Evaluating Antitumor and Antioxidant Activities of Yellow Monascus Pigments from Monascus ruber Fermentation
Abstract
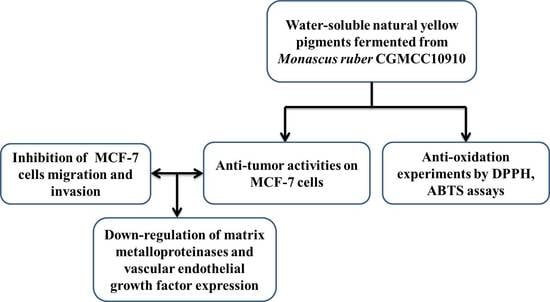
1. Introduction
2. Results and Discussion
2.1. Antioxidant Activities of Water-Soluble NYMPs
2.2. Cytotoxicity of Water-Soluble NYMPs on MCF-7 Cells
2.3. Regulating Effect of Water-Soluble NYMPs on the Expression of MMP-2, MMP-9, and VEGF
3. Materials and Methods
3.1. Microorganisms and Chemicals
3.2. Monascus Pigment Fermentation
3.3. Isolation of Water-Soluble NYMPs
3.4. Antioxidant Activity Determination
3.5. MCF-7 Cell Line and Culture
3.6. Measurement of Cytotoxicity and Inhibition Activity in Cell Proliferation
3.7. Transwell Assay
3.8. Wound-Healing Assay
3.9. Invasion Assay
3.10. Western Blotting
3.11. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chen, G.; Wu, Z.-Q. Production and biological activities of yellow pigments from Monascus fungi. World J. Microbiol. Biotechnol. 2016, 32, 136–144. [Google Scholar] [CrossRef] [PubMed]
- Patakova, P. Monascus secondary metabolites: Production and biological activity. J. Ind. Microbiol. Biotechnol. 2013, 40, 169–181. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Zhang, Y.; Chen, D.; Chen, H.; Lin, L.; Zheng, C.; Guo, Y. Monascus pigment Rubropunctatin: A potential dual agent for cancer chemotherapy and phototherapy. J. Agric. Food Chem. 2016, 64, 2541–2548. [Google Scholar] [CrossRef] [PubMed]
- Hsu, W.-H.; Pan, T.-M. Monascus purpureus-fermented products and oral cancer: A review. Appl. Microbiol. Biotechnol. 2012, 93, 1831–1842. [Google Scholar] [CrossRef] [PubMed]
- Su, N.-W.; Lin, Y.-L.; Lee, M.-H.; Ho, C.-Y. Ankaflavin from Monascus-fermented red rice exhibits selective cytotoxic effect and induces cell death on Hep G2 cells. J. Agric. Food Chem. 2005, 53, 1949–1954. [Google Scholar] [CrossRef] [PubMed]
- Akihisa, T.; Tokuda, H.; Ukiya, M.; Kiyota, A.; Yasukawa, K.; Sakamoto, N.; Kimura, Y.; Suzuki, T.; Takayasu, J.; Nishino, H. Anti-tumor-initiating effects of Monascin, an azaphilonoid pigment from the extract of Monascus pilosus fermented rice (red-mold rice). Chem. Biodivers. 2005, 2, 1305–1309. [Google Scholar] [CrossRef] [PubMed]
- Camphausen, K.; Chiu, H.-W.; Fang, W.-H.; Chen, Y.-L.; Wu, M.-D.; Yuan, G.-F.; Ho, S.-Y.; Wang, Y.-J. Monascuspiloin enhances the radiation sensitivity of human prostate cancer cells by stimulating endoplasmic reticulum stress and inducing autophagy. PLoS ONE 2012, 7, e40462. [Google Scholar] [CrossRef]
- Hsu, L.-C.; Hsu, Y.-W.; Liang, Y.-H.; Liaw, C.-C.; Kuo, Y.-H.; Pan, T.-M. Induction of apoptosis in human breast adenocarcinoma cells MCF-7 by monapurpyridine A, a new azaphilone derivative from Monascus purpureus NTU 568. Molecules 2012, 17, 664–673. [Google Scholar] [CrossRef]
- Hsu, Y.-W.; Hsu, L.-C.; Chang, C.-L.; Liang, Y.-H.; Kuo, Y.-H.; Pan, T.-M. New anti-inflammatory and anti-proliferative constituents from fermented red mold rice Monascus purpureus NTU 568. Molecules 2010, 15, 7815–7824. [Google Scholar] [CrossRef]
- Ho, B.-Y.; Pan, T.-M. The Monascus metabolite monacolin K reduces tumor progression and metastasis of Lewis lung carcinoma cells. J. Agric. Food Chem. 2009, 57, 8258–8265. [Google Scholar] [CrossRef]
- Shi, Y.-C.; Pan, T.-M.; Liao, V.-H. Monascin from Monascus-fermented products reduces oxidative stress and amyloid-beta toxicity via DAF-16/FOXO in caenorhabditis elegans. J. Agric. Food Chem. 2016, 64, 7114–7120. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Huang, T.; Chen, G.; Wu, Z.-Q. Production of water-soluble yellow pigments via high glucose stress fermentation of Monascus ruber CGMCC 10910. Appl. Microbiol. Biotechnol. 2017, 101, 3121–3130. [Google Scholar] [CrossRef] [PubMed]
- Barchuk, A.; Bespalov, A.; Huhtala, H.; Chimed, T.; Laricheva, I.; Belyaev, A.; Bray, F.; Anttila, A.; Auvinen, A. Breast and cervical cancer incidence and mortality trends in Russia 1980–2013. Cancer Epidemiol. 2018, 55, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, C.; Wilson, C.; Hutchinson, A.D.; Grunfeld, E.A. The relationship between anticipated response and subsequent experience of cancer treatment-related side effects: A meta-analysis comparing effects before and after treatment exposure. Cancer Treat. Rev. 2018, 68, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Kasinathan, A.; Ganesan, R.; Balasubramanian, A.; Bhaskaran, J.; Suresh, S.; Srinivasan, R.; Aravind, K.B.; Sivalingam, N. Curcumin induces apoptosis and cell cycle arrest via the activation of reactive oxygen species–independent mitochondrial apoptotic pathway in Smad4 and p53 mutated colon adenocarcinoma HT29 cells. Nutr. Res. 2018, 51, 67–81. [Google Scholar] [CrossRef] [PubMed]
- García, E.R.; Gutierrez, E.A.; Melo, F.C.S.A.D.; Novaes, R.D.; Gonçalves, R.V. Flavonoids effects on hepatocellular carcinoma in murine models: A systematic review. Evid. Based Complement. Altern. Med. 2018, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Russo, M.; Spagnuolo, C.; Russo, G.L.; Skalicka-Wozniak, K.; Daglia, M.; Sobarzo-Sanchez, E.; Nabavi, S.F.; Nabavi, S.M. Nrf2 targeting by sulforaphane: A potential therapy for cancer treatment. Crit. Rev. Food Sci. 2018, 58, 1391–1405. [Google Scholar] [CrossRef]
- Chikara, S.; Nagaprashantha, L.D.; Singhal, J.; Horne, D.; Awasthi, S.; Singhal, S.S. Oxidative stress and dietary phytochemicals: Role in cancer chemoprevention and treatment. Cancer Lett. 2018, 413, 122–134. [Google Scholar] [CrossRef]
- Huang, G.-J.; Yang, C.-M.; Chang, Y.-S.; Amagaya, S.; Wang, H.-C.; Hou, W.-C.; Huang, S.-S.; Hu, M.-L. Hispolon suppresses SK-Hep1 human hepatoma cell metastasis by inhibiting matrix metalloproteinase-2/9 and urokinase-plasminogen activator through the PI3K/Akt and ERK signaling pathways. J. Agric. Food Chem. 2010, 58, 9468–9475. [Google Scholar] [CrossRef]
- Fujii, T.; Yajima, R.; Tatsuki, H.; Oosone, K.; Kuwano, H. Anticancer effect of rapamycin on MCF-7 via downregulation of VEGF expression. Vitr. Cell Dev. Biol. Anim. 2016, 52, 45–48. [Google Scholar] [CrossRef]
- Barr, M.P.; Bouchier-Hayes, D.J.; Harmey, J.H. Vascular endothelial growth factor is an autocrine survival factor for breast tumour cells under hypoxia. Int. J. Oncol. 2008, 32, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.; Tan, H.-L.; Chen, G.; Wang, L.; Wu, Z.-Q. Rising temperature stimulates the biosynthesis of water-soluble fluorescent yellow pigments and gene expression in Monascus ruber CGMCC10910. AMB Express. 2017, 7, 134. [Google Scholar] [CrossRef] [PubMed]
- Srianta, I.; Zubaidah, E.; Estiasih, T.; Iuchi, Y.; Harijono, H.; Yamada, M. Antioxidant activity of pigments derived from Monascus purpureus-fermented rice, corn, and sorghum. Int. Food Res. J. 2017, 24, 1186–1191. [Google Scholar] [CrossRef]
- Huang, Q.; Zhang, H.; Xue, D. Enhancement of antioxidant activity of Radix Puerariae and red yeast rice by mixed fermentation with Monascus purpureus. Food Chem. 2017, 226, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Jongrungruangchok, S.; Kittakoop, P.; Yongsmith, B.; Bavovada, R.; Tanasupawat, S.; Lartpornmatulee, N.; Thebtaranonth, Y. Azaphilone pigments from a yellow mutant of the fungus Monascus kaoliang. Phytochemistry 2004, 65, 2569–2575. [Google Scholar] [CrossRef]
- Hsu, Y.-W.; Hsu, L.-C.; Liang, Y.-H.; Kuo, Y.-H.; Pan, T.-M. Monaphilones A–C, three new antiproliferative azaphilone derivatives from Monascus purpureus NTU 568. J. Agric. Food Chem. 2010, 58, 8211–8216. [Google Scholar] [CrossRef]
- Chang, W.-T.; Chuang, C.-H.; Lee, W.-J.; Huang, C.-S. Extract of Monascus purpureus CWT715 fermented from sorghum liquor biowaste inhibits migration and invasion of SK-Hep-1 human hepatocarcinoma cells. Molecules 2016, 21, 1691. [Google Scholar] [CrossRef]
- Zheng, Y.-Q.; Xin, Y.-W.; Shi, X.-N.; Guo, Y.-H. Anti-cancer effect of rubropunctatin against human gastric carcinoma cells BGC-823. Appl. Microbiol. Biot. 2010, 88, 1169–1177. [Google Scholar] [CrossRef]
- Hsu, Y.-W.; Hsu, L.-C.; Liang, Y.-H.; Kuo, Y.-H.; Pan, T.-M. New bioactive orange pigments with yellow fluorescence from Monascus-fermented dioscorea. J. Agric. Food Chem. 2011, 59, 4512–4518. [Google Scholar] [CrossRef]
- Park, H.-J.; Kim, I.-S. Antioxidant activities and anticancer effects of red yeast rice grown in the medium containing garlic. Food Sci. Biotechnol. 2011, 20, 297–302. [Google Scholar] [CrossRef]
- Gialeli, C.; Theocharis, A.D.; Karamanos, N.K. Roles of matrix metalloproteinases in cancer progression and their pharmacological targeting. FEBS J. 2011, 278, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Duffy, M.J.; Maguire, T.M.; Hill, A.; McDermott, E.; O’Higgins, N. Metalloproteinases: Role in breast carcinogenesis, invasion and metastasis. Breast Cancer Res. 2000, 2, 252–257. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Kim, H.J.; Park, K.G.; Kim, Y.N.; Kwon, T.K.; Park, J.Y.; Lee, K.U.; Kim, J.G.; Lee, I.K. alpha-Lipoic acid inhibits matrix metalloproteinase-9 expression by inhibiting NF-kappa B transcriptional activity. Exp. Mol. Med. 2007, 39, 106–113. [Google Scholar] [CrossRef] [PubMed]
- Jezierska, A.; Motyl, T. Matrix metalloproteinase-2 involvement in breast cancer progression: A mini-review. Med. Sci. Monit. 2009, 15, 32–40. [Google Scholar] [CrossRef]
- El-Far, S.W.; Helmy, M.W.; Khattab, S.N.; Bekhit, A.A.; Hussein, A.A.; Elzoghby, A.O. Phytosomal bilayer-enveloped casein micelles for codelivery of monascus yellow pigments and resveratrol to breast cancer. Nanomedicine 2018, 13, 481–499. [Google Scholar] [CrossRef]
- Wu, Z.-Q.; Wang, M.-H.; Tang, R.; Chen, G. Adsorption and separation of Monascus yellow pigments in Monascus fermentation broth. Mod. Food Sci. Technol. 2017, 33, 150–157. [Google Scholar] [CrossRef]
- Wang, L.; Wu, Y.-N.; Huang, T.; Shi, K.; Wu, Z.-Q. Chemical compositions, antioxidant and antimicrobial activities of essential oils of Psidium guajava L. Leaves from different geographic regions in China. Chem. Biodivers. 2017, 14, e1700114. [Google Scholar] [CrossRef]
- Justus, C.R.; Leffler, N.; Ruiz-Echevarria, M.; Yang, L.V. In vitro Cell Migration and Invasion Assays. J. Vis. Exp. 2014, 1–8. [Google Scholar] [CrossRef]
Sample Availability: Samples of natural yellow Monascus pigments and reduced yellow Monascus pigments are not available from the authors. |






| Samples (mg/mL) | 0.025 | 0.075 | 0.100 | 0.500 | 1.000 |
|---|---|---|---|---|---|
| RYMPs b | d 3.59 ± 0.14 | 6.57 ± 0.75 | 8.91 ± 0.19 | 20.75 ± 0.13 | 32.84 ± 1.02 |
| NYMPs a | 9.54 ± 0.23 | 21.23 ± 1.37 | 26.74 ± 0.36 | 69.03 ± 0.92 | 86.33 ± 1.50 |
| BHT c | 14.25 ± 1.45 | 37.48 ± 2.63 | 47.07 ± 2.40 | 94.42 ± 0.17 | 95.14 ± 0.58 |
| Samples (mg/mL) | 0.025 | 0.050 | 0.075 | 0.100 | 0.250 |
|---|---|---|---|---|---|
| RYMPs b | d 13.95 ± 0.94 | 25.33 ± 0.32 | 38.00 ± 0.86 | 45.77 ± 0.33 | 84.92 ± 1.77 |
| NYMPs a | 38.00 ± 1.71 | 60.67 ± 1.00 | 81.92 ± 0.33 | 87.03 ± 0.11 | 99.95 ± 0.09 |
| BHT c | 94.42 ± 2.48 | 99.70 ± 0.38 | 99.45 ± 0.31 | 99.76 ± 0.28 | 98.61 ± 0.10 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tan, H.; Xing, Z.; Chen, G.; Tian, X.; Wu, Z. Evaluating Antitumor and Antioxidant Activities of Yellow Monascus Pigments from Monascus ruber Fermentation. Molecules 2018, 23, 3242. https://doi.org/10.3390/molecules23123242
Tan H, Xing Z, Chen G, Tian X, Wu Z. Evaluating Antitumor and Antioxidant Activities of Yellow Monascus Pigments from Monascus ruber Fermentation. Molecules. 2018; 23(12):3242. https://doi.org/10.3390/molecules23123242
Chicago/Turabian StyleTan, Hailing, Ziyi Xing, Gong Chen, Xiaofei Tian, and Zhenqiang Wu. 2018. "Evaluating Antitumor and Antioxidant Activities of Yellow Monascus Pigments from Monascus ruber Fermentation" Molecules 23, no. 12: 3242. https://doi.org/10.3390/molecules23123242
APA StyleTan, H., Xing, Z., Chen, G., Tian, X., & Wu, Z. (2018). Evaluating Antitumor and Antioxidant Activities of Yellow Monascus Pigments from Monascus ruber Fermentation. Molecules, 23(12), 3242. https://doi.org/10.3390/molecules23123242