Pseudoginsengenin DQ Exhibits Therapeutic Effects in Cisplatin-Induced Acute Kidney Injury via Sirt1/NF-κB and Caspase Signaling Pathway without Compromising Its Antitumor Activity in Mice
Abstract
1. Introduction
2. Results
2.1. PDQ Protects against CDDP-Induced Renal Dysfunction
2.2. PDQ Attenuates CDDP-Induced Histopathological Damage in Kidney
2.3. PDQ Reduces CDDP-Induced Tubular Cell Apoptosis in Kidney
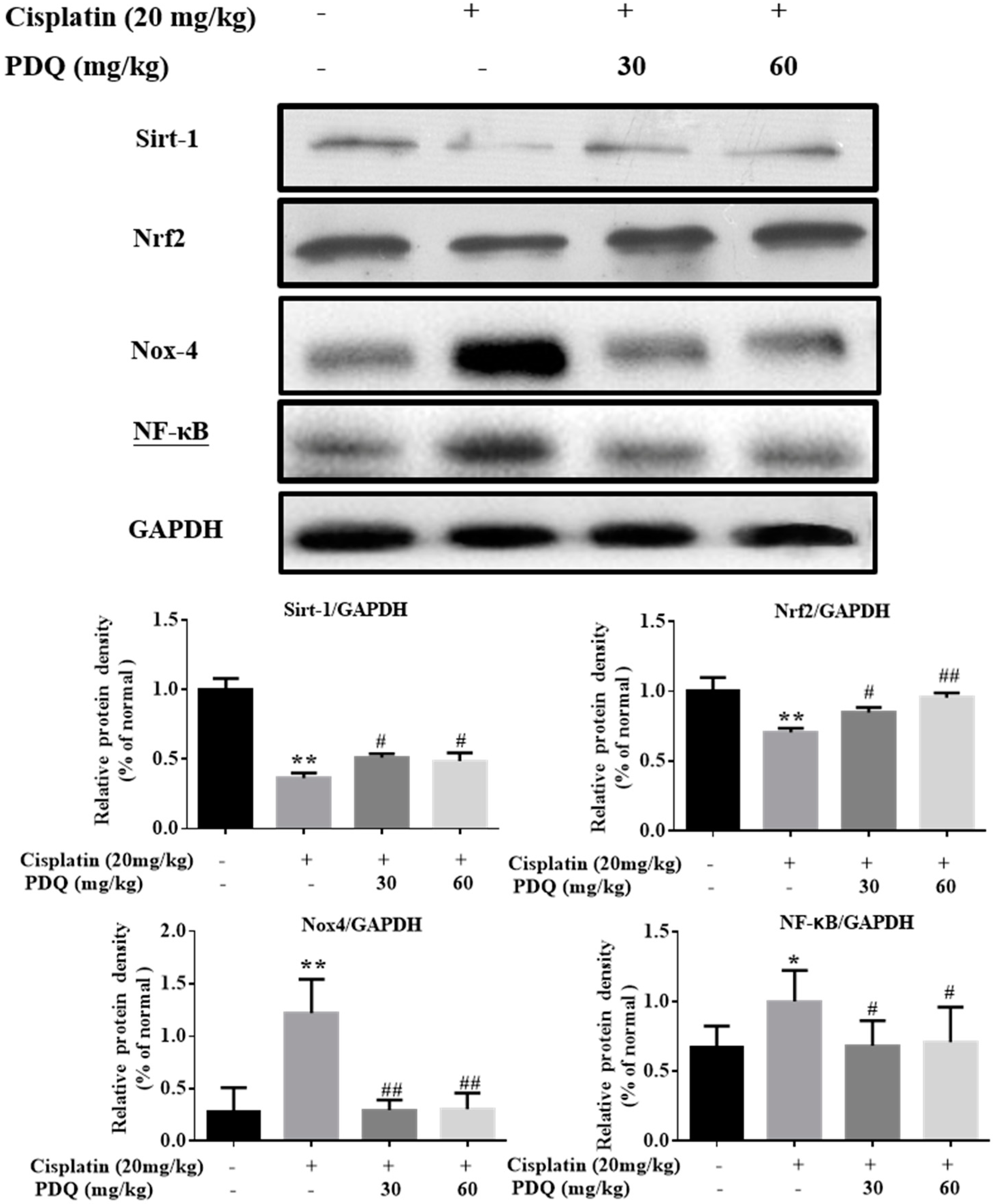
2.4. PDQ Ameliorates CDDP-Induced Oxidative Stress in Kidney
2.5. PDQ Suppresses CDDP-Induced Inflammation in Kidney
2.6. PDQ Does Not Compromise the In Vivo Antitumor Activity of CDDP
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Animals and Experimental Protocol
4.3. Assessment of Biochemical Parameters
4.4. Measurement of Serum TNF-α and IL-1β Levels
4.5. Histological Assays
4.6. Hoechst 33258 and TUNEL Staining
4.7. Immunohistochemistry (IHC) and Immunofluorescence Analysis
4.8. Western Blotting Analysis
4.9. Establishment of Xenograft Tumor Model
4.10. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Miller, R.P.; Tadagavadi, R.K.; Ramesh, G.; Reeves, W.B. Mechanisms of cisplatin nephrotoxicity. Toxins 2010, 2, 2490–2518. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Hernandez, F.J.; Lopez-Novoa, J.M. Potential utility of pparalpha activation in the prevention of ischemic and drug-induced acute renal damage. Kidney Int. 2009, 76, 1022–1024. [Google Scholar] [CrossRef] [PubMed]
- Karasawa, T.; Steyger, P.S. An integrated view of cisplatin-induced nephrotoxicity and ototoxicity. Toxicol. Lett. 2015, 237, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Li, Z.; Li, P.; Lin, M.; Han, L.; Wang, F.; Liu, J. Determination of pseudoginsengenin dq in rat plasma by uplc-ms/ms and application of the method in a pharmacokinetic study. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2013, 933, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Meng, Q.; Zhang, J.; Bi, Y.; Jiang, N. Study on the structure-function relationship of 20(s)-panaxadiol and its epimeric derivatives in myocardial injury induced by isoproterenol. Fitoterapia 2010, 81, 783–787. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Liang, K.; Zhang, J.; Yu, G.; Lv, G.; Zhang, F.; Chen, X.; Tian, J.; Fu, F. The pseudoginsenoside f11 ameliorates cisplatin-induced nephrotoxicity without compromising its anti-tumor activity in vivo. Sci. Rep. 2014, 4, 4986. [Google Scholar] [CrossRef] [PubMed]
- Karimi, A.; Absalan, F.; Khorsandi, L.; Valizadeh, A.; Mansouri, E. Sodium hydrogen sulfide (nahs) ameliorates alterations caused by cisplatin in filtration slit diaphragm and podocyte cytoskeletal in rat kidney. J. Nephropathol. 2017, 6, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.Z.; Zhou, H.; Yan, Y. Xiap underlies apoptosis resistance of renal cell carcinoma cells. Mol. Med. Rep. 2018, 17, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Hu, J.; Yan, M.; Xing, J.; Liu, W.; Li, W. Caspase-mediated anti-apoptotic effect of ginsenoside rg5, a main rare ginsenoside, on acetaminophen-induced hepatotoxicity in mice. J. Agric. Food Chem. 2017, 65, 9226–9236. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Li, Y.; Han, X.; Sun, Y.; Zhang, L.; Liu, W.; Liu, X.; Li, W.; Liu, Y. Kidney protection effect of ginsenoside re and its underlying mechanisms on cisplatin-induced kidney injury. Cell. Physiol. Biochem. 2018, 48, 2219–2229. [Google Scholar] [CrossRef] [PubMed]
- Malik, S.; Bhatia, J.; Suchal, K.; Gamad, N.; Dinda, A.K.; Gupta, Y.K.; Arya, D.S. Nobiletin ameliorates cisplatin-induced acute kidney injury due to its anti-oxidant, anti-inflammatory and anti-apoptotic effects. Exp. Toxicol. Pathol. 2015, 67, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Qi, Z.L.; Wang, Z.; Li, W.; Hou, J.G.; Liu, Y.; Li, X.D.; Li, H.P.; Wang, Y.P. Nephroprotective effects of anthocyanin from the fruits of panax ginseng (gfa) on cisplatin-induced acute kidney injury in mice. Phytother. Res. 2017, 31, 1400–1409. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Li, W.; Wang, L.; Li, W.; Sun, H.; He, X.; Zhang, J. The protective effects of sika deer antler protein on cisplatin-induced nephrotoxicity. Cell. Physiol. Biochem. 2017, 43, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wu, Q.; Lu, Y.; Wan, J.; Dai, H.; Zhou, X.; Lv, S.; Chen, X.; Zhang, X.; Hang, C.; et al. Cerebroprotection by salvianolic acid b after experimental subarachnoid hemorrhage occurs via nrf2- and sirt1-dependent pathways. Free Radic. Biol. Med. 2018, 124, 504–516. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Tao, X.; Yin, L.; Xu, L.; Xu, Y.; Qi, Y.; Han, X.; Song, S.; Zhao, Y.; Lin, Y.; et al. Protective effects of dioscin against cisplatin-induced nephrotoxicity via the microrna-34a/sirtuin 1 signalling pathway. Br. J. Pharmacol. 2017, 174, 2512–2527. [Google Scholar] [CrossRef] [PubMed]
- Dhanda, S.; Kaur, S.; Sandhir, R. Preventive effect of n-acetyl-l-cysteine on oxidative stress and cognitive impairment in hepatic encephalopathy following bile duct ligation. Free Radic. Biol. Med. 2013, 56, 204–215. [Google Scholar] [CrossRef] [PubMed]
- Do, M.T.; Kim, H.G.; Choi, J.H.; Jeong, H.G. Metformin induces microrna-34a to downregulate the sirt1/pgc-1alpha/nrf2 pathway, leading to increased susceptibility of wild-type p53 cancer cells to oxidative stress and therapeutic agents. Free Radic. Biol. Med. 2014, 74, 21–34. [Google Scholar] [PubMed]
- Li, W.; Yan, M.H.; Liu, Y.; Liu, Z.; Wang, Z.; Chen, C.; Zhang, J.; Sun, Y.S. Ginsenoside rg5 ameliorates cisplatin-induced nephrotoxicity in mice through inhibition of inflammation, oxidative stress, and apoptosis. Nutrients 2016, 8. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.Z.; Ren, S.; Yan, X.T.; Sun, Y.S.; Li, H.P.; Li, W.; Liu, Y.Y.; Wang, Z. Improvement of cisplatin-induced renal dysfunction by schisandra chinensis stems via anti-inflammation and anti-apoptosis effects. J. Ethnopharmacol. 2018, 217, 228–237. [Google Scholar] [CrossRef] [PubMed]
- Yao, X.; Panichpisal, K.; Kurtzman, N.; Nugent, K. Cisplatin nephrotoxicity: A review. Am. J. Med. Sci. 2007, 334, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-L.; Li, T.; Li, J.-H.; Miao, S.-Y.; Xiao, X.-Z. The effects of resveratrol on inflammation and oxidative stress in a rat model of chronic obstructive pulmonary disease. Molecules 2017, 22, 1529. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.; Wu, W.-F.; Dong, L.; Ren, G.-L.; Li, H.-D.; Yang, Q.; Li, X.-F.; Xu, T.; Li, Z.; Wu, B.-M.; et al. Protocatechuic aldehyde attenuates cisplatin-induced acute kidney injury by suppressing nox-mediated oxidative stress and renal inflammation. Front. Pharmacol. 2016, 7, 479. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Yao, Y.; Huang, H.; Hao, H.; Ying, M. Xanthohumol attenuates cisplatin-induced nephrotoxicity through inhibiting nf-kappab and activating nrf2 signaling pathways. Int. Immunopharmacol. 2018, 61, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Zhou, P.H.; Xu, C.G.; Zhou, X.J.; Hu, W.; Zhang, J. Baicalein attenuates renal fibrosis by inhibiting inflammation via down-regulating nf-κb and mapk signal pathways. J. Mol. Histol. 2015, 46, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.N.; Li, Y.Z.; Li, W.; Yan, X.T.; Yang, G.; Zhang, J.; Zhao, L.C.; Yang, L.M. Nephroprotective effects of saponins from leaves of panax quinquefolius against cisplatin-induced acute kidney injury. Int. J. Mol. Sci. 2017, 18, 1407. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.N.; Liu, Z.; Wang, Z.; Ren, S.; Tang, S.; Wang, Y.P.; Xiao, S.Y.; Chen, C.; Li, W. Supplementation of american ginseng berry extract mitigated cisplatin-evoked nephrotoxicity by suppressing ros-mediated activation of mapk and NF-κB signaling pathways. Food Chem. Toxicol. 2017, 110, 62–73. [Google Scholar] [CrossRef] [PubMed]
- Suliman, F.A.; Khodeer, D.M.; Ibrahiem, A.; Mehanna, E.T.; El-Kherbetawy, M.K.; Mohammad, H.M.F.; Zaitone, S.A.; Moustafa, Y.M. Renoprotective effect of the isoflavonoid biochanin a against cisplatin induced acute kidney injury in mice: Effect on inflammatory burden and p53 apoptosis. Int. Immunopharmacol. 2018, 61, 8–19. [Google Scholar] [CrossRef] [PubMed]
- Ahn, D.S.; Lee, H.J.; Hwang, J.; Han, H.; Kim, B.; Shim, B.; Kim, S.-H. Lambertianic acid sensitizes non-small cell lung cancers to trail-induced apoptosis via inhibition of xiap/nf-κb and activation of caspases and death receptor 4. Int. J. Mol. Sci. 2018, 19, 1476. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Yuan, X.; Chen, Y.; Zheng, Q.; Xu, L.; Wu, Y. Endoplasmic reticulum stress mediated mdrv p10.8 protein-induced cell cycle arrest and apoptosis through the perk/eif2α pathway. Front. Microbiol. 2018, 9, 1327. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Sample of pseudoginsengenin DQ is available from the authors. |









| Groups | Dosage (mg/kg) | Body Weights (g) | Kidney Index (mg/g) | Spleen Index (mg/g) | |
|---|---|---|---|---|---|
| Initial | Final | ||||
| Normal | - | 30.37 ± 2.44 | 32.07 ± 1.51 | 16.25 ± 1.43 | 4.17 ± 0.56 |
| Cisplatin | - | 29.28 ± 1.57 | 27.41 ± 2.04 | 21.34 ± 1.96 ** | 1.71 ± 0.30 ** |
| PDQ | 30 | 31.32 ± 2.19 | 29.16 ± 2.54 | 19.28 ± 0.52 | 2.24 ± 0.25 # |
| PDQ | 60 | 31.44 ± 1.72 | 30.42 ± 1.73 | 18.03 ± 1.67 # | 2.47 ± 0.23 # |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qi, Z.; Li, Z.; Li, W.; Liu, Y.; Wang, C.; Lin, H.; Liu, J.; Li, P. Pseudoginsengenin DQ Exhibits Therapeutic Effects in Cisplatin-Induced Acute Kidney Injury via Sirt1/NF-κB and Caspase Signaling Pathway without Compromising Its Antitumor Activity in Mice. Molecules 2018, 23, 3038. https://doi.org/10.3390/molecules23113038
Qi Z, Li Z, Li W, Liu Y, Wang C, Lin H, Liu J, Li P. Pseudoginsengenin DQ Exhibits Therapeutic Effects in Cisplatin-Induced Acute Kidney Injury via Sirt1/NF-κB and Caspase Signaling Pathway without Compromising Its Antitumor Activity in Mice. Molecules. 2018; 23(11):3038. https://doi.org/10.3390/molecules23113038
Chicago/Turabian StyleQi, Zeng, Zhuo Li, Wei Li, Yunhe Liu, Cuizhu Wang, Hongqiang Lin, Jinping Liu, and Pingya Li. 2018. "Pseudoginsengenin DQ Exhibits Therapeutic Effects in Cisplatin-Induced Acute Kidney Injury via Sirt1/NF-κB and Caspase Signaling Pathway without Compromising Its Antitumor Activity in Mice" Molecules 23, no. 11: 3038. https://doi.org/10.3390/molecules23113038
APA StyleQi, Z., Li, Z., Li, W., Liu, Y., Wang, C., Lin, H., Liu, J., & Li, P. (2018). Pseudoginsengenin DQ Exhibits Therapeutic Effects in Cisplatin-Induced Acute Kidney Injury via Sirt1/NF-κB and Caspase Signaling Pathway without Compromising Its Antitumor Activity in Mice. Molecules, 23(11), 3038. https://doi.org/10.3390/molecules23113038






