3.5. Typical Procedure D for the Synthesis of Unprotected 3-iodo Glucosylquinolinone β-2g
A mixture of 3-iodo β-N-glucosylquinolinones (100 mg, 1.0 equiv.) and K2CO3 (12 mg, 0.5 equiv.) in methanol (3 mL) was placed in a small balloon and the mixture was stirred under argon at room temperature for 30 min to 1 h. The crude mixture was then filtered through celite, washed with 10 mL of methanol, and filtered for only 1 min. The filtrate was concentrated under reduced pressure at 25 °C for 1–2 h.
(2R,3R,4S,5R,6R)-2-(acetoxymethyl)-6-(3-bromo-2-oxoquinolin-1(2H)-yl)tetrahydro-2H-pyran-3,4,5-triyl triacetate 2a: Following procedure A, A 50-mL round tube flash was charged with 1a N-(2,3,4,6-tetra-O-acetyl-1-deoxy-α-d-glucopyranosyl)-quinolin-2-one (500 mg, 1 equiv.) and freshly crystallized NBS (469 mg, 2.5 equiv.). Under an argon atmosphere, 30 mL of anhydrous DMF was added. The mixture was heated to 70 °C and stirred until reaction completeness (72 h), ascertained by TLC. The crude was diluted with EtOAc and extracted with saturated NH4Cl (50 mL × 3). The organic layer was washed with water, dried by MgSO4, and concentrated under vacuum. The residue was purified by silica gel column chromatography (cyclohexane/EtOAc 6/4) to afford the desired product 2a as white powder (345 mg, 59%); m.p. = 213–215 °C. + 103.0 (c, 1.0 in CHCl3). 1H-NMR (300 MHz, CDCl3) δ 8.08 (s, 1H), 7.95 (d, J = 8.8 Hz, 1H), 7.58 (dd, J = 7.3 Hz, 8.6 Hz, 1H), 7.48 (d, J = 7.4 Hz, 1H), 7.29 (t, J = 7.9 Hz, 1H), 6.89 (d, J = 10.0 Hz, 1H), 5.89 (t, J = 9.3 Hz, 1H), 5.46 (t, J = 9.3 Hz, 1H), 5.36 (t, J = 9.8 Hz, 1H), 4.30 (dd, J = 12.4, 4.4 Hz, 1H), 4.23 (dd, J = 12.5, 2.0 Hz, 1H), 4.08–4.00 (m, 1H), 2.09 (s, 6H), 2.00 (s, 3H), 1.81 (s, 3H); 13C-NMR (75 MHz, CDCl3) δ 170.5 (C), 169.9 (C), 169.8 (C), 169.3 (C), 158.8 (C), 142.8 (CH), 137.1 (C), 130.8 (CH), 128.9 (CH), 123.9 (CH), 121.1 (C), 116.9 (CH), 82.4 (CH), 75.3 (CH), 73.8 (CH), 68.0 (CH), 67.9 (CH), 61.8 (CH2), 20.9 (CH3), 20.7 (CH3), 20.7 (CH3), 20.3 (CH3). FT-IR (neat, cm−1) 1757, 1652, 1366, 1229, 1032, 905, 837. HRMS (ESI, m/z) calcd. for C23H24NO10BrNa [M + Na]+: 576.0481 found 576.0485.
(2S,3S,4R,5S,6R)-2-(acetoxymethyl)-6-(3-bromo-2-oxoquinolin-1(2H)-yl)tetrahydro-2H-pyran-3,4,5-triyl triacetate 2b: Following procedure A, a 30-mL round tube flash was charged with 1b N-(2,3,4,6-tetra-O-acetyl-1-deoxy-α-d-glucopyranosyl)-quinolin-2-one (200 mg, 1 equiv.) and freshly crystallized NBS (188 mg, 2.5 equiv.). Under an argon atmosphere, 12 mL of anhydrous DMF was added. The mixture was heated to 70 °C and stirred until reaction completeness (72 h), ascertained by TLC. The crude was diluted with EtOAc and extracted with saturated NH4Cl (50 mL × 3). The organic layer was washed with water, dried by MgSO4, and concentrated under vacuum. The residue was purified by silica gel column chromatography (heptane/EtOAc 6/4) to afford the desired product 2b as white powder (173 mg, 74%); 1H-NMR (300 MHz, CDCl3) δ 8.11 (s, 1H), 7.76 (d, J = 8.6 Hz, 1H), 7.54 (dd, J = 8.6 Hz, 7.4, 1H), 7.47 (d, J = 7.6 Hz, 1H), 7.26 (t, J = 7.4 Hz, 1H), 5.37 (t, J = 6.4 Hz, 1H), 5.24 (t, J = 7.8 Hz, 1H), 4.71–4.62 (m, 1H), 4.39 (dd, J = 12.4, 4.8 Hz, 1H), 4.16 (dd, J = 12.4, 2.7 Hz, 1H), 2.10 (s, 3H), 2.08 (s, 3H), 2.05 (s, 3H), 1.73 (s, 3H); 13C-NMR (75 MHz, CDCl3) δ 170.7 (C), 169.9 (C), 169.8 (C), 169.8 (C), 159.5 (C), 142.4 (CH), 139.4 (C), 130.7 (CH), 128.1 (CH), 123.7 (CH), 121.3 (C), 117.1 (C), 116.6 (CH), 80.8 (CH), 73.4 (CH), 72.5 (CH), 70.2 (CH), 68.0 (CH), 61.7 (CH2), 21.0 (CH3), 20.9 (CH3), 20.9 (CH3), 20.4 (CH3).
(2R,3R,4S,5R,6R)-2-(acetoxymethyl)-6-(3-bromo-4-(4-methoxyphenyl)-2-oxoquinolin-1(2H)-yl)tetrahydro-2H-pyran-3,4,5-triyl triacetate 2c: Following procedure A, a 10-mL round tube flash was charged with 1c (50 mg, 1 equiv.) and freshly crystallized NBS (39 mg, 2.5 equiv.). Under an argon atmosphere, 3 mL of anhydrous DMF was added. The mixture was heated to 70 °C and stirred until reaction completeness (4 h), ascertained by TLC. The crude was diluted with EtOAc and extracted with saturated NH4Cl (50 mL × 3). The organic layer was washed with water, dried by MgSO4, and concentrated under vacuum. The residue was purified by silica gel column chromatography (cyclohexane/EtOAc 5/5) to afford the desired product 2c as pale yellow solid (34 mg, 60%); m.p.: 105–107 °C; Rf = 0.48 (Ethyl/Cyclohexane: 5/5); + 35.55 (c, 1.0 in CHCl3); IR (neat): 2963, 1756, 1650, 1604, 1554, 1511, 1455, 1366, 1260, 1248, 1033,1013,802, 763 cm−1; 1H-NMR (300 MHz, CDCl3) δ 8.00 (d, J = 8.5 Hz, 1H), 7.59–7.53 (m, 1H), 7.22–7.13 (m, 4H), 7.02 (dd, J = 17.8, 9.3 Hz, 3H), 5.97 (t, J = 9.3 Hz, 1H), 5.44 (dt, J = 30.0, 9.5 Hz, 2H), 4.33–4.24 (m, 2H), 4.17–4.09 (m, 1H), 3.90 (s, 3H), 2.11 (s, 6H), 2.02 (s, 4H), 1.85 (s, 2H); 13C-NMR (75 MHz, CDCl3) δ 170.56, (C) 169.95 (C), 169.77 (C), 169.65 (C), 169.28 (C), 160.00 (C), 158.88 (C), 152.51 (C), 136.55 (C), 130.55 (CH), 130.08 (CH), 129.90 (CH), 129.42 (C), 129.25 (CH), 123.56 (CH), 122.35 (C), 116.76 (CH), 114.23 (2CH), 82.55 (CH), 75.33 (CH), 73.96 (CH), 68.05 (CH), 61.85 (CH2), 55.49 (CH) 20.86 (CH3), 20.75 (CH3), 20.73 (2CH3), 20.37 (CH3); HR-MS (ESI positive, m/z): found 682.0894 ([M + Na]+), calc. for C30H30NO11NaBr (M + Na): 682.0900.
(2R,3R,4S,5R,6R)-2-(acetoxymethyl)-6-(3-iodo-2-oxoquinolin-1(2H)-yl)tetrahydro-2H-pyran-3,4,5-triyl triacetate 2d: Following procedure B, a solution of β-3-bromo N-glycosylquinolinone 2a (0.722 mmol, 400 mg), NaI (1.443 mmol, 217 mg), trans-N,N′-dimethylcyclohexane-1,2-diamine (21 mg, 0,14 mmol), and CuI (14 mg, 0.072 mmol) was stirred at 110 °C overnight. The crude product was purified by silica gel flash chromatography (Cyclohexane/EtOAc 5/5), and the product 2d was isolated as a pale yellow solid (362 mg, 83%); m.p.: 222.7–223.8 °C; Rf = 0.56 (Cyclohexane/EtOAc: 5/5); + 96.29 (c, 1.0 in CHCl3); IR (neat): 1746, 1648, 1596, 1365, 1217, 1077, 914, 751 cm−1; 1H-NMR (300 MHz, CDCl3) δ 8.35 (s, 1H), 7.95 (d, J = 8.6 Hz, 1H), 7.58 (t, J = 7.9 Hz, 1H), 7.45 (d, J = 7.7 Hz, 1H), 7.31–7.24 (m, 1H), 6.89 (d, J = 9.8 Hz, 1H), 5.88 (t, J = 9.3 Hz, 1H), 5.49–5.29 (m, 2H), 4.33–4.19 (m, 2H), 4.07–3.97 (m, 1H), 2.10 (s, 6H), 2.00 (s, 3H), 1.80 (s, 3H); 13C-NMR (75 MHz, CDCl3) δ 170.55, 169.90, 169.76, 169.24, 159.22, 150.33, 137.80, 131.02, 128.61, 123.69, 122.32, 116.94, 92.17, 82.92, 75.30, 73.77, 68.03, 67.94, 61.82, 20.85, 20.74, 20.70, 20.27; HR-MS (ESI positive, m/z): found 624.0339 ([M + Na]+), calc. for C23H24NO10NaI (M + Na): 624.0343.
(2S,3S,4R,5S,6R)-2-(acetoxymethyl)-6-(3-iodo-2-oxoquinolin-1(2H)-yl)tetrahydro-2H-pyran-3,4,5-triyl triacetate 2e: Following procedure B, α-3-bromo N-glycosylquinolinone 2b (0.288 mmol, 160 mg), NaI (0.577 mmol, 87 mg), trans-N,N′-dimethylcyclohexane-1,2-diamine (8.2 mg, 0,057 mmol), and CuI (5.5 mg, 0.028 mmol) were stirred at 110 °C overnight. The crude product was purified by silica gel flash chromatography (EtOAc/heptan: 5/5), and the product 2e was isolated as a pale yellow solid (124 mg, 73%): m.p.: 111.4–123.4 °C; Rf = 0.26 (EtOAc/heptan: 5/5); + 370 (c, 1.0 in CHCl3); IR (neat): 1739, 1641, 1596, 1367, 1260, 1205, 1031, 817, 798 cm−1; 1H-NMR (300 MHz, CDCl3) δ 8.37 (s, 1H), 7.77 (d, J = 8.7 Hz, 1H), 7.53 (t, J = 7.9 Hz, 1H), 7.44 (d, J = 7.8 Hz, 1H), 7.23 (d, J = 7.6 Hz, 1H), 6.70 (d, J = 6.2 Hz, 1H), 6.01 (t, J = 6.7 Hz, 1H), 5.36 (t, J = 6.3 Hz, 1H), 5.23 (t, J = 7.7 Hz, 1H), 4.70–4.63 (m, 1H), 4.39 (dd, J = 12.4, 4.9 Hz, 1H), 4.17 (dd, J = 12.4, 3.0 Hz, 1H), 2.13–2.04 (m, 9H), 1.73 (s, 3H); 13C-NMR (75 MHz, CDCl3) δ 170.73 (C), 169.93 (C), 169.76 (2C), 160.03 (C), 149.95 (CH), 140.12 (C), 130.86 (CH), 127.84 (CH), 123.48 (CH), 122.48 (C), 116.70(CH), 93.58 (C), 80.99 (CH), 73.35 (CH), 72.45 (CH), 70.17 (CH), 68.06 (CH), 61.67 (CH2), 21.06 (CH3), 20.94 (CH3), 20.89 (CH3), 20.46 (CH3); HR-MS (ESI positive, m/z): found 624.0344 ([M + Na]+), calc. for C23H24NO10NaI (M + Na): 624.0343.
(2R,3R,4S,5R,6R)-2-(acetoxymethyl)-6-(3-iodo-4-(4-methoxyphenyl)-2-oxoquinolin-1(2H)-yl)tetrahydro-2H-pyran-3,4,5-triyl triacetate 2f: Following procedure B, N-glycosylquinolinone β-2c (0.136 mmol, 90 mg), NaI (0.272 mmol, 41 mg), trans-N,N′-dimethylcyclohexane-1,2-diamine (4 mg, 0,027 mmol), and CuI (3 mg, 0.0135 mmol) were stirred at 110 °C overnight. The crude product was purified by silica gel flash chromatography (Cyclohexane/EtOAc 6/4), and the product 2f was isolated as a yellow solid (67 mg, yield 70%); m.p.: 106.6–121.8 °C; Rf = 0.48 (Cyclohexane/EtOAc 6/4); + 22.35 (c, 1.0 in CHCl3); IR (neat): 1756, 1652, 1604, 1511, 1367, 1249, 1217, 1113, 1097, 1035, 765 cm−1; 1H-NMR (300 MHz, Acetone) δ 8.34 (d, J = 8.6 Hz, 1H), 7.70–7.63 (m, 1H), 7.26–7.06 (m, 5H), 7.02 (d, J = 9.9 Hz, 1H), 5.98 (t, J = 10.6 Hz, 1H), 5.59 (t, J = 9.5, 1H), 5.47 (t, J = 9.2 Hz, 1H), 4.37–4,28 (m, 3H), 4.06 (d, J = 7.1 Hz, 1H), 3.92 (s, 3H), 2.11–2.01 (m, 9H), 1.97 (s, 3H); 13C-NMR (75 MHz, Acetone) δ 170.84, 170.25, 169.48, 167.30, 161.09, 154.96, 137.19, 131.79, 130.85, 130.54, 130.05, 124.11, 117.14, 115.16 (3), 83.93, 76.03, 74.54, 69.00 (2), 62.72, 55.89, 20.84 (2), 20.72, 20.30. HR-MS (ESI positive, m/z): found 730.0739 ([M + Na]+), calc. for C30H30NO11NaI (M + Na): 730.0761.
3-iodo-1-((2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)quinolin-2(1H)-one 2g: Following procedure D, a mixture of β-2d (100 mg, 1.0 equiv.) and K2CO3 (12 mg, 0.5 equiv.) in methanol (3 mL) was stirred under argon at room temperature for 30 min to 1 h. The crude mixture was then filtered through celite, washed with 10 mL of methanol, and filtered for only 1 min. The filtrate was concentrated under reduced pressure at 25 °C for 1–2 h. The product 2g was isolated as a pale brown solid (100 mg, yield 99%); m.p.: 155.2–171.8 °C; Rf = 0.4 (Ethyl/MeOH: 9/1); + 45 (c, 1.0 in MeOH); IR (neat): 3360, 1629, 1589, 1451, 1314, 1277, 1082, 766, 749 cm−1; 1H-NMR (300 MHz, MeOD-d4) δ 8.59 (s, 1H), 8.10 (d, J = 8.9 Hz, 1H), 7.58 (t, J = 9.7 Hz, 2H), 7.27 (t, J = 7.4 Hz, 1H), 6.57 (d, J = 9.8 Hz, 1H), 4.27 (t, J = 9.0 Hz, 1H), 3.94 (d, J = 11.8 Hz, 1H), 3.81 (dd, J = 12.5, 4.6 Hz, 1H), 3.62–3.53 (m, 3H); 3C-NMR (75 MHz, MeOD) δ 161.39, 151.53, 132.47, 131.55, 129.90, 129.46, 124.33, 124.02, 119.17, 86.94, 81.86, 79.54, 71.30, 70.75, 62.62; HR-MS (ESI positive, m/z): found 455.9928 ([M + Na]+), calc. for C15H16NO6NaI (M + Na): 455.9920.
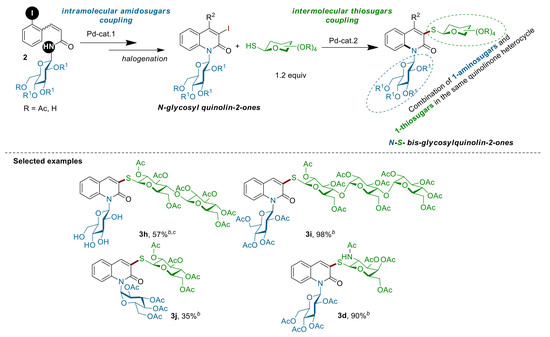
(2R,3S,4S,5R,6S)-2-(acetoxymethyl)-6-((2-oxo-1-((2R,3R,5R,6R)-3,4,5-triacetoxy-6-(acetoxymethyl)tetrahydro-2H-pyran-2-yl)-1,2-dihydroquinolin-3-yl)thio)tetrahydro-2H-pyran-3,4,5-triyl triacetate 3a: Following procedure C, 3-iodo N-β-glycosylquinolinone β-2d (0.0831 mmol, 50 mg) and β-thiogalactose 1a (0.2077 mmol, 76 mg) were stirred at room temperature for 3 h. The crude product was purified by silica gel flash chromatography (Cyclohexane/EtOAc 4/6). The product 3a was isolated as pale yellow solid (46 mg, yield 70%); m.p.: 222.7–223.8 °C; Rf = 0.4 (Cyclohexan/EtOAc: 4/6); + 16.21 (c, 1.0 in CHCl3); IR (neat): 1745, 1648, 1595, 1367, 1208, 1034, 794, 733,702 cm−1; 1H-NMR (300 MHz, Acetone) δ 8.28 (d, J = 8.7 Hz, 1H), 7.98 (s, 1H), 7.69–7.58 (m, 2H), 7.33 (t, J = 7.5 Hz, 1H), 6.94 (d, J = 9.7 Hz, 1H), 5.94 (t, J = 9.3 Hz, 1H), 5.60–5.26 (m, 6H), 4.50 (t, J = 6.3 Hz, 1H), 4.41–4.26 (m, 3H), 4.18 (d, J = 6.3 Hz, 2H), 2.19 (s, 3H), 2.10–1.92 (m, 21H); 13C-NMR (75 MHz, Acetone-d6) δ 169.87(C), 169.75 (C), 169.67 (C), 169.25 (C), 169.21 (C), 169.15 (C), 169.04 (C), 168.38 (C), 159.72 (C), 135.91 (C), 135.60 (CH), 129.62 (CH), 128.41 (CH), 127.91 (C), 123.16 (CH), 121.09 (C), 117.49 (CH), 81.87 (CH), 81.25 (CH), 74.84 (CH), 74.51 (CH), 73.38 (CH), 71.56 (CH), 67.88 (CH), 67.81 (CH), 67.74 (CH), 66.74 (CH), 61.96 (CH2), 61.55 (CH2), 19.74 (6CH3), 19.61 (CH3), 19.20 (CH3). HR-MS (ESI positive, m/z): found 860.2037 ([M + Na]+), calc. for C37H43NO19NaS (M + Na): 860.2048.
(2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-((2-oxo-1-((2R,3R,5R,6R)-3,4,5-triacetoxy-6-(acetoxymethyl)tetrahydro-2H-pyran-2-yl)-1,2-dihydroquinolin-3-yl)thio)tetrahydro-2H-pyran-3,4,5-triyl triacetate 3b: Following procedure C, 3-iodo N-β-glycosylquinolinone β-2d (0.0831 mmol, 50 mg) and β-thioglucose 1b (0.2077 mmol, 76 mg) were stirred at room temperature for 3 h. The crude product was purified by silica gel flash chromatography (diethylether/Pentan: 9/1), and the product 3b was isolated as a pale yellow solid (62 mg, yield 89%); m.p.: 221.8–223 °C; Rf = 0.33 (diethylether/Pentan: 9/1); + 23.07 (c, 1.0 in CHCl3); IR (neat): 1756, 1649, 1595, 1366, 1206, 1032, 914, 798,764 cm−1; 1H-NMR (300 MHz, Acetone) δ 8.27 (d, J = 8.8 Hz, 1H), 7.92 (s, 1H), 7.70–7.58 (m, 2H), 7.33 (t, J = 7.5 Hz, 1H), 6.94 (d, J = 9.8 Hz, 1H), 5.94 (t, J = 9.4 Hz, 1H), 5.58 (dd, J = 18.7, 9.2 Hz, 1H), 5.51–5.38 (m, 3H), 5.21–5.09 (m, 2H), 4.40–4.16 (m, 6H), 2.08–1.96 (m, 24H); 13C-NMR (75 MHz, Acetone) δ 169.75 (C), 169.37 (C), 169.20 (C), 169.14 (2C), 168.97 (C), 168.91 (C), 168.38 (C), 159.68 (C), 135.86 (C), 135.31 (CH), 129.63 (CH), 128.46 (CH), 127.84 (C), 123.16 (CH), 121.08 (C), 117.48 (CH), 81.30 (2CH), 75.47 (CH), 74.85 (CH), 73.42 (2CH), 69.69 (CH), 68.49 (CH), 67.88 (CH), 67.81 (CH), 62.34 (CH2), 61.55 (CH2), 19.73 (3CH3), 19.68 (2CH3), 19.61 (2CH3), 19.20 (CH3); HR-MS (ESI positive, m/z): found 860.2056 ([M + Na]+), calc. for C37H43NO19NaS (M + Na): 860.2048.
(2R,3R,4S,5R,6S)-2-((benzoyloxy)methyl)-6-((2-oxo-1-((2R,3R,5R,6R)-3,4,5-triacetoxy-6-(acetoxymethyl)tetrahydro-2H-pyran-2-yl)-1,2-dihydroquinolin-3-yl)thio)tetrahydro-2H-pyran-3,4,5-triyl tribenzoate 3c: Following procedure C, 3-iodo N-β-glycosylquinolinone β-2d (0.0831 mmol, 50 mg) and O-benzoylated 1-thio-β-d-glucopyranose 1d (0.2077 mmol, 128 mg) were stirred at room temperature for 3 h. The crude product was purified by silica gel flash chromatography (Cyclohexan/EtOAc: 5/5), and the product 3c was isolated as a pale yellow solid (52 mg, yield 58%); m.p.: 221–249 °C; Rf = 0.4 (Cyclohexan/EtOAc: 5/5); + 86.95 (c, 1.0 in CHCl3); IR (neat): 1728, 1648, 1597, 1452, 1367, 1277,1215, 1111,1086, 1068,801,766,708 cm−1; 1H-NMR (300 MHz, Acetone-d6) δ 8.24 (d, J = 8.4 Hz, 1H), 8.05–7.92 (m, 7H), 7.88–7.83 (m, 2H), 7.63–7.38 (m, 14H), 7.19 (t, J = 7.3 Hz, 1H), 6.88 (d, J = 9.7 Hz, 1H), 6.21 (t, J = 9.3 Hz, 1H), 5.93–5.78 (m, 4H), 5.57–5.38 (m, 2H), 4.89–4.72 (m, 2H), 4.59 (dd, J = 12.3, 6.2 Hz, 1H), 4.39–4.25 (m, 3H), 2.09–2.02 (m, 9H), 1.93 (s, 3H);13C-NMR (75 MHz, Acetone-d6) δ 169.73, 169.40, 169.14, 168.23, 168.84, 165.58, 165.17, 164.97, 164.77, 159.64, 135.85, 135.75, 135.04, 133.59, 133.54, 133.45, 133.18, 129.79, 129.5–128.53 (m), 123.06, 120.95, 117.36, 81.94, 81.30, 75.86, 74.81, 74.20, 73.27, 70.44, 69.59, 67.85, 67.77, 63.41, 61.55, 19.72, 19.58, 19.01; HR-MS (ESI positive, m/z): found 1086.2856 ([M + H]+), calc. for C57H52NO19S (M + H): 1086.2854.
(2R,3R,5R,6R)-2-(3-(((2S,3R,4R,5S,6R)-3-acetamido-4,5-diacetoxy-6-(acetoxymethyl)tetrahydro-2H-pyran-2-yl)thio)-2-oxoquinolin-1(2H)-yl)-6-(acetoxymethyl)tetrahydro-2H-pyran-3,4,5-triyl triacetate 3d: Following procedure C, 3-iodo N-β-glycosylquinolinone β-2d (0.083 mmol, 50 mg) and N-acetyl-O-acetylated 1-thio-β-d-glucopyranose 1c (0.2077mmol, 76 mg) were stirred at room temperature for 3 h. The crude product was purified by silica gel flash chromatography (dichloromethan/EtOAc: 2/8). The product 3d was isolated as a white solid (62 mg, yield 90%); m.p.: 258.9–261.5 °C; Rf = 0.28 (dichloromethan/EtOAc: 2/8); + 20 (c, 1.0 in CHCl3); IR (neat): 1740, 1649, 1596, 1367, 1260, 1213,1076, 796,748 cm−1; 1H-NMR (400 MHz, CDCl3) δ 7.97 (s, 1H), 7.89 (d, J = 8.7 Hz, 1H), 7.51 (t, J = 7.5 Hz, 2H), 7.27–7.21 (m, 1H), 6.86 (d, J = 9.9 Hz, 1H), 6.75 (s, 1H), 5.88 (t, J = 9.4 Hz, 1H), 5.47–5.30 (m, 3H), 5.22–5.17 (m, 1H), 5.06 (t, J = 9.7 Hz, 1H), 4.25–4.03 (m, 6H), 3.81–3.74 (m, 1H), 2.09–1.85 (m, 24H); 13C-NMR (101 MHz, CDCl3) δ 170.77 (2C), 170.62 (C), 170.47 (C), 169.85 (C), 169.56 (C), 169.43 (C), 168.66, (C) 161.38 (C), 143.41 (CH), 136.63(C), 130.77 (CH), 129.30 (CH), 124.77 (C), 123.84 (CH), 120.99 (C), 116.84 (CH), 90.76 (CH), 82.98 (CH), 81.62 (CH), 76.03 (CH), 75.13 (CH), 73.85 (CH), 73.65 (CH), 70.79 (CH), 68.81 (CH), 68.04 (CH), 67.80 (CH), 62.44 (CH2), 61.62 (CH2), 23.51 (CH3), 23.10 (CH3), 21.00 (CH3), 20.77 (CH3), 20.65 (CH3), 20.63 (CH3), 20.59 (CH3), 19.98 (CH3); HR-MS (ESI positive, m/z): found 859.2211 ([M + Na]+), calc. for C37H44N2O18S (M + Na): 859.2208.
(2R,4S,5R,6S)-2-(acetoxymethyl)-6-((2-oxo-1-((2R,3R,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)-1,2-dihydroquinolin-3-yl)thio)tetrahydro-2H-pyran-3,4,5-triyl triacetate 3e: Following procedure C, 3-iodo N-β-glycosylquinolinone β-2g (0.115 mmol, 50 mg) and β-thiogalactose 1a (0.2885 mmol, 106 mg) were stirred at room temperature for 3 h. After purification of the crude reaction by HPLC preparative (conditions: H2O + 0.1% AF/ACN gradient from 20% to 50% in 20 min), the product 3e was isolated as a pale yellow solid (52 mg, yield 68%); m.p.: 244–252.8 °C; Rf = 0.4 (Cyclohexane/EtOAc: 5/5); − 16 (c, 1.0 in CHCl3); IR (neat): 1750, 1633, 1561, 1367, 1211, 1046, 732 cm−1; 1H-NMR (300 MHz, Acetone-d6) δ 8.10 (d, J = 8.1 Hz, 1H), 7.92 (s, 1H), 7.65 (d, J = 7.6 Hz, 1H), 7.48 (t, J = 7.7 Hz, 1H), 7.28 (t, J = 7.5 Hz, 1H), 6.59 (d, J = 9.7 Hz, 1H), 5.55 (d, J = 2.9 Hz, 1H), 5.47–5.27 (m, 4H), 4.54 (t, J = 6.1 Hz, 1H), 4,33 (t, J=8 Hz,1H), 4.24–4.12 (m, 3H), 3.97–3.56 (m, 7H), 2.08–2.03 (m, 12H);13C-NMR (75 MHz, Acetone-d6) δ 169.95 (C), 169.79 (C), 169.36 (C), 159.99 (C), 135.96 (C), 133.46 (CH), 129.20 (C), 128.81 (CH), 128.02 (CH), 122.70 (CH), 121.35 (C), 118.83 (C), 117.83 (CH), 84.04 (CH), 81.38 (CH), 80.44 (CH), 78.57 (CH), 74.45 (CH), 71.65 (CH), 70.16 (CH), 69.28 (CH), 67.67(CH), 66.68(CH), 62.05 (CH2), 61.57 (CH2), 19.80 (2CH3), 19.74 (CH3), 19.63 (CH3); HR-MS (ESI positive, m/z): found 692.1636 ([M + Na]+), calc. for C29H35NO15NaS (M + Na): 692.1625.
(2R,3R,4S,6S)-2-(acetoxymethyl)-6-((4-(4-methoxyphenyl)-2-oxo-1-((2R,3R,5R,6R)-3,4,5-triacetoxy-6-(acetoxymethyl)tetrahydro-2H-pyran-2-yl)-1,2-dihydroquinolin-3-yl)thio)tetrahydro-2H-pyran-3,4,5-triyl triacetate 3f: Following procedure C, 3-iodo-N-β-glycosylquinolinone β-2f (0.071 mmol, 50 mg) and β-thioglucose 1a (0.1767 mmol, 65 mg) were stirred at room temperature for 3 h. The crude product was purified by silica gel flash chromatography (EtOAc/Cyclohexane: 5/5), and the product 3f was isolated as a pale brown solid (31 mg, yield 47%); m.p.: 143.4–150.3 °C; Rf = 0.36 (EtOAc/Cyclohexane: 5/5); − 1.42 (c, 1.0 in CHCl3); IR (neat): 1756, 1367, 1277, 1260, 1035, 766, 749 cm−1; 1H-NMR (400 MHz, Acetone d6) δ 8.31 (d, J = 8.7 Hz, 1H), 7.63 (t, J = 8.4 Hz, 1H), 7.27 (dd, J = 9.1, 1.9 Hz, 1H), 7.19–7.11 (m, 3H), 7.07–7.01 (m, 3H), 6.04 (t, J = 9.5 Hz, 1H), 5.59 (t, J = 9.4 Hz, 1H), 5.49 (t, J = 10.3 Hz, 1H), 5.38–5.32 (m, 2H), 5.16 (t, J = 9.6 Hz, 1H), 5.06 (t, J = 9.8 Hz, 1H), 4.98–4.92 (m, 2H), 4.85 (t, J = 9.7 Hz, 1H), 4.35–4.30 (m, 2H), 4.17 (dd, J = 12.5, 2.3 Hz, 1H), 3.89 (s, 3H), 2.07 (d, J = 4.5 Hz, 11H), 2.00 (s, 3H), 1.96 (d, J = 8.2 Hz, 10H); 13C-NMR (101 MHz, Acetone) δ 170.88, 170.66, 170.62, 170.24, 170.18, 170.06, 169.99, 169.94, 169.90, 169.69, 169.47, 131.62, 131.57, 131.24, 129.86, 129.63, 123.66, 122.91, 118.33, 114.67, 114.16, 87.75, 82.88, 82.18, 76.70, 75.95, 75.85, 74.49, 74.36, 74.19, 71.92, 70.34, 69.45, 68.98, 68.77, 68.59, 62.97, 62.53, 55.59, 20.79, 20.65, 20.55, 20.50, 20.40; HR-MS (ESI positive, m/z): found 966.2470 ([M + Na]+), calc. for C44H49NO20NaS (M + Na): 966.2466.
(2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-(((2R,3R,4S,5R,6S)-4,5-diacetoxy-2-(acetoxymethyl)-6-((2-oxo-1-((2R,3R,5R,6R)-3,4,5-triacetoxy-6-(acetoxymethyl)tetrahydro-2H-pyran-2-yl)-1,2-dihydroquinolin-3-yl)thio)tetrahydro-2H-pyran-3-yl)oxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate 3g: Following procedure C, 3-iodo N-β-glycosylquinolinone β-2d (0.083 mmol, 50 mg) and β-thiocellebiose 1e (0.2077 mmol, 136 mg) were stirred at room temperature for 3 h. After purification of the crude reaction by HPLC preparative (conditions: H2O + 0.1% AF/ACN gradient from 40% to 100% in 15 min), the product 3g was isolated as a pale yellow solid (92 mg, yield 98%): m.p.: 154.3–157.8 °C; Rf = 0.53 (dichloromethane/EtOAc: 6/4); − 15 (c, 1.0 in CHCl3); IR (neat): 1755, 1649, 1595, 1397, 1229, 1035,910 cm−1; 1H-NMR (400 MHz, CDCl3) δ 7.90 (d, J = 8.6 Hz, 1H), 7.63 (s, 1H), 7.52–7.43 (m, 2H), 7.26–7.22 (m, 1H), 6.81 (d, J = 9.9 Hz, 1H), 5.86 (t, J = 9.4 Hz, 1H), 5.41 (t, J = 9.3 Hz, 1H), 5.33 (t, J = 9.8 Hz, 1H), 5.24 (t, J = 8.7 Hz, 1H), 5.12–5.03 (m, 3H), 4.96 (d, J = 10.0 Hz, 1H), 4.90 (t, J = 8.6 Hz, 1H), 4.64–4.52 (m, 1H), 4.49 (d, J = 7.8 Hz, 1H), 4.42 (d, J = 11.3 Hz, 1H), 4.33 (dd, J = 12.5, 4.5 Hz, 1H), 4.26–4.19 (m, 1H), 4.09 (dd, J = 11.9, 5.9 Hz, 1H), 4.05–3.96 (m, 2H), 3.79–3.71 (m, 2H), 3.68–3.61 (m, 1H), 2.08–2.03 (m, 10H), 2.01–1.93 (m, 23H). 13C-NMR (101 MHz, CDCl3) δ 170.57 (C), 170.49 (C), 170.33 (C), 170.27 (C), 169.86 (C), 169.81 (C), 169.73 (C), 169.48 (C), 169.40 (C), 169.14 (C), 169.08 (C), 160.26 (C), 137.79 (CH), 136.19 (C), 130.16 (CH), 128.73 (CH), 126.93 (C), 123.62 (CH), 121.04 (C), 116.84 (CH), 100.93 (CH), 82.07 (CH), 81.52 (CH), 76.94 (CH), 76.69 (CH), 75.21 (CH), 73.79 (CH), 73.62 (CH), 73.01 (CH), 72.15 (CH),71.76 (CH), 70.32 (CH), 67.95 (2CH), 67.84 (CH), 62.52 (CH2), 61.75 (CH2), 61.61 (CH2), 20.83 (CH3), 20.78 (CH3), 20.75 (CH3), 20.73 (CH3), 20.69 (CH3), 20.62 (4CH3), 20.23 (CH3), 1.10 (CH3); HR-MS (ESI positive, m/z): found 1148.2888 ([M + Na]+), calc. for C49H59NO27NaS (M + Na): 1148.2893.
(2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-(((2R,3R,4S,5R,6S)-4,5-diacetoxy-2-(acetoxymethyl)-6-((2-oxo-1-((2R,3R,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)-1,2-dihydroquinolin-3-yl)thio)tetrahydro-2H-pyran-3-yl)oxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate 3h: Following procedure C, 3-iodo N-β-glycosylquinolinone β-2g (0.115 mmol, 50 mg) and β-thiocellobiose 1e (0.2885 mmol, 189 mg) were stirred at room temperature for 3 h. After purification of the crude reaction by HPLC preparative (conditions: H2O + 0.1% AF/ACN gradient from 20% to 50% in 15 min), the product 3h was isolated as a white solid (63 mg, yield 57%); m.p.: 249.2–251.9 °C; Rf = 0.52 (EtOAc/MeOH: 9/1); + 7.14 (c, 1.0 in CHCl3); IR (neat): 1740, 1629, 1590, 1366, 1212, 1033, 907, 751 cm−1; 1H-NMR (300 MHz, Acetone-d6) δ 8.09 (d, J = 8.0 Hz, 1H), 7.82 (s, 1H), 7.65 (d, J = 7.6 Hz, 1H), 7.48 (t, J = 7.90 Hz, 1H), 7.28 (t, J = 7.3 Hz, 1H), 6.58 (d, J = 9.7 Hz, 1H), 5.43–5.23 (m, 4H), 5.14–5,03 (m, 2H), 4.94–4.84 (m, 2H), 4.60–4.30 (m, 6H), 4.25–4.14 (m, 3H), 3.96–3.56 (m, 8H), 2.05–1.92 (m, 16H); 13C-NMR (75 MHz, Acetone-d6) δ 169.97 (C), 169.83 (C), 169.41 (C), 169.31 (C), 169.23 (C), 169.03 (C), 168.82 (C), 159.87 (C), 135.88 (C), 132.79 (CH), 130.77 (C), 128.77 (CH), 128.00 (CH), 122.63 (CH), 121.34 (C), 117.80 (CH), 100.53 (CH), 84.05 (CH), 80.55 (CH), 80.46 (CH), 78.56 (CH), 76.73 (CH), 76.45 (CH), 73.33 (CH), 72.78 (CH), 71.57 (CH), 71.53(CH), 70.25 (CH), 69.80 (CH), 69.35 (CH), 68.03 (CH), 62.69 (CH2), 61.67 (CH2), 61.57 (CH2), 19.93 (CH3), 19.84 (CH3), 19.76 (CH3), 19.71 (CH3), 19.69 (CH3), 19.61 (CH3), 19.52 (CH3); HR-MS (ESI positive, m/z): found 980.2461 ([M + Na]+), calc. for C41H51NO23NaS (M + Na): 980.2470.
(2R,3R,4S,5R,6R)-2-(acetoxymethyl)-6-(((2R,3S,4S,5R,6R)-4,5-diacetoxy-2-(acetoxymethyl)-6-(((2R,3S,4S,5R,6S)-4,5-diacetoxy-2-(acetoxymethyl)-6-((2-oxo-1-((2R,3R,5R,6R)-3,4,5-triacetoxy-6-(acetoxymethyl)tetrahydro-2H-pyran-2-yl)-1,2-dihydroquinolin-3-yl)thio)tetrahydro-2H-pyran-3-yl)oxy)tetrahydro-2H-pyran-3-yl)oxy)tetrahydro-2H-pyran-3,4,5-triyl triacetate 3i: Following procedure C, 3-iodo N-β-glycosylquinolinone β-2d (0.083 mmol, 50 mg) and β-thiomalthotriose 1f (0.2077mmol, 196 mg) were stirred at room temperature for 3 h. After purification of the crude reaction by HPLC preparative (conditions: H2O + 0.1% AF/MeOH gradient from 50% to 100% in 15 min), the product 3i was isolated as a white solid (116.25 mg, yield 98%); m.p.: 110.8–134.3 °C; Rf = 0.19 (Cyclohexane/EtOAc: 4/6); + 40 (c, 1.0 in CHCl3); IR (neat): 1756, 1649, 1367, 1260, 1208, 1011, 794, 708, 702 cm−1; 1H-NMR (300 MHz, CDCl3) δ 7.93 (d, J = 8.6 Hz, 1H), 7.73 (s, 1H), 7.54 (t, J = 9.1 Hz, 2H), 7.31 (d, J = 7.4 Hz, 1H), 6.85 (d, J = 9.9 Hz, 1H), 5.91 (t, J = 9.5 Hz, 1H), 5.45–5.27 (m, 8H), 5.12–4.94 (m, 4H), 4.86 (dd, J = 10.5, 4.0 Hz, 1H), 4.75 (dd, J = 10.3, 4.0 Hz, 1H), 4.49–4.18 (m, 8H), 3.99–3.89 (m, 4H), 2.12–1.96 (m, 42H); 13C-NMR (75 MHz, CDCl3) δ 170.68, (C) 170.59 (C), 170.52 (C), 170.39 (C), 170.18 (C), 170.12 (C), 170.00 (C), 169.86 (C), 169.75 (C), 169.69 (C), 169.61 (C), 169.51 (C), 169.39 (C), 168.97 (C), 144.13 (C), 136.14 (C), 130.09 (CH), 126.52 (C), 123.61 (CH), 120.99 (C), 116.67 (CH), 95.90 (CH), 95.75 (CH), 81.76 (CH), 81.43 (CH), 76.15 (CH), 75.98 (CH), 75.76 (CH), 75.13 (CH), 74.17 (CH), 73.72 (CH), 72.67 (CH), 71.71 (CH), 70.81 (CH), 70.36 (CH), 70.06 (CH), 69.38 (CH), 69.02 (CH), 68.59 (CH), 67.89 (CH), 68.19 (CH), 67.78 (CH), 67.56 (CH), 63.50 (CH2), 62.46 (CH2), 61.66 (CH2), 61.42 (CH2), 20.84 (2CH3), 20.81 (CH3), 20.77 (CH3), 20.56 (9CH3), 20.17 (CH3); HR-MS (ESI positive, m/z): found 1436.3730 ([M + Na]+), calc. for C61H75NO35NaS (M + Na): 1436.3738.
(2R,3R,4S,5R,6S)-2-(acetoxymethyl)-6-((2-oxo-1-(((2R,3S,4R,5S,6S)-3,4,5-triacetoxy-6-(acetoxymethyl)tetrahydro-2H-pyran-2-yl)-1,2-dihydroquinolin-3-yl)thio)tetrahydro-2H-pyran-3,4,5-triyl triacetate 3j: Following procedure C, 3-iodo N-α-glycosylquinolinone α-2e (0.083 mmol, 50 mg) and β-thioglucose 1a (0.2077 mmol, 76 mg) were stirred at room temperature for 3 h. After purification of the crude reaction by HPLC preparative (conditions: H2O + 0.1% AF/ACN gradient from 40 to 100% in 15 min), the product 3j was isolated as a pale yellow solid (26 mg, yield 35%); m.p.: 224–225 °C; Rf = 0.35 (EtOAc/heptan: 7/3); + 4.44 (c, 1.0 in CHCl3); IR (neat): 1755, 1641, 1367, 1259, 1031, 913, 799, 748 cm−1; 1H-NMR (400 MHz, Acetone-d6) δ 7.87–7.77 (m, 2H), 7.54 (d, J = 7.8 Hz, 1H), 7.44 (t, J = 8.07 Hz, 1H), 7.19 (t, J = 7.5 Hz, 1H), 6.71 (d, J = 5.9 Hz, 1H), 5.84 (t, J = 6.12 Hz,1H), 5.33–5.22 (m, 3H), 5.08 (t, J = 7.3 Hz, 1H), 5.01 (dd, J = 18.4, 9.4 Hz, 2H), 4.57–4.52 (m, 1H), 4.34 (dd, J = 12.5, 5.8 Hz, 1H), 4.18–4.05 (m, 4H), 1.97 (s, 2H), 1.93–1.88 (m, 13H), 1.86–1.82 (m, 9H); 13C-NMR (101 MHz, Acetone-d6) δ 170.71 (C), 170.70 (C), 170.29 (C), 170.19 (C), 170.03 (C), 170.01 (C), 169.96 (C), 169.92 (C), 161.31 (C), 138.86 (C), 136.24 (CH), 130.31 (CH), 129.88 (C), 128.96 (CH), 124.01 (CH), 122.29 (C), 117.77 (CH),82.25 (CH), 80.36 (CH), 76.37 (CH), 74.67 (CH), 74.37 (CH), 70.52 (CH), 70.41, 69.38, 68.58, 63.26 (CH2), 62.23 (CH2), 20.83 (CH3), 20.79 (CH3), 20.70 (CH3), 20.64 (CH3), 20.62 (CH3), 20.60 (CH3), 20.53 (CH3), 20.30 (CH3); HR-MS (ESI positive, m/z): found 860.2042 ([M + Na]+), calc. for C37H43NO19NaS (M + Na): 860.2048.
1-((2R,3R,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)-3-(((2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)thio)quinolin-2(1H)-one 4a: Following procedure D, a mixture of 3b (20 mg, 1.0 equiv.) and K2CO3 (2 mg, 0.5 equiv.) in methanol (2 mL) was stirred under argon at room temperature for 1 h. The crude mixture was then filtered through celite, washed with 10 mL of methanol, and filtered again. The filtrate was concentrated under reduced pressure at 25. The product 4a was isolated as a white solid (12 mg, yield 98%); m.p.: 187–197.8 °C; Rf = 0.52 (EtOAc/MeOH: 9/1); − 63.15 (c, 1.0 in CHCl3); IR (neat): 1277, 1281, 798, 766, 749 cm−1; 1H-NMR (400 MHz, DMSO-d6) δ 7.95 (d, J = 8.6 Hz, 1H), 7.87 (s, 1H), 7.58 (d, J = 7.6 Hz, 1H), 7.45 (t, J = 8.0 Hz, 1H), 7.24 (t, J = 7.5 Hz, 1H), 6.31 (d, J = 9.7 Hz, 1H), 4.75 (d, J = 9.6 Hz, 1H), 4.04 (t, J = 9 Hz; 2H), 3.74 (d, J = 10.6 Hz, 3H), 3.24–3.13 (m, 15H); 13C-NMR (101 MHz, DMSO-d6) δ 160.08 (C), 135.24 (C), 133.12 (CH), 130.37 (C), 128.51 (CH), 128.23 (CH), 122.82 (CH), 121.52 (C), 117.63 (CH), 84.15 (CH), 84.02 (CH), 81.38 (CH), 80.89 (CH), 78.52 (CH), 78.18 (CH), 72.78 (CH), 70.22 (CH), 69.82 (CH), 68.91 (CH), 61.20 (CH2), 61.08 (CH2); HR-MS (ESI positive, m/z): found 524.1205 ([M + Na]+), calc. for C21H27NO11NaS (M + Na): 524.1203.