Optimization of Ultrasonic-assisted Extraction of Fatty Acids in Seeds of Brucea Javanica (L.) Merr. from Different Sources and Simultaneous Analysis Using High-Performance Liquid Chromatography with Charged Aerosol Detection
Abstract
:1. Introduction
2. Results and Discussion
2.1. Validation of HPLC-CAD Method
2.1.1. Calibration Curves, Limits of Detection, and Quantification
2.1.2. Precision
2.1.3. Accuracy
2.2. Effect of Independent Variables on the Content of FAs and the Oil Yield
2.3. Model Fitting
2.4. Analysis of Response Surface
2.5. Optimization of Extraction Parameters and Validation of the Model
2.6. Effect of Extraction Times on Content of FAs and Oil Yield
2.7. Comparison of Content of FAs
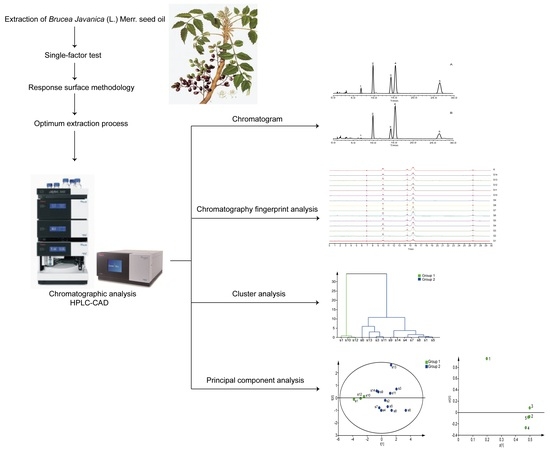
2.8. HPLC Fingerprint, Cluster, and Principal Component Analysis
3. Materials and Methods
3.1. Materials and Standards
3.2. Extraction of Oil
3.3. Preparation of Sample Solution
3.4. HPLC-CAD Analysis
3.5. Validation of HPLC-CAD Method
3.5.1. Calibration Curves, Limits of Detection, and Quantification
3.5.2. Precision
3.5.3. Accuracy
3.6. Content of FAs and Oil Yield Determination
3.7. Optimization of UAE
3.8. Determination of Extraction Times
3.9. Data Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of interest
Abbreviations
| ANOVA | Analysis of variance |
| BBD | Box-Behnken design |
| BJO | Brucea javanica (L.) Merr. seed oil |
| CAD | Charged aerosol detector |
| FAs | Fatty acids |
| RSM | Response surface methodology |
| TCM | Traditional Chinese Medicine |
References
- Su, Z.; Huang, H.; Li, J.; Zhu, Y.; Huang, R.; Qiu, S.X. Chemical composition and cytotoxic activities of petroleum ether fruit extract of fruits of Brucea javanica (Simarubaceae). Trop. J. Pharm. Res. 2013, 12, 735–742. [Google Scholar] [CrossRef]
- Lv, W.; Zhao, S.; Yu, H.; Li, N.; Garamus, V.M.; Chen, Y.; Yin, P.; Zhang, R.; Gong, Y.; Zou, A. Brucea javanica oil-loaded nanostructure lipid carriers (BJO NLCs): Preparation, characterization and in vitro evaluation. Colloid Surf. A. 2016, 504, 312–319. [Google Scholar] [CrossRef]
- Wan, Y.; Li, Y.; Wu, J.; Shen, Q. Characterisation and evaluation of self-microemulsifying drug delivery system of Brucea javanica oil. Micro Nano Lett. 2012, 7, 256–261. [Google Scholar]
- Shi, W.-R.; Liu, Y.; Wang, X.-T.; Huang, Q.-Y.; Cai, X.-R.; Wu, S.-R. Antitumor efficacy and mechanism in hepatoma H22-bearing mice of Brucea javanica oil. Evid. Based Compl. Alt. 2015, 4, 1–8. [Google Scholar]
- Wang, X.; Zhao, X.-E.; Yang, B.; Dong, H.; Liu, D.; Huang, L. A Combination of ultrasonic-assisted extraction with RRLC-QQQ method for the determination of Artemisinin in the Chinese herb Artemisia annua L. Phytochem. Anal. 2011, 3, 280–284. [Google Scholar] [CrossRef] [PubMed]
- Tung, Y.T.; Chang, W.C.; Chen, P.S.; Chang, T.C.; Chang, S.T. Ultrasound-assisted extraction of phenolic antioxidants from Acacia confusa flowers and buds. J. Sep. Sci. 2011, 7, 844–851. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Xu, Z.; Zheng, B.; Lo, Y.M. Optimization of ultrasonic-assisted extraction of pomegranate (Punica granatum L.) seed oil. Ultrason. Sonochem. 2013, 20, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.-S.; Wang, L.-J.; Li, D.; Jiao, S.-S.; Chen, X.D.; Mao, Z.-H. Ultrasound-assisted extraction of oil from flaxseed. Sep. Purif. Technol. 2008, 1, 192–198. [Google Scholar] [CrossRef]
- Zhao, L.-C.; Liang, J.; Li, W.; Cheng, K.-M.; Xia, X.; Deng, X.; Yang, G.-L. The use of response surface methodology to optimize the ultrasound-assisted extraction of five anthraquinones from Rheum palmatum L. Molecules 2011, 7, 5928–5937. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Wang, Z.; Sun, Y.-S.; Chen, L.; Han, L.-K.; Zheng, Y.-N. Application of response surface methodology to optimise ultrasonic-assisted extraction of four chromones in Radix Saposhnikoviae. Phytochem. Anal. 2011, 22, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Hammi, K.M.; Hammami, M.; Rihouey, C.; Le Cerf, D.; Ksouri, R.; Majdoub, H. Optimization extraction of polysaccharide from Tunisian Zizyphus lotus fruit by response surface methodology: Composition and antioxidant activity. Food Chem. 2016, 212, 476–484. [Google Scholar] [CrossRef] [PubMed]
- Vajic, U.-J.; Grujic-Milanovic, J.; Zivkovic, J.; Savikin, K.; Godevac, D.; Miloradovic, Z.; Bugarski, B.; Mihailovic-Stanojevic, N. Optimization of extraction of stinging nettle leaf phenolic compounds using response surface methodology. Ind. Crops Prod. 2015, 74, 912–917. [Google Scholar] [CrossRef]
- Qu, Y.; Li, C.; Zhang, C.; Zeng, R.; Fu, C. Optimization of infrared-assisted extraction of Bletilla striata polysaccharides based on response surface methodology and their antioxidant activities. Carbohyd. Polym. 2016, 148, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; He, L.; Hu, M. Optimized ultrasonic-assisted extraction of flavonoids from Prunella vulgaris L. and evaluation of antioxidant activities in vitro. Innovative Food Sci. Emerging Technol. 2011, 12, 18–25. [Google Scholar] [CrossRef]
- Teleszko, M.; Wojdylo, A.; Rudzinska, M.; Oszmianski, J.; Golis, T. Analysis of lipophilic and hydrophilic bioactive compounds content in Sea Buckthorn (Hippophae rhamnoides L.) Berries. J. Agric. Food Chem. 2015, 63, 4120–4129. [Google Scholar] [CrossRef] [PubMed]
- Ertas, A.; Boga, M.; Yilmaz, M.A.; Yesil, Y.; Hasimi, N.; Kaya, M.S.; Temel, H.; Kolak, U. Chemical compositions by using LC–MS/MS and GC-MS and biological activities of Sedum sediforme (Jacq.) Pau. J. Agric. Food Chem. 2014, 62, 4601–4609. [Google Scholar] [CrossRef] [PubMed]
- Topkafa, M.; Kara, H.; Sherazi, S.T.H. Evaluation of the triglyceride composition of Pomegranate seed oil by RP-HPLC followed by GC–MS. J. Am. Oil Chem. Soc. 2015, 92, 791–800. [Google Scholar] [CrossRef]
- Guarrasi, V.; Mangione, M.R.; Sanfratello, V.; Martorana, V.; Bulone, D. Quantification of underivatized fatty acids from vegetable oils by HPLC with UV detection. J. Chrom. Sci. 2010, 48, 663–668. [Google Scholar] [CrossRef]
- Bravi, E.; Perretti, G.; Montanari, L. Fatty acids by high-performance liquid chromatography and evaporative light-scattering detector. J. Chromatogr. A. 2006, 1134, 210–214. [Google Scholar] [CrossRef] [PubMed]
- Turtygin, A.V.; Deineka, V.I.; Deineka, L.A. Determination of the triglyceride composition of pomegranate seed oil by reversed-phase HPLC and spectrophotometry. J. Anal. Chem. 2013, 68, 558–563. [Google Scholar] [CrossRef]
- Ichihara, K.I.; Kohsaka, C.; Tomari, N.; Kiyono, T.; Wada, J.; Hirooka, K.; Yamamoto, Y. Fatty acid analysis of triacylglycerols: Preparation of fatty acid methyl esters for gas chromatography. Anal. Biochem. 2016, 495, 6–8. [Google Scholar] [CrossRef] [PubMed]
- Thao, N.; Aparicio, M.; Saleh, M.A. Accurate mass GC/LC-quadrupole time of flight mass spectrometry analysis of fatty acids and triacylglycerols of Spicy fruits from the Apiaceae family. Molecules 2015, 20, 21421–21432. [Google Scholar]
- Dong, T.; Yu, L.; Gao, D.; Yu, X.; Miao, C.; Zheng, Y.; Lian, J.; Li, T.; Chen, S. Direct quantification of fatty acids in wet microalgal and yeast biomass via a rapid in situ fatty acid methyl ester derivatization approach. Appl. Microbiol. Biot. 2015, 99, 10237–10247. [Google Scholar] [CrossRef] [PubMed]
- Dixon, R.W.; Peterson, D.S. Development and testing of a detection method for liquid chromatography based on aerosol charging. Anal. Chem. 2002, 74, 2930–2937. [Google Scholar] [CrossRef] [PubMed]
- Peng, M.; Zhang, T.; Ding, Y.; Yi, Y.; Yang, Y.; Le, J. Structure-based prediction of CAD response factors of dammarane-type tetracyclic triterpenoid saponins and its application to the analysis of saponin contents in raw and processed Panax notoginseng. Rsc Adv. 2016, 6, 36987–37005. [Google Scholar] [CrossRef]
- Lisa, M.; Lynen, F.; Holcapek, M.; Sandra, P. Quantitation of triacylglycerols from plant oils using charged aerosol detection with gradient compensation. J. Chromatogr. A 2007, 1176, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Kielbowicz, G.; Trziszka, T.; Wawrzenczyk, C. Separation and quantification of phospholipid and neutral lipid classes by HPLC-CAD: Application to egg yolk lipids. J. Liq. Chromatogr. R. T. 2015, 38, 898–903. [Google Scholar] [CrossRef]
- Bai, C.-C.; Han, S.-Y.; Chai, X.-Y.; Jiang, Y.; Li, P.; Tu, P.-F. Sensitive determination of saponins in Radix et Rhizoma Notoginseng by charged aerosol detector coupled with HPLC. J. Liq. Chromatogr. R. T. 2009, 32, 242–260. [Google Scholar] [CrossRef]
- Roy, C.E.; Kauss, T.; Prevot, S.; Barthelemy, P.; Gaudin, K. Analysis of fatty acid samples by hydrophilic interaction liquid chromatography and charged aerosol detector. J. Chromatogr. A 2015, 1383, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Ilko, D.; Braun, A.; Germershaus, O.; Meinel, L.; Holzgrabe, U. Fatty acid composition analysis in polysorbate 80 with high performance liquid chromatography coupled to charged aerosol detection. Eur. J. Pharm. Biophar. 2015, 94, 569–574. [Google Scholar] [CrossRef] [PubMed]
- Gong, F.; Liang, Y.Z.; Xie, P.S.; Chau, F.T. Information theory applied to chromatographic fingerprint of herbal medicine for quality control. J. Chromatogr. A 2003, 1002, 25–40. [Google Scholar] [CrossRef]
- Xie, P.S.; Chen, S.B.; Liang, Y.Z.; Wang, X.H.; Tian, R.T.; Upton, R. Chromatographic fingerprint analysis—A rational approach for quality assessment of traditional Chinese herbal medicine. J. Chromatogr. A 2016, 1112, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Ge, F.-H.; Lei, H.-P. Study on the supercritical carbon dioxide extraction of Brucea Javanica oil. J. Chin. Med. Mater. 2006, 29, 383–387. [Google Scholar]
- Guo, H.; Hu, C.; Qian, J.; Wu, D. Determination of underivatized long chain fatty acids using HPLC with an evaporative light-scattering detector. J. Am. Oil Chem. Soc. 2012, 89, 183–187. [Google Scholar] [CrossRef]
Sample Availability: Not available. |





| Compound | Retention Time (min) | Linear Range (mg/mL) | Equation | R2 | LOD (μg/mL) | LOQ (μg/mL) |
|---|---|---|---|---|---|---|
| Linolenic acid | 6.95 | 0.0012–0.0349 | y = 63.533x + 0.0164 | 0.9999 | 0.504 | 1.080 |
| Linoleic acid | 9.95 | 0.0109–0.5452 | y = 44.152x + 0.8404 | 0.9983 | 0.368 | 2.624 |
| Palmitic acid | 14.45 | 0.0084–0.4204 | y = 53.760x + 0.2189 | 0.9996 | 1.400 | 4.032 |
| Oleic acid | 15.56 | 0.0161–0.8032 | y = 46.574x + 1.3419 | 0.9983 | 1.376 | 3.856 |
| Stearic acid | 26.65 | 0.0068–0.2030 | y = 81.835x + 0.0361 | 0.9996 | 1.958 | 6.800 |
| Compound | Concentration (mg/mL) | Intra-day (n = 6) | Inter-day (n = 3) |
|---|---|---|---|
| RSD (%) | RSD (%) | ||
| Linolenic acid | 0.0012 | 1.80 | 1.19 |
| 0.0116 | 1.07 | 1.36 | |
| 0.0349 | 1.71 | 1.99 | |
| Linoleic acid | 0.0112 | 1.82 | 2.17 |
| 0.1118 | 0.37 | 1.42 | |
| 0.3353 | 1.04 | 2.29 | |
| Palmitic acid | 0.0088 | 1.33 | 1.21 |
| 0.0877 | 0.48 | 1.94 | |
| 0.2630 | 1.36 | 2.02 | |
| Oleic acid | 0.0231 | 1.97 | 2.06 |
| 0.2310 | 0.26 | 1.44 | |
| 0.6931 | 1.00 | 2.05 | |
| Stearic acid | 0.0066 | 1.48 | 2.71 |
| 0.0664 | 0.44 | 2.86 | |
| 0.1992 | 0.98 | 2.64 |
| Compound | Initial(mg) | Amount(mg) | Add(mg) | Found ± SD(mg) | Recovery (%) | RSD (%, n = 3) |
|---|---|---|---|---|---|---|
| Linolenic acid | 8.87 | 0.0177 | 0.0089 | 0.0265 ± 0.0006 | 98.30 | 2.79 |
| 8.96 | 0.0179 | 0.0090 | 0.0272 ± 0.0002 | 103.68 | ||
| 8.71 | 0.0174 | 0.0087 | 0.0264 ± 0.0003 | 102.55 | ||
| 8.48 | 0.0170 | 0.0170 | 0.0331 ± 0.0005 | 95.19 | 2.17 | |
| 8.71 | 0.0174 | 0.0174 | 0.0346 ± 0.0005 | 98.65 | ||
| 8.63 | 0.0173 | 0.0173 | 0.0336 ± 0.0006 | 94.91 | ||
| 8.11 | 0.0162 | 0.0243 | 0.0398 ± 0.0006 | 96.71 | 2.20 | |
| 8.04 | 0.0161 | 0.0241 | 0.0404 ± 0.0004 | 101.03 | ||
| 8.23 | 0.0165 | 0.0247 | 0.0408 ± 0.0004 | 98.46 | ||
| Linoleic acid | 8.87 | 1.2306 | 0.6153 | 1.8451 ± 0.0116 | 99.87 | 2.66 |
| 8.96 | 1.2431 | 0.6216 | 1.8414 ± 0.0037 | 96.25 | ||
| 8.71 | 1.2084 | 0.6042 | 1.7817 ± 0.0133 | 94.88 | ||
| 8.48 | 1.1765 | 1.1765 | 2.3642 ± 0.0061 | 100.95 | 2.21 | |
| 8.71 | 1.2084 | 1.2084 | 2.4752 ± 0.0070 | 104.83 | ||
| 8.63 | 1.1973 | 1.1973 | 2.4545 ± 0.0041 | 105.00 | ||
| 8.11 | 11252 | 1.6878 | 2.8308 ± 0.0307 | 101.06 | 1.84 | |
| 8.04 | 1.1155 | 1.6732 | 2.8386 ± 0.0064 | 102.98 | ||
| 8.23 | 1.1418 | 1.7127 | 2.9375 ± 0.1431 | 104.84 | ||
| Palmitic acid | 8.87 | 0.6905 | 0.3453 | 1.0414 ± 0.0146 | 101.63 | 1.49 |
| 8.96 | 0.6975 | 0.3478 | 1.0604 ± 0.0014 | 104.03 | ||
| 8.71 | 0.6781 | 0.3390 | 1.0212 ± 0.0005 | 101.21 | ||
| 8.48 | 0.6602 | 0.6602 | 1.2946 ± 0.0044 | 96.10 | 1.82 | |
| 8.71 | 0.6781 | 0.6781 | 1.3456 ± 0.0027 | 98.44 | ||
| 8.63 | 0.6718 | 0.6718 | 1.3411 ± 0.0080 | 99.62 | ||
| 8.11 | 0.6314 | 0.9470 | 1.6031 ± 0.0235 | 102.60 | 0.94 | |
| 8.04 | 0.6259 | 0.9389 | 1.6065 ± 0.0033 | 104.44 | ||
| 8.23 | 0.6407 | 0.9611 | 1.6301 ±0.0197 | 102.94 | ||
| Oleic acid | 8.87 | 2.9146 | 1.4573 | 4.3238 ± 0.0055 | 96.70 | 2.71 |
| 8.96 | 2,9442 | 1.4721 | 4.4206 ± 0.0068 | 100.29 | ||
| 8.71 | 2.8620 | 1.4310 | 4.3215 ± 0.0102 | 101.99 | ||
| 8.48 | 2.7864 | 2.7864 | 5.4940 ± 0.0036 | 97.17 | 1.15 | |
| 8.71 | 2.8620 | 2.8620 | 5.6369 ± 0.0138 | 96.96 | ||
| 8.63 | 2.8357 | 2.8357 | 5.5337 ± 0.0057 | 95.14 | ||
| 8.11 | 2.6649 | 3.9973 | 6.6010 ± 0.0695 | 98.47 | 1.29 | |
| 8.04 | 2.6419 | 3.9628 | 6.6026 ± 0.0303 | 99.95 | ||
| 8.23 | 2.7043 | 4.0564 | 6.8029 ± 0.3343 | 101.04 | ||
| Stearic acid | 8.87 | 0.3869 | 0.1930 | 0.5709 ± 0.0018 | 95.80 | 2.94 |
| 8.96 | 0.3899 | 0.1950 | 0.5799 ± 0.0020 | 97.42 | ||
| 8.71 | 0.3791 | 0.1896 | 0.5713 ± 0.0036 | 101.41 | ||
| 8.48 | 0.3690 | 0.3690 | 0.7205 ± 0.0037 | 95.24 | 2.33 | |
| 8.71 | 0.3791 | 0.3791 | 0.7566 ± 0.0038 | 99.60 | ||
| 8.63 | 0.3756 | 0.3756 | 0.7376 ± 0.0109 | 96.38 | ||
| 8.11 | 0.3529 | 0.5294 | 0.8560 ± 0.0113 | 95.03 | 1.19 | |
| 8.04 | 0.3499 | 0.5249 | 0.8498 ± 0.0070 | 95.24 | ||
| 8.23 | 0.3582 | 0.5373 | 0.8799 ± 0.0062 | 97.10 |
| Source | Content of FAs (mg/g) | Oil Yield (%) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| SS | DF | MS | F | P | SS | DF | MS | F | P | |
| Model | 32.63 | 9 | 3.63 | 4.98 | 0.0229 | 178.73 | 9 | 19.86 | 13.52 | 0.0012 |
| X1 | 1.06 | 1 | 1.06 | 1.46 | 0.2669 | 3.24 | 1 | 3.24 | 2.20 | 0.1812 |
| X2 | 5.53 | 1 | 5.53 | 7.60 | 0.0282 | 18.79 | 1 | 18.79 | 12.79 | 0.009 |
| X3 | 10.76 | 1 | 10.76 | 14.80 | 0.0063 | 147.32 | 1 | 147.32 | 100.27 | <0.0001 |
| X1X2 | 0.89 | 1 | 0.89 | 1.23 | 0.3045 | 3.06 | 1 | 3.06 | 2.08 | 0.192 |
| X1X3 | 0.05 | 1 | 0.05 | 0.07 | 0.8039 | 0.39 | 1 | 0.39 | 0.27 | 0.622 |
| X2X3 | 0.69 | 1 | 0.69 | 0.95 | 0.3629 | 2.13 | 1 | 2.13 | 1.45 | 0.2675 |
| X12 | 4.57 | 1 | 4.57 | 6.29 | 0.0405 | 2.92 | 1 | 2.92 | 1.99 | 0.2012 |
| X22 | 0.44 | 1 | 0.44 | 0.60 | 0.4635 | 0.18 | 1 | 0.18 | 0.12 | 0.7378 |
| X32 | 9.35 | 1 | 9.35 | 12.86 | 0.0089 | 0.80 | 1 | 0.80 | 0.55 | 0.4837 |
| Residual | 5.09 | 7 | 0.73 | 10.28 | 7 | 1.47 | ||||
| Lack of Fit | 1.65 | 3 | 0.55 | 0.64 | 0.6278 | 8.06 | 3 | 2.69 | 4.84 | 0.0808 |
| Pure Error | 3.44 | 4 | 0.86 | 2.22 | 4 | 0.56 | ||||
| Cor Total | 37.72 | 16 | 189.01 | 16 | ||||||
| R2 | 0.8650 | 0.9456 | ||||||||
| Number | Origin | Linolenic Acid (mg/g) | Linoleic Acid (mg/g) | Palmitic Acid (mg/g) | Oleic Acid (mg/g) | Stearic Acid (mg/g) | Total Contents of FAs (mg/g) |
|---|---|---|---|---|---|---|---|
| S1 | Guangdong-1 | 0.005 ± 0.001 a | 4.588 ± 0.132 | 3.242 ± 0.138 | 16.105 ± 0.489 | 1.469 ± 0.066 | 25.409 |
| S2 | Guangdong-2 | 0.092 ± 0.004 | 13.254 ± 0.101 | 6.805 ± 0.117 | 34.278 ± 1.213 | 3.817 ± 0.048 | 58.246 |
| S3 | Guangdong-3 | 0.178 ± 0.009 | 14.064 ± 0.082 | 6.977 ± 0.209 | 30.492 ± 0.717 | 3.950 ± 0.078 | 55.661 |
| S4 | Guangdong-4 | 0.012 ± 0.001 | 21.593 ± 0.156 | 11.584 ± 0.124 | 65.924 ± 2.023 | 7.497 ± 0.129 | 106.610 |
| S5 | Guangdong-5 | 0.064 ± 0.003 | 17.259 ± 0.984 | 8.735 ± 0.274 | 49.433 ± 2.123 | 4.169 ± 0.140 | 79.660 |
| S6 | Guangdong-6 | 0.058 ± 0.003 | 20.321 ± 0.331 | 9.755 ± 0.227 | 44.498 ± 0.564 | 6.095 ± 0.100 | 80.727 |
| S7 | Guangxi-1 | 0.054 ± 0.003 | 25.486 ± 0.902 | 12.803 ± 0.473 | 77.477 ± 3.706 | 6.876 ± 0.164 | 122.696 |
| S8 | Guangxi-2 | 0.056 ± 0.001 | 27.270 ± 0.693 | 12.936 ± 0.219 | 69.677 ± 0.772 | 6.885 ± 3.840 | 116.824 |
| S9 | Guangxi-3 | 0.280 ± 0.004 | 22.283 ± 0.568 | 11.176 ± 0.209 | 50.379 ± 2.704 | 6.060 ± 0.204 | 90.178 |
| S10 | Fujian-1 | 0.070 ± 0.002 | 8.284 ± 0.052 | 4.660 ± 0.028 | 22.655 ± 0.054 | 1.646 ± 0.033 | 37.315 |
| S11 | Fujian-2 | 0.165 ± 0.006 | 16.404 ± 0.170 | 8.522 ± 0.136 | 36.787 ± 0.378 | 4.580 ± 0.069 | 66.458 |
| S12 | Hebei | 0.056 ± 0.002 | 8.623 ± 0.086 | 4.020 ± 0.011 | 24.894 ± 0.227 | 1.907 ± 0.034 | 39.500 |
| S13 | Yunnan | 0.255 ± 0.008 | 9.007 ± 0.078 | 5.297 ± 0.153 | 20.355 ± 0.578 | 2.441 ± 0.081 | 37.355 |
| S14 | Hainan | 0.150 ± 0.004 | 11.178 ± 0.010 | 6.398 ± 0.088 | 30.067 ± 0.537 | 2.646 ± 0.076 | 50.439 |
| Independent Variable | Units | Symbol | Coded Levels | ||
|---|---|---|---|---|---|
| −1 | 0 | 1 | |||
| Sonication time | min | X1 | 20 | 30 | 40 |
| Liquid-solid ratio | mL/g | X2 | 16:1 | 20:1 | 24:1 |
| Ethanol concentration | % | X3 | 90 | 95 | 100 |
| Run | Factor 1 (X1) | Factor 2 (X2) | Factor 3 (X3) | Response 1 (Y1) | Response 2 (Y2) |
|---|---|---|---|---|---|
| Sonication Time (min) | Liquid-solid Ratio (mL/g) | Ethanol Concentration (%) | Content of FAs (mg/g) | Oil Yield (%) | |
| 1 | −1 | 0 | −1 | 8.71 | 5.00 |
| 2 | 0 | 0 | 0 | 9.81 | 8.33 |
| 3 | 0 | 1 | 1 | 11.40 | 14.25 |
| 4 | 1 | 1 | 0 | 14.16 | 14.25 |
| 5 | 0 | 0 | 0 | 11.73 | 9.17 |
| 6 | −1 | 1 | 0 | 12.08 | 9.75 |
| 7 | 0 | −1 | 1 | 9.32 | 10.83 |
| 8 | 1 | 0 | 1 | 12.05 | 14.17 |
| 9 | 0 | −1 | −1 | 8.53 | 4.50 |
| 10 | 0 | 0 | 0 | 11.70 | 10.00 |
| 11 | 1 | 0 | −1 | 8.81 | 4.17 |
| 12 | 0 | 1 | −1 | 8.95 | 5.00 |
| 13 | −1 | 0 | 1 | 11.51 | 13.75 |
| 14 | 0 | 0 | 0 | 10.31 | 8.13 |
| 15 | 0 | 0 | 0 | 10.04 | 8.75 |
| 16 | 1 | −1 | 0 | 11.14 | 8.33 |
| 17 | −1 | −1 | 0 | 10.95 | 7.33 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, Z.; Li, L.; Li, N.; Zhang, T.; Pu, Y.; Zhang, X.; Zhang, Y.; Wang, B. Optimization of Ultrasonic-assisted Extraction of Fatty Acids in Seeds of Brucea Javanica (L.) Merr. from Different Sources and Simultaneous Analysis Using High-Performance Liquid Chromatography with Charged Aerosol Detection. Molecules 2017, 22, 931. https://doi.org/10.3390/molecules22060931
Wu Z, Li L, Li N, Zhang T, Pu Y, Zhang X, Zhang Y, Wang B. Optimization of Ultrasonic-assisted Extraction of Fatty Acids in Seeds of Brucea Javanica (L.) Merr. from Different Sources and Simultaneous Analysis Using High-Performance Liquid Chromatography with Charged Aerosol Detection. Molecules. 2017; 22(6):931. https://doi.org/10.3390/molecules22060931
Chicago/Turabian StyleWu, Zhuona, Ling Li, Ning Li, Tong Zhang, Yiqiong Pu, Xitong Zhang, Yue Zhang, and Bing Wang. 2017. "Optimization of Ultrasonic-assisted Extraction of Fatty Acids in Seeds of Brucea Javanica (L.) Merr. from Different Sources and Simultaneous Analysis Using High-Performance Liquid Chromatography with Charged Aerosol Detection" Molecules 22, no. 6: 931. https://doi.org/10.3390/molecules22060931