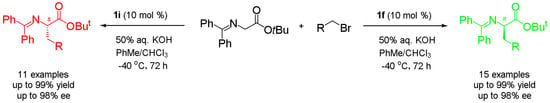
Synthesis of Both Enantiomers of Chiral Phenylalanine Derivatives Catalyzed by Cinchona Alkaloid Quaternary Ammonium Salts as Asymmetric Phase Transfer Catalysts
Abstract
1. Introduction
2. Results and Discussion
2.1. Condition Screening
2.2. Substrate Expansion
2.3. Application
3. Materials and Methods
3.1. Instruments and Reagents
3.2. General Methods
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Ahn, S.; Kahsai, A.W.; Pani, B.; Wang, Q.T.; Zhao, S.; Wall, A.L.; Strachan, R.T.; Staus, D.P.; Wingler, L.M.; Sun, L.D.; et al. Allosteric “beta-blocker” isolated from a DNA-encoded small molecule library. Proc. Natl. Acad. Sci. USA 2017, 114, 1708–1713. [Google Scholar] [CrossRef] [PubMed]
- Matsuura, T.; Cahnman, H.J. Model reactions for the biosynthesis of thyroxine. I. Structural influence of the side chain in analogs of diiodotyrosine on their conversion to analogs of thyroxine. J. Am. Chem. Soc. 1959, 81, 871–878. [Google Scholar] [CrossRef]
- Salamonczyk, G.M.; Oza, V.B.; Sih, C.J. A concise synthesis of thyroxine (T4) and 3,5,3′-triiodo-L-thyronine (T3). Tetrahedron Lett. 1997, 38, 6965–6968. [Google Scholar] [CrossRef]
- Neu, V.; Bielow, C.; Reinert, K.; Huber, C.G. Ultrahigh-performance liquid chromatography-ultraviolet absorbance detection-high-resolution-mass spectrometry combined with automated data processing for studying the kinetics of oxidative thermal degradation of thyroxine in the solid state. J. Chromatogr. A 2014, 1371, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Goodreid, J.D.; Santos, E.S.; Batey, R.A. A lanthanide(III) triflate mediated macrolactonization/solid-phase synthesis approach for depsipeptide synthesis. Org. Lett. 2015, 17, 2182–2185. [Google Scholar] [CrossRef] [PubMed]
- Farrell, D.J.; Couturier, C.; Hryniewicz, W. Distribution and antibacterial susceptibility of macrolide resistance genotypes in Streptococcus pneumoniae: PROTEKT Year 5 (2003–2004). Int. J. Antimicrob. Agents. 2008, 31, 245–249. [Google Scholar] [CrossRef] [PubMed]
- Wagner, M.M.; Paul, D.C.; Shih, C.; Jordan, M.A.; Wilson, L.; Williams, D.C. In vitro pharmacology of cryptophycin 52 (LY355703) in human tumor cell lines. Cancer Chemother. Pharmacol. 1999, 43, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Patel, V.F.; Andis, S.L.; Kennedy, J.H.; Ray, J.E.; Schultz, R.M. Novel cryptophycin antitumor agents: Synthesis and cytotoxicity of fragment “B” analogues. J. Med. Chem. 1999, 42, 2588–2603. [Google Scholar] [CrossRef] [PubMed]
- Jew, S.; Jeong, B.-S.; Lee, J.-H.; Yoo, M.-S.; Lee, Y.-J.; Park, B.; Kim, M.G.; Park, H. Highly enantioselective synthesis of α-alkyl-alanines via the catalytic phase-transfer alkylation of 2-naphthyl aldimine tert-butyl ester by using O(9)-allyl-N(1)-2′,3′,4′-trifluorobenzylhydrocinchonidinium bromide. J. Org. Chem. 2003, 68, 4514–4516. [Google Scholar] [CrossRef] [PubMed]
- Waser, M.; Gratzer, K.; Herchl, R.; Muller, N. Design, synthesis, and application of tartaric acid derived N-spiro quaternary ammonium salts as chiral phase-transfer catalysts. Org. Biomol. Chem. 2012, 10, 251–254. [Google Scholar] [CrossRef] [PubMed]
- Tiffner, M.; Zielke, K.; Mayr, J.; Haring, M.; Diaz, D.D.; Waser, M. Phase-transfer catalysis with ionene polymers. ChemistrySelect 2016, 1, 4030–4033. [Google Scholar] [CrossRef]
- Liu, Y.; Arumugam, N.; Almansour, A.I.; Kumar, R.S.; Maruoka, K. Practical synthesis of both enantiomeric amino acid, Mannich, and aldol derivatives by Aasymmetric organocatalysis. Chem. Rec. 2017, 17, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Arakawa, Y.; Haraguchi, N.; Itsuno, S. An immobilization method of chiral quaternary ammonium salts onto polymer supports. Angew. Chem. 2008, 120, 8356–8359. [Google Scholar] [CrossRef]
- Jew, S.; Park, H. Cinchona-based phase-transfer catalysts for asymmetric synthesis. Chem. Commun. 2009, 46, 7090–7103. [Google Scholar] [CrossRef] [PubMed]
- Shirakawa, S.; Maruoka, K. Recent developments in asymmetric phase-transfer reactions. Angew. Chem. Int. Ed. 2013, 52, 4312–4348. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Kumatabara, Y.; Shirakawa, S. Chiral quaternary phosphonium salts as phase-transfer catalysts for environmentally benign asymmetric transformations. Green Chem. 2016, 18, 331–341. [Google Scholar] [CrossRef]
- Boratynski, P.J.; Kowalczyk, R.; Kobylanska, A.; Bakowicz, J. Tricyclic quaternary ammonium salts derived from cinchona alkaloids. J. Org. Chem. 2016, 81, 12489–12493. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, M.J.; Bennett, W.D.; Wu, S. The stereoselective synthesis of α-amino acids by phase-transfer catalysis. J. Am. Chem. Soc. 1989, 111, 2353–2355. [Google Scholar] [CrossRef]
- Lygo, B.; Wainwright, P.G. A new class of asymmetric phase-transfer catalysts derived from cinchona alkaloids-Application in the enantioselective synthesis of α-amino acids. Tetrahedron Lett. 1997, 38, 8595–8598. [Google Scholar] [CrossRef]
- Corey, E.J.; Xu, F.; Noe, M.C. A rational approach to catalytic enantioselective enolate alkylation using a structurally rigidified and defined chiral quaternary ammonium salt under phase transfer conditions. J. Am. Chem. Soc. 1997, 119, 12414–12415. [Google Scholar] [CrossRef]
- McAllister, L.A.; Bechle, B.M.; Dounay, A.B.; Evrard, E.; Gan, X.; Ghosh, S.; Kim, J.Y.; Parikh, V.D.; Tuttle, J.B.; Verhoest, P.R. A general strategy for the synthesis of cyclic N-aryl hydroxamic acids via partial nitro group reduction. J. Org. Chem. 2011, 76, 3484–3497. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.T.; Zhao, S.; Jin, L.; Chen, X. Synthesis of Fmoc-protected (S)-3,5-dibromophenylalanine in the presence of a phase transfer catalyst or a chiral catalyst. Chin. J. Org. Chem. 2016, 36, 2242–2246. [Google Scholar] [CrossRef]
- O’Donnell, M.J.; Boniece, J.M.; Earp, S.E. The synthesis of amino acids by phase-transfer reaction. Tetrahedron Lett. 1978, 30, 2641–2644. [Google Scholar] [CrossRef]
- Jin, K.J.; Collum, D.B. Solid-state and solution structures of glycinimine-derived lithium enolates. J. Am. Chem. Soc. 2015, 137, 14446–14455. [Google Scholar] [CrossRef] [PubMed]
- Cann, P.; Levacher, V.; Bourguignon, J.; Dupas, G. Enantioselective catalytic phase transfer Michael addition of methyl 4-cyano-4-(3,4-dichlorophenyl)butanoates on acrylates. Lett. Org. Chem. 2004, 1, 129–133. [Google Scholar] [CrossRef]
- Corey, E.J.; Noe, M.C. Preparation of O-allyl-N-(9-anthracenylmethyl) cinchonidinium bromide as a phase transfer catalyst for catalyst for the enantionselective alkylation of glycine benzophenone imine tert-butyl ester. Org. Synth. 2003, 80, 38–45. [Google Scholar]
- Yin, D.; Zhang, L.; Tang, G.; Tang, X.; Wang, Y. Enantioselective synthesis of no-carrier added (NCA) 6-[18F]fluoro-L-dopa. J. Radioanal. Nucl. Chem. 2003, 257, 179–185. [Google Scholar] [CrossRef]
- Mizuta, S.; Shibata, N.; Hibino, M.; Nagano, S.; Nakamura, S.; Toru, T. Ammonium bromides/KF catalyzed trifluoromethylation of carbonyl compounds with (trifluoromethyl)trimethylsilane and its application in the enantioselective trifluoromethylation reaction. Tetrahedron 2007, 68, 8521–8528. [Google Scholar] [CrossRef]
- He, W.; Wang, Q.J.; Wang, Q.F.; Zhang, B.L.; Sun, X.L.; Zhang, S.Y. Synthesis of Novel Chiral Phase-Transfer Catalysts and Their Application to Asymmetric Synthesis of a-Amino Acid Derivatives. Synlett 2009, 8, 1311–1314. [Google Scholar]
- Lv, J.; Zhang, L.P.; Liu, L.; Wang, Y.M. A new class of acetophenone-based cinchona alkaloids as phase-transfer catalysts: Application to the enantioselective synthesis of α-amino acids. Chem. Lett. 2007, 36, 1354–1355. [Google Scholar] [CrossRef]
- Chinchilla, R.; Nájera, C.; Ortega, F.J. Improved conditions for the asymmetric synthesis of a-amino acids using Cinchona-derived anthracenylmethyl ammonium salts as chiral phase-transfer organocatalysts. Tetrahedron Asymmetry 2006, 17, 3423–3429. [Google Scholar] [CrossRef]
- Gratzer, K.; Waser, M. Investigations concerning the syntheses of TADDOL-derived secondary amines and their use to access novel chiral organocatalysts. Synthesis 2012, 44, 3661–3670. [Google Scholar] [PubMed]
- Ooi, T.; Kameda, M.; Maruoka, K. Design of N-spiro C2-symmetric chiral quaternary ammonium bromides as novel chiral phase-transfer catalysts: Synthesis and application to practical asymmetric synthesis of α-amino acids. J. Am. Chem. Soc. 2003, 125, 5139–5151. [Google Scholar] [CrossRef] [PubMed]
- Peng, W.W.; Wan, J.W.; Xie, B.; Ma, X.B. 9-Amino-(9-deoxy)cinchona alkaloid-derived new chiral phase-transfer catalysts. Org. Biomol. Chem. 2014, 12, 8336–8345. [Google Scholar] [CrossRef] [PubMed]
- Feng, D.; Wan, J.W.; Teng, F.; Ma, X.B. Heterogeneous simplified Maruoka phase-transfer catalyst tethered on pol(styrene-co-acrylamide) microsphere: Structure-activity relationship in enantioselective α-alkylation. Catal. Commun. 2017, 100, 127–133. [Google Scholar] [CrossRef]
- Wang, X.; Lv, J.; Liu, L.; Wang, Y.M.; Wu, Y. A novel N-acetophenone cinchona ammonium salts as chiral phase transfer catalysts for the alkylation of Schiff base in water. J. Mol. Catal. A 2007, 276, 102–109. [Google Scholar] [CrossRef]
- Shi, Q.H.; Lee, Y.-J.; Kim, M.-J.; Park, M.-K.; Lee, K.; Song, H.R.; Cheng, M.S.; Jeong, B.-S.; Park, H.; Jew, S. Highly efficient polymer supported phase-transfer catalysts containing hydrogen bond inducing functional groups. Tetrahedron Lett. 2008, 49, 1380–1383. [Google Scholar] [CrossRef]
- Wang, X.; Yin, L.; Yang, T.; Wang, Y.M. Synthesis of new dimeric-PEG-supported cinchona ammonium salts as chiral phase transfer catalysts for the alkylation of Schiff bases with water as the solvent. Tetrahedron Asymmetry 2007, 18, 108–114. [Google Scholar] [CrossRef]
- Lee, J.H.; Yoo, M.S.; Jung, J.-H.; Jew, S.; Parka, H.; Jeong, B.S. Polymeric chiral phase-transfer catalysts derived from cinchona alkaloids for enantioselective synthesis of α-amino acids. Tetrahedron 2007, 63, 7906–7915. [Google Scholar] [CrossRef]
- Ooi, T.; Kameda, M.; Maruoka, K. Molecular design of a C2-symmetric chiral phase-transfer catalyst for practical asymmetric synthesis of α-amino acids. J. Am. Chem. Soc. 1999, 121, 6519–6520. [Google Scholar] [CrossRef]
- Yoo, M.S.; Jeong, B.S.; Lee, J.H.; Park, H.G.; Jew, S.S. Evidence of the electronic factor for the highly enantioselective catalytic efficiency of cinchona-derived phase-transfer catalysts. Org. Lett. 2005, 7, 1129–1131. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds 4a–4o, 4a’–4o’ and 5–13 are available from the authors. |




| Entry a | 1 | Temp | Time/h | Yield b | ee c |
|---|---|---|---|---|---|
| 1 | 1a | rt | 24 | 81% | 88% |
| 2 | 1b | rt | 24 | 77% | 81% |
| 3 | 1c | rt | 24 | 93% | 77% |
| 4 | 1d | rt | 24 | 97% | 82% |
| 5 | 1e | rt | 24 | 72% | 54% |
| 6 | 1f | rt | 24 | 94% | 94% |
| 7 | 1g | rt | 24 | 74% | 78% |
| 8 | 1h | rt | 24 | 99% | 81% |
| 9 | 1f | −20 °C | 48 | 97% | 96% |
| 10 | 1f | −40 °C | 72 | 95% | 97% |
| 11 | 1f | −60 °C | 72 | 87% | 92% |
| 12d | 1f | −40 °C | 72 | 89% | 96% |
| 13e | 1f | −40 °C | 60 | 99% | 96% |
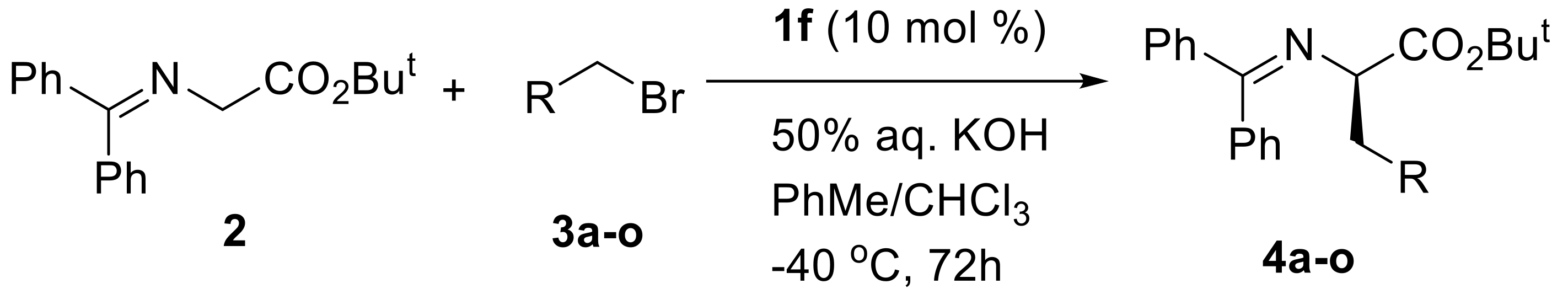
| Entry a | R | Product | Yield b | ee c |
|---|---|---|---|---|
| 1 | 3,5-Cl2C6H3 | 4a | 95% | 97% (R) |
| 2 | 3,5-F2C6H3 | 4b | 95% | 94% (R) |
| 3 | 3,5-Br2C6H3 | 4c | 95% | 93% (R) |
| 4 | 3-Cl,5-FC6H3 | 4d | 93% | 97% (R) |
| 5 | 2-Cl,6-FC6H3 | 4e | 68% | 98% (R) |
| 6 | 3-Cl,4-FC6H3 | 4f | 86% | 97% (R) |
| 7 | 3-Br,5-FC6H3 | 4g | 83% | 96% (R) |
| 8 | 3,5-(MeO)2C6H3 | 4h | 85% | 96% (R) |
| 9 | 3-FC6H4 | 4i | 77% | 95% (R) |
| 10 | 3-ClC6H4 | 4j | 76% | 96% (R) |
| 11 | 3-BrC6H4 | 4k | 98% | 95% (R) |
| 12 | 4-BrC6H4 | 4l | 95% | 95% (R) |
| 13 | 3-IC6H4 | 4m | 82% | 96% (R) |
| 14 | 4-NO2C6H4 | 4n | 99% | 95% (R) |
| 15 | 1-naphthyl | 4o | 81% | 96% (R) |
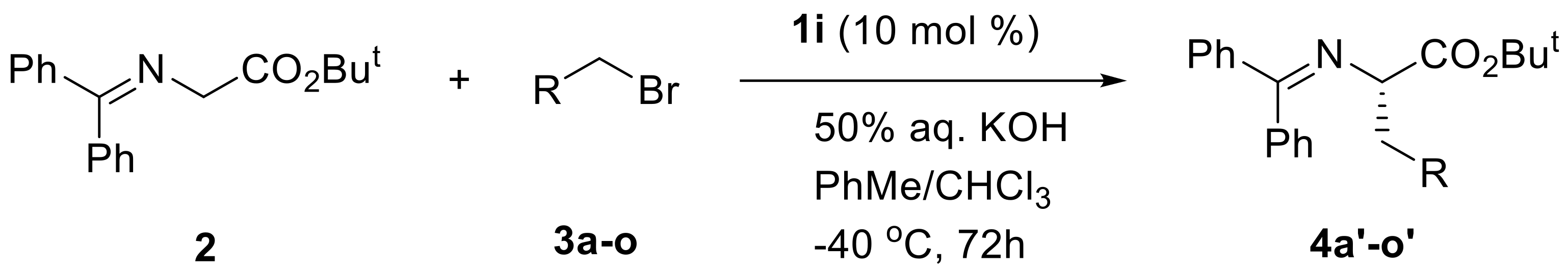
| Entry a | R | Product | Yieldb | ee c |
|---|---|---|---|---|
| 1 | 3,5-Cl2C6H3 | 4a’ | 98% | 97% (S) |
| 2 | 3,5-F2C6H3 | 4b’ | 99% | 94% (S) |
| 3 | 3,5-Br2C6H3 | 4c’ | 83% | 95% (S) |
| 4 | 3-Cl,5-FC6H3 | 4d’ | 97% | 98% (S) |
| 5 | 2-Cl,6-FC6H3 | 4e’ | 74% | 99% (S) |
| 6 | 3-Cl,4-FC6H3 | 4f’ | 88% | 97% (S) |
| 7 | 3-Br,5-FC6H3 | 4g’ | 86% | 98% (S) |
| 8 | 3,5-(MeO)2C6H3 | 4h’ | 97% | 98% (S) |
| 9 | 3-FC6H4 | 4i’ | 82% | 95% (S) |
| 10 | 3-ClC6H4 | 4j’ | 71% | 97% (S) |
| 11 | 3-BrC6H4 | 4k’ | 93% | 93% (S) |
| 12 | 4-BrC6H4 | 4l’ | 95% | 94% (S) |
| 13 | 3-IC6H4 | 4m’ | 86% | 94% (S) |
| 14 | 4-NO2C6H4 | 4n’ | 96% | 94% (S) |
| 15 | 1-naphthyl | 4o’ | 85% | 97% (S) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jin, L.; Zhao, S.; Chen, X. Synthesis of Both Enantiomers of Chiral Phenylalanine Derivatives Catalyzed by Cinchona Alkaloid Quaternary Ammonium Salts as Asymmetric Phase Transfer Catalysts. Molecules 2018, 23, 1421. https://doi.org/10.3390/molecules23061421
Jin L, Zhao S, Chen X. Synthesis of Both Enantiomers of Chiral Phenylalanine Derivatives Catalyzed by Cinchona Alkaloid Quaternary Ammonium Salts as Asymmetric Phase Transfer Catalysts. Molecules. 2018; 23(6):1421. https://doi.org/10.3390/molecules23061421
Chicago/Turabian StyleJin, Lei, Shuai Zhao, and Xin Chen. 2018. "Synthesis of Both Enantiomers of Chiral Phenylalanine Derivatives Catalyzed by Cinchona Alkaloid Quaternary Ammonium Salts as Asymmetric Phase Transfer Catalysts" Molecules 23, no. 6: 1421. https://doi.org/10.3390/molecules23061421
APA StyleJin, L., Zhao, S., & Chen, X. (2018). Synthesis of Both Enantiomers of Chiral Phenylalanine Derivatives Catalyzed by Cinchona Alkaloid Quaternary Ammonium Salts as Asymmetric Phase Transfer Catalysts. Molecules, 23(6), 1421. https://doi.org/10.3390/molecules23061421