The Microencapsulation of Maqui (Aristotelia chilensis (Mol.) Stuntz) Juice by Spray-Drying and Freeze-Drying Produces Powders with Similar Anthocyanin Stability and Bioaccessibility
Abstract
:1. Introduction
2. Results and Discussion
2.1. The Encapsulation of Anthocyanins from Maqui Juice (MJ) by SD and FD
2.2. Characterization of the MJ Powders Obtained by SD and FD
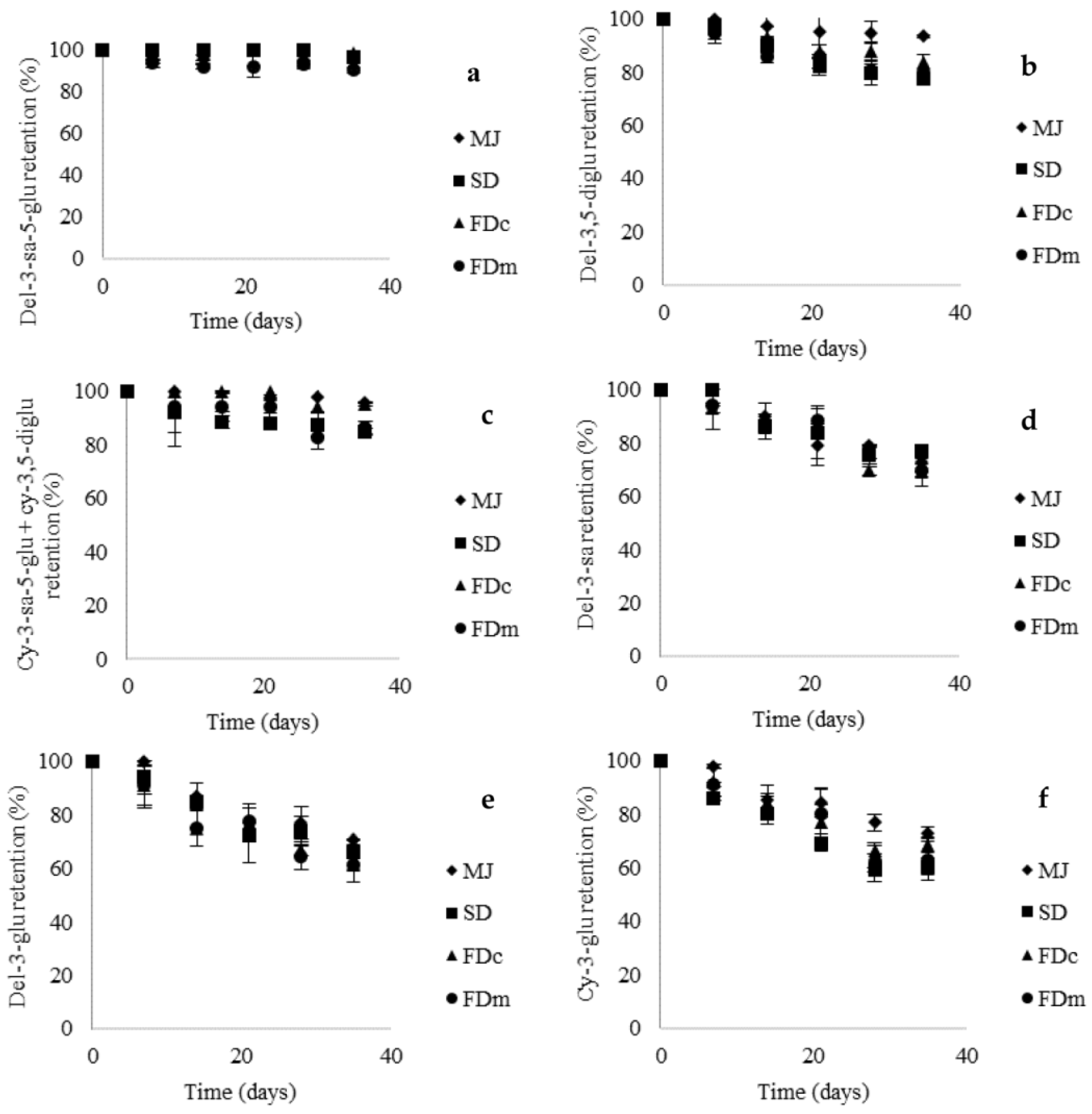
2.3. Stability of MJ Powders in Yogurt
2.4. Bioaccessibility of MJ Powders after an In Vitro Digestion
3. Materials and Methods
3.1. Raw Material and Encapsulating Agents
3.2. Preparation of MJ Powders by SD
3.3. Preparation of MJ Powders by FD
3.4. Characterization of the MJ Powders Obtained by SD and FD
3.4.1. Total Anthocyanin Determination in MJ Powders
3.4.2. Surface Anthocyanin Determination in MJ Powders
3.4.3. Determination of Anthocyanins by HPLC
3.4.4. Physical Analysis of MJ Powders and Reconstitution
3.4.5. Powder Morphology
3.4.6. Particle Size
3.5. Stability of Anthocyanins from MJ Powders in Yogurt
3.6. Bioaccessibility of MJ Powders Using an In Vitro Digestion Model
3.6.1. Mouth Digestion
3.6.2. Stomach Digestion
3.6.3. Gut Digestion
3.7. Statistical Analysis
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| aw | water activity |
| BA | bioaccessibility |
| cy-3-glu | cyanidin-3-glucoside |
| cy-3-sa | cyanidin-3-sambubioside |
| cy-3-sa-5-glu | cyanidin-3-sambubioside-5-glucoside |
| cy-3,5-diglu | cyanidin-3,5-diglucoside |
| del-3-sa-5-glu | delphinidin-3-sambubioside-5-glucoside |
| del-3,5-diglu | delphinidin-3,5-diglucoside |
| del-3-glu | delphinidin-3-glucoside |
| del-3-sa | delphinidin-3-sambubioside |
| FD | freeze-drying |
| GI | gastrointestinal |
| EA | encapsulating agents |
| EE | encapsulation efficiency |
| HPLC-DAD | high-performance liquid chromatography–photo diode array detector |
| MD | maltodextrin |
| MJ | maqui juice |
| RSM | response surface methodology |
| SD | spray-drying |
| SPI | soy protein isolate |
| TA | total anthocyanins |
| SEM | scanning electron microscopy |
References
- Fredes, C.; Robert, P. The powerful colour of the maqui (Aristotelia chilensis [Mol.] Stuntz) fruit. J. Berry Res. 2014, 4, 175–182. [Google Scholar] [CrossRef]
- ODEPA—Oficina de Estudios y Políticas Agrarias. Boletín de frutas y hortalizas procesadas. Available online: http://www.odepa.gob.cl/wp-content/files_mf/1490990342BFrutaprocesada 0317.pdf (accessed on 9 May 2017).
- Fredes, C.; Montenegro, G.; Zoffoli, J.P.; Santander, F.; Robert, P. Comparison of total phenolic, total anthocyanin and antioxidant activity of polyphenol-rich fruits grown in Chile. Cienc. Investig. Agrar. 2014, 41, 49–60. [Google Scholar] [CrossRef]
- Fredes, C.; Osorio, M.J.; Parada, J.; Robert, P. Stability and bioaccessibility of anthocyanins from maqui (Aristotelia chilensis [Mol.] Stuntz) juice microparticles. LWT-Food Sci. Technol. 2018, 91, 549–556. [Google Scholar] [CrossRef]
- Mahdavi, S.A.; Jafari, S.M.; Ghorbani, M.; Assadpoor, E. Spray-drying microencapsulation of anthocyanins by natural biopolymers: A review. Drying Technol. 2014, 32, 509–518. [Google Scholar] [CrossRef]
- Robert, P.; García, P.; Fredes, C. Drying and preservation of polyphenols. In Advances in Technologies for Producing Food-Relevant Polyphenols; Cuevas-Valenzuela, J., Vergara Salinas, J.R., Pérez-Correa, J.R., Eds.; CRC Press, Taylor and Francis Group: Boca Raton, FL, USA, 2017; pp. 281–302. [Google Scholar]
- Desai, K.; Park, H. Recent developments in microencapsulation of food ingredients. Drying Technol. 2005, 23, 1361–1394. [Google Scholar] [CrossRef]
- Laokuldilok, T.; Kanha, N. Effects of processing conditions on powder properties of black glutinous rice (Oryza sativa L.) bran anthocyanins produced by spray drying and freeze drying. LWT Food Sci. Technol. 2015, 64, 405–411. [Google Scholar] [CrossRef]
- Darniadi, S.; Ho, P.; Murray, B.S. Comparison of blueberry powder produced via foam-mat freeze drying versus spray-drying: Evaluation of foam and powder properties. J. Sci. Food Agric. 2017. [Google Scholar] [CrossRef] [PubMed]
- Robert, P.; Gorena, T.; Romero, N.; Sepulveda, E.; Chavez, J.; Saenz, C. Encapsulation of polyphenols and anthocyanins from pomegranate (Punica granatum) by spray drying. Int. J. Food Sci. Technol. 2010, 45, 1386–1394. [Google Scholar] [CrossRef]
- Fredes, C.; Yousef, G.G.; Robert, P.; Grace, M.H.; Lila, M.A.; Gómez, M.; Gebauer, M.; Montenegro, G. Anthocyanin profiling of wild maqui berries (Aristotelia chilensis [Mol.] Stuntz) from different geographical regions in Chile. J. Sci. Food Agric. 2014, 94, 2639–2648. [Google Scholar] [CrossRef] [PubMed]
- Rojo, L.E.; Ribnicky, D.; Logendra, S.; Poulev, A.; Rojas-Silva, P.; Kuhn, P.; Dorna, R.; Grace, M.H.; Lila, M.A.; Raskin, I. In vitro and in vivo anti-diabetic effects of anthocyanins from maqui berry (Aristotelia chilensis). Food Chem. 2012, 131, 387–396. [Google Scholar] [CrossRef] [PubMed]
- Céspedes, C.L.; Valdez-Morales, M.; Avila, J.G.; El-Hafidi, M.; Alarcón, J.; Paredes-López, O. Phytochemical profile and the antioxidant activity of Chilean wild black-berry fruits, Aristotelia chilensis (Mol) Stuntz (Elaeocarpaceae). Food Chem. 2010, 119, 886–895. [Google Scholar] [CrossRef]
- Escribano-Bailón, M.T.; Alcalde-Eon, C.; Muñoz, O.; Rivas-Gonzalo, J.C.; Santos-Buelga, C. Anthocyanins in berries of Maqui (Aristotelia chilensis (Mol.) Stuntz). Phytochem. Anal. 2006, 17, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Gironés-Vilaplana, A.; Baenas, N.; Villaño, D.; Speisky, H.; García-Viguera, C.; Moreno, D.A. Evaluation of Latin-American fruits rich in phytochemicals with biological effects. J. Funct. Foods. 2014, 7, 599–608. [Google Scholar] [CrossRef]
- Villacrez, J.L.; Carriazo, J.G.; Osorio, C. Microencapsulation of Andes Berry (Rubus glaucus Benth.) aqueous extract by spray drying. Food Bioprocess Technol. 2014, 7, 1445–1456. [Google Scholar] [CrossRef]
- Fang, Z.; Bhandari, B. Effect of spray drying and storage on the stability of bayberry polyphenols. Food Chem. 2011, 129, 1139–1147. [Google Scholar] [CrossRef] [PubMed]
- Santana, A.; Cano-Higuita, D.; De Oliveira, R.; Telis, V. Influence of different combinations of wall materials on the microencapsulation of jussara pulp (Euterpe edulis) by spray drying. Food Chem. 2016, 212, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Fang, Z.; Bhandari, B. Encapsulation of polyphenols—A review. Trends Food Sci. Technol. 2010, 21, 510–523. [Google Scholar] [CrossRef]
- Dutra Alvim, I.; Abreu Stein, A.; Paes Koury, I.; Balardin Hellmeister Dantas, F.; de Camargo Vianna Cruz, C.L. Comparison between the spray drying and spray chilling microparticles contain ascorbic acid in a baked product application. LWT Food Sci. Technol. 2016, 65, 689–694. [Google Scholar] [CrossRef]
- Robert, P.; Torres, V.; García, P.; Vergara, C.; Sáenz, C. The encapsulation of purple cactus pear (Opuntia ficus-indica) pulp by using polysaccharide-proteins as encapsulating agents. LWT Food Sci. Technol. 2015, 60, 1039–1045. [Google Scholar] [CrossRef]
- Coisson, J.; Travaglia, F.; Piana, G.; Capasso, M.; Arlorio, M. Euterpe oleracea juice as a functional pigment for yogurt. Food Res. Int. 2005, 38, 893–897. [Google Scholar] [CrossRef]
- Sung, J.-M.; Kim, Y.-B.; Kum, J.-S.; Choi, Y.-S.; Seo, D.-H.; Choi, H.-W.; Park, J.-D. Effects of freeze-dried mulberry on antioxidant activities and fermented characteristics of yogurt during refrigerated storage. Korean J. Food Sci. Anim. Resour. 2015, 35, 807–814. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, H.L.; de Oliveira, A.V.; de Brito, E.S.; Ribeiro, P.R.B.; Souza Filho, M.M.; Azeredo, H.M.C. Stabilizing effect of montmorillonite on acerola juice anthocyanins. Food Chem. 2018, 245, 966–973. [Google Scholar] [CrossRef] [PubMed]
- Lila, M.; Ribnicky, D.; Rojo, L.; Rojas-Silva, P.; Oren, A.; Havenaar, R.; Janle, E.; Raskin, I.; Yousef, G.; Grace, M. Complementary approaches to gauge the bioavailability and distribution of ingested berry polyphenolics. J. Agric. Food Chem. 2012, 60, 5763–5771. [Google Scholar] [CrossRef] [PubMed]
- Lucas-Gonzalez, R.; Navarro-Coves, S.; Pérez-Álvarez, J.A.; Fernández-López, J.; Muñoz, L.A.; Viuda-Martos, M. Assessment of polyphenolic profile stability and changes in the antioxidant potential of maqui berry (Aristotelia chilensis (Molina) Stuntz) during in vitro gastrointestinal digestion. Ind. Crops Prod. 2016, 94, 774–782. [Google Scholar] [CrossRef]
- Flores, F.; Singh, R.; Kerr, W.; Pegg, R.; Kong, F. Total phenolics content and antioxidant capacities of microencapsulated blueberry anthocyanins during in vitro digestion. Food Chem. 2014, 153, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Mosele, J.I.; Macià, A.; Romero, M.P.; Motilva, M.J. Stability and metabolism of Arbutus unedo bioactive compounds (phenolics and antioxidants) under in vitro digestion and colonic fermentation. Food Chem. 2016, 201, 120–130. [Google Scholar] [CrossRef] [PubMed]
- Oidtmann, J.; Schantz, M.; Mäder, K.; Baum, M.; Berg, S.; Betz, M.; Kulozik, U.; Leick, S.; Rehage, H.; Schwarz, K.; et al. Preparation and comparative release characteristics of three anthocyanin encapsulation systems. J. Agric. Food Chem. 2012, 60, 844–851. [Google Scholar] [CrossRef] [PubMed]
- McDougall, G.; Fyffe, S.; Dobson, P.; Stewart, D. Anthocyanins from red wine. Their stability under simulated gastrointestinal digestion. Phytochemistry 2005, 66, 2540–2548. [Google Scholar] [CrossRef] [PubMed]
- AOAC—Association of Official Analytical Chemists. Fruits & Fruit Products. In Official Methods of Analysis; Cunniff, P., Ed.; AOAC International: Gaithersburg, MD, USA, 1996. [Google Scholar]
- Cai, Y.Z.; Corke, H. Production and properties of spray-dried Amaranthus betacyanin pigments. J. Food Sci. 2000, 65, 1248–1252. [Google Scholar] [CrossRef]
- Jinapong, N.; Suphantharika, M.; Jamnong, P. Production of instant soymilk powders by ultrafiltration, spray drying and fluidized bed agglomeration. J. Food Eng. 2008, 84, 194–205. [Google Scholar] [CrossRef]
- Aravena, G.; García, O.; Muñoz, O.; Pérez-Correa, J.R.; Parada, J. The impact of cooking and delivery modes of thymol and carvacrol on retention and bioaccessibility in starchy foods. Food Chem. 2016, 196, 848–852. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |

| Run | Inlet Air Temperature (°C) | MJ/EA Ratio | Yield (%) | Recovery (%) | EE (%) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| TA | Del-3-sa-5-glu | Del-3,5-diglu | Cy-3-sa-5-glu+Cy-3,5-diglu | Del-3-sa | Del-3-glu | Cy-3-glu | |||||
| 1 | 120 | 1:2 | 76.3 | 88.1 | 77.7 | 84.0 | 79.4 | 81.1 | 79.9 | 77.1 | 73.4 |
| 2 | 180 | 1:2 | 74.6 | 93.3 | 78.8 | 83.1 | 79.0 | 80.6 | 79.0 | 77.1 | 74.2 |
| 3 | 120 | 1:6 | 69.1 | 90.3 | 91.9 | 93.5 | 92.7 | 91.7 | 90.3 | 89.3 | 88.8 |
| 4 | 180 | 1:6 | 72.1 | 93.3 | 89.4 | 90.9 | 88.8 | 88.1 | 87.2 | 84.6 | 84.6 |
| 5 | 114 | 1:4 | 73.7 | 91.4 | 90.7 | 92.6 | 91.3 | 90.6 | 87.8 | 87.7 | 87.4 |
| 6 | 186 | 1:4 | 73.9 | 96.1 | 90.0 | 90.4 | 88.9 | 89.1 | 87.8 | 86.4 | 86.1 |
| 7 | 150 | 1:1.6 | 74.8 | 100.0 | 74.7 | 76.6 | 71.1 | 71.5 | 71.5 | 66.5 | 65.3 |
| 8 | 150 | 1:6.4 | 70.5 | 82.6 | 91.6 | 92.2 | 90.9 | 90.8 | 88.9 | 87.7 | 87.4 |
| 9 | 150 | 1:4 | 71.0 | 91.8 | 89.7 | 91.0 | 88.8 | 88.8 | 86.9 | 83.8 | 84.7 |
| 10 | 150 | 1:4 | 69.4 | 90.1 | 87.3 | 89.5 | 87.6 | 86.6 | 85.1 | 82.2 | 83.2 |
| 11 | 150 | 1:4 | 71.5 | 94.0 | 89.3 | 90.6 | 89.3 | 88.6 | 86.6 | 84.8 | 86.1 |
| 12 | 150 | 1:4 | 69.5 | 90.1 | 90.1 | 90.9 | 89.7 | 89.6 | 86.0 | 85.7 | 85.0 |
| R2 | R2 (Adjusted for d.f.) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Yield (%) | 136.3140 | −0.6917 | −6.1670 | 0.0021 | 0.0196 | 0.2692 | 0.902 | 0.820 | |
| Recovery (%) | 100.6240 | −0.1919 | 1.5889 | 0.0010 | −0.0092 | −0.1968 | 0.429 | 0.00 | |
| EE% | TA | 57.4578 | −0.0461 | 14.7790 | 0.0003 | −0.0150 | −1.1579 | 0.979 | 0.961 |
| del-3-sa-5-glu | 90.3751 | −0.3418 | 11.3291 | 0.0011 | −0.0071 | −0.9569 | 0.963 | 0.933 | |
| del-3,5-diglu | 78.7725 | −0.3441 | 15.8078 | 0.0012 | −0.0146 | −1.2780 | 0.972 | 0.949 | |
| cy-3-sa-5-glu+cy-3,5-diglu | 88.4993 | −0.4300 | 14.0942 | 0.0015 | −0.0129 | −1.1456 | 0.930 | 0.872 | |
| del-3-glu | 94.0431 | −0.4748 | 11.6651 | 0.0016 | −0.0092 | −0.9285 | 0.951 | 0.909 | |
| cy-3-sa | 102.4870 | −0.7099 | 15.2545 | 0.0025 | −0.0196 | −1.1305 | 0.927 | 0.866 | |
| cy-3-glu | 70.4168 | −0.3790 | 18.4930 | 0.0015 | −0.0208 | −1.4470 | 0.971 | 0.947 |
| MJ | SD | FD | ||||
|---|---|---|---|---|---|---|
| mg/g | BA % | mg/g | BA % | mg/g | BA % | |
| Del-3-sa-5-glu | 7.4 ± 0.02 | 35.1 ± 3.35 b,A | 1.3 ± 0.04 | 43.2 ± 0.61 c,AB | 1.4 ± 0.07 | 40.8 ± 2.00 cd,B |
| Del-3,5-diglu | 3.3 ± 0.01 | 36.4 ± 1.12 b,A | 0.6 ± 0.02 | 34.6 ± 1.16 b,A | 0.6 ± 0.03 | 36.2 ± 1.9 c,A |
| Cy-3-sa-5-glu + cy-3,5 diglu | 3.4 ± 0.01 | 48.9 ± 1.58 c,A | 0.7 ± 0.01 | 73.5 ± 3.04 d,B | 0.6 ± 0.03 | 76.5 ± 2.39 e,B |
| Del-3-sa | 1.5 ± 0.02 | 21.9 ± 2.83 ª,A | 0.3 ± 0.01 | 24.8 ± 0.85 a,AB | 0.3 ± 0.01 | 28.5 ± 2.56 b,B |
| Del-3-glu | 2.4 ± 0.01 | 21.2 ± 0.50 ª,A | 0.4 ± 0.01 | 20.8 ± 0.41 a,A | 0.4 ± 0.02 | 22.6 ± 0.47 a,B |
| Cy-3-glu | 1.2 ± 0.02 | 24.0 ± 2.29 ª,A | 0.2 ± 0.01 | 43.8 ± 2.93 c,B | 0.2 ± 0.01 | 44.8 ± 1.00 d,B |
| 3-O-monoglycosylated anthocyanins | 5.0 ± 0.01 | 23.3 ± 3.29 a,A | 0.9 ± 0.02 | 27.2 ± 0.57 a,AB | 0.9 ± 0.04 | 30.8 ± 2.09 a,B |
| 3,5-O-diglycosylated anthocyanins | 14.1 ± 0.04 | 39.5 ± 3.18 b,A | 2.6 ± 0.06 | 50.1 ± 2.07 b,B | 2.6 ± 0.12 | 48.4 ± 0.90 b,B |
| TA HPLC | 17.5 ± 0.04 | 35.2 ± 3.21 A | 3.3 ± 0.20 | 44.1 ± 1.68 B | 3.3 ± 0.41 | 43.8 ± 0.13 B |
| MJ Powder | SD | FDc | Fdm |
|---|---|---|---|
| EE TA | 92.5 ± 0.02 a | 93.0 ± 0.01 a | 93.9 ± 0.01 a |
| EE 3-O-monoglycosylated anthocyanins | 89.5 ± 2.2 a,A | 91.4 ± 1.3 ab,A | 92.3 ± 1.4 b,A |
| EE 3,5-O-diglycosylated anthocyanins | 93.1 ± 1.4 a,B | 94.6 ± 0.6 ab,B | 95.4 ± 0.9 b,B |
| Recovery (%) | 99.8 ± 0.01 b | 91.9 ± 0.01 a | 91.8 ± 0.02 a |
| Yield (%) | 64.1 ± 0.01 a | 94.6 ± 0.01 b | 94.6 ± 0.01 b |
| aw | 0.3 ± 0.01 b | 0.1 ± 0.01 a | 0.1 ± 0.01 a |
| Moisture content (%) | 6.4 ± 0.02 b | 3.2 ± 0.01 a | 3.2 ± 0.02 a |
| Hygroscopicity (%) | 39.4 ± 0.03 a | 48.0 ± 0.02 b | 53.1 ± 0.01 b |
| Bulk density (g/mL) | 0.4 ± 0.01 b | 0.3 ± 0.01 a | 0.3 ± 0.01 a |
| Solubility (%) | 70.4 ± 0.01 b | 59.9 ± 0.02 a | 59.1 ± 0.02 a |
| Dispersibility (%) | 99.1 ± 0.01 a | 100.0 ± 0.01 a | 97.7 ± 0.01 a |
| pH | 5.3 ± 0.02 b | 5.2 ± 0.01 a | 5.2 ± 0.02 a |
| MJ (Control) | SD | FDc | FDm | |||||
|---|---|---|---|---|---|---|---|---|
| t1/2 (day) | Retention t35 (%) | t1/2 (day) | Retention t35 (%) | t1/2 (day) | Retention t35 (%) | t1/2 (day) | Retention t35 (%) | |
| Del-3-sa-5-glu | nd | 100 ± 0.02 | nd | 97 ± 0.01 | nd | 99 ± 0.01 | nd | 90 ± 0.02 |
| Del-3,5-diglu | nd | 94 ± 0.02 | nd | 78 ± 0.02 | nd | 84 ± 0.03 | nd | 79 ± 0.03 |
| Cy-3-sa-5-glu+cy-3,5-diglu | nd | 96 ± 0.03 | nd | 85 ± 0.01 | nd | 95 ± 0.01 | nd | 86 ± 0.01 |
| Del-3-sa | 75 ± 8 b,A | 73 ± 0.03 | 75 ± 11 b,A | 77 ± 0.01 | 67 ± 11 b,A | 69 ± 0.02 | 70 ± 4 b,A | 70 ± 0.02 |
| Del-3-glu | 65 ± 9 a,B | 71 ± 0.02 | 59 ± 5 ab,AB | 66 ± 0.02 | 48 ± 8 a,A | 62 ± 0.01 | 46 ± 4 a,A | 62 ± 0.01 |
| Cy-3-glu | 68 ± 7 a,C | 73 ± 0.01 | 44 ± 2 a,A | 61 ± 0.02 | 59 ± 9 a,BC | 69 ± 0.02 | 50 ± 7 a,B | 63 ± 0.02 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fredes, C.; Becerra, C.; Parada, J.; Robert, P. The Microencapsulation of Maqui (Aristotelia chilensis (Mol.) Stuntz) Juice by Spray-Drying and Freeze-Drying Produces Powders with Similar Anthocyanin Stability and Bioaccessibility. Molecules 2018, 23, 1227. https://doi.org/10.3390/molecules23051227
Fredes C, Becerra C, Parada J, Robert P. The Microencapsulation of Maqui (Aristotelia chilensis (Mol.) Stuntz) Juice by Spray-Drying and Freeze-Drying Produces Powders with Similar Anthocyanin Stability and Bioaccessibility. Molecules. 2018; 23(5):1227. https://doi.org/10.3390/molecules23051227
Chicago/Turabian StyleFredes, Carolina, Camila Becerra, Javier Parada, and Paz Robert. 2018. "The Microencapsulation of Maqui (Aristotelia chilensis (Mol.) Stuntz) Juice by Spray-Drying and Freeze-Drying Produces Powders with Similar Anthocyanin Stability and Bioaccessibility" Molecules 23, no. 5: 1227. https://doi.org/10.3390/molecules23051227
APA StyleFredes, C., Becerra, C., Parada, J., & Robert, P. (2018). The Microencapsulation of Maqui (Aristotelia chilensis (Mol.) Stuntz) Juice by Spray-Drying and Freeze-Drying Produces Powders with Similar Anthocyanin Stability and Bioaccessibility. Molecules, 23(5), 1227. https://doi.org/10.3390/molecules23051227








