Mollicellins O–R, Four New Depsidones Isolated from the Endophytic Fungus Chaetomium sp. Eef-10
Abstract
:1. Introduction
2. Results
2.1. Purification and Characterization
2.2. Structure Elucidation of Compounds 1–4
2.3. Antibacterial Assay
2.4. Cytotoxicity Assay
2.5. Antioxidant Assay
3. Discussion
4. Materials and Methods
4.1. General Experimental Procedures
4.2. Fungal Material
4.3. Fermentation, Extraction, and Isolation
4.4. Compound Identification
4.5. Antibacterial Assay
4.6. Cytotoxicity Assay
4.7. Antioxidant Activiity Assay
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Chang, H.S.; Lin, C.H.; Chen, Y.S.; Wang, H.C.; Chan, H.Y.; Hsieh, S.Y.; Wu, H.C.; Cheng, M.J.; Yuan, G.F.; Lin, S.Y.; et al. Secondary metabolites of the endophytic fungus Lachnum abnorme from Ardisia cornudentata. Int. J. Mol. Sci. 2016, 17, 1512. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, A.S.; Polonio, J.C.; Costa, A.T.; Menicoze dos Santos, C.; Rhoden, S.A.; Azevedo, J.L.; Pamphile, J.A. Bioprospection of culturable endophytic fungi associated with the ornamental plant Pachystachys lutea. Curr. Microbiol. 2018, 75, 588–596. [Google Scholar] [CrossRef] [PubMed]
- Kusari, S.; Singh, S.; Jayabaskaran, C. Rethinking production of Taxol (paclitaxel) using endophyte biotechnology. Trends Biotechnol. 2014, 32, 304–311. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.J.; Min, C.L.; Ge, M.; Zuo, R.H. An endophytic sanguinarine-producing fungus from Macleaya cordata, Fusarium proliferatum BLH51. Curr. Microbiol. 2014, 68, 336–341. [Google Scholar] [CrossRef] [PubMed]
- Pan, F.; Su, T.J.; Cai, S.M.; Wu, W. Fungal endophyte-derived Fritillaria unibracteata var. wabuensis: Diversity, antioxidant capacities in vitro and relations to phenolic, flavonoid or saponin compounds. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Tong, Q.; Zhu, H.; Tan, D.; Zhang, J.; Xue, Y.; Yao, G.; Luo, Z.; Wang, J.; Wang, Y.; et al. Nine new cytochalasan alkaloids from Chaetomium globosum TW1-1 (Ascomycota, Sordariales). Sci. Rep. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Ancheeva, E.; Mandi, A.; Kiraly, S.B.; Kurtan, T.; Hartmann, R.; Akone, S.H.; Weber, H.; Daletos, G.; Proksch, P. Chaetolines A and B, pyrano[3,2-f]isoquinoline alkaloids from cultivation of Chaetomium sp. in the presence of autoclaved Pseudomonas aeruginosa. J. Nat. Prod. 2018, 26, 2392–2398. [Google Scholar] [CrossRef]
- Yang, M.H.; Gu, M.L.; Han, C.; Guo, X.J.; Yin, G.P.; Yu, P.; Kong, L.Y. Aureochaeglobosins A–C, three [4 + 2] adducts of chaetoglobosin and aureonitol derivatives from Chaetomium globosum. Org. Lett. 2018, 20, 3345–3348. [Google Scholar] [CrossRef]
- Yu, F.X.; Chen, Y.; Yang, Y.H.; Li, G.H; Zhao, P.J. A new epipolythiodioxopiperazine with antibacterial and cytotoxic activities from the endophytic fungus Chaetomium sp. M336. Nat. Prod. Rep. 2018, 32, 689–694. [Google Scholar] [CrossRef]
- Yamada, T.; Doi, M.; Shigeta, H.; Muroga, Y.; Hosoe, S.; Numata, A.; Tanaka, R. Absolute stereostructures of cytotoxic metabolites, chaetomugilins A-C, produced by a Chaetomium species separated from a marine fish. Cheminform 2008, 39, 4192–4195. [Google Scholar] [CrossRef]
- Ge, H.M.; Zhang, W.Y.; Ding, G.; Saparpakorn, P.; Song, Y.C.; Hannongbua, S.; Tan, R.X. Chaetoglobins A and B, two unusual alkaloids from endophytic Chaetomium globosum culture. Chem. Commun. 2009, 40, 5978–5980. [Google Scholar] [CrossRef]
- Yamada, T.; Muroga, Y.; Jinno, M.; Kajimoto, T.; Usami, Y.; Numata, A.; Tanaka, R. New class azaphilone produced by a marine fish-derived Chaetomium globosum. The stereochemistry and biological activities. Bioorg. Med. Chem. 2011, 19, 4106–4113. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Wang, J.; Zhu, H.; Wang, J.; Xue, Y.; Wei, G.; Guo, Y.; Tan, D.; Zhang, J.; Yin, C.; et al. Chaephilones A and B, two new azaphilone derivatives isolated from Chaetomium globosum. Chem. Biodivers. 2016, 13, 422–426. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Liao, Y.; Chen, R.; Hou, Y.; Ke, W.; Zhang, B.; Gao, M.; Shao, Z.; Chen, J.; Li, F. Chlorinated azaphilone pigments with antimicrobial and cytotoxic activities isolated from the deep sea derived fungus Chaetomium sp. NA-S01-R1. Mar. Drugs 2018, 16, 61. [Google Scholar] [CrossRef] [PubMed]
- Sekita, S.; Yoshihira, K.; Natori, S. Structures of chaetoglobosin A and B, cytotoxic metabolites of Chaetomium globosum. Cheminform 1973, 4, 628–631. [Google Scholar] [CrossRef]
- Oikawa, H.; Murakami, Y.; Ichihara, A. New plausible precursors of chaetoglobosin an accumulated by treatment of chaetomium subaffine with cytochrome P-450 inhibitors. Tetrahedron Lett. 1991, 32, 4533–4536. [Google Scholar] [CrossRef]
- Cui, C.M.; Li, X.M.; Li, C.S.; Proksch, P.; Wang, B.G. Cytoglobosins A–G, cytochalasans from a marine-derived endophytic fungus, Chaetomium globosum QEN-14. J. Nat. Prod. 2010, 73, 729–733. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.S.; Zhang, Y.Y.; Yan, W.; Cao, L.L.; Xiao, Y.; Ye, Y.H. Chaetomium globosum CDW7, a potential biological control strain and its antifungal metabolites. FEMS Microbiol. Lett. 2017, 364. [Google Scholar] [CrossRef]
- Li, G.Y.; Li, B.G.; Yang, T.; Liu, G.Y; Zhang, G.L. Chaetoindicins A–C, three Isoquinoline alkaloids from the fungus Chaetomium indicum. Cheminform 2006, 37, 3613–3615. [Google Scholar] [CrossRef]
- Kanokmedhakul, S.; Kanokmedhakul, K.; Phonkerd, N.; Soytong, K.; Kongsaeree, P.; Suksamrarn, A. Antimycobacterial anthraquinone-chromanone compound and diketopiperazine alkaloid from the fungus Chaetomium globosum KMITL-N0802. Planta Med. 2002, 68, 834–836. [Google Scholar] [CrossRef]
- Wang, S.; Li, X.M.; Teuscher, F.; Diesel, A.; Ebel, R.; Proksch, P.; Wang, B.G. Chaetopyranin, a benzaldehyde derivative, and other related metabolites from Chaetomium globosum, an endophytic fungus derived from the marine red alga Polysiphonia urceolata. J. Nat. Prod. 2006, 69, 1622–1625. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Jiang, J.; Hu, S.; Ma, H.R.; Zhu, H.C.; Tong, Q.Y.; Cheng, L.; Hao, X.C.; Zhang, G.; Zhang, Y.H. Secondary metabolites from endophytic fungus Chaetomium sp. induce colon cancer cell apoptotic death. Fitoterapia 2017, 121, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Yoshimura, S.; Kinoshita, T.; Abe, F.; Nitta, K.; Ezaki, M.; Sakamoto, K.; Hashimoto, M.; Fujie, A.; Hino, M.; et al. FR207944, an antifungal antibiotic from Chaetomium sp. No. 217, 2: Isolation and structure elucidation. Biosci. Biotechnol. Biochem. 2005, 69, 1029–1032. [Google Scholar] [CrossRef] [PubMed]
- Stark, A.A.; Kobbe, B.; Matsuo, D.; Buchi, G.; Wogan, G.N.; Demain, A.L. Mollicellins: Mutagenic and antibacterial mycotoxins. Appl. Environ. Microbiol. 1978, 36, 412–420. [Google Scholar] [PubMed]
- Li, G.Y.; Li, B.G.; Yang, T.; Liu, G.Y.; Zhang, G.L. Secondary metabolites from the fungus Chaetomium brasiliense. Helv. Chim. Acta 2008, 91, 124–129. [Google Scholar] [CrossRef]
- Khumkomkhet, P.; Kanokmedhakul, S.; Kanokmedhakul, K.; Hahnvajanawong, C.; Soytong, K. Antimalarial and cytotoxic depsidones from the fungus Chaetomium brasiliense. J. Nat. Prod. 2009, 72, 1487–1491. [Google Scholar] [CrossRef] [PubMed]
- Bilal, S.; Ali, L.; Khan, A.L.; Shahzad, R.; Asaf, S.; Imran, M.; Kang, S.M.; Kim, S.K.; Lee, I.J. Endophytic fungus Paecilomyces formosus LHL10 produces sester-terpenoid YW3548 and cyclic peptide that inhibit urease and alpha-glucosidase enzyme activities. Arch. Microbiol. 2018, 200, 1493–1502. [Google Scholar] [CrossRef]
- Ibrahim, S.R.M.; Mohame, G.A.; Al Haidari, R.A.; El-Kholy, A.A.; Zayed, M.F.; Khayat, M.T. Biologically active fungal depsidones: Chemistry, biosynthesis, structural characterization, and bioactivities. Fitoterapia 2018, 129, 317–365. [Google Scholar] [CrossRef]
- Shan, T.; Tian, J.; Wang, X.; Mou, Y.; Mao, Z.; Lai, D.; Dai, J.; Peng, Y.; Zhou, L.; Wang, M. Bioactive spirobisnaphthalenes from the endophytic fungus Berkleasmium sp. J. Nat. Prod. 2014, 77, 2151–2160. [Google Scholar] [CrossRef]
- Wang, J.; Zhao, J.; Liu, H.; Zhou, L.; Liu, Z.; Wang, J.; Han, J.; Yu, Z.; Yang, F. Chemical analysis and biological activity of the essential oils of two Valerianaceous species from China: Nardostachys chinensis and Valeriana officinalis. Molecules 2010, 15, 6411–6422. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
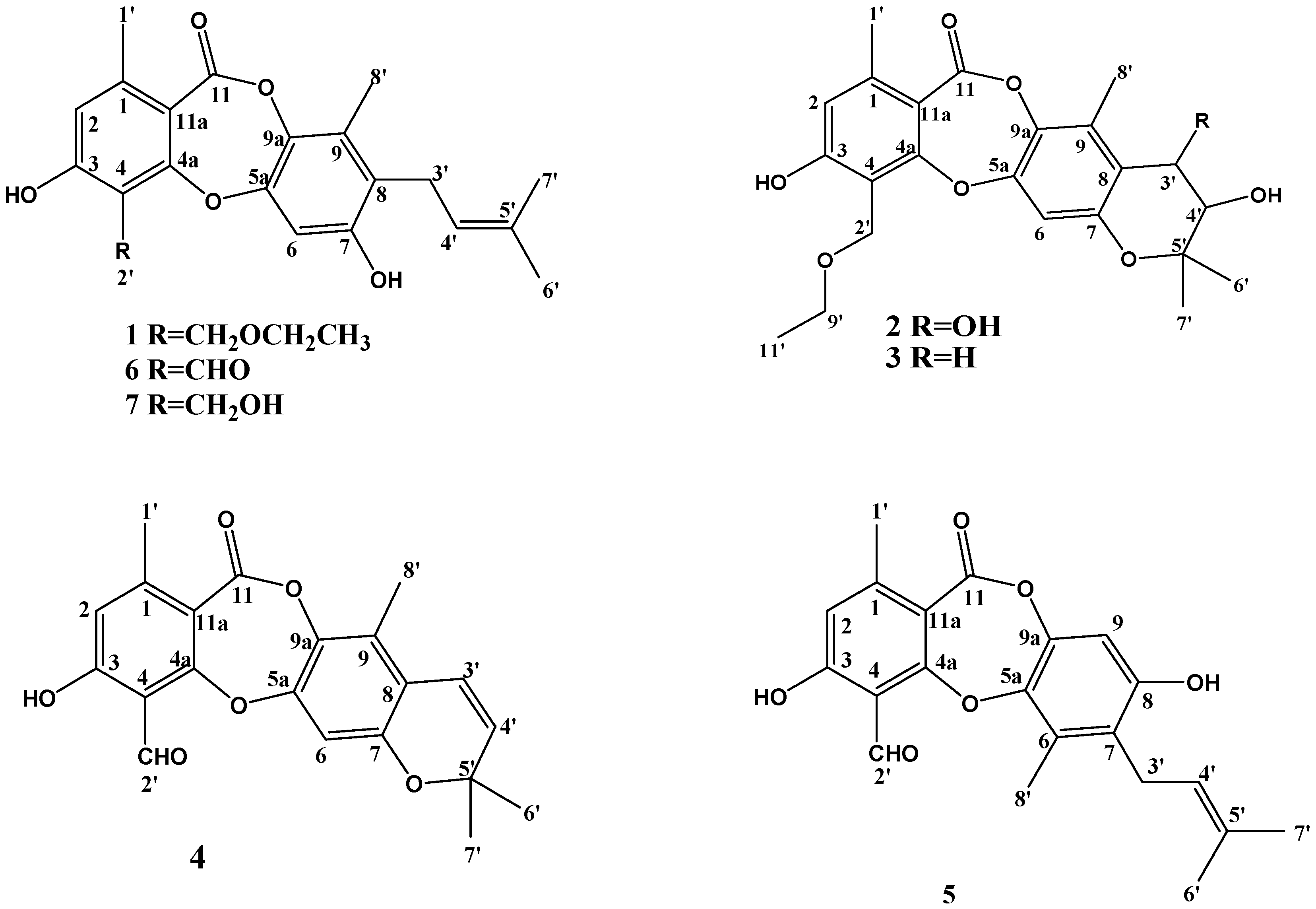




| Position | 1 | 2 | 3 | 4 | 5 | 6 | 7 |
|---|---|---|---|---|---|---|---|
| 1 | 144.60 | 144.74 | 150.66 | 154.10 | 153.72 | 151.66 | 144.02 |
| 2 | 116.32 | 116.76 | 116.43 | 118.14 | 117.93 | 117.60 | 116.45 |
| 3 | 161.16 | 161.81 | 161.31 | 165.90 | 166.05 | 164.04 | 161.35 |
| 4 | 114.41 | 114.66 | 114.54 | 111.69 | 111.90 | 112.06 | 116.59 |
| 4a | 162.45 | 162.43 | 162.33 | 165.37 | 166.56 | 164.53 | 161.37 |
| 5a | 149.90 | 153.32 | 144.57 | 151.07 | 143.03 | 148.56 | 149.96 |
| 6 | 106.36 | 101.39 | 108.11 | 107.3 | 130.95 | 105.07 | 106.21 |
| 7 | 152.57 | 157.92 | 150.98 | 151.41 | 143.35 | 152.58 | 152.70 |
| 8 | 125.73 | 127.03 | 118.38 | 119.49 | 123.01 | 125.56 | 125.82 |
| 9 | 129.90 | 128.85 | 129.40 | 127.22 | 105.82 | 129.47 | 129.99 |
| 9a | 137.00 | 138.01 | 137.35 | 137.34 | 154.62 | 135.24 | 137.05 |
| 11 | 164.02 | 163.81 | 163.93 | 162.51 | 162.82 | 162.76 | 164.08 |
| 11a | 114.00 | 113.68 | 113.97 | 113.97 | 114.21 | 112.94 | 113.95 |
| 1′ | 21.03 | 21.03 | 21.01 | 22.07 | 22.27 | 21.80 | 20.92 |
| 2′ | 62.35 | 62.77 | 62.61 | 194.92 | 194.70 | 191.53 | 56.12 |
| 3′ | 25.76 | 72.87 | 30.34 | 119.11 | 25.86 | 25.32 | 25.78 |
| 4′ | 123.08 | 99.27 | 69.64 | 132.46 | 126.01 | 122.39 | 123.15 |
| 5′ | 131.81 | 71.19 | 77.62 | 76.75 | 132.20 | 131.35 | 131.85 |
| 6′ | 25.86 | 26.15 | 25.82 | 27.81 | 25.95 | 25.85 | 25.91 |
| 7′ | 17.94 | 25.50 | 20.43 | 27.81 | 18.06 | 18.21 | 17.97 |
| 8′ | 12.64 | 12.36 | 12.24 | 11.75 | 13.49 | 12.81 | 12.67 |
| 9′ | 66.27 | 66.47 | 66.36 | - | - | - | - |
| 11′ | 15.53 | 15.58 | 15.50 | - | - | - | - |
| Position | 1 | 2 | 3 | 4 | 5 | 6 | 7 |
|---|---|---|---|---|---|---|---|
| 2 | 6.68 (s) | 6.71 (s) | 6.69 (s) | 6.80 (s) | 6.67 (s) | 6.80 (s) | 6.64 (s) |
| 6 | 6.86 (s) | 6.72 (s) | 6.71 (s) | 6.85 (s) | - | 6.79 (s) | 6.80 (s) |
| 9 | - | - | - | - | 6.78 (s) | - | - |
| 1′ | 2.36 (s) | 2.37 (s) | 2.37 (s) | 2.51 (s) | 2.49 (s) | 2.40 (s) | 2.36 (s) |
| 2′ | 4.77 (s) | 4.81 (s) | 4.82 (s) | 10.68 (s) | 10.77 (s) | 10.49 (s) | 4.99 (s) |
| 3′a | 3.33 (d, 6.2) | 5.42 (d, 3.8) | 2.86 (dd, 16.8, 5.6) | 6.58 (d,10.2) | 3.37 (d, 6.9) | 3.22 (d, 6.9) | 3.33 (d, 6.9) |
| 3′b | - | - | 2.52 (dd, 16.9, 7.7) | - | - | - | - |
| 4′ | 5.02 (t, 6.2) | 4.29 (d, 3.8) | 3.78 (dd, 7.6, 5.6) | 5.83 (d,10.2) | 5.03 (m) | 4.95 (t, 7.0) | 5.02 (m) |
| 6′ | 1.62 (s) | 1.19 (s) | 1.18 (s) | 1.37 (s) | 1.63 (s) | 1.60 (s) | 1.63 (s) |
| 7′ | 1.74 (s) | 1.25 (s) | 1.30 (s) | 1.37 (s) | 1.75 (s) | 1.70 (s) | 1.75 (s) |
| 8′ | 2.23 (s) | 2.35 (s) | 2.18 (s) | 2.30 (s) | 2.37 (s) | 2.18 (s) | 2.23 (s) |
| 9′ | 3.60 (q, 7.0) | 3.64 (q, 10.2) | 3.63 (q, 7.0) | - | - | - | - |
| 11′ | 1.19 (t, 7.0) | 1.21 (t, 10.2) | 1.21 (t, 7.0) | - | - | - | - |
| OH-3 | 9.02 (br s) | - | - | 12.25 (s) | 12.22 (s) | - | - |
| OH-7 | 9.02 (br s) | - | - | - | - | 9.70 (s) | - |
| OH-8 | - | - | - | - | 8.82 (s) | - | - |
| Compound | IC50 (µg/mL) | |
|---|---|---|
| S. aureus ATCC29213 | S. aureus N50 | |
| Mollicellin O (1) | 79.44 | 76.35 |
| Mollicellin H (6) | 5.14 | 6.21 |
| Mollicellin I (7) | 70.14 | 63.15 |
| Streptomycin sulfate | 1.05 | - a |
| Compounds | IC50 (µg/mL) | |
|---|---|---|
| HepG2 | Hela | |
| Mollicellin G (5) | 19.64 | 13.97 |
| Mollicellin H (6) | 6.83 | - a |
| Mollicellin I (7) | - a | 21.35 |
| Camptothecin | 3.6 | 6.3 |
| Compound | IC50 (µg/mL) |
|---|---|
| Mollicellin O (1) | 71.92 ± 0.09 |
| BHT | 0.15 ± 0.03 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ouyang, J.; Mao, Z.; Guo, H.; Xie, Y.; Cui, Z.; Sun, J.; Wu, H.; Wen, X.; Wang, J.; Shan, T. Mollicellins O–R, Four New Depsidones Isolated from the Endophytic Fungus Chaetomium sp. Eef-10. Molecules 2018, 23, 3218. https://doi.org/10.3390/molecules23123218
Ouyang J, Mao Z, Guo H, Xie Y, Cui Z, Sun J, Wu H, Wen X, Wang J, Shan T. Mollicellins O–R, Four New Depsidones Isolated from the Endophytic Fungus Chaetomium sp. Eef-10. Molecules. 2018; 23(12):3218. https://doi.org/10.3390/molecules23123218
Chicago/Turabian StyleOuyang, Jinkui, Ziling Mao, Hui Guo, Yunying Xie, Zehua Cui, Jian Sun, Huixiong Wu, Xiujun Wen, Jun Wang, and Tijiang Shan. 2018. "Mollicellins O–R, Four New Depsidones Isolated from the Endophytic Fungus Chaetomium sp. Eef-10" Molecules 23, no. 12: 3218. https://doi.org/10.3390/molecules23123218




