. Pharm. Res. 2016, 6, 23–33. [Google Scholar]
- Tintori, C.; Magnani, M.; Schenone, S.; Botta, M. Docking, 3D-QSAR studies and in silico ADME prediction on c-Src tyrosine kinase inhibitors. Eur. J. Med. Chem. 2009, 44, 990–1000. [Google Scholar] [CrossRef] [PubMed]
- Danchev, N.; Bijev, A.; Yaneva, D.; Vladimirova, S.; Nikolova, I. Synthesis, acute toxicity, and analgesic activity of new derivatives of pyrrole. Arch. Pharm. (Weinheim) 2006, 339, 670–674. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Yu, H.; Lai, T.; Chiou, W.; Don, M. Synthesis and anti-inflammatory effect of four rutaecarpine metabolites. J. Chin. Med. 2011, 22, 37–46. [Google Scholar]
- Eung, S.L.; Seung, I.K.; Seung, H.L.; Tae, C.J.; Tae, C.M.; Hyeun, W.C.; Jahng, Y. Synthesis and COX inhibitory activities of rutaecarpine derivatives. Bull. Korean Chem. Soc. 2005, 26, 1975–1980. [Google Scholar]
- Bocheva, A.; Bijev, A.; Nankov, A. Further evaluation of a series of anti-inflammatory N-pyrrolylcarboxylic acids: Effects on the nociception in rats. Arch. Pharm. (Weinheim) 2006, 339, 141–144. [Google Scholar] [CrossRef] [PubMed]
- Lessigiarska, I.; Nankov, A.; Bocheva, A.; Pajeva, I.; Bijev, A. 3D-QSAR and preliminary evaluation of anti-inflammatory activity of series of N-pyrrolylcarboxylic acids. Farmaco 2005, 60, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Nakao, A.; Ohkawa, N.; Nagasaki, T.; Kagari, T.; Doi, H.; Shimozato, T.; Ushiyama, S.; Aoki, K. Tetrahydropyridine derivatives with inhibitory activity on the production of proinflammatory cytokines: part 1. Bioorg. Med. Chem. Lett. 2009, 19, 4607–4610. [Google Scholar] [CrossRef] [PubMed]
- Gallelli, L.; Galasso, O.; Falcone, D.; Southworth, S.; Greco, M.; Ventura, V.; Romualdi, P.; Corigliano, A.; Terracciano, R.; Savino, R.; et al. The effects of nonsteroidal anti-inflammatory drugs on clinical outcomes, synovial fluid cytokine concentration and signal transduction pathways in knee osteoarthritis. A randomized open label trial. Osteoarthr. Cartil. 2013, 21, 1400–1408. [Google Scholar] [CrossRef] [PubMed]
- Page, T.H.; Turner, J.J.O.; Brown, A.C.; Timms, E.M.; Inglis, J.J.; Brennan, F.M.; Foxwell, B.M.J.; Ray, K.P.; Feldmann, M. Nonsteroidal Anti-Inflammatory Drugs Increase TNF Production in Rheumatoid Synovial Membrane Cultures and Whole Blood. J. Immunol. 2010, 185, 3694–3701. [Google Scholar] [CrossRef] [PubMed]
- Cole, G.M.; Frautschy, S.A. Mechanisms of action of non-steroidal anti-inflammatory drugs for the prevention of Alzheimer’s disease. CNS Neurol. Disord. Drug Targets 2010, 9, 140–148. [Google Scholar] [CrossRef] [PubMed]
- Rao, P.P.N.; Kabir, S.N.; Mohamed, T. Nonsteroidal Anti-Inflammatory Drugs (NSAIDs): Progress in Small Molecule Drug Development. Pharmaceuticals 2010, 3, 1530–1549. [Google Scholar] [CrossRef] [PubMed]
- Lucetti, D.L.; Lucetti, E.C.; Bandeira, M.; Veras, H.N.; Silva, A.H.; Leal, L.; Lopes, A.A.; Alves, V.C.; Silva, G.S.; Brito, G.; et al. Anti-inflammatory effects and possible mechanism of action of lupeol acetate isolated from Himatanthus drasticus (Mart.) Plumel. J. Inflamm. 2010, 7, 60. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.; Qadri, S.M.; Liu, L. Inhibition of nitric oxide synthesis enhances leukocyte rolling and adhesion in human microvasculature. J. Inflamm. 2012, 9, 28. [Google Scholar] [CrossRef] [PubMed]
- Shao, Y.; Cheng, Z.; Li, X.; Chernaya, V.; Wang, H.; Yang, X.-F. Immunosuppressive/anti-inflammatory cytokines directly and indirectly inhibit endothelial dysfunction—A novel mechanism for maintaining vascular function. J. Hematol. Oncol. 2014, 7, 80. [Google Scholar] [CrossRef] [PubMed]
- Rashad, A.E.; Mohamed, M.S.; Zaki, M.E.A.; Fatahala, S.S. Synthesis and biological evaluation of some pyrrolo[2,3-d]pyrimidines. Arch. Pharm. (Weinheim) 2006, 339, 664–669. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, M.S.; Rashad, A.E.; Zaki, M.E.A.; Fatahala, S.S. Synthesis and antimicrobial screening of some fused heterocyclic pyrroles. Acta Pharm. 2005, 55, 237–249. [Google Scholar] [PubMed]
- Mohamed, M.S.; Kamel, R.; Fathallah, S.S. Synthesis of new pyrroles of potential anti-inflammatory activity. Arch. Pharm. (Weinheim) 2011, 344, 830–839. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, M.S.; Ali, S.A.; Abdelaziz, D.H.A.; Fathallah, S.S. Synthesis and Evaluation of Novel Pyrroles and Pyrrolopyrimidines as Antihypoglycemic Agents. BioMed Res. Int. 2014, 2014, 249780. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, M.S.; Kamel, R.; Abd El-hameed, R.H. Evaluation of the anti-inflammatory activity of some pyrrolo[2,3-d]pyrimidine derivatives. Med. Chem. Res. 2012, 22, 2244–2252. [Google Scholar] [CrossRef]
- Zheng, G.Z.; Mao, Y.; Lee, C.H.; Pratt, J.K.; Koenig, J.R.; Perner, R.J.; Cowart, M.D.; Gfesser, G.A.; McGaraughty, S.; Chu, K.L.; et al. Adenosine kinase inhibitors: Polar 7-substitutent of pyridopyrimidine derivatives improving their locomotor selectivity. Bioorg. Med. Chem. Lett. 2003, 13, 3041–3044. [Google Scholar] [CrossRef]
- Zheng, G.Z.; Lee, C.H.; Pratt, J.K.; Perner, R.J.; Jiang, M.Q.; Gomtsyan, A.; Matulenko, M.A.; Mao, Y.; Koenig, J.R.; Kim, K.H.; et al. Pyridopyrimidine analogues as novel adenosine kinase inhibitors. Bioorg. Med. Chem. Lett. 2001, 11, 2071–2074. [Google Scholar] [CrossRef]
- Rashad, A.E.; Sayed, H.H.; Shamroukh, A.H.; Awad, H.M. Preparation of Some Fused Pyridopyrimidine and Pyridothienotriazine Derivatives for Biological Evaluation. Phosphorus Sulfur Silicon Relat. Elem. 2005, 180, 2767–2777. [Google Scholar] [CrossRef]
- Nofal, Z.M.; Fahmy, H.H.; Zarea, E.S.; El-eraky, W. Drug Synthesis Synthesis of New Pyrimidine Derivatives With Evaluation of Their Anti-Inflammatory and Analgesic Activities. Acta Pol. Pharm. 2011, 68, 507–517. [Google Scholar] [PubMed]
- Harrak, Y.; Rosell, G.; Daidone, G.; Plescia, S.; Schillaci, D.; Pujol, M.D. Synthesis and biological activity of new anti-inflammatory compounds containing the 1,4-benzodioxine and/or pyrrole system. Bioorg. Med. Chem. 2007, 15, 4876–4890. [Google Scholar] [CrossRef] [PubMed]
- Khedr, M.A.; Shehata, T.M.; Mohamed, M.E. Repositioning of 2,4-Dichlorophenoxy acetic acid as a potential anti-inflammatory agent: In Silico and Pharmaceutical Formulation study. Eur. J. Pharm. Sci. 2014, 65, 130–138. [Google Scholar] [CrossRef] [PubMed]
- Housby, J.N.; Cahill, C.M.; Chu, B.; Prevelige, R.; Bickford, K.; Stevenson, M.A.; Calderwood, S.K. Non-steroidal anti-inflammatory drugs inhibit the expression of cytokines and induce HSP70 in human monocytes. Cytokine 1999, 11, 347–358. [Google Scholar] [CrossRef] [PubMed]
- De Cássia da Silveira e Sá, R.; Andrade, L.; de Sousa, D. A Review on Anti-Inflammatory Activity of Monoterpenes. Molecules 2013, 18, 1227–1254. [Google Scholar] [CrossRef] [PubMed]
- Da Silveira E Sá, R.D.C.; Andrade, L.N.; de Oliveira, R.D.R.B.; De Sousa, D.P. A review on anti-inflammatory activity of phenylpropanoids found in essential oils. Molecules 2014, 19, 1459–1480. [Google Scholar] [CrossRef] [PubMed]
- Strober, W.; Fuss, I.J. Proinflammatory Cytokines in the Pathogenesis of Inflammatory Bowel Diseases. Gastroenterology 2011, 140, 1756–1767. [Google Scholar] [CrossRef] [PubMed]
- Shaikh, P.Z. Cytokines & their physiologic and pharmacologic functions in inflammation: A review. Int. J. Pharm. Life Sci. 2011, 2, 1247–1263. [Google Scholar]
- Arshad, S.H.; Holgate, S. The role of IgE in allergen-induced inflammation and the potential for intervention with a humanized monoclonal anti-IgE antibody. Clin. Exp. Allergy 2001, 31, 1344–1351. [Google Scholar] [CrossRef] [PubMed]
- Zampeli, E.; Tiligada, E. The role of histamine H4 receptor in immune and inflammatory disorders. Br. J. Pharmacol. 2009, 157, 24–33. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, M.; Jørgensen, P.N.; Christoffersen, C.T.; Jensen, K.G.; Balle, T.; Bang-Andersen, B. Discovery of novel α₁-adrenoceptor ligands based on the antipsychotic sertindole suitable for labeling as PET ligands. Bioorg. Med. Chem. 2013, 21, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Tang, L.; Jennings, T.A.; Eaton, J.W. Mast cells mediate acute inflammatory responses to implanted biomaterials. Proc. Natl. Acad. Sci. USA 1998, 95, 8841–8846. [Google Scholar] [CrossRef] [PubMed]
- Elenkov, I.J. Neurohormonal-cytokine interactions: Implications for inflammation, common human diseases and well-being. Neurochem. Int. 2008, 52, 40–51. [Google Scholar] [CrossRef] [PubMed]
- Limban, C.; Missir, A.V.; Fahelelbom, K.M.S.; Al-Tabakha, M.M.; Caproiu, M.T.; Sadek, B. Novel N-phenylcarbamothioylbenzamides with anti-inflammatory activity and prostaglandin E 2 inhibitory properties. J. Drug Des. Dev. Ther. 2013, 7, 883–892. [Google Scholar]
- Mastorakos, G.; Karoutsou, E.I.; Mizamtsidi, M. Corticotropin releasing hormone and the immune/inflammatory response. Eur. J. Endocrinol. 2006, 155, S77–S84. [Google Scholar] [CrossRef]
- Wojdasiewicz, P.; Poniatowski, Ł.A.; Szukiewicz, D. The role of inflammatory and anti-inflammatory cytokines in the pathogenesis of osteoarthritis. Mediat. Inflamm. 2014, 2014, 561459. [Google Scholar] [CrossRef] [PubMed]
- Dinarello, C.A. Interleukin 1 and interleukin 18 as mediators of inflammation and the aging process. Am. J. Clin. Nutr. 2006, 83, 447S–455S. [Google Scholar] [PubMed]
- Teeling, J.L.; Cunningham, C.; Newman, T.A.; Perry, V.H. The effect of non-steroidal anti-inflammatory agents on behavioural changes and cytokine production following systemic inflammation: Implications for a role of COX-1. Brain Behav. Immun. 2010, 24, 409–419. [Google Scholar] [CrossRef] [PubMed]
- Toledo, T.R.; Dejani, N.N.; Monnazzi, L.G.S.; Kossuga, M.H.; Berlinck, R.G.S.; Sette, L.D.; Medeiros, A.I. Potent anti-inflammatory activity of pyrenocine A isolated from the marine-derived fungus Penicillium paxilli Ma(G)K. Mediat. Inflamm. 2014, 2014, 767061. [Google Scholar] [CrossRef] [PubMed]
- Buceta, M.; Domínguez, E.; Castro, M.; Brea, J.; Álvarez, D.; Barcala, J.; Valdés, L.; Álvarez-Calderón, P.; Domínguez, F.; Vidal, B.; et al. A new chemical tool (C0036E08) supports the role of adenosine A2B receptors in mediating human mast cell activation. Biochem. Pharmacol. 2008, 76, 912–921. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.-J.; Schwed, J.S.; Weizel, L.; De Jonghe, S.; Stark, H.; Herdewijn, P. Synthesis and evaluation of novel ligands for the histamine H₄ receptor based on a pyrrolo[2,3-d]pyrimidine scaffold. Bioorg. Med. Chem. Lett. 2013, 23, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Cocco, M.T.; Congiu, C.; Lilliu, V.; Onnis, V. Synthesis and in vitro antitumoral activity of new hydrazinopyrimidine-5-carbonitrile derivatives. Bioorg. Med. Chem. 2006, 14, 366–372. [Google Scholar] [CrossRef] [PubMed]
- Caram’s, B.; Lopez-Armada, M.J.; Cillero-Pastor, B.; Lires-Dean, M.; Vaamonde, C.; Galdo, F.; Blanco, F.J. Differential effects of tumor necrosis factor-α and interleukin-1β on cell death in human articular chondrocytes. Osteoarthr. Cartil. 2008, 16, 715–722. [Google Scholar] [CrossRef] [PubMed]
- Ozturk Bilgin, O.; Doguc, D.K.; Altuntas, I.; Sutcu, R.; Delibas, N. Effects of subchronic treatment with ibuprofen and nimesulide on spatial memory and NMDAR subunits expression in aged rats. Iran. J. Pharm. Res. 2013, 12, 877–885. [Google Scholar] [PubMed]
- Pouliot, M.; Cleland, L.; Caughey, C.E.; Pouliot, M.; Cleland, L.C.; James, M.J. Regulation of Tumor Necrosis Factor-α and IL-1β Synthesis by Thromboxane A2 in Nonadherent Human Monocytes. J. Immunol. 1997, 351–358. [Google Scholar]
- Ren, K.; Torres, R. Role of interleukin-1beta during pain and inflammation. Brain Res. Rev. 2009, 60, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Budhiraja, A.; Kadian, K.; Kaur, M.; Aggarwal, V.; Garg, A.; Sapra, S.; Nepali, K.; Suri, O.P.; Dhar, K.L. Synthesis and biological evaluation of naphthalene, furan and pyrrole based chalcones as cytotoxic and antimicrobial agents. Med. Chem. Res. 2011, 21, 2133–2140. [Google Scholar] [CrossRef]
- Sidney, L.E.; Heathman, T.R.J.; Britchford, E.R.; Abed, A.; Rahman, C.V.; Buttery, L.D.K. Investigation of Localized Delivery of Diclofenac Sodium from Poly(d,l-Lactic Acid-co-Glycolic Acid)/Poly(Ethylene Glycol) Scaffolds Using an In Vitro Osteoblast Inflammation Model. Tissue Eng. Part A 2015, 21, 362–373. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, M.S.; El-Domany, R.A.; Abd El-Hameed, R.H.; El-hameed, R.H.A.B.D.; Mohamed, M.S.; El-Domany, R.A.; Abd El-Hameed, R.H. Synthesis of certain pyrrole derivatives as antimicrobial agents. Acta Pharm. 2009, 59, 145–158. [Google Scholar] [CrossRef] [PubMed]
- Hussein, W.M.; Fatahala, S.S.; Mohamed, Z.M.; McGeary, R.P.; Schenk, G.; Ollis, D.L.; Mohamed, M.S. Synthesis and kinetic testing of tetrahydropyrimidine-2-thione and pyrrole derivatives as inhibitors of the metallo-β-lactamase from Klebsiella pneumonia and Pseudomonas aeruginosa. Chem. Biol. Drug Des. 2012, 80, 500–515. [Google Scholar] [CrossRef] [PubMed]
- Winter, C.A.; Risley, E.A.; Nuss, G.W. Anti-Inflammatory and Antipyretic Activities of Indomethacin, 1-(P-Chlorobenzoyl)-5-Methoxy-2-Methylindole-3-Acetic Acid. J. Pharmacol. Exp. Ther. 1963, 141, 369–376. [Google Scholar] [PubMed]
- Kawakita, Y.; Miwa, K.; Seto, M.; Banno, H.; Ohta, Y.; Tamura, T.; Yusa, T.; Miki, H.; Kamiguchi, H.; Ikeda, Y.; et al. Design and synthesis of pyrrolo[3,2-d]pyrimidine HER2/EGFR dual inhibitors: Improvement of the physicochemical and pharmacokinetic profiles for potent in vivo anti-tumor efficacy. Bioorg. Med. Chem. 2012, 20, 6171–6180. [Google Scholar] [CrossRef] [PubMed]
- CACC. Guide to the Care and Use of Experimental Animals; CCAC: Ottawa, ON, Canada, 1993; Volume 1. [Google Scholar]
- Standing, J.F.; Tibboel, D.; Korpela, R.; Olkkola, K.T. Diclofenac pharmacokinetic meta-analysis and dose recommendations for surgical pain in children aged 1–12 years. Paediatr. Anaesth. 2011, 21, 316–324. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.-N.; Li, H.; Yao, H.; Liu, X.; Li, L.; Leung, K.-S.; Kung, H.-F.; Lu, D.; Wong, M.-H.; Lin, M.C.-M. In Silico Identification and In Vitro and In Vivo Validation of Anti-Psychotic Drug Fluspirilene as a Potential CDK2 Inhibitor and a Candidate Anti-Cancer Drug. PLoS ONE 2015, 10, e0132072. [Google Scholar] [CrossRef] [PubMed]
- Ishizaka, T. IgE and mechanisms of IgE—Mediated hypersensitivity. Ann. Allergy 1982, 48, 313–319. [Google Scholar] [PubMed]
- El Bakali, J.; Muccioli, G.G.; Renault, N.; Pradal, D.; Body-Malapel, M.; Djouina, M.; Hamtiaux, L.; Andrzejak, V.; Desreumaux, P.; Chavatte, P.; et al. 4-Oxo-1,4-dihydropyridines as selective CB2 cannabinoid receptor ligands: structural insights into the design of a novel inverse agonist series. J. Med. Chem. 2010, 53, 7918–7931. [Google Scholar] [CrossRef] [PubMed]
- Krasilnikova, A.A.; Shestopalov, M.A.; Brylev, K.A.; Kirilova, I.A.; Khripko, O.P.; Zubareva, K.E.; Khripko, Y.I.; Podorognaya, V.T.; Shestopalova, L.V.; Fedorov, V.E.; et al. Prospects of molybdenum and rhenium octahedral cluster complexes as X-ray contrast agents. J. Inorg. Biochem. 2015, 144, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Mojtaba, E.; Somayeh, B.; Payman, A.; Davood, K. Does systemic inflammation and allergen-specific IgE are related to each other in presence asthma. Int. J. Biosci. 2011, 1, 89–94. [Google Scholar]
- Hsin, L.; Tian, X.; Webster, E.L.; Coop, A.; Caldwell, T.M.; Jacobson, A.E.; Chrousos, G.P.; Gold, P.W.; Habib, K.E.; Ayala, A.; et al. CRHR 1 Receptor Binding and Lipophilicity of Pyrrolopyrimidines, Potential Nonpeptide Corticotropin-Releasing Hormone Type 1 Receptor Antagonists. Bioorg. Med. Chem. 2002, 10, 175–183. [Google Scholar] [CrossRef]
- Donald, S.Y. Effects of Drugs on Clinical Laboratory Tests, 5th ed.; AACC Press: Washington, DC, USA, 2000. [Google Scholar]
- Sample Availability: Samples of the compounds are Available on request.





| Compound Nos. | Ar | X | R | Compound No. | Ar | X | R |
|---|---|---|---|---|---|---|---|
| Ia | 3,4-dichlorophenyl | Ph | ------ | 3f | antipyrinyl | Ph | H |
| Ib | p-methoxyphenyl | Ph | ------ | 3g | p-tolyl | H | OCH3 |
| Ic | antipyrinyl | Ph | ------ | 3h | p-methoxyphenyl | H | OCH3 |
| IId | p-tolyl | H | ------ | 3i | antipyrinyl | H | OCH3 |
| IIe | p-methoxyphenyl | H | ------ | 3j | 3,4-dichlorophenyl | Ph | OCH3 |
| IIf | antipyrinyl | H | ------ | 3k | p-methoxyphenyl | Ph | OCH3 |
| 1a | p-tolyl | H | ------ | 3l | antipyrinyl | Ph | OCH3 |
| 1b | p-methoxyphenyl | H | ------ | 4a | p-tolyl | H | H |
| 1c | antipyrinyl | H | ------ | 4b | p-methoxyphenyl | H | H |
| 1d | p-tolyl | Ph | ------ | 4c | antipyrinyl | H | H |
| 1e | p-methoxyphenyl | Ph | ------ | 4d | 3,4-dichlorophenyl | Ph | H |
| 1f | antipyrinyl | Ph | ------ | 4e | p-methoxyphenyl | Ph | H |
| 2a | ------------ | ------ | H | 4f | antipyrinyl | Ph | H |
| 2b | ------------ | ------ | OCH3 | 4g | p-tolyl | H | OCH3 |
| 3a | p-tolyl | H | H | 4h | p-methoxyphenyl | H | OCH3 |
| 3b | p-methoxyphenyl | H | H | 4i | antipyrinyl | H | OCH3 |
| 3c | antipyrinyl | H | H | 4j | 3,4-dichlorophenyl | Ph | OCH3 |
| 3d | 3,4-dichlorophenyl | Ph | H | 4k | p-methoxyphenyl | Ph | OCH3 |
| 3e | p-methoxyphenyl | Ph | H | 4l | antipyrinyl | Ph | OCH3 |
| Compound | 1 h | 2 h | 3 h | 4 h | ||||
|---|---|---|---|---|---|---|---|---|
| Paw Size (mm) | %Inh | Paw Size (mm) | %Inh | Paw Size (mm) | %Inh | Paw Size (mm) | %Inh | |
| Control + ve | 5.86 ± 0.31 | 0 | 5.99 ± 0.43 | 0 | 6.11 ± 0.42 | 0 | 6.01 ± 0.46 | 0 |
| Diclofenac | 4.11 ± 0.19 *** | 29.86 | 3.93 ± 0.21 *** | 34.39 | 3.64 ± 0.20 *** | 40.43 | 3.35 ± 0.27 *** | 44.26 |
| 3b | 5.09 ± 0.36 * | 13.20 | 4.93 ± 0.51 ** | 17.71 | 4.69 ± 0.31 *** | 23.24 | 4.61 ± 0.27 *** | 23.30 |
| 3c | 5.49 ± 0.37 | 6.31 | 4.81 ± 0.32 *** | 19.70 | 4.76 ± 0.12 *** | 22.10 | 4.61 ± 0.34 *** | 23.29 |
| 3d | 5.36 ± 0.34 | 8.53 | 5.62 ± 0.46 | 6.18 | 5.76 ± 0.38 | 5.73 | 5.63 ± 0.48 | 6.32 |
| 3g | 5.49 ± 0.17 | 6.31 | 5.41 ± 0.24 | 9.68 | 5.40 ± 0.47 | 11.62 | 5.32 ± 0.48 | 11.48 |
| 3h | 4.91 ± 0.25 ** | 16.21 | 4.73 ± 0.43 *** | 21.04 | 4.71 ± 0.46 *** | 22.91 | 4.63 ± 0.48 *** | 22.96 |
| 3i | 4.59 ± 0.56 *** | 21.67 | 4.43 ± 0.42 *** | 26.04 | 4.32 ± 0.39 *** | 29.30 | 4.13 ± 0.53 *** | 31.28 |
| 3j | 5.69 ± 0.41 | 2.90 | 4.86 ± 0.40 ** | 18.87 | 4.55 ± 0.52 *** | 25.53 | 4.21 ± 0.43 *** | 29.95 |
| 3k | 5.44 ± 0.25 | 7.17 | 5.39 ± 0.37 | 10.02 | 5.34 ± 0.52 | 12.60 | 5.18 ± 0.50 | 13.81 |
| 3l | 4.69 ± 0.28 *** | 19.97 | 4.19 ± 0.24 *** | 30.05 | 3.89 ± 0.37 *** | 36.33 | 3.81 ± 0.34 *** | 36.61 |
| 4k | 5.51 ± 0.34 | 5.97 | 5.47 ± 0.32 | 8.69 | 5.36 ± 0.31 | 12.28 | 5.14 ± 0.43 | 14.48 |
| Compound | ΔG Free Binding Energy (Kcal/mol) | Clash | Interacted Residues |
|---|---|---|---|
| Diclofenac | −16.85 | 5.2 | Tyr 355 |
| 3b | −10.70 | 3.91 | Try 355 and Ser 530 |
| 3c | −10.12 | 3.11 | Ser 530 and Tyr 355 |
| 3d | −7.95 | 7.47 | Tyr 355 |
| 3g | −7.06 | 7.21 | Arg 120 |
| 3h | −10.52 | 3.45 | Arg 120 |
| 3i | −11.25 | 3.50 | Tyr 385 and Ser 530 |
| 3j | −11.42 | 2.87 | Tyr 355 |
| 3k | −7.62 | 6.95 | Ser 119 |
| 3l | −12.23 | 2.51 | Ser 530 and Tyr 355 |
| 4k | −8.01 | 7.72 | Ser 119 and Tyr 115 |
| Active Compound/Drug | Histamine (μg/L) | IgE (IU/mL) | CRP (mg/L) | IL1-β (ng/L) | TNF-α (pg/mL) |
|---|---|---|---|---|---|
| Control | 0.936 ± 0.024 | 3.7 ± 0.07 | 10.4 ± 0.89 | 11.8 ± 0.06 | 28.83 ± 0.19 |
| Diclofenac | 0.732 ± 0.009 | 1.7 ± 0.31 *** | 7.6 ± 0.51 * | 8.9 ± 0.05 * | 26.28 ± 1.46 |
| 3c | 0.784 ± 0.016 | 1.5 ± 0.28 *** | 6.6 ± 0.80 * | 11.58 ± 0.42 | 31.1 ± 0.10 |
| 3i | 0.338 ± 0.195 ** | 1.68 ± 0.06 *** | 10.8 ± 0.0.37 | 11.18 ± 0.90 | 20.3 ± 1.42 * |
| 3j | 0.762 ± 0.114 | 2.14 ± 0.05 *** | 11.4 ± 1.20 | 7.44 ± 0.89 *** | 26.88 ± 3.25 |
| 3l | 0.819 ± 0.05 | 1.5 ± 0.07 *** | 12.4 ± 0.51 | 7.6 ± 0.34 *** | 16.1 ± 1.50 ** |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Said Fatahala, S.; Hasabelnaby, S.; Goudah, A.; Mahmoud, G.I.; Helmy Abd-El Hameed, R. Pyrrole and Fused Pyrrole Compounds with Bioactivity against Inflammatory Mediators. Molecules 2017, 22, 461. https://doi.org/10.3390/molecules22030461
Said Fatahala S, Hasabelnaby S, Goudah A, Mahmoud GI, Helmy Abd-El Hameed R. Pyrrole and Fused Pyrrole Compounds with Bioactivity against Inflammatory Mediators. Molecules. 2017; 22(3):461. https://doi.org/10.3390/molecules22030461
Chicago/Turabian StyleSaid Fatahala, Samar, Sherifa Hasabelnaby, Ayman Goudah, Ghada I. Mahmoud, and Rania Helmy Abd-El Hameed. 2017. "Pyrrole and Fused Pyrrole Compounds with Bioactivity against Inflammatory Mediators" Molecules 22, no. 3: 461. https://doi.org/10.3390/molecules22030461