Effect of Sipjeondaebo-Tang on the Pharmacokinetics of S-1, an Anticancer Agent, in Rats Evaluated by Population Pharmacokinetic Modeling
Abstract
:1. Introduction
2. Results
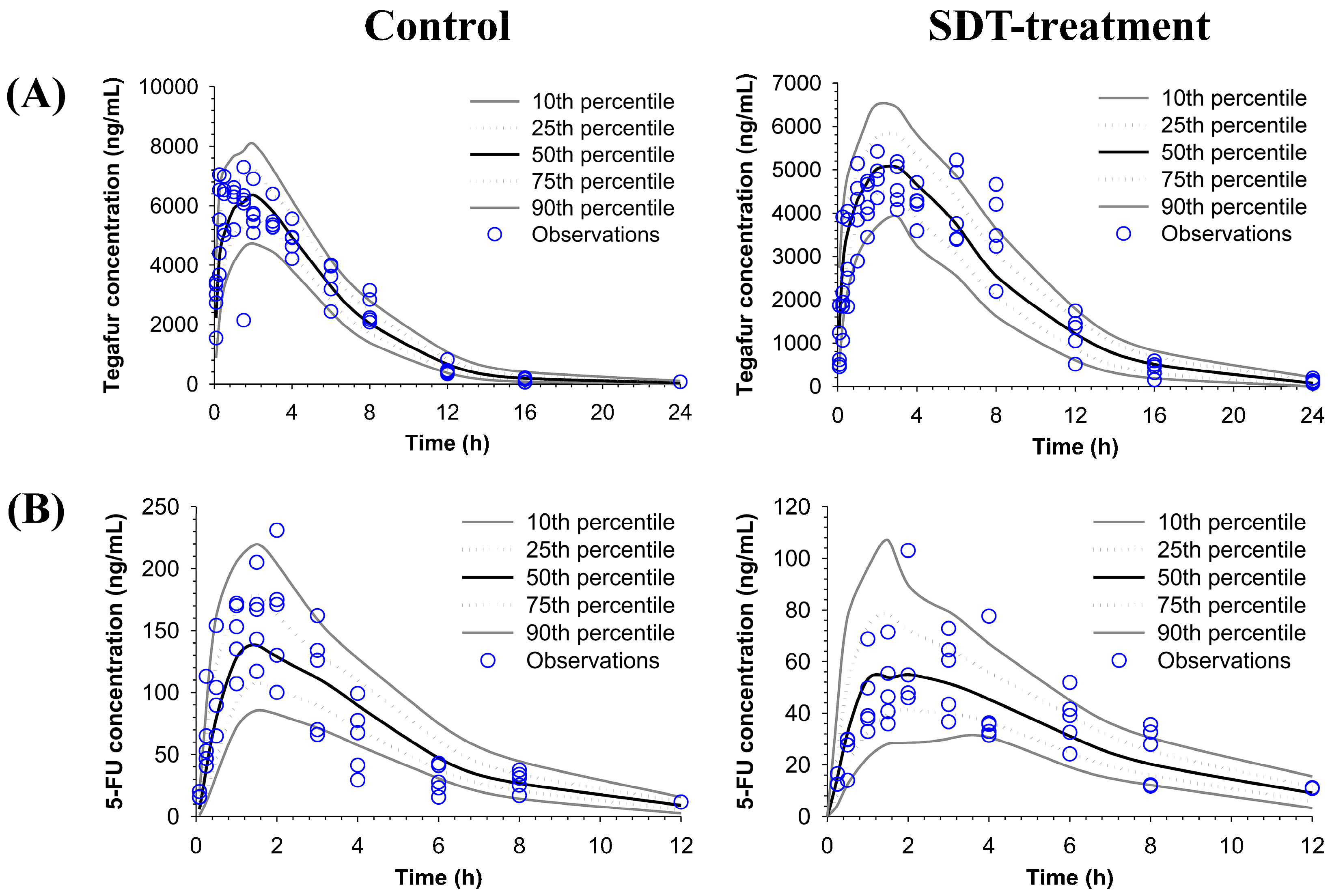
2.1. Pharmacokinetics of S-1 after Pretreatment with a Single Dose of SDT
2.2. Pharmacokinetics of S-1 after Pretreatment with Multiple Doses of SDT
2.3. Population Pharmacokinetic (POP-PK) Modeling
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Animal Study
4.3. LC/MS/MS
4.4. Noncompartmental Analysis
4.5. Population Pharmacokinetic Modeling
4.6. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Shirasaka, T.; Shimamato, Y.; Ohshimo, H.; Yamaguchi, M.; Kato, T.; Yonekura, K.; Fukushima, M. Development of a novel form of an oral 5-fluorouracil derivative (S-1) directed to the potentiation of the tumor selective cytotoxicity of 5-fluorouracil by two biochemical modulators. Anticancer Drugs 1996, 7, 548–557. [Google Scholar] [CrossRef] [PubMed]
- Osugi, H.; Takada, N.; Takemura, M.; Kaseno, S.; Lee, S.; Ueno, M.; Tanaka, Y.; Fukuhara, K.; Fujiwara, Y.; Kinoshita, H. Oral fluoropyrimidine anticancer drug TS-1 for gastric cancer patients with peritoneal dissemination. Oncol. Rep. 2002, 9, 811–815. [Google Scholar] [CrossRef] [PubMed]
- Diasio, R.B. Clinical implications of dihydropyrimidine dehydrogenase inhibition. Oncology 1999, 13, 17–21. [Google Scholar] [PubMed]
- Sakuramoto, S.; Sasako, M.; Yamaguchi, T.; Kinoshita, T.; Fujii, M.; Nashimoto, A.; Furukawa, H.; Nakajima, T.; Ohashi, Y.; Imamura, H.; et al. Adjuvant chemotherapy for gastric cancer with S-1, an oral fluoropyrimidine. N. Engl. J. Med. 2007, 357, 1810–1820. [Google Scholar] [CrossRef] [PubMed]
- Sugimachi, K.; Maehara, Y.; Horikoshi, N.; Shimada, Y.; Sakata, Y.; Mitachi, Y.; Taguchi, T. An early phase ii study of oral S-1, a newly developed 5-fluorouracil derivative for advanced and recurrent gastrointestinal cancers. The s-1 gastrointestinal cancer study group. Oncology 1999, 57, 202–210. [Google Scholar] [CrossRef] [PubMed]
- Koizumi, W.; Kurihara, M.; Nakano, S.; Hasegawa, K. Phase II study of S-1, a novel oral derivative of 5-fluorouracil, in advanced gastric cancer. For the S-1 cooperative gastric cancer study group. Oncology 2000, 58, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Sakata, Y.; Ohtsu, A.; Horikoshi, N.; Sugimachi, K.; Mitachi, Y.; Taguchi, T. Late phase ii study of novel oral fluoropyrimidine anticancer drug S-1 (1 m tegafur-0.4 m gimestat-1 m otastat potassium) in advanced gastric cancer patients. Eur. J. Cancer 1998, 34, 1715–1720. [Google Scholar] [CrossRef]
- Uesaka, K.; Fukutomi, A.; Boku, N.; Kanemoto, H.; Konishi, M.; Matsumoto, I.; Kaneoka, Y.; Shimizu, Y.; Nakamori, S.; Sakamoto, H.; et al. Randomized phase iii trial of adjuvant chemotherapy with gemcitabine versus S-1 for patients with resected pancreatic cancer (jaspac-01 study). J. Clin. Oncol. 2013, 31, 145. [Google Scholar] [CrossRef]
- Sudo, K.; Nakamura, K.; Yamaguchi, T. S-1 in the treatment of pancreatic cancer. World J. Gastroenterol. 2014, 20, 15110. [Google Scholar] [CrossRef] [PubMed]
- Chuah, B.; Goh, B.C.; Lee, S.C.; Soong, R.; Lau, F.; Mulay, M.; Dinolfo, M.; Lim, S.E.; Soo, R.; Furuie, T.; et al. Comparison of the pharmacokinetics and pharmacodynamics of S-1 between caucasian and east asian patients. Cancer Sci. 2011, 102, 478–483. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.H.; Park, G.Y.; Shin, S.; Kwon, D.R.; Seo, W.S.; Shin, J.C.; Choi, J.H.; Joo, S.H.; Weon, K.Y.; Min, B.S.; et al. Pharmacokinetic alteration of baclofen by multiple oral administration of herbal medicines in rats. Evid. Based Comp. Altern. Med. 2014, 2014, 402126. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, I.; Emi, Y.; Kakeji, Y.; Uchida, J.; Fukushima, M.; Maehara, Y. Increased antitumor activity in combined treatment TS-1 and docetaxel. A preclinical study using gastric cancer xenografts. Oncology 2005, 68, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Yasui-Furukori, N.; Hashimoto, K.; Kubo, K.; Tomita, T. Interaction between paliperidone extended release and TS-1®, an oral anticancer drug containing a 5-fluorouracil derivative, in a schizophrenic patient. Neuropsychiatr. Dis. Treat. 2013, 9, 317–320. [Google Scholar] [CrossRef] [PubMed]
- Cheon, C.; Park, S.; Park, Y.L.; Huang, C.W.; Ko, Y.; Jang, B.H.; Shin, Y.C.; Ko, S.G. Sipjeondaebo-tang in patients with cancer with anorexia: A protocol for a pilot, randomised, controlled trial. BMJ Open 2016, 6, e011212. [Google Scholar] [CrossRef] [PubMed]
- Jeon, W.Y.; Shin, I.S.; Shin, H.K.; Lee, M.Y. Gastroprotective effect of the traditional herbal medicine, sipjeondaebo-tang water extract, against ethanol-induced gastric mucosal injury. BMC Comp. Altern. Med. 2014, 14, 373. [Google Scholar] [CrossRef] [PubMed]
- Takeda, H.; Sadakane, C.; Hattori, T.; Katsurada, T.; Ohkawara, T.; Nagai, K.; Asaka, M. Rikkunshito, an herbal medicine, suppresses cisplatin-induced anorexia in rats via 5-HT2 receptor antagonism. Gastroenterology 2008, 134, 2004–2013. [Google Scholar] [CrossRef] [PubMed]
- Cheng, K.C.; Li, Y.X.; Cheng, J.T. The use of herbal medicine in cancer-related anorexia/cachexia treatment around the world. Curr. Pharm. Des. 2012, 18, 4819–4826. [Google Scholar] [CrossRef] [PubMed]
- Niwa, K.; Hashimoto, M.; Morishita, S.; Lian, Z.; Tagami, K.; Mori, H.; Tamaya, T. Preventive effects of juzen-taiho-to on N-methyl-N-nitrosourea and estradiol-17β-induced endometrial carcinogenesis in mice. Carcinogenesis 2001, 22, 587–591. [Google Scholar] [CrossRef] [PubMed]
- Ohnishi, Y.; Fujii, H.; Kimura, F.; Mishima, T.; Murata, J.; Tazawa, K.; Fujimaki, M.; Okada, F.; Hosokawa, M.; Saiki, I. Inhibitory effect of a traditional chinese medicine, juzen-taiho-to, on progressive growth of weakly malignant clone cells derived from murine fibrosarcoma. Jpn. J. Cancer Res. 1996, 87, 1039–1044. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, K.; Ueda, H.; Ichio, Y.; Yokota, M. Improvement of cisplatin toxicity and lethality by juzen-taiho-to in mice. Biol. Pharm. Bull. 1995, 18, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Comets, E.; Ikeda, K.; Hoff, P.; Fumoleau, P.; Wanders, J.; Tanigawara, Y. Comparison of the pharmacokinetics of S-1, an oral anticancer agent, in western and japanese patients. J. Pharmacokinet. Pharmacodyn. 2003, 30, 257–283. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, H.; Komatsu, T.; Takemoto, K.; Shimada, N.; Nakajima, M.; Yokoi, T. Rat cytochrome p450 1a and 3a enzymes involved in bioactivation of tegafur to 5-fluorouracil and autoinduced by tegafur in liver microsomes. Drug Metab. Dispos. 2001, 29, 794–797. [Google Scholar] [PubMed]
- Ikeda, K.; Yoshisue, K.; Matsushima, E.; Nagayama, S.; Kobayashi, K.; Tyson, C.A.; Chiba, K.; Kawaguchi, Y. Bioactivation of tegafur to 5-fluorouracil is catalyzed by cytochrome P-450 2A6 in human liver microsomes in vitro. Clin. Cancer Res. 2000, 6, 4409–4415. [Google Scholar] [PubMed]
- Diasio, R.B. The role of dihydropyrimidine dehydrogenase (DPD) modulation in 5-FU pharmacology. Oncology 1998, 12, 23–27. [Google Scholar] [PubMed]
- Yi, H.; Cho, H.J.; Cho, S.M.; Lee, D.G.; El-Aty, A.A.; Yoon, S.J.; Bae, G.W.; Nho, K.; Kim, B.; Lee, C.H.; et al. Pharmacokinetic properties and antitumor efficacy of the 5-fluorouracil loaded peg-hydrogel. BMC Cancer 2010, 10, 211. [Google Scholar] [CrossRef] [PubMed]
- Kobuchi, S.; Ito, Y.; Okada, K.; Imoto, K.; Kuwano, S.; Takada, K. Pre-therapeutic assessment of plasma dihydrouracil/uracil ratio for predicting the pharmacokinetic parameters of 5-fluorouracil and tumor growth in a rat model of colorectal cancer. Biol. Pharm. Bull. 2013, 36, 907–916. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.-H.; Park, J.-K.; Kang, J.-H.; Chung, S.-J.; Shim, C.-K.; Kuh, H.-J. Stability of 5-FU and tegafur in biological fluids of rats. J. Pharm. Investig. 2004, 34, 161–168. [Google Scholar]
- European Medicines Agency (EMA). Assessment Report for Teysuno (Tegafur/Gimeracil/Oteracil); EMA: London, UK, 2010. [Google Scholar]
- Hukkanen, J.; Jacob, P., 3rd; Benowitz, N.L. Effect of grapefruit juice on cytochrome P450 2A6 and nicotine renal clearance. Clin. Pharmacol. Ther. 2006, 80, 522–530. [Google Scholar] [CrossRef] [PubMed]
- Yano, J.K.; Hsu, M.H.; Griffin, K.J.; Stout, C.D.; Johnson, E.F. Structures of human microsomal cytochrome P450 2A6 complexed with coumarin and methoxsalen. Nat. Struct. Mol. Biol. 2005, 12, 822–823. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, M.; Itoh, M.; Yamanaka, H.; Fukami, T.; Tokudome, S.; Yamamoto, Y.; Yamamoto, H.; Yokoi, T. Isoflavones inhibit nicotine c-oxidation catalyzed by human CYP2A6. J. Clin. Pharmacol. 2006, 46, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Peng, J.; Wang, X.; Zhu, X.; Cheng, B.; Gao, J.; Jiang, M.; Bai, G.; Hou, Y. Carboxymethylcellulose sodium improves the pharmacodynamics of 1-deoxynojirimycin by changing its absorption characteristics and pharmacokinetics in rats. Pharmazie 2012, 67, 168–173. [Google Scholar] [PubMed]
- Bauer, R.J.; Guzy, S.; Ng, C. A survey of population analysis methods and software for complex pharmacokinetic and pharmacodynamic models with examples. AAPS J. 2007, 9, E60–E83. [Google Scholar] [CrossRef] [PubMed]
- Bulitta, J.B.; Bingolbali, A.; Shin, B.S.; Landersdorfer, C.B. Development of a new pre- and post-processing tool (sadapt-tran) for nonlinear mixed-effects modeling in S-adapt. AAPS J. 2011, 13, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Bulitta, J.B.; Landersdorfer, C.B. Performance and robustness of the monte carlo importance sampling algorithm using parallelized S-adapt for basic and complex mechanistic models. AAPS J. 2011, 13, 212–226. [Google Scholar] [CrossRef] [PubMed]
- Brendel, K.; Comets, E.; Laffont, C.; Laveille, C.; Mentre, F. Metrics for external model evaluation with an application to the population pharmacokinetics of gliclazide. Pharm. Res. 2006, 23, 2036–2049. [Google Scholar] [CrossRef] [PubMed]
- Bulitta, J.B.; Duffull, S.B.; Kinzig-Schippers, M.; Holzgrabe, U.; Stephan, U.; Drusano, G.L.; Sorgel, F. Systematic comparison of the population pharmacokinetics and pharmacodynamics of piperacillin in cystic fibrosis patients and healthy volunteers. Antimicrob. Agents Chemother. 2007, 51, 2497–2507. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |


| Parameter | Single Dose | Multiple Dose | |||
|---|---|---|---|---|---|
| Control (n = 5) | SDT (n = 5) | Control (n = 5) | SDT (n = 5) | ||
| Tegafur | t1/2 (h) | 2.3 ± 0.7 | 3.5 ± 0.7 * | 2.2 ± 1.4 | 3.3 ± 0.8 |
| Tmax (h) | 1.5 ± 0.4 | 1.3 ± 0.4 | 1.2 ± 0.7 | 3.2 ± 1.6 * | |
| Cmax (ng/mL) | 9328.0 ± 3099.3 | 7202.0 ± 2374.7 | 6828.0 ± 384.0 | 4960.0 ± 431.9 * | |
| AUCall (ng·h/mL) | 55,372.7 ± 20215.9 | 42,705.1 ± 11087.4 | 43,496.9 ± 4673.0 | 46,842.4 ± 8127.5 | |
| AUCinf (ng·h/mL) | 55,712.7 ± 20247.5 | 42,945.5 ± 11033.1 | 43,748.1 ± 4835.6 | 47,461.3 ± 8163.9 | |
| CL/F (mL/min/kg) | 1.7 ± 0.8 | 2.1 ± 0.6 | 1.9 ± 0.2 | 1.8 ± 0.3 | |
| Vz/F (L/kg) | 0.3 ± 0.1 | 0.6 ± 0.2 * | 0.4 ± 0.2 | 0.5 ± 0.1 | |
| 5-FU | t1/2 (h) | 2.6 ± 2.1 | 2.2 ± 1.0 | 3.0 ± 0.9 | 3.1 ± 0.8 |
| Tmax (h) | 1.7 ± 0.8 | 1.7 ± 1.1 | 1.8 ± 0.3 | 2.4 ± 0.5 | |
| Cmax (ng/mL) | 178.2 ± 42.0 | 209.5 ± 94.9 | 172.2 ± 40.4 | 64.3 ± 23.0 * | |
| AUCall (ng·h/mL) | 613.7 ± 233.4 | 892.4 ± 439.7 | 639.3 ± 190 | 362.7 ± 96.2 * | |
| AUCinf (ng·h/mL) | 924.9 ± 537.6 | 955.8 ± 438.9 | 750.2 ± 145.8 | 429.6 ± 83.2 * | |
| AUCmeta/AUCparent (%) | 2.5 ± 0.9 | 3.3 ± 1.0 | 2.7 ± 0.7 | 1.5 ± 0.5 * | |
| Gimeracil | t1/2 (h) | 1.2 ± 0.2 | 0.9 ± 0.3 | 0.7 ± 0.1 | 0.8 ± 0.2 |
| Tmax (h) | 0.6 ± 0.4 | 0.8 ± 0.8 | 0.5 ± 0.0 | 0.7 ± 0.5 | |
| Cmax (ng/mL) | 422.2 ± 136.5 | 358 ± 121.4 | 347.8 ± 42.7 | 142.3 ± 41.8 * | |
| AUCall (ng·h/mL) | 768.3 ± 312.9 | 678.7 ± 190.9 | 449.5 ± 74.2 | 180.2 ± 41.5 * | |
| AUCinf (ng·h/mL) | 984.8 ± 335.4 | 758.7 ± 186.7 | 526.6 ± 85.4 | 247.4 ± 57.4 * | |
| CL/F (mL/min/kg) | 26.8 ± 8.8 | 33.7 ± 9.4 | 46.9 ± 7.8 | 101.9 ± 22.6 * | |
| Vz/F (L/kg) | 2.9 ± 1.3 | 2.6 ± 1.4 | 2.8 ± 0.4 | 6.8 ± 0.9 * | |
| Parameter | Symbol | Unit | Population Mean (BSV) |
|---|---|---|---|
| Absorption rate constant for tegafur in control group | Ka,Con | 1/h | 0.296 (0.0101) |
| Absorption rate constant for tegafur in SDT pretreatment group | Ka,Pretre | 1/h | 0.197 (0.0125) |
| Formation rate constant of 5-FU precursor from gut compartment in control group | Ka,Met,Con | 1/h | 0.122 (0.308) |
| Formation rate constant of 5-FU precursor from gut compartment in SDT pretreatment group | Ka,Met,Pretre | 1/h | 0.0595 (0.655) |
| Formation rate constant of 5-FU from 5-FU precursor | KConv | 1/h | 2.88 (0.747) |
| Clearance for tegafur | CLTeg/F | L/h/kg | 0.0813 (0.00221) |
| Fraction of 5-FU clearance for 5-FU precursor formation | FMet | - | 0.342 (0.0162) |
| Clearance for 5-FU in control group | CL5FU,Con/F | L/h/kg | 3.52 (0.163) |
| Clearance for 5-FU in SDT pretreatment group | CL5FU,Pretre/F | L/h/kg | 5.93 (0.025) |
| Distribution clearance for tegafur | CLdTeg/F | L/h/kg | 0.184 (0.105) |
| Distribution clearance for 5-FU | CLd5FU/F | L/h/kg | 1.87 (0.168) |
| Central volume of distribution for tegafur | V1,Teg/F | L/kg | 0.0464 (0.729) |
| Central volume of distribution for 5-FU | V1,5FU/F | L/kg | 0.623 (0.291) |
| Peripheral volume of distribution for tegafur | V2,Teg/F | L/kg | 0.137 (0.0141) |
| Peripheral volume of distribution for 5-FU | V2,5FU/F | L/kg | 0.294 (0.184) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, T.H.; Shin, S.; Shin, J.C.; Bulitta, J.B.; Weon, K.-Y.; Yoo, S.D.; Park, G.-Y.; Jeong, S.W.; Kwon, D.R.; Min, B.S.; et al. Effect of Sipjeondaebo-Tang on the Pharmacokinetics of S-1, an Anticancer Agent, in Rats Evaluated by Population Pharmacokinetic Modeling. Molecules 2017, 22, 1488. https://doi.org/10.3390/molecules22091488
Kim TH, Shin S, Shin JC, Bulitta JB, Weon K-Y, Yoo SD, Park G-Y, Jeong SW, Kwon DR, Min BS, et al. Effect of Sipjeondaebo-Tang on the Pharmacokinetics of S-1, an Anticancer Agent, in Rats Evaluated by Population Pharmacokinetic Modeling. Molecules. 2017; 22(9):1488. https://doi.org/10.3390/molecules22091488
Chicago/Turabian StyleKim, Tae Hwan, Soyoung Shin, Jeong Cheol Shin, Jürgen B. Bulitta, Kwon-Yeon Weon, Sun Dong Yoo, Gi-Young Park, Seok Won Jeong, Dong Rak Kwon, Byung Sun Min, and et al. 2017. "Effect of Sipjeondaebo-Tang on the Pharmacokinetics of S-1, an Anticancer Agent, in Rats Evaluated by Population Pharmacokinetic Modeling" Molecules 22, no. 9: 1488. https://doi.org/10.3390/molecules22091488
APA StyleKim, T. H., Shin, S., Shin, J. C., Bulitta, J. B., Weon, K.-Y., Yoo, S. D., Park, G.-Y., Jeong, S. W., Kwon, D. R., Min, B. S., Woo, M. H., & Shin, B. S. (2017). Effect of Sipjeondaebo-Tang on the Pharmacokinetics of S-1, an Anticancer Agent, in Rats Evaluated by Population Pharmacokinetic Modeling. Molecules, 22(9), 1488. https://doi.org/10.3390/molecules22091488








