Analysis of Metabolites in White Flowers of Magnolia Denudata Desr. and Violet Flowers of Magnolia Liliiflora Desr.
Abstract
:1. Introduction
2. Results and Discussion
2.1. LC-MS and HPLC Analysis
2.2. Total Phenolic and Anthocyanin Contents
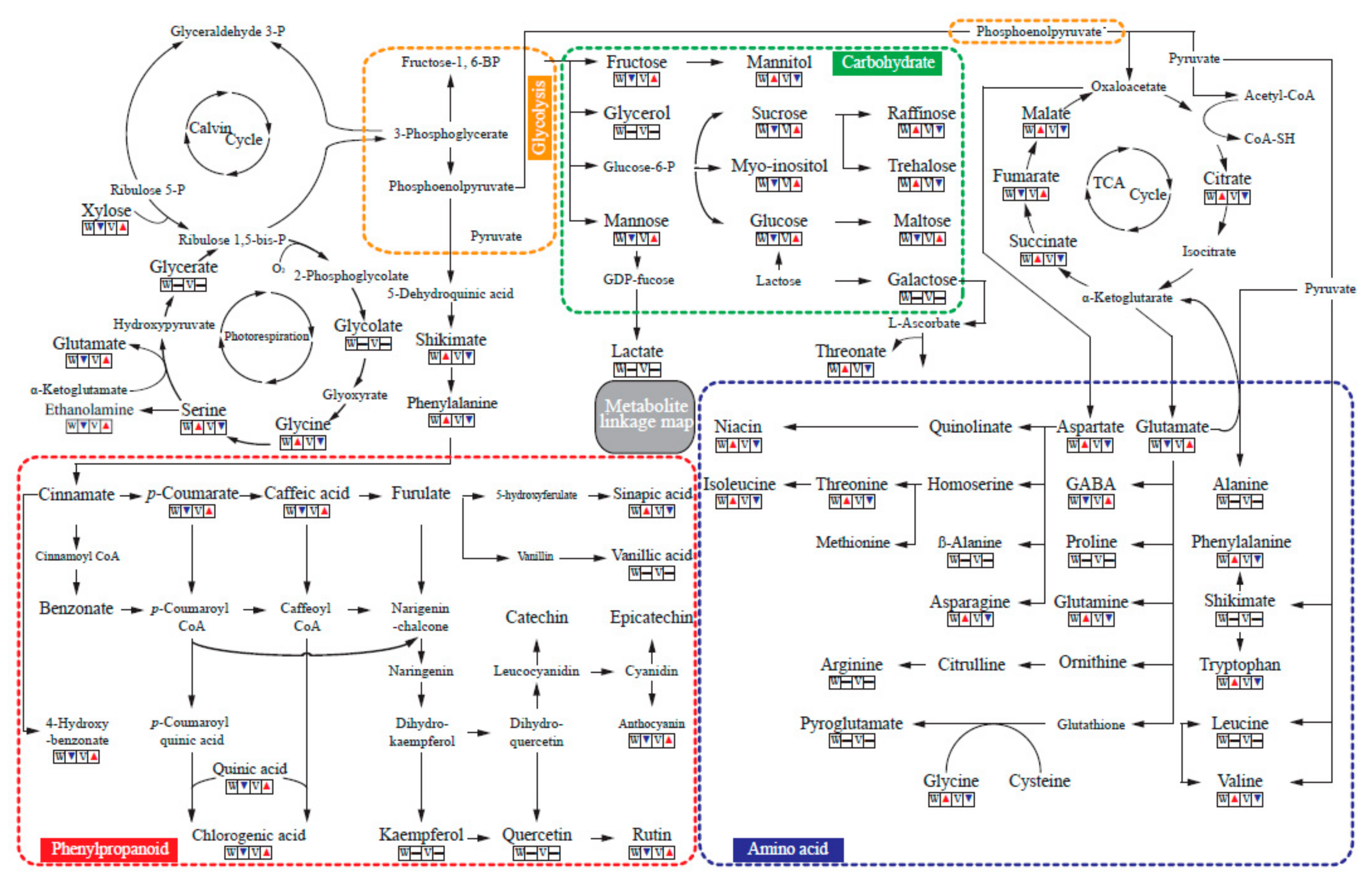
2.3. GC-TOFMS Analysis
2.4. In-Vitro Antioxidant Assays
2.4.1. 1,1-Diphenyl-2-picrylhydrazyl (DPPH) Radical Scavenging Activity
2.4.2. Dismutase-Like Activity
2.5. 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium Bromide (MTT) Assay for Cell Viability
2.6. Nitric Oxide (NO) Assay
3. Materials and Methods
3.1. Plant Materials
3.2. LC-MS Analysis for the Quantification of Phenylpropanoid Contents
3.3. Phenylpropanoid HPLC Analysis
3.4. Total Phenolics
3.5. Total Anthocyanin Content
3.6. GC-TOFMS Analysis
3.7. Superoxide Dismutase (SOD)-Like Activity
3.8. DPPH Assay
3.9. MTT Assay for Cell Viability
3.10. Nitric Oxide (NO) Assay
3.11. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Sowndhararajan, K.; Cho, H.; Yu, B.; Kim, S. Comparison of essential oil compositions of fresh and dried fruits of Magnolia kobus DC. J. Appl. Pharm. Sci. 2016, 6, 146–149. [Google Scholar] [CrossRef]
- Azuma, H.; Toyota, M.; Asakawa, Y. Intraspecific variation of floral scent chemistry in Magnolia kobus DC. (Magnoliaceae). J. Plant Res. 2001, 114, 411–422. [Google Scholar] [CrossRef]
- Seo, J.-J.; Lee, S.-H.; Lee, Y.-S.; Kwon, B.-M.; Ma, Y.; Hwang, B.-Y.; Hong, J.-T.; Oh, K.-W. Anxiolytic-like effects of obovatol isolated from Magnolia obovata: Involvement of GABA/benzodiazepine receptors complex. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2007, 31, 1363–1369. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.-J.; Lee, Y.M.; Lee, C.-K.; Jung, J.K.; Han, S.B.; Hong, J.T. Therapeutic applications of compounds in the Magnolia family. Pharmacol. Ther. 2011, 130, 157–176. [Google Scholar] [CrossRef] [PubMed]
- Teng, C.M.; Chen, C.C.; Ko, F.N.; Lee, L.G.; Huang, T.F.; Chen, Y.P.; Hsu, H.Y. Two antiplatelet agents from Magnolia officinalis. Thromb. Res. 1988, 50, 757–765. [Google Scholar] [CrossRef]
- Kassuya, C.A.L.; Cremoneze, A.; Barros, L.F.L.; Simas, A.S.; da Rocha Lapa, F.; Mello-Silva, R.; Stefanello, M.É.A.; Zampronio, A.R. Antipyretic and anti-inflammatory properties of the ethanolic extract, dichloromethane fraction and costunolide from Magnolia ovata (Magnoliaceae). J. Ethnopharmacol. 2009, 124, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Pang, Y.; Liu, S.; Tan, Z.; Tang, S.; Zhou, C.; Wang, M.; Xiao, W. Antidiarrhea and antioxidant activities of honokiol extract from magnoliae officinalis cortex in mice. Trop. J. Pharm. Res. 2014, 13, 1643–1651. [Google Scholar] [CrossRef]
- Watanabe, K. Pharmacology of magnolia bark with special reference to gastrointestinal functions. Gendai Toyo Igaku 1986, 7, 54–59. [Google Scholar]
- Hamasaki, Y.; Kobayashi, I.; Zaitu, M.; Tsuji, K.; Kita, M.; Hayasaki, R.; Muro, E.; Yamamoto, S.; Matsuo, M.; Ichimaru, T. Magnolol inhibits leukotriene synthesis in rat basophilic leukemia-2H3 cells. Planta Med. 1999, 65, 222–226. [Google Scholar] [CrossRef] [PubMed]
- Jo, Y.-H.; Seo, G.-U.; Yuk, H.-G.; Lee, S.-C. Antioxidant and tyrosinase inhibitory activities of methanol extracts from Magnolia denudata and Magnolia denudata var. purpurascens flowers. Food Res. Int. 2012, 47, 197–200. [Google Scholar] [CrossRef]
- Li, J.; Tanaka, M.; Kurasawa, K.; Ikeda, T.; Nohara, T. Lignan and neolignan derivatives from Magnolia denudata. Chem. Pharm. Bull. 2005, 53, 235–237. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.H.; Jiang, X.L.; Cai, C.G. Extraction technology and determination of total flavonoid in Magnolia liliiflora petals. Adv. Mater. Res. 2014, 941–944, 1003–1007. [Google Scholar] [CrossRef]
- Vinson, J.A.; Dabbagh, Y.A.; Serry, M.M.; Jang, J. Plant flavonoids, especially tea flavonols, are powerful antioxidants using an in vitro oxidation model for heart disease. J. Agric. Food Chem. 1995, 43, 2800–2802. [Google Scholar] [CrossRef]
- Dixon, R.A.; Paiva, N.L. Stress-induced phenylpropanoid metabolism. Plant Cell 1995, 7, 1085–1097. [Google Scholar] [CrossRef] [PubMed]
- Winkel-Shirley, B. Flavonoid biosynthesis. A colorful model for genetics, biochemistry, cell biology, and biotechnology. Plant Physiol. 2001, 126, 485–493. [Google Scholar] [CrossRef] [PubMed]
- Tattini, M.; Galardi, C.; Pinelli, P.; Massai, R.; Remorini, D.; Agati, G. Differential accumulation of flavonoids and hydroxycinnamates in leaves of Ligustrum vulgare under excess light and drought stress. New Phytol. 2004, 163, 547–561. [Google Scholar] [CrossRef] [Green Version]
- Tanaka, Y.; Sasaki, N.; Ohmiya, A. Biosynthesis of plant pigments: Anthocyanins, betalains and carotenoids. Plant J. 2008, 54, 733–749. [Google Scholar] [CrossRef] [PubMed]
- Castaneda-Ovando, A.; de Lourdes Pacheco-Hernández, M.; Páez-Hernández, M.E.; Rodríguez, J.A.; Galán-Vidal, C.A. Chemical studies of anthocyanins: A review. Food Chem. 2009, 113, 859–871. [Google Scholar] [CrossRef]
- Clifford, M.N. Anthocyanins–nature, occurrence and dietary burden. J. Sci. Food Agric. 2000, 80, 1063–1072. [Google Scholar] [CrossRef]
- Nijveldt, R.J.; Van Nood, E.; Van Hoorn, D.E.; Boelens, P.G.; Van Norren, K.; Van Leeuwen, P.A. Flavonoids: A review of probable mechanisms of action and potential applications. Am. J. Clin. Nutr. 2001, 74, 418–425. [Google Scholar] [CrossRef] [PubMed]
- Guardia, T.; Rotelli, A.E.; Juarez, A.O.; Pelzer, L.E. Anti-inflammatory properties of plant flavonoids. Effects of rutin, quercetin and hesperidin on adjuvant arthritis in rat. Farmaco 2001, 56, 683–687. [Google Scholar] [CrossRef]
- Villas-Boas, S.G.; Nielsen, J.; Smedsgaard, J.; Hansen, M.A.; Roessner-Tunali, U. Metabolome Analysis: An Introduction, 1st ed.; JohnWiley & Sons: Hoboken, NJ, USA, 2007; pp. 1–319. [Google Scholar]
- Roessner, U.; Luedemann, A.; Brust, D.; Fiehn, O.; Linke, T.; Willmitzer, L.; Fernie, A.R. Metabolic profiling allows comprehensive phenotyping of genetically or environmentally modified plant systems. Plant Cell 2001, 13, 11–29. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Liang, Y.; Dunn, W.B.; Shen, H.; Kell, D.B. Comparative evaluation of software for deconvolution of metabolomics data based on GC-TOF-MS. TrAC Trends Anal. Chem. 2008, 27, 215–227. [Google Scholar] [CrossRef]
- Dunn, W.B.; Broadhurst, D.; Ellis, D.I.; Brown, M.; Halsall, A.; O’hagan, S.; Spasic, I.; Tseng, A.; Kell, D.B. A GC-TOF-MS study of the stability of serum and urine metabolomes during the UK Biobank sample collection and preparation protocols. Int. J. Epidemiol. 2008, 37, i23–i30. [Google Scholar] [CrossRef] [PubMed]
- Becker, S.; Kortz, L.; Helmschrodt, C.; Thiery, J.; Ceglarek, U. LC–MS-based metabolomics in the clinical laboratory. J. Chromatogr. B 2012, 883, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Lu, N.; Chen, J.-H.; Wei, D.; Chen, F.; Chen, G. Global metabolic regulation of the snow alga Chlamydomonas nivalis in response to nitrate or phosphate deprivation by a metabolome profile analysis. Int. J. Mol. Sci. 2016, 17. [Google Scholar] [CrossRef] [PubMed]
- Lei, Z.; Huhman, D.V.; Sumner, L.W. Mass spectrometry strategies in metabolomics. J. Biol. Chem. 2011, 286, 25435–25442. [Google Scholar] [CrossRef] [PubMed]
- Merly, L.; Smith, S.L. Murine RAW 264.7 cell line as an immune target: Are we missing something? Immunopharmacol. Immunotoxicol. 2017, 39, 55–58. [Google Scholar] [CrossRef] [PubMed]
- Lipavská, H.E.L.E.N.; Konrádová, H.A.N. Invited review: Somatic embryogenesis in conifers: The role of carbohydrate metabolism. In Vitro Cell. Dev. Biol. Plant 2004, 40, 23–30. [Google Scholar] [CrossRef]
- Lam, H.-M.; Coschigano, K.; Oliveira, I.; Melo-Oliveira, R.; Coruzzi, G. The molecular-genetics of nitrogen assimilation into amino acids in higher plants. Annu. Rev. Plant Biol. 1996, 47, 569–593. [Google Scholar] [CrossRef] [PubMed]
- Yoon, H. Effects of aging on the phenolic content and antioxidant activities of magnolia (Magnolia denudata) flower extracts. Food Sci. Biotechnol. 2014, 23, 1715–1718. [Google Scholar] [CrossRef]
- Hongbing, G.; Huan, W.; Fengguo, D. Determination of free phenolics and combined phenolics in foliage of Magnolia sieboldii K. Koch. Funct. Mater. 2016, 23, 437–442. [Google Scholar] [CrossRef]
- Pyo, M.-K.; Koo, Y.-K.; YunChoi, H.-S. Anti-platelet effect of the phenolic constituents isolated from the leaves of Magnolia obovata. Nat. Prod. Sci. 2002, 8, 147–151. [Google Scholar]
- Porter, E.A.; Kite, G.C.; Veitch, N.C.; Geoghegan, I.A.; Larsson, S.; Simmonds, M.S. Phenylethanoid glycosides in tepals of Magnolia salicifolia and their occurrence in flowers of Magnoliaceae. Phytochemistry 2015, 117, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Chua, L.S. A review on plant-based rutin extraction methods and its pharmacological activities. J. Ethnopharmacol. 2013, 150, 805–817. [Google Scholar] [CrossRef] [PubMed]
- Choi, I.; Park, Y.; Choi, H.; Lee, E.H. Anti-adipogenic activity of rutin in 3T3-L1 cells and mice fed with high-fat diet. BioFactors 2006, 26, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Kessler, M.; Ubeaud, G.; Jung, L. Anti-and pro-oxidant activity of rutin and quercetin derivatives. J. Pharm. Pharmacol. 2003, 55, 131–142. [Google Scholar] [CrossRef] [PubMed]
- Selloum, L.; Bouriche, H.; Tigrine, C.; Boudoukha, C. Anti-inflammatory effect of rutin on rat paw oedema, and on neutrophils chemotaxis and degranulation. Exp. Toxicol. Pathol. 2003, 54, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Jung, C.H.; Lee, J.Y.; Cho, C.H.; Kim, C.J. Anti-asthmatic action of quercetin and rutin in conscious guinea-pigs challenged with aerosolized ovalbumin. Arch. Pharm. Res. 2007, 30, 1599–1607. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Z.; Ning, Z. Scavenging and antioxidant properties of compound derived from chlorogenic acid in South-China honeysuckle. LWT Food Sci. Technol. 2008, 41, 1189–1203. [Google Scholar] [CrossRef]
- Nakagawa, K.; Ninomiya, M.; Okubo, T.; Aoi, N.; Juneja, L.R.; Kim, M.; Yamanaka, K.; Miyazawa, T. Tea catechin supplementation increases antioxidant capacity and prevents phospholipid hydroperoxidation in plasma of humans. J. Agric. Food Chem. 1999, 47, 3967–3973. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Wang, S.; Zhang, J.; Yang, G.; Zhao, M.; Ma, W.; Zhang, X.; Li, X.; Han, B.; Chen, N. Effects of ecological factors on secondary metabolites and inorganic elements of Scutellaria baicalensis and analysis of geoherblism. Sci. China Life Sci. 2013, 56, 1047–1056. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Markom, M.; Hasan, M.; Daud, W.R.W.; Singh, H.; Jahim, J.M. Extraction of hydrolysable tannins from Phyllanthus niruri Linn.: Effects of solvents and extraction methods. Sep. Purif. Technol. 2007, 52, 487–496. [Google Scholar] [CrossRef]
- Szakiel, A.; Pączkowski, C.; Henry, M. Influence of environmental abiotic factors on the content of saponins in plants. Phytochem. Rev. 2011, 10, 471–491. [Google Scholar] [CrossRef]
- Aharoni, A.; Galili, G. Metabolic engineering of the plant primary–secondary metabolism interface. Curr. Opin. Biotechnol. 2011, 22, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Masoumian, M.; Arbakariya, A.; Syahida, A.; Maziah, M. Flavonoids production in Hydrocotyle bonariensis callus tissues. J. Med. Plants Res. 2011, 5, 1564–1574. [Google Scholar]
- Zulak, K.G.; Weljie, A.M.; Vogel, H.J.; Facchini, P.J. Quantitative 1H NMR metabolomics reveals extensive metabolic reprogramming of primary and secondary metabolism in elicitor-treated opium poppy cell cultures. BMC Plant Biol. 2008, 8, 5. [Google Scholar] [CrossRef] [PubMed]
- Payyavula, R.S.; Singh, R.K.; Navarre, D.A. Transcription factors, sucrose, and sucrose metabolic genes interact to regulate potato phenylpropanoid metabolism. J. Exp. Bot. 2013, 64, 5115–5131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferri, M.; Righetti, L.; Tassoni, A. Increasing sucrose concentrations promote phenylpropanoid biosynthesis in grapevine cell cultures. J. Plant Physiol. 2011, 168, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Park, C.H.; Kim, Y.S.; Li, X.; Kim, H.H.; Arasu, M.V.; Al-Dhabi, N.A.; Lee, S.Y.; Park, S.U. Influence of Different Carbohydrates on Flavonoid Accumulation in Hairy Root Cultures of Scutellaria baicalensis. Nat. Prod. Commun. 2016, 11, 799–802. [Google Scholar] [PubMed]
- Rice-Evans, C. Flavonoid antioxidants. Curr. Med. Chem. 2001, 8, 797–807. [Google Scholar] [CrossRef] [PubMed]
- Rice-Evans, C.; Miller, N.; Paganga, G. Antioxidant properties of phenolic compounds. Trends Plant Sci. 1997, 2, 152–159. [Google Scholar] [CrossRef]
- Torel, J.; Cillard, J.; Cillard, P. Antioxidant activity of flavonoids and reactivity with peroxy radical. Phytochemistry 1986, 25, 383–385. [Google Scholar] [CrossRef]
- Kang, Y.-H.; Park, Y.-K.; Lee, G.-D. The nitrite scavenging and electron donating ability of phenolic compounds. Korean J. Food Sci. Technol. 1996, 28, 232–239. [Google Scholar]
- Natella, F.; Nardini, M.; Di Felice, M.; Scaccini, C. Benzoic and cinnamic acid derivatives as antioxidants: Structure− activity relation. J. Agric. Food Chem. 1999, 47, 1453–1459. [Google Scholar] [CrossRef] [PubMed]
- Kähkönen, M.P.; Heinonen, M. Antioxidant activity of anthocyanins and their aglycons. J. Agric. Food Chem. 2003, 51, 628–633. [Google Scholar] [CrossRef] [PubMed]
- Park, C.H.; Baskar, T.B.; Park, S.-Y.; Kim, S.-J.; Valan Arasu, M.; Al-Dhabi, N.A.; Kim, J.K.; Park, S.U. Metabolic profiling and antioxidant assay of metabolites from three radish cultivars (Raphanus sativus). Molecules 2016, 21, 157. [Google Scholar] [CrossRef] [PubMed]
- Park, C.H.; Yeo, H.J.; Baskar, T.B.; Kim, J.K.; Park, S.U. Metabolic Profiling and Chemical-Based Antioxidant Assays of Green and Red Lettuce (Lactuca sativa). Nat. Prod. Commun. 2018, 13, 315–322. [Google Scholar]
- Akerele, O. Nature’s medicinal bounty: Don’t throw it away. World Health Forum 1993, 14, 390–395. [Google Scholar] [PubMed]
- Ekor, M. The growing use of herbal medicines: Issues relating to adverse reactions and challenges in monitoring safety. Front. Pharmacol. 2014, 4, 177. [Google Scholar] [CrossRef] [PubMed]
- Park, C.H.; Yeo, H.J.; Park, Y.J.; Morgan, A.; Valan Arasu, M.; Al-Dhabi, N.A.; Park, S.U. Influence of indole-3-acetic acid and gibberellic acid on phenylpropanoid accumulation in common buckwheat (Fagopyrum esculentum Moench) sprouts. Molecules 2017, 22, 374. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Kim, J.K.; Park, S.-Y.; Zhao, S.; Kim, Y.B.; Lee, S.; Park, S.U. Comparative analysis of flavonoids and polar metabolite profiling of tanno-original and tanno-high rutin buckwheat. J. Agric. Food Chem. 2014, 62, 2701–2708. [Google Scholar] [CrossRef] [PubMed]
- Cho, E.-K.; Choi, Y.-J. Physiological activities of hot water extracts from Ecklonia cava Kjellman. J. Life Sci. 2010, 20, 1675–1682. [Google Scholar] [CrossRef]
- Yang, E.-J.; Yim, E.-Y.; Song, G.; Kim, G.-O.; Hyun, C.-G. Inhibition of nitric oxide production in lipopolysaccharide-activated RAW 264.7 macrophages by Jeju plant extracts. Interdiscip. Toxicol. 2009, 2, 245–249. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of compounds are not available from the authors. |




| No. | Retention Time | Phenolics | [M − H]− (m/z) | White | Violet |
|---|---|---|---|---|---|
| 1 | 20.98 | 4-hydroxybenzoic acid | 137.6 | 0.27 ± 0.02 | 1.20 ± 0.46 * |
| 2 | 22.85 | Chlorogenic acid | 353.8 | 0.47 ± 0.10 | 1.69 ± 0.42 * |
| 3 | 27.78 | Caffeic acid | 179.9 | tr 1 | 0.04 ± 0.00 * |
| 4 | 41.71 | p-coumaric acid | 163.4 | tr | 0.26 ± 0.04 * |
| 5 | 61.99 | Rutin | 609.0 | 10.47 ± 0.34 | 13.62 ± 1.45 * |
| 6 | 73.66 | Quercetin | 301.1 | 0.38 ± 0.00 | 0.37 ± 0.00 |
| 7 | 80.36 | Kaempferol | 285.5 | 0.12 ± 0.00 | 0.13 ± 0.02 |
| Total | 11.71 ± 0.46 | 17.31 ± 2.39 * |
| Total Phenolics (mg/g) | Total Anthocyanin (mg/g) | |
|---|---|---|
| Violet Flowers | 16.94 ± 0.71 * | 0.78 ± 0.16 * |
| White Flowers | 14.07 ± 0.55 | 0.21 ± 0.08 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, C.H.; Park, S.-Y.; Lee, S.Y.; Kim, J.K.; Park, S.U. Analysis of Metabolites in White Flowers of Magnolia Denudata Desr. and Violet Flowers of Magnolia Liliiflora Desr. Molecules 2018, 23, 1558. https://doi.org/10.3390/molecules23071558
Park CH, Park S-Y, Lee SY, Kim JK, Park SU. Analysis of Metabolites in White Flowers of Magnolia Denudata Desr. and Violet Flowers of Magnolia Liliiflora Desr. Molecules. 2018; 23(7):1558. https://doi.org/10.3390/molecules23071558
Chicago/Turabian StylePark, Chang Ha, Soo-Yun Park, Sook Young Lee, Jae Kwang Kim, and Sang Un Park. 2018. "Analysis of Metabolites in White Flowers of Magnolia Denudata Desr. and Violet Flowers of Magnolia Liliiflora Desr." Molecules 23, no. 7: 1558. https://doi.org/10.3390/molecules23071558





