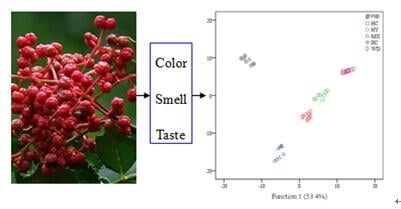
Rapid Determination of the Geographical Origin of Chinese Red Peppers (Zanthoxylum Bungeanum Maxim.) Based on Sensory Characteristics and Chemometric Techniques
Abstract
:1. Introduction
2. Results
2.1. Discriminant Analysis Based on Color
2.2. Discriminant Analysis Based on Smells
2.2.1. Response Curves of Electronic Nose
2.2.2. PCA Analysis
2.2.3. Evaluation of Geographic Origin
2.3. Discriminant Analysis Based on Tastes
2.3.1. Profiles of Tastes
2.3.2. Evaluation of Geographic Origin
3. Discussion
4. Experimental Section
4.1. Plant Materials
4.2. Pericarp Color Analysis
4.3. Powder Color Analysis
4.4. Smell Analysis
4.5. Taste Analysis
4.6. Multivariate Data Processing
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Gong, Y.W.; Huang, Y.F.; Zhou, L.G.; Shi, X.Y.; Guo, Z.J. Chemical composition and antifungal activity of the fruit oil of Zanthoxylum bungeanum Maxim. (Rutaceae) from China. J. Essent. Oil Res. 2009, 21, 174–178. [Google Scholar] [CrossRef]
- Zhang, J.; Jiang, L.; Zhang, J.H.; Jiang, L.F. Acid-catalyzed esterification of Zanthoxylum bungeanum seed oil with high free fatty acids for biodiesel production. Bioresour. Technol. 2008, 99, 8995–8998. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Hui, T.; Wang, F.; Li, S.; Cui, B.; Cui, Y.; Peng, Z. Chinese red pepper (Zanthoxylum bungeanum Maxim.) leaf extract as natural antioxidants in salted silver carp (Hypophthalmichthys molitrix) in dorsal and ventral muscles during processing. Food Control 2015, 56, 9–17. [Google Scholar] [CrossRef]
- Li, K.; Zhou, R.; Jia, W.W.; Li, Z.; Li, J.; Zhang, P.; Xiao, T. Zanthoxylum bungeanum essential oil induces apoptosis of HaCaT human keratinocytes. J. Ethnopharmacol. 2016, 186, 351–361. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.C.; Li, R.; Tan, J.; Jiang, Z.T. Polyphenolics composition of the leaves of Zanthoxylum bungeanum Maxim. grown in Hebei, China, and their radical scavenging activities. J. Agric. Food Chem. 2013, 61, 1772–1778. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.; Han, S.; Jiang, J.; Lin, Y.; Chen, H.; Yuan, Y. Antitumor compound identification from Zanthoxylum bungeanum essential oil based on composition–activity relationship. Chem. Res. Chin. Univ. 2013, 29, 1068–1071. [Google Scholar]
- Yang, X.G. Aroma constituents and alkylamides of red and green huajiao (Zanthoxylum bungeanum and Zanthoxylum schinifolium). J. Agric. Food Chem. 2008, 56, 1089–1096. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Bai, J.; Qiu, C.; Zhong, K.; Zhao, Z.; Gao, H.; Huang, Y. Geographical origin discrimination of Zanthoxylum bungeanum from southwest China using HPLC and PCA: identification and determination of marker compounds. Modern Food Sci. Technol. 2015, 31, 308–316. [Google Scholar]
- Berrueta, L.A.; Alonso-Salces, R.M.; Héberger, K. Supervised pattern recognition in food analysis. J. Chromatogr. A 2007, 1158, 196–214. [Google Scholar] [CrossRef] [PubMed]
- Drivelos, S.A.; Georgiou, C.A. Multi-element and multi-isotoperatio analysis to determine the geographical origin of foods in the European Union. Trend. Anal. Chem. 2012, 40, 38–51. [Google Scholar] [CrossRef]
- Camin, F.; Boner, M.; Bontempo, L.; Fauhl-Hassek, C.; Kelly, S.D.; Riedl, J.; Rossmann, A. Stable isotope techniques for verifying the declared geographical origin of food in legal cases. Trends Food Sci. Tech. 2017, 61, 176–187. [Google Scholar] [CrossRef]
- Nietner, T.; Pfister, M.; Glomb, M.A.; Fauhl-Hassek, C. Authentication of the botanical and geographical origin of distillers dried grains and solubles (DDGS) by FT-IR spectroscopy. J. Agric. Food Chem. 2013, 61, 7225–7233. [Google Scholar] [CrossRef] [PubMed]
- Luykx, D.M.A.M.; van Ruth, S.M. An overview of analytical methods for determining the geographical origin of food products. Food Chem. 2008, 107, 897–911. [Google Scholar] [CrossRef]
- Ma, G.C.; Zhang, Y.B.; Zhang, J.Y.; Wang, J.Q.; Chen, L.Y.; Zhang, M.L.; Liu, T.; Liu, X.; Lu, C.Y. Determining the geographical origin of Chinese green tea by linear discriminant analysis of trace metals and rare earth elements: Taking Dongting Biluochun as an example. Food Control 2016, 59, 714–720. [Google Scholar] [CrossRef]
- Carrerá, M.; Gallardo, J.M. Determination of the geographical origin of all commercial hake species by stable isotope ratio (SIR) analysis. J. Agric. Food Chem. 2017, 65, 1070–1077. [Google Scholar] [CrossRef] [PubMed]
- Chung, I.M.; Kim, J.K.; Lee, J.K.; Kim, S.H. Discrimination of geographical origin of rice (Oryza sativa L.) by multielement analysis using inductively coupled plasma atomic emission spectroscopy and multivariate analysis. J. Cereal Sci. 2015, 65, 252–259. [Google Scholar] [CrossRef]
- Priscila del Campo, C.; Garde-Cerdán, T.; Sánchez, A.; Maggi, M.; Carmona, M.; Alonso, G.L. Determination of free amino acids and ammonium ion in saffron (Crocus sativus L.) from different geographical origins. Food Chem. 2009, 114, 1542–1548. [Google Scholar] [CrossRef]
- Marseglia, A.; Palla, G.; Caligiani, A. Presence and variation of γ-aminobutyric acid and other free amino acids in cocoa beans from different geographical origins. Food Res. Int. 2014, 63, 360–366. [Google Scholar] [CrossRef]
- Guo, J.; Yue, T.; Yuan, Y.; Wang, Y. Chemometric classification of apple juices according to variety and geographical origin based on polyphenolic profiles. J. Agric. Food Chem. 2013, 61, 6949–6963. [Google Scholar] [CrossRef] [PubMed]
- Karabagias, I.K.; Vavoura, M.V.; Nikolaou, C.; Badeka, A.V.; Kontakos, S.; Kontominas, M.G. Floral authentication of greek unifloral honeys based on the combination of phenolic compounds, physicochemical parameters and chemometrics. Food Res. Int. 2014, 62, 753–760. [Google Scholar] [CrossRef]
- Lerma-García, M.J.; Lantano, C.; Chiavaro, E.; Cerretani, L.; Herrero-Martínez, J.M.; Simó-Alfonso, E.F. Classification of extra virgin olive oils according to their origin using phenolic compound profiles obtained by capillary electrochromatography. Food Res. Int. 2009, 42, 1446–1452. [Google Scholar] [CrossRef]
- Fechner, D.C.; Moresi, A.L.; Ruiz Díaz, J.D.; Pellerano, R.G.; Vazquez, F.A. Multivariate classification of honeys from Corrientes (Argentina) according to geographical origin based on physicochemical properties. Food Biosci. 2016, 15, 49–54. [Google Scholar] [CrossRef]
- Hoffmann, J.F.; Carvalho, I.R.; Barbieri, R.L.; Rombaldi, C.V.; Chaves, F.C. Butia spp. (Arecaceae) LC-MS-based metabolomics for species and geographical origin discrimination. J. Agric. Food Chem. 2017, 65, 523–532. [Google Scholar] [CrossRef] [PubMed]
- Hong, X.; Wang, J.; Qiu, S. Authenticating cherry tomato juices-Discussion of different data standardization and fusion approaches based on electronic nose and tongue. Food Res. Int. 2014, 60, 173–179. [Google Scholar] [CrossRef]
- Zapotoczny, P. Discrimination of wheat grain varieties using image analysis and neural networks. Part I. Single kernel texture. J. Cereal Sci. 2011, 54, 60–68. [Google Scholar] [CrossRef]
- Haddi, Z.; Alami, H.; El Bari, N.; Tounsi, M.; Barhoumi, H.; Maaref, A.; Jaffrezic-Renault, N.; Bouchikhi, B. Electronic nose and tongue combination for improved classification of Moroccan virgin olive oil profiles. Food Res. Int. 2013, 54, 1488–1498. [Google Scholar] [CrossRef]
- Phat, C.; Moon, B.; Lee, C. Evaluation of umami taste in mushroom extracts by chemical analysis, sensory evaluation, and an electronic tongue system. Food Chem. 2016, 192, 1068–1077. [Google Scholar] [CrossRef] [PubMed]
- Di Rosa, A.R.; Leone, F.; Cheli, F.; Chiofalo, V. Fusion of electronic nose, electronic tongue and computer vision for animal source food authentication and quality assessment—A review. J. Food Eng. 2017, 210, 62–75. [Google Scholar] [CrossRef]
- Trinderup, C.H.; Dahl, A.; Jensen, K.; Carstensen, J.M.; Conradsen, K. Comparison of a multispectral vision system and a colorimeter for the assessment of meat color. Meat Sci. 2015, 102, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Terouzi, W.; De Luca, M.; Bolli, A.; Oussama, A.; Patumi, M.; Ioele, G.; Ragno, G. A discriminant method for the classification of Moroccan olive varieties by using the FT-IR analysis of the mesocarp section. Vibr. Spectr. 2011, 56, 123–128. [Google Scholar] [CrossRef]
- Yang, X.; Rui, G.; Zhong, Z.; Gan, P.; Sheng, J. Study on chemical components of supercritical CO2 extract from nine species Zanthoxylum bungeanum Maxim by GC/MS/AMDIS. Food Ferment. Ind. 2011, 279, 158–162. [Google Scholar]
- Luo, K.; Zhu, L.; Kan, J.; Huang, X. Comparative study of the essential oil content of Zanthoxylum Schinifolium Sieb.et Zucc and Zanthosylum Bungeanum Maxim from different producing areas. Sci. Tech. Food Ind. 2012, 33, 103–106. [Google Scholar]
- Qi, H.; Wu, Y.; Wang, Y.; Wang, H. Ultraviolet spectrum fingerprint study on Zanthosylum Bungeanum. China Condiment. 2015, 40, 62–65. [Google Scholar]
- Yam, K.L.; Papadakis, S.E. A simple digital imaging method for measuring and analyzing color of food surfaces. J. Food Eng. 2004, 61, 137–142. [Google Scholar] [CrossRef]
Sample Availability: Not available. |




| Samples | Pericarp | Powder | ||||
|---|---|---|---|---|---|---|
| R | G | B | L * | a * | b * | |
| HC | 93.9 ± 2.7 ab | 63.7 ± 3.1 b | 58.8 ± 1.9 a | 30.4 ± 4.4 b | 16.5 ± 0.6 a | 22.4 ± 1.3 b |
| HY | 81.9 ± 4.6 b | 59.9 ± 2.3 b | 58.4 ± 1.8 a | 37.2 ± 4.3 ab | 15.3 ± 0.6 ab | 26.1 ± 1.4 b |
| MX | 85.9 ± 3.9 b | 63.7 ± 4.7 b | 63.9 ± 4.0 a | 37.9 ± 4.5 ab | 16.9 ± 0.5 a | 22.8 ± 1.3 b |
| RC | 104.6 ± 4.3 a | 77.7 ± 4.7 a | 62.6 ± 2.9 a | 46.2 ± 5.6 a | 13.7 ± 0.5 b | 34.7 ± 3.3 a |
| WD | 88.9 ± 6.5 b | 63.7 ± 3.3 b | 62.3 ± 2.2 a | 44.1 ± 6.5 ab | 14.5 ± 0.9 b | 27.0 ± 3.4 b |
| Groups | Number of Samples | LDA | ANN | SVM | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| RBF | Linear | |||||||||
| Training Set | Test Set | AC-tr (%) | AC-te (%) | AC-tr (%) | AC-te (%) | AC-tr (%) | AC-te (%) | AC-tr (%) | AC-te (%) | |
| Pericarp color | ||||||||||
| HC | 7 | 4 | 85.7 | 100 | 100 | 100 | 71.4 | 100 | 85.7 | 100 |
| HY | 6 | 2 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| MX | 6 | 2 | 66.7 | 50 | 83.3 | 100 | 66.7 | 100 | 66.7 | 100 |
| RC | 8 | 4 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| WD | 6 | 3 | 66.7 | 66.7 | 83.3 | 33.3 | 50 | 66.7 | 33.3 | 66.7 |
| total | 84.8 | 86.7 | 93.9 | 86.7 | 78.8 | 93.3 | 78.8 | 93.3 | ||
| Powder color | ||||||||||
| HC | 7 | 4 | 42.9 | 100 | 100 | 100 | 57.1 | 100 | 57.1 | 100 |
| HY | 6 | 2 | 57.1 | 100 | 100 | 50 | 100 | 100 | 83.3 | 100 |
| MX | 6 | 2 | 100 | 100 | 100 | 100 | 66.7 | 100 | 50 | 100 |
| RC | 8 | 4 | 100 | 75 | 100 | 100 | 100 | 100 | 100 | 100 |
| WD | 6 | 3 | 83.3 | 66.7 | 100 | 100 | 83.3 | 66.7 | 83.3 | 66.7 |
| total | 69.7 | 86.7 | 100 | 93.3 | 78.8 | 93.3 | 69.7 | 93.3 | ||
| Samples | Number | LDA (%) | ANN (%) | SVM | ||
|---|---|---|---|---|---|---|
| RBF (%) | Linear (%) | |||||
| Training set | HC | 7 | 100 | 100 | 100 | 100 |
| HY | 6 | 83.3 | 83.3 | 66.7 | 50 | |
| MX | 6 | 100 | 100 | 100 | 100 | |
| RC | 8 | 100 | 100 | 100 | 100 | |
| WD | 6 | 100 | 100 | 100 | 100 | |
| total | 97 | 97 | 87.9 | 90.9 | ||
| Test set | HC | 4 | 100 | 100 | 100 | 100 |
| HY | 2 | 100 | 100 | 50 | 100 | |
| MX | 2 | 100 | 100 | 100 | 100 | |
| RC | 4 | 75 | 75 | 100 | 100 | |
| WD | 3 | 100 | 100 | 100 | 100 | |
| total | 93.3 | 93.3 | 93.3 | 100 | ||
| Groups | Number of Samples | LDA (%) | ANN (%) | SVM RBF (%) | SVM Linear (%) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Training Set | Test Set | Training Set | Test Set | Training Set | Test Set | Training Set | Test Set | Training Set | Test Set | |
| HC | 7 | 4 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| HY | 6 | 2 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| MX | 6 | 2 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| RC | 8 | 4 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| WD | 6 | 3 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| Sample | Number of Samples | Longitude (E) | Latitude (N) | Climate Type | Agrotype |
|---|---|---|---|---|---|
| HC | 11 | E110°7′–110°37′ | N35°18′–35°52′ | Warm temperate continental monsoon | brown |
| HY | 8 | E102°16′–103°00′ | N29°05′–29°43′ | Subtropical humid monsoon | yellow brown |
| MX | 8 | E102°56′–104°10′ | N31°25′–32°16′ | Subtropical monsoon | dark brown |
| RC | 12 | E110°36′–110°42′ | N34°36′–34°48′ | Warm sub-humid continental | cinnamon |
| WD | 9 | E104°34′–105°38′ | N32°47′–33°42′ | north subtropical semi-humid | yellow brown |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yin, X.; Xu, X.; Zhang, Q.; Xu, J. Rapid Determination of the Geographical Origin of Chinese Red Peppers (Zanthoxylum Bungeanum Maxim.) Based on Sensory Characteristics and Chemometric Techniques. Molecules 2018, 23, 1001. https://doi.org/10.3390/molecules23051001
Yin X, Xu X, Zhang Q, Xu J. Rapid Determination of the Geographical Origin of Chinese Red Peppers (Zanthoxylum Bungeanum Maxim.) Based on Sensory Characteristics and Chemometric Techniques. Molecules. 2018; 23(5):1001. https://doi.org/10.3390/molecules23051001
Chicago/Turabian StyleYin, Xiangqian, Xiaoxue Xu, Qiang Zhang, and Jianguo Xu. 2018. "Rapid Determination of the Geographical Origin of Chinese Red Peppers (Zanthoxylum Bungeanum Maxim.) Based on Sensory Characteristics and Chemometric Techniques" Molecules 23, no. 5: 1001. https://doi.org/10.3390/molecules23051001