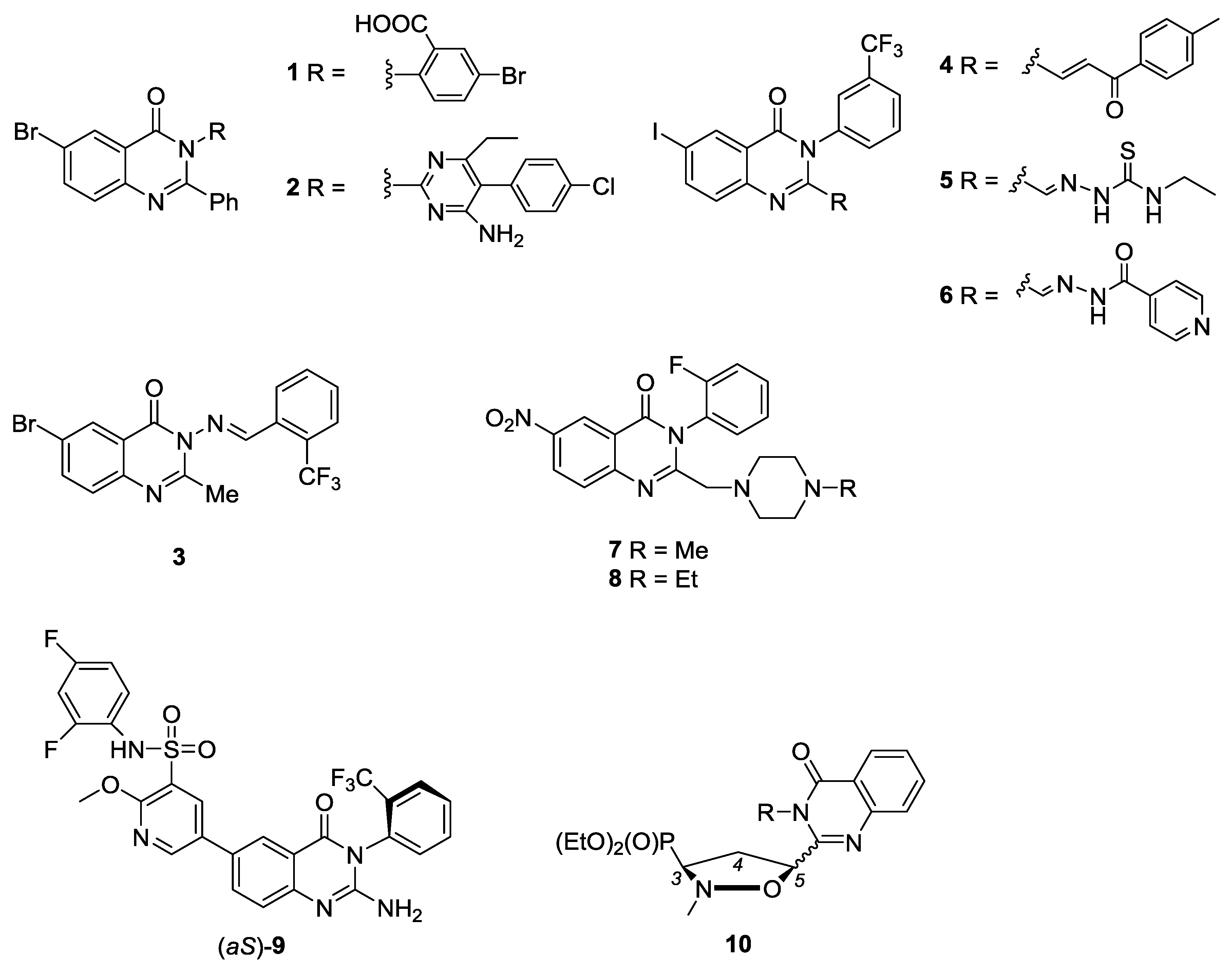
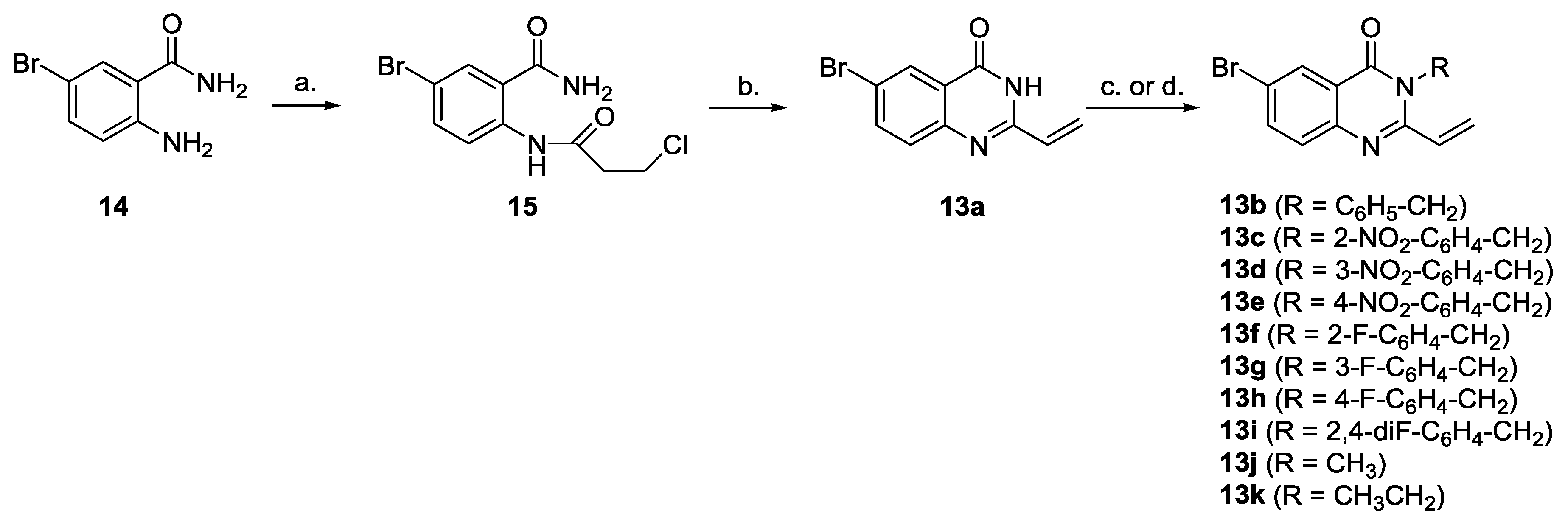
3.3. General Procedure for the Synthesis of N3-Benzylated 6-bromo-2-vinyl-3H-quinazolin-4-ones 13b–13i
To a solution of 6-bromo-2-vinyl-3H-quinazolin-4-one (0.251 g, 1.00 mmol) in acetonitrile (15 mL) potassium hydroxide (0.168 g, 3.00 mmol) was added. After 15 min. the respective benzyl bromide (1.10 mmol) was added and the reaction mixture was stirred under reflux for 4 h. The solvent was removed and the residue was re-dissolved in methylene chloride (10 mL) and extracted with water (3 × 10 mL). An organic phase was dried (MgSO4), concentrated, and the crude product was purified on a silica gel column with a methylene chloride:hexane (7:3, v/v) mixture followed by crystallization (chloroform-petroleum ether or ethyl acetate) to give pure quinazolinones.
3-Benzyl-6-bromo-2-vinylquinazolin-4(3H)-one (13b). A white amorphous solid, m.p. = 71–72 °C (re-crystallized from ethyl acetate). IR (KBr, cm−1) νmax: 3097, 3036, 2926, 1608, 1571, 1553, 1470, 1290, 1153, 1112, 989, 841, 678, 581, 437. 1H-NMR (200 MHz, CDCl3): δ = 8.30–8.27 (m, 1H), 7.88–7.82 (m, 1H), 7.77–7.73 (m, 1H), 7.57–7.50 (m, 2H), 7.48–7.37 (m, 3H), 6.91 (dd, 3J = 17.2 Hz, 3J = 9.9 Hz, 1H, CH=CH2), 6.73 (dd, 3J = 17.2 Hz, 2J = 2.5 Hz, 1H, CH=CH2), 5.82 (dd, 3J = 9.9 Hz, 2J = 2.5 Hz, 1H, CH=CH2), 5.67 (s, 2H, N-CH2). 13C-NMR (151 MHz, CDCl3): δ = 165.16 (C=O), 160.28, 150.36, 137.00, 136.88, 136.02, 129.45, 128.67, 128.45, 126.09, 124.36, 120.03, 116.67, 68.69 (N-CH2). Anal. Calcd. for C17H13BrN2O: C, 59.84; H, 3.84; N, 8.21. Found: C, 59.92; H, 3.68; N, 8.29.
6-Bromo-3-(2-nitrobenzyl)-2-vinylquinazolin-4(3H)-one (13c). A yellowish amorphous solid, m.p. 117–119 °C (re-crystallized from chloroform-petroleum ether). IR (KBr, cm−1) νmax: 3078, 2980, 2925, 2854, 1689, 1613, 1573, 1526, 1489, 1390, 1354, 1287, 1242, 1116, 1053, 1024, 968, 836, 791, 730. 1H-NMR (600 MHz, CDCl3): δ = 8.33 (d, 4J = 2.2 Hz, 1H, HC5), 8.18–8.16 (m, 1H), 7.90 (dd, 3J = 8.9 Hz, 4J = 2.2 Hz, 1H, HC7), 7.81 (d, 3J = 8.9 Hz, 1H, HC8), 7.80–7.78 (m, 1H), 7.71–7.69 (m, 1H), 7.56–7.53 (m, 1H), 6.87 (dd, 3J = 17.2 Hz, 3J = 10.4 Hz, 1H, CH=CH2), 6.64 (dd, 3J = 17.2 Hz, 2J = 1.5 Hz, 1H, CH=CH2), 6.10 (s, 2H, N-CH2), 5.80 (dd, 3J = 10.4 Hz, 2J = 1.5 Hz, 1H, CH=CH2). 13C-NMR (151 MHz, CDCl3): δ = 164.62 (C=O), 160.12, 150.32, 147.84, 137.31, 136.31, 133.78, 132.33, 129.51, 129.05, 128.87, 125.76, 125.07, 124.92, 120.38, 116.31, 65.14 (N-CH2). Anal. Calcd. for C17H12BrN3O3: C, 52.87; H, 3.13; N, 10.88. Found: C, 52.50; H, 2.80; N, 10.80.
6-Bromo-3-(3-nitrobenzyl)-2-vinylquinazolin-4(3H)-one (13d). A yellowish amorphous solid, m.p. 147–149 °C (re-crystallized from chloroform-petroleum ether). IR (KBr, cm−1) νmax: 3112, 3071, 2924, 2854, 1610, 1573, 1529, 1456, 1382, 1294, 1119, 836, 668. 1H-NMR (600 MHz, CDCl3): δ = 8.44 (brs, 1H), 8.32–8.30 (m, 1H), 8.26–8.24 (m, 1H), 7.91–7.88 (m, 2H), 7.80–7.78 (m, 1H), 7.64–7.61 (m, 1H), 6.91 (dd, 3J = 17.2 Hz, 3J = 10.4 Hz, 1H, CH=CH2), 6.73 (dd, 3J = 17.2 Hz, 2J = 1.4 Hz, 1H, CH=CH2), 5.84 (dd, 3J = 10.4 Hz, 2J = 1.4 Hz, 1H, CH=CH2), 5.78 (s, 2H, N-CH2). 13C-NMR (151 MHz, CDCl3): δ = 164.69 (C=O), 160.05, 150.49, 148.51, 138.14, 137.28, 136.68, 134.10, 129.73, 129.60, 125.84, 124.47, 123.37, 123.27, 120.35, 116.34, 67.22 (N-CH2). Anal. Calcd. for C17H12BrN3O3: C, 52.87; H, 3.13; N, 10.88. Found: C, 52.81; H, 2.98; N, 10.60.
6-Bromo-3-(4-nitrobenzyl)-2-vinylquinazolin-4(3H)-one (13e). Off-white amorphous solid, m.p. 121–123 °C (re-crystallized from chloroform-petroleum ether). IR (KBr, cm−1) νmax: 3418, 2924, 2853, 1739, 1608, 1573, 1524, 1343, 1292, 1117, 945, 833, 661. 1H-NMR (200 MHz, CDCl3): δ = 8.30–8.26 (m, 3H), 7.96–7.92 (m, 1H), 7.81–7.76 (m, 1H), 7.73–7.67 (m, 2H), 6.88 (dd, 3J = 17.2 Hz, 3J = 10.1 Hz, 1H, CH=CH2), 6.65 (dd, 3J = 17.2 Hz, 2J = 2.1 Hz, 1H, CH=CH2), 5.80 (dd, 3J = 10.1 Hz, 2J = 2.1 Hz, 1H, CH=CH2), 5.77 (s, 2H, N-CH2). 13C-NMR (151 MHz, CDCl3): δ = 164.69 (C=O), 160.03, 150.41, 147.93, 143.27, 137.37, 136.59, 129.58, 128.50, 125.80, 124.58, 123.93, 120.44, 116.33, 67.14 (N-CH2). Anal. Calcd. for C17H12BrN3O3: C, 52.87; H, 3.13; N, 10.88. Found: C, 52.81; H, 2.98; N, 10.60.
6-Bromo-3-(2-fluorobenzyl)-2-vinylquinazolin-4(3H)-one (13f). A white amorphous solid, m.p. 91–92 °C (re-crystallized from ethyl acetate). IR (KBr, cm−1) νmax: 3078, 2967, 1568, 1555, 1486, 1420, 1348, 1293, 1238, 1115, 938, 828, 760, 675. 1H-NMR (600 MHz, CDCl3): δ = 8.31 (d, 4J = 2.2 Hz, 1H, HC5), 7.89 (dd, 3J = 8.9 Hz, 4J = 2.2 Hz, 1H, HC7), 7.68 (d, 3J = 8.9 Hz, 1H, HC8), 7.61–7.58 (m, 1H), 7.42–7.38 (m, 1H), 7.23–7.20 (m, 1H), 7.18–7.15 (m, 1H), 6.92 (dd, 3J = 17.2 Hz, 3J = 10.4 Hz, 1H, CH=CH2), 6.78 (dd, 3J = 17.2 Hz, 2J = 1.7 Hz, 1H, CH=CH2), 5.84 (dd, 3J = 10.4 Hz, 2J = 1.7 Hz, 1H, CH=CH2), 5.78 (s, 2H, N-CH2). 13C-NMR (151 MHz, CDCl3): δ = 164.99 (C=O), 161.20 (d, 1J(CF) = 248.8 Hz, C2′), 160.24, 150.39, 137.03, 136.81, 130.70 (d, 3J(CCCF) = 4.0 Hz, C4′), 130.39 (d, 3J(CCCF) = 7.9 Hz, C6′), 129.47, 126.04, 124.44, 124.25 (d, 4J(CCCCF) = 3.4 Hz, C5′), 123.24 (d, 2J(CCF) = 14.5 Hz, C3′), 120.06, 116.59, 115.63 (d, 2J(CCF) = 21.5 Hz, C1′), 62.51 (d, 3J(CCF) = 4.4 Hz, N-CH2). Anal. Calcd. for C17H12BrFN2O: C, 56.85; H, 3.37; N, 7.80. Found: C, 56.62; H, 2.99; N, 7.79.
6-Bromo-3-(3-fluorobenzyl)-2-vinylquinazolin-4(3H)-one (13g). A yellowish amorphous solid, m.p. 78–80 °C (re-crystallized from ethyl acetate). IR (KBr, cm−1) νmax: 3070, 2980, 2925, 2853, 1614, 1568, 1490, 1416, 1336, 1286, 1115, 1055, 967, 836, 790. 1H-NMR (600 MHz, CDCl3): δ = 8.32 (d, 4J = 2.2 Hz, 1H, HC5), 7.89 (dd, 3J = 8.9 Hz, 4J = 2.2 Hz, 1H, HC7), 7.80 (d, 3J = 8.9 Hz, 1H, HC8), 7.43–7.39 (m, 1H), 7.34–7.32 (m, 1H), 7.28–7.26 (m, 1H), 7.11–7.08 (m, 1H), 6.92 (dd, 3J = 17.1 Hz, 3J = 10.4 Hz, 1H, CH=CH2), 6.73 (dd, 3J = 17.1 Hz, 2J = 1.7 Hz, 1H, CH=CH2), 5.84 (dd, 3J = 10.4 Hz, 2J = 1.7 Hz, 1H, CH=CH2), 5.69 (s, 2H, N-CH2). 13C-NMR (151 MHz, CDCl3): δ = 164.95 (C=O), 162.95(d, 1J(CF) = 246.6 Hz, C3′), 160.19, 150.43, 138.50 (d, 3J(CCCF) = 7.5 Hz, C5′), 137.13, 136.79, 130.25 (d, J = 8.1 Hz, C1′), 129.53, 125.98, 124.42, 123.74 (d, 4J(CCCCF) = 3.1 Hz, C6′), 120.18, 116.54, 115.33 (d, 2J(CCF) = 21.0 Hz, C4′), 115.14 (d, 2J(CCF) = 22.0 Hz, C2′), 67.77 (d, 4JCCCCF = 1.5 Hz, N-CH2). Anal. Calcd. for C17H12BrFN2O: C, 56.85; H, 3.37; N, 7.80. Found: C, 56.99; H, 2.97; N, 7.84.
6-Bromo-3-(4-fluorobenzyl)-2-vinylquinazolin-4(3H)-one (13h). A yellowish amorphous solid, m.p. 100–103 °C (re-crystallized from chloroform-petroleum ether). IR (KBr, cm−1) νmax: 3064, 2926, 1608, 1565, 1510, 1487, 1354, 1219, 1118, 989, 832, 697, 501. 1H-NMR (200 MHz, CDCl3): δ = 8.26 (d, 4J = 2.1 Hz, 1H, HC5), 7.85 (dd, 3J = 8.9 Hz, 4J = 2.1 Hz, 1H, HC7), 7.75 (d, 3J = 8.9 Hz, 1H, HC8), 7.55–7.48 (m, 2H), 7.15–7.06 (m, 2H), 6.90 (dd, 3J = 17.2 Hz, 3J = 11.5 Hz, 1H, CH=CH2), 6.72 (dd, 3J = 17.2 Hz, 2J = 2.3 Hz, 1H, CH=CH2), 5.82 (dd, 3J = 11.5 Hz, 2J = 2.3 Hz, 1H, CH=CH2), 5.63 (s, 2H, N-CH2). 13C-NMR (151 MHz, CDCl3): δ = 165.04 (C=O), 162.82 (d, 1J(CF) = 247.1 Hz, C4′), 160.20, 150.40, 137.04, 136.88, 131.84 (d, 4J(CCCCF) = 3.1 Hz, C1′), 130.39 (d, 3J(CCCF) = 8.6 Hz, C2′, C6′), 129.50, 125.99, 124.28, 120. 08, 116.60, 115.62 (d, 2J(CCF) = 21.9 Hz, C3′, C5′), 67.94 (N-CH2). Anal. Calcd. for C17H12BrFN2O: C, 56.85; H, 3.37; N, 7.80. Found: C, 56.50; H, 3.01; N, 7.76.
6-Bromo-3-(2,4-difluorobenzyl)-2-vinylquinazolin-4(3H)-one (13i). A yellowish amorphous solid, m.p. 88–90 °C (re-crystallized from chloroform-petroleum ether). IR (KBr, cm−1) νmax: 3079, 2923, 2852, 1614, 1565, 1508, 1488, 1416, 1349, 1279, 1099, 955, 834, 728, 538. 1H-NMR (600 MHz, CDCl3): δ = 8.27 (d, 4J = 2.1 Hz, 1H, HC5), 7.88 (dd, 3J = 8.9 Hz, 4J = 2.1 Hz, 1H, HC7), 7.78 (d, 3J = 8.9 Hz, 1H, HC8), 7.59–7.55 (m, 1H), 6.96–6.89 (m, 2H), 6.89 (dd, 3J = 17.2 Hz, 3J = 10.0 Hz, 1H, CH=CH2), 6.72 (dd, 3J = 17.2 Hz, 2J = 2.4 Hz, 1H, CH=CH2), 5.82 (dd, 3J = 10.0 Hz, 2J = 2.4 Hz, 1H, CH=CH2), 5.68 (s, 2H, N-CH2). 13C-NMR (151 MHz, CDCl3) δ: 164.86 (C=O), 163.23 (dd, 1J(CF) = 250.4 Hz, 3J(CCCF) = 12.0 Hz, C2′), 161.45 (dd, 1J(CF) = 251.8 Hz, 3J(CCCF) = 12.1 Hz, C4′), 160.15, 150.38, 137.06, 136.80, 131.83 (dd, 3J(CCCF) = 9.8 Hz, 3J(CCCF) = 5.4 Hz, C6′), 129.49, 125.95, 124.39, 120.12, 119.29 (dd, 2J(CCF) = 15.0 Hz, 4J(CCCCF) = 4.0 Hz, C1′), 116.48, 111.50 (dd, 2J(CCF) = 21.7 Hz, 4J(CCCCF) = 4.1 Hz, C5′), 104.16 (dd, 2J(CCF) = 25.3 Hz, 2J(CCF) = 25.3 Hz, C3′), 61.93 (d, 3J(CCCF) = 3.4 Hz, N-CH2) Anal. Calcd. for C17H11BrF2N2O: C, 54.13; H, 2.94; N, 7.43. Found: C, 54.42; H, 3.14; N, 7.04.
3.5. General Procedure for the Synthesis of Isoxazolidines trans-11 and cis-11
A solution of the nitrone (0.195 g, 1.00 mmol) and the respective 6-bromo-2-vinylquinazolin-4(3H)-one (1.00 mmol) in toluene (2 mL) was stirred at 70 °C until the disappearance (TLC) of the starting nitrone. Solvents were evaporated in vacuo and crude products were subjected to chromatography on silica gel columns with chloroform:methanol (100:1, 50:1, 20:1, v/v) mixtures.
Diethyl trans-[5-(6-bromo-4-oxo-3,4-dihydroquinazolin-2-yl)-2-methylisoxazolidin-3-yl]phosphonate (trans-11a). Colorless oil. IR (film, cm−1) νmax: 3316, 3171, 3090, 2980, 2974, 2783, 1660, 1625, 1486, 1412, 1301, 1234, 1054, 968, 834, 775, 575. 1H-NMR (600 MHz, CDCl3): δ = 10.63 (s, 1H, NH), 8.44 (d, 4J = 2.3 Hz, 1H, HC5′), 7.84 (dd, 3J = 8.6 Hz, 4J = 2.3 Hz, 1H, HC7′), 7.54 (d, 3J = 8.6 Hz, 1H, HC8′), 5.02 (dd, 3J(H5–H4β) = 8.4 Hz, 3J(H5–H4α) = 6.2 Hz, 1H, HC5), 4.30–4.21 (m, 4H, 2 × CH2OP), 3.25–3.21 (m, 1H, HC3), 3.04 (s, 3H, CH3N), 3.12 (dddd, 3J(H4β–P) = 16.7 Hz, 2J(H4β–H4α) = 12.9 Hz, 3J(H4β–H5) = 8.6 Hz, 3J(H4β–H3) = 8.3 Hz, 1H, HβC4), 2.94 (dddd, 3J(H4α–P) = 12.9 Hz, 2J(H4α–H4β) = 12.9 Hz, 3J(H4α–H3) = 10.0 Hz, 3J(H4α–H5) = 6.2 Hz, 1H, HαC4), 1.40 (t, 3J = 7.1 Hz, 3H, CH3CH2OP), 1.38 (t, 3J = 7.0 Hz, 3H, CH3CH2OP). 13C-NMR (151 MHz, CDCl3): δ = 160.30 (C=O), 156.50, 147.35, 137.66, 129.26, 128.96, 123.27, 120.46, 74.68 (d, 3J(CCCP) = 8.6 Hz, C5), 64.60 (d, 1J(CP) = 168.1 Hz, C3), 63.51 (d, 2J(COP) = 6.6 Hz, CH2OP), 62.61 (d, 2J(COP) = 6.9 Hz, CH2OP), 46.07 (CH3N), 40.92 (C4), 16.55 (d, 3J(CCOP) = 6.2 Hz, CH3CH2OP), 16.48 (d, 3J(CCOP) = 5.5 Hz, CH3CH2OP). 31P-NMR (243 MHz, CDCl3): δ = 20.51. Anal. Calcd. for C16H21BrN3O5P: C, 43.07; H, 4.74; N, 9.42. Found: C, 43.09; H, 4.52; N, 9.35.
Diethyl cis-[5-(6-bromo-4-oxo-3,4-dihydroquinazolin-2-yl)-2-methylisoxazolidin-3-yl]phosphonate (cis-11a). Colorless oil. IR (film, cm−1) νmax: 3090, 2959, 2925, 2865, 1661, 1626, 1601, 1461, 1336, 1234, 1054, 969, 834, 575. 1H-NMR (600 MHz, CDCl3): δ = 10.63 (s, 1H, NH), 8.44 (d, 4J = 2.3 Hz, 1H, HC5′), 7.84 (dd, 3J = 8.6 Hz, 4J = 2.3 Hz, 1H, HC7′), 7.54 (d, 3J = 8.6 Hz, 1H, HC8′), 5.07 (dd, 3J(H5–H4α) = 9.2 Hz, 3J(H5–H4β) = 4.3 Hz, 1H, HC5), 4.23–4.12 (m, 4H, 2 × CH2OP), 3.21 (dddd, 3J(H4β–P) = 19.8 Hz, 2J(H4β–H4α) = 10.9 Hz, 3J(H4β–H3) = 6.8 Hz, 3J(H4β–H5) = 4.3 Hz, 1H, HβC4), 3.12 (ddd, 3J(H3–H4β) = 6.8 Hz, 3J(H3–H4α) = 9.6 Hz, 2J(H3–P) = 4.4 Hz 1H, HC3), 3.00 (s, 3H, CH3N), 2.83 (dddd, 3J(H4α–P) = 13.1 Hz, 2J(H4α–H4β) = 10.9 Hz, 3J(H4α–H3) = 9.6 Hz, 3J(H4α–H5) = 9.2 Hz, 1H, HαC4), 1.32 (t, 3J = 7.0 Hz, 3H, CH3CH2OP), 1.27 (t, 3J = 7.1 Hz, 3H, CH3CH2OP). 13C-NMR (151 MHz, CDCl3): δ = 160.33 (C=O), 156.50, 147.35, 137.65, 129.26, 128.96, 123.27, 120.46, 75.58 (d, 3J(CCCP) = 6.6 Hz, C5), 63.58 (d, 1J(CP) = 169.1 Hz, C3), 63.18 (d, 2J(COP) = 6.5 Hz, CH2OP), 63.15 (d, 2J(COP) = 6.4 Hz, CH2OP), 45.58 (d, 3J(CNCP) = 5.8 Hz, CH3N), 37.71 (C4), 16.38 (d, 3J(CCOP) = 5.3 Hz, CH3CH2OP), 16.37 (d, 3J(CCOP) = 5.4 Hz, CH3CH2OP). 31P-NMR (243 MHz, CDCl3): δ = 20.92. Anal. Calcd. for C16H21BrN3O5P: C, 43.07; H, 4.74; N, 9.42. Found: C, 43.12; H, 4.35; N, 9.33.
Diethyl trans-[5-(3-benzyl-6-bromo-4-oxo-3,4-dihydroquinazolin-2-yl)-2-methylisoxazolidin-3-yl]phosphonate (trans-11b). A yellowish oil. IR (film, cm−1) νmax: 3040, 2980, 2926, 2853, 1613, 1568, 1490, 1418, 1353, 1239, 1056, 1025, 835, 822, 699. 1H-NMR (600 MHz, CDCl3): δ = 8.33 (d, 4J = 2.3 Hz, 1H, HC5′), 7.90 (dd, 3J = 8.9 Hz, 4J = 2.3 Hz, 1H, HC7′), 7.83 (d, 3J = 8.9 Hz, 1H, HC8′), 7.55–7.53 (m, 2H), 7.45–7.42 (m, 2H), 7.40–7.37 (m, 1H), 5.66 (s, 2H, N-CH2), 5.25 (dd, 3J(H5–H4β) = 6.8 Hz, 3J(H5–H4α) = 6.4 Hz, 1H, HC5), 4.33–4.19 (m, 4H, 2 × CH2OP), 3.42–3.39 (m, 1H, HC3), 3.07–2.94 (m, 2H, HαC4, HβC4), 3.05 (s, 3H, CH3N), 1.42 (t, 3J = 7.0 Hz, 3H, CH3CH2OP), 1.39 (t, 3J = 6.9 Hz, 3H, CH3CH2OP). 13C-NMR (151 MHz, CDCl3): δ = 166.08 (C=O), 163.35, 149.91, 137.20, 135.69, 129.67, 128.67, 128.65, 128.52, 128.45, 125.97, 120.69, 80.15 (d, 3J(CCCP) = 8.5 Hz, C5), 69.14 (N-CH2), 64.41 (d, 1J(CP) = 168.8 Hz, C3), 63.26 (d, 2J(COP) = 6.5 Hz, CH2OP), 62.42 (d, 2J(COP) = 7.2 Hz, CH2OP), 46.62 (CH3N), 37.90 (C4), 16.58 (d, 3J(CCOP) = 5.6 Hz, CH3CH2OP), 16.52 (d, 3J(CCOP) = 5.6 Hz, CH3CH2OP). 31P-NMR (243 MHz, CDCl3): δ = 22.04. Anal. Calcd. for C23H27BrN3O5P: C, 51.50; H, 5.07; N, 7.83. Found: C, 51.78; H, 5.11; N, 7.70.
Diethyl trans-{5-[6-bromo-3-(2-nitrobenzyl)-4-oxo-3,4-dihydroquinazolin-2-yl]-2-methylisoxazolidin-3-yl}phosphonate (trans-11c). A yellowish oil. IR (film, cm−1) νmax: 3444, 3077, 2979, 2925, 2853, 1690, 1614, 1575, 1527, 1489, 1413, 1355, 1337, 1055, 1024, 835, 791, 730, 574. 1H-NMR (600 MHz, CDCl3): δ = 8.36 (d, 4J = 2.0 Hz, 1H, HC5′), 8.20–8.18 (m, 1H), 7.95 (dd, 3J = 8.9 Hz, 4J = 2.3 Hz, 1H, HC7′), 7.87 (d, 4J = 8.9 Hz, 1H, HC8′), 7.78–7.76 (m, 1H), 7.72–7.69 (m, 1H), 7.57–7.55 (m, 1H), 6.10 (AB, JAB = 14.6 Hz, 1H, N-CH2b), 6.07 (AB, JAB = 14.6 Hz, 1H, N-CH2a), 5.21 (dd, 3J(H5–H4β) = 8.0 Hz, 3J(H5–H4α) = 6.1 Hz, 1H, HC5), 4.31–4.22 (m, 4H, 2 × CH2OP), 3.36–3.33 (m, 1H, HC3), 3.00 (s, 3H, CH3N), 2.99 (dddd, 2J(H4β–P) = 16.6 Hz, 3J(H4β–H4α) = 12.6 Hz, 3J(H4β–H3) = 8.2 Hz, 3J(H4β–H5) = 8.0 Hz, 1H, HβC4), 2.38 (dddd, 2J(H4α–H4β) = 12.6 Hz, 3J(H4α–P) = 10.2 Hz, 3J(H4α–H3) = 8.8 Hz, 3J (H4α–H5) = 6.1 Hz, 1H, HαC4), 1.41 (t, 3J = 7.1 Hz, 3H, CH3CH2OP), 1.39 (t, 3J = 7.1 Hz, 3H, CH3CH2OP). 13C-NMR (151 MHz, CDCl3): δ = 165.56 (C=O), 163.42, 150.09, 147.81, 137.49, 133.80, 132.03, 129.85, 129.14, 129.00, 125.66, 125.15, 121.03, 116.40, 80.08 (d, 3J(CCCP) = 7.8 Hz, C5), 65.64 (N-CH2), 64.30 (d, 1J(CP) = 168.5 Hz, C3), 63.21 (d, 2J(COP) = 6.3 Hz, CH2OP), 62.49 (d, 2J(COP) = 6.8 Hz, CH2OP), 46.61 (d, 3J(CNCP) = 4.2 Hz, CH3N), 37.91 (C4), 16.54 (d, 3J(CCOP) = 5.6 Hz, CH3CH2OP), 16.49 (d, 3J(CCOP) = 5.5 Hz, CH3CH2OP). 31P-NMR (243 MHz, CDCl3): δ = 21.85. Anal. Calcd. for C23H26BrN4O7P: C, 47.52; H, 4.51; N, 9.64. Found: C, 47.22; H, 4.40; N, 9.35.
Diethyl trans-{5-[6-bromo-3-(3-nitrobenzyl)-4-oxo-3,4-dihydroquinazolin-2-yl]-2-methylisoxazolidin-3-yl}phosphonate (trans-11d). A yellowish oil. IR (film, cm−1) νmax: 3078, 2980, 2926, 2854, 1613, 1566, 1531, 1489, 1416, 1348, 1242, 1115, 1053, 1024, 966, 836, 805, 733, 671. 1H-NMR (600 MHz, CDCl3): δ = 8.44–8.42 (m, 1H), 8.32 (d, 4J = 2.0 Hz, 1H, HC5′), 8.24–8.22 (m, 1H), 7.92 (dd, 3J = 8.9 Hz, 4J = 2.0 Hz, 1H, HC7′), 7.90–7.88 (m, 1H), 7.84 (d, 3J = 8.9 Hz, 1H, HC8′), 7.63–7.60 (m, 1H), 5.77 (s, 2H, N-CH2), 5.29 (dd, 3J(H5–H4β) = 8.0 Hz, 3J(H5–H4α) = 6.4 Hz, 1H, HC5), 4.34–4.23 (m, 4H, 2 × CH2OP), 3.44–3.37 (m, 1H, HC3), 3.10–2.94 (m, 2H, HαC4, HβC4), 3.07 (s, 3H, CH3N), 1.43 (t, 3J = 7.1 Hz, 3H, CH3CH2OP), 1.41 (t, 3J = 7.0 Hz, 3H, CH3CH2OP). 13C-NMR (151 MHz, CDCl3): δ = 165.59 (C=O), 163.05, 150.03, 148.49, 137.81, 137.49, 134.31, 129.78, 129.76, 125.74, 123.45, 123.29, 121.03, 116.42, 80.00 (d, 3J(CCCP) = 8.4 Hz, C5), 65.61 (N-CH2), 64.38 (d, 1J(CP) = 168.5 Hz, C3), 63.26 (d, 2J(COP) = 6.5 Hz, CH2OP), 62.50 (d, 2J(COP) = 6.8 Hz, CH2OP), 46.57 (CH3N), 37.77 (C4), 16.56 (d, 3J(CCOP) = 5.6 Hz, CH3CH2OP), 16.50 (d, 3J(CCOP) = 5.9 Hz, CH3CH2OP). 31P-NMR (243 MHz, CDCl3): δ = 21.52. Anal. Calcd. for C23H26BrN4O7P: C, 47.52; H, 4.51; N, 9.64. Found: C, 47.71; H, 4.51; N, 9.44.
Diethyl trans-{5-[6-bromo-3-(4-nitrobenzyl)-4-oxo-3,4-dihydroquinazolin-2-yl]-2-methylisoxazolidin-3-yl}phosphonate (trans-11e). A yellowish oil. IR (film, cm−1) νmax: 3069, 2969, 2925, 2854, 1610, 1571, 1523, 1490, 1343, 1285, 1241, 1114, 1027, 968, 837. 1H-NMR (600 MHz, CDCl3): δ = 8.35 (d, 4J = 2.0 Hz, 1H, HC5′), 1H), 8.30–8.28 (m, 2H), 7.95 (dd, 3J = 8.9 Hz, 4J = 2.0 Hz, 1H, HC7′), 7.86 (d, 3J = 8.9 Hz, 1H, HC8′), 7.72–7.71 (m, 2H), 5.76 (s, 1H, N-CH2), 5.24 (dd, 3J(H5–H4β) = 6.4 Hz, 3J(H5–H4α) = 6.0 Hz, 1H, HC5), 4.32–4.22 (m, 4H, 2 × CH2OP), 3.37–3.33 (m, 1H, HC3), 3.07–2.98 (m, 1H, HβC4), 3.03 (s, 3H, CH3-N), 2.94 (dddd, 3J(H4α–P) = 12.4 Hz, 2J(H4α–H4β) = 12.4 Hz, 3J(H4α–H3) = 9.2 Hz, 3J(H4α–H5) = 6.0 Hz, 1H, HαC4), 1.41 (t, 3J = 7.0 Hz, 3H, CH3CH2OP), 1.39 (t, 3J = 7.0 Hz, 3H, CH3CH2OP). 13C-NMR (151 MHz, CDCl3): δ = 165.57 (C=O), 163.06, 150.04, 147.95, 142.93, 137.55, 129.83, 128.67, 125.69, 123.92, 121.09, 116.42, 79.97 (d, 3J(CCCP) = 8.6 Hz, C5), 67.55 (s, N-CH2), 64.40 (d, 1J(CP) = 168.3 Hz, C3), 63.28 (d, 2J(COP) = 6.5 Hz, CH2OP), 62.46 (d, 2J(COP) = 7.1 Hz, CH2OP), 46.55 (CH3N), 37.77 (C4), 16.56 (d, 3J(CCOP) = 5.7 Hz, CH3CH2OP), 16.50 (d, 3J(CCOP) = 5.8 Hz, CH3CH2OP). 31P-NMR (243 MHz, CDCl3): δ = 21.85. Anal. Calcd. for C23H26BrN4O7P: C, 47.52; H, 4.51; N, 9.64. Found: C, 47.75; H, 4.54; N, 9.39.
Diethyl trans-{5-[6-bromo-3-(2-fluorobenzyl)-4-oxo-3,4-dihydroquinazolin-2-yl]-2-methylisoxazolidin-3-yl}phosphonate (trans-11f). Data presented below were extracted from spectra of a 88:12 mixture of trans-11f and cis-11f. Yellowish oil. IR (film, cm−1) νmax: 3069, 2981, 2929, 2909, 1614, 1567, 1490, 1456, 1353, 1285, 1116, 1025, 964, 869, 760, 691. 1H-NMR (600 MHz, CDCl3): δ = 8.31 (d, 4J = 2.1 Hz, 1H, HC5′), 7.90 (dd, 3J = 8.9 Hz, 4J = 2.1 Hz, 1H, HC7′), 7.84 (d, 3J = 8.9 Hz, 1H, HC8′), 7.58–7.55 (m, 1H), 7.40–7.36 (m, 1H), 7.21–7.18 (m, 1H), 7.16–7.13 (m, 1H), 5.73 (AB, JAB = 12.4 Hz, 1H, N-CH2b), 5.71 (AB, JAB = 12.4 Hz, 1H, N-CH2a), 5.25 (dd, 3J(H5–H4β) = 7.9 Hz, 3J(H5–H4α) = 6.2 Hz, 1H, HC5), 4.33–4.18 (m, 4H, 2 × CH2OP), 3.42–3.39 (m, 1H, C3), 3.05 (s, 3H, CH3-N), 3.03–2.96 (m, 1H, HβC4), 2.96 (dddd, 3J(H4α–P) = 12.5 Hz, 2J(H4α–H4β) = 12.5 Hz, 3J(H4α–H3) = 8.9 Hz, 3J(H4α–H5) = 6.2 Hz, 1H, HαC4), 1.42 (t, 3J = 7.0 Hz, 3H, CH3CH2OP), 1.39 (t, 3J = 7.1 Hz, 3H, CH3CH2OP). 13C-NMR (151 MHz, CDCl3): δ = 165.94 (C=O), 163.31, 161.19 (d, 1J(CF) = 248.8 Hz, C2″), 149.96, 137.25, 130.81 (d, 3J(CCCF) = 3.5 Hz, C4″), 130.52 (d, 3J(CCCF) = 8.0 Hz, C6″), 129.68, 125.94, 124.26 (d, 4J(CCCCF) = 3.4 Hz, C5″), 122.93 (d, 2J(CCF) = 14.5 Hz, C3″), 120.75, 116.65, 115.66 (d, 2J(CCF) = 21.0 Hz, C1″), 80.14 (dd, 3J(CCCP) = 8.1 Hz, C5), 64.42 (d, 1J(CP) = 168.3 Hz, C3), 63.25 (d, 2J(COP) = 6.5 Hz, CH2OP), 63.03 (d, 3J = 4.3 Hz, N-CH2), 62.43 (d, 2J(COP) = 7.2 Hz, CH2OP), 46.59 (CH3N), 37.89 (C4), 16.55 (d, 3J(CCOP) = 5.7 Hz, CH3CH2OP), 16.50 (d, 3J(CCOP) = 5.8 Hz, CH3CH2OP). 31P-NMR (243 MHz, CDCl3): δ = 22.01. Anal. Calcd. for C23H26BrFN3O5P: C, 49.83; H, 4.73; N, 7.58. Found: C, 49.49; H, 4.53; N, 7.71 (obtained on a 88:12 mixture of trans-11f and cis-11f).
Diethyl trans-{5-[6-bromo-3-(3-fluorobenzyl)-4-oxo-3,4-dihydroquinazolin-2-yl]-2-methylisoxazolidin-3-yl}phosphonate (trans-11g). Yellowish oil. IR (film, cm−1) νmax: 3071, 2979, 2926, 2853, 1613, 1572, 1490, 1452, 1415, 1345, 1285, 1255, 1114, 1055, 1025, 966, 836, 789, 749. 1H-NMR (200 MHz, CDCl3): δ = 8.35–8.33 (m, 1H), 7.91–7.89 (m, 1H), 7.84–7.82 (m, 1H), 7.41–7.37 (m, 1H), 7.30–7.28 (m, 1H), 7.26–7.24 (m, 1H), 7.08–7.05 (m, 1H), 5.64 (s, 1H, N-CH2), 5.24 (dd, 3J(H5–H4β) = 7.9 Hz, 3J(H5–H4α) = 6.5 Hz, 1H, HC5), 4.33–4.17 (m, 4H, 2 × CH2OP), 3.39–3.36 (m, 1H, C3), 3.04 (s, 3H, CH3-N), 3.05–2.99 (m, 1H, HβC4), 2.95 (dddd, 3J(H4α–P) = 12.7 Hz, 2J(H4α–H4β) = 12.7 Hz, 3J(H4α–H3) = 9.7 Hz, 3J(H4α–H5) = 6.5 Hz, 1H, HαC4), 1.41 (t, 3J = 7.0 Hz, 3H, CH3CH2OP), 1.39 (t, 3J = 6.9 Hz, 3H, CH3CH2OP). 13C-NMR (151 MHz, CDCl3): δ = 165.84 (C=O), 163.22, 162.91, (d, 1J(CF) = 246.7 Hz, C3′), 149.97, 138.18 (d, 3J(CCCF) = 7.6 Hz, C5′), 137.31, 130.26 (d, J = 7.9 Hz, C1′), 129.72, 125.84, 123.80 (d, 4J(CCCCF) = 2.4 Hz, C6′), 120.83, 116.59, 115.45 (d, 2J(CCF) = 21.0 Hz, C4′), 115.21 (d, 2J(CCF) = 22.0 Hz, C2′), 80.06 (dd, 3J(CCCP) = 8.0 Hz, C5), 68.19 (d, 3J = 1.7 Hz, N-CH2), 64.42 (d, 1J(CP) = 168.3 Hz, C3), 63.24 (d, 2J(COP) = 6.4 Hz, CH2OP), 62.44 (d, 2J(COP) = 7.3 Hz, CH2OP), 46.57 (d, 3J(CNCP) = 3.7 Hz, CH3N), 37.84 (s, C4), 16.55 (d, 3J(CCOP) = 5.6 Hz, CH3CH2OP), 16.49 (d, 3J(CCOP) = 5.9 Hz, CH3CH2OP). 31P-NMR (121.5 MHz, CDCl3): δ = 21.97. Anal. Cald. for C23H26BrFN3O5P: C, 49.83; H, 4.73; N, 7.58. Found: C, 49.49; H, 4.53; N, 7.71.
Diethyl trans{5-[6-bromo-3-(4-fluorobenzyl)-4-oxo-3,4-dihydroquinazolin-2-yl]-2-methylisoxazolidin-3-yl}phosphonate (trans-11h). A yellowish oil. IR (film, cm−1) νmax: 3072, 2981, 2926, 2853, 1611, 1568, 1512, 1490, 1430, 1351, 1285, 1227, 1160, 1114, 1099,1056, 1026, 965, 835.1H-NMR (600 MHz, CDCl3): δ = 8.31 (d, 4J = 2.2 Hz, 1H, HC5′), 7.91 (dd, 3J = 8.9 Hz, 4J = 2.2 Hz, 1H, HC7′), 7.84 (d, 3J = 8.9 Hz, 1H, HC8′), 7.55–7.53 (m, 2H), 7.14–7.11 (m, 2H), 5.63 (s, 1H, N-CH2), 5.26 (dd, 3J(H5–H4β) = 6.4 Hz, 3J(H5–H4α) = 6.4 Hz, 1H, HC5), 4.34–4.22 (m, 4H, 2 × CH2OP), 3.41–3.39 (m, 1H, C3), 3.07–3.00 (m, 1H, HβC4), 3.06 (s, 3H, CH3-N), 2.98 (dddd, 3J(H4α–P) = 13.4 Hz, 2J(H4α–H4β) = 13.4 Hz, 3J(H4α–H3) = 10.4 Hz, 3J(H4α–H5) = 6.4 Hz, 1H, HαC4), 1.42 (t, 3J = 7.0 Hz, 3H, CH3CH2OP), 1.40 (t, 3J = 7.1 Hz, 3H, CH3CH2OP). 13C-NMR (151 MHz, CDCl3): δ = 165.94 (C=O), 163.26, 162.84 (d, 1J(CF) = 247.6 Hz, C4″), 149.94, 137.25, 131.52 (d, 4J(CCCCF) = 3.1 Hz, C1″), 130.49 (d, 3J(CCCF) = 8.0 Hz, C2″, C6″), 129.69, 125.89, 120.75, 116.65, 115.62 (d, 2J(CCF) = 21.9 Hz, C3″, C5″), 80.09 (d, 3J(CCP) = 8.1 Hz, C5), 68.40 (N-CH2), 64.43 (d, 1J(CP) = 168.3 Hz, C3), 63.26 (d, 2J(COP) = 6.5 Hz, CH2OP), 62.45 (d, 2J(COP) = 6.8 Hz, CH2OP), 46.59 (CH3N), 37.87 (C4), 16.56 (d, 3J(CCOP) = 5.8 Hz, CH3CH2OP), 16.46 (d, 3J(CCOP) = 5.3 Hz, CH3CH2OP). 31P-NMR (243 MHz, CDCl3): δ = 22.00. Anal. Calcd. for C23H26BrFN3O5P: C, 49.83; H, 4.73; N, 7.58. Found: C, 50.20; H, 4.62; N, 7.32.
Diethyl trans-{5-[6-bromo-3-(2,4-difluorobenzyl)-4-oxo-3,4-dihydroquinazolin-2-yl]-2-methylisoxazolidin-3-yl}phosphonate (trans-11i). Data presented below were extracted from spectra of a 92:8 mixture of trans-11i and cis-11i. Yellowish oil. IR (film, cm−1) νmax: 3079, 2959, 2924, 2853, 1738, 1689, 1613, 1565, 1509, 1490, 1416, 1351, 1280, 1141, 1100, 961, 836, 798. 1H-NMR (600 MHz, CDCl3): δ = 8.30 (d, 4J = 2.1 Hz, 1H, HC5′), 7.92 (dd, 3J = 8.8 Hz, 4J = 2.1 Hz, 1H, HC7′), 7.85 (d, 3J = 8.8 Hz, 1H, HC8′), 7.59–7.55 (m, 1H), 6.95–6.89 (m, 2H), 5.68 (AB, JAB = 13.3 Hz, 1H, N-CH2b), 5.66 (AB, JAB = 13.3 Hz, 1H, N-CH2a), 5.25 (dd, 3J(H5–H4β) = 7.7 Hz, 3J(H5–H4α) = 6.5 Hz, 1H, HC5), 4.33–4.18 (m, 4H, 2 × CH2OP), 3.41–3.38 (m, 1H, HC3), 3.07–3.01 (m, 1H, HβC4), 3.05 (s, 3H, CH3-N), 2.98 (dddd, 3J(H4α–P) = 12.8 Hz, 2J(H4α–H4β) = 12.8 Hz, 3J(H4α–H3) = 9.7 Hz, 3J(H4α–H5) = 6.5 Hz, 1H, HαC4), 1.41 (t, 3J = 7.0 Hz, 3H, CH3CH2OP), 1.40 (t, 3J = 7.0 Hz, 3H, CH3CH2OP). 13C-NMR (151 MHz, CDCl3): δ = 165.82 (C=O), 163.32 (dd, 1J(CF) = 250.9 Hz, 3J(CCCF) = 12.1 Hz, C2″), 163.20, 161.50 (dd, 1J(CF) = 251.4 Hz, 3J(CCCF) = 12.2 Hz, C4″), 149.87, 137.31, 132.09 (dd, 3J(CCCF) = 9.8 Hz, 3J(CCCF) = 4.9 Hz, C6″), 129.71, 125.87, 120.81, 118.98 (dd, 2J(CCF) = 14.4 Hz, 4J(CCCCF) = 3.4 Hz, C1″), 116.58, 111.51 (dd, 2J(CCF) = 21.1 Hz, 4J(CCCCF) = 3.5 Hz, C5″), 104.21 (dd, 2J(CCF) = 25.3 Hz, 2J(CCF) = 25.4 Hz, C3″), 80.09 (dd, 3J(CCCP) = 7.9 Hz, C5), 64.43 (d, 1J(CP) = 168.3 Hz, C3), 63.26 (d, 2J(COP) = 6.5 Hz, CH2OP), 62.47 (d, 3J(CCCF) = 2.7 Hz, N-CH2), 62.43 (d, 2J(COP) = 6.3 Hz, CH2OP), 46.57 (d, 3J(CNCP) = 4.0 Hz, CH3N), 37.89 (C4), 16.56 (d, 3J(CCOP) = 5.7 Hz, CH3CH2OP), 16.50 (d, 3J(CCOP) = 5.6 Hz, CH3CH2OP). 31P-NMR (243 MHz, CDCl3): δ = 21.98. Anal. Calcd. for C23H25BrF2N3O5P × 0.75 H2O: C, 47.15; H, 4.56; N, 7.17. Found: C, 46.84; H, 4.18; N, 6.96 (obtained on a 92:8 mixture of trans-11i and cis-11i).
Diethyl trans-{5-[6-bromo-3-methyl-4-oxo-3,4-dihydroquinazolin-2-yl]-2-methylisoxazolidin-3-yl}phosphonate (trans-11j). A yellowish oil. IR (film, cm−1) νmax: 3521, 3477, 2976, 2912, 2855, 1687, 1606, 1470, 1308, 1265, 1050, 1023, 972, 849, 574. 1H-NMR (600 MHz, CDCl3): δ = 8.42 (d, 4J = 2.3 Hz, 1H, HC5′), 7.82 (dd, 3J = 8.7 Hz, 4J = 2.3 Hz, 1H, HC7′), 7.56 (d, 3J = 8.7 Hz, 1H, HC8′), 5.18 (dd, 3J(H5–H4β) = 7.6 Hz, 3J(H5–H4α) = 5.7 Hz, 1H, HC5), 4.29–4.23 (m, 4H, 2 × CH2OP), 3.75 (s, 3H, CH3), 3.71 (dddd, 3J(H4α–P) = 12.6 Hz, 2J(H4α–H4β) = 11.3 Hz, 3J(H4α–H3) = 9.1 Hz, 3J(H4α–H5) = 5.7 Hz, 1H, HαC4), 3.35 (ddd, 3J(H3–H4α) = 9.1 Hz, 3J(H3–H4β) = 7.6 Hz, 2J(H3-P) = 2.8 Hz, 1H, HC3), 2.87 (s, 3H, CH3N), 2.80 (dddd, 3J(H4β–P) = 15.3 Hz, 2J(H4β–H4α) = 11.3 Hz, 3J(H4β–H3) = 7.6 Hz, 3J(H4β–H5) = 7.6 Hz, 1H, HβC4), 1.41 (t, 3J = 7.0 Hz, 3H, CH3CH2OP), 1.40 (t, 3J = 7.1 Hz, 3H, CH3CH2OP). 13C-NMR (151 MHz, CDCl3): δ = 161.31 (C=O), 152.70, 145.34, 137.33, 129.44, 129.37, 122.31, 121.02, 76.29 (d, 3J(CCCP) = 7.8 Hz, C5), 64.35 (d, 1J(CP) = 170.1 Hz, C3), 62.89 (d, 2J(COP) = 6.6 Hz, CH2OP), 62.73 (d, 2J(COP) = 7.2 Hz, CH2OP), 47.15 (d, 3J(CNCP) = 6.4 Hz CH3N), 34.38 (C4), 30.97 (CH3), 16.56 (d, 3J(CCOP) = 4.9 Hz, CH3CH2OP), 16.53 (d, 3J(CCOP) = 5.1 Hz, CH3CH2OP). 31P-NMR (243 MHz, CDCl3): δ = 21.93. Anal. Calcd. for C17H23BrN3O5P: C, 44.36; H, 5.04; N, 9.13. Found: C, 44.15; H, 4.75; N, 9.03.
Diethyl trans-{5-[6-bromo-3-ethyl-4-oxo-3,4-dihydroquinazolin-2-yl]-2-methylisoxazolidin-3-yl}phosphonate (trans-11k). Colorless oil. IR (film, cm−1) νmax: 3055, 2981, 2929, 2854, 1687, 1613, 1569, 1493, 1430, 1383, 1285, 1241, 1117, 1056, 1024, 967, 836. 1H-NMR (600 MHz, CDCl3): δ = 8.32 (d, 4J = 2.2 Hz, 1H, HC5′), 7.90 (dd, 3J = 8.8 Hz, 4J = 2.2.Hz, 1H, HC7′), 7.82 (d, 3J = 8.8 Hz, 1H, HC8′), 5.23 (dd, 3J(H5–H4β) = 8.0 Hz, 3J(H5–H4α) = 6.0 Hz, 1H, HC5), 4.68 (q, 3J = 7.2 Hz, 2H, CH3CH2), 4.33–4.21 (m, 4H, 2 × CH2OP), 3.45–3.43 (m, 1H, HC3), 3.07 (s, 3H, CH3N), 3.06–3.01 (m, 1H, HβC4), 2.97 (dddd, 3J(H4α–P) = 12.6 Hz, 2J(H4α–H4β) = 12.6 Hz, 3J(H4α–H3) = 9.4 Hz, 3J(H4α–H5) = 6.0 Hz, 1H, HαC4), 1.55 (t, 3J = 7.2 Hz, 3H, CH3CH2), 1.42 (t, 3J = 7.0 Hz, 3H, CH3CH2OP), 1.39 (t, 3J = 6.8 Hz, 3H, CH3CH2OP). 13C-NMR (151 MHz, CDCl3): δ =166.33 (C=O), 163.63, 149.78, 137.01, 129.63, 126.03, 120.47, 116.78, 80.22 (d, 3J(CCCP) = 7.9 Hz, C5), 64.38 (d, 1J(CP) = 168.0 Hz, C3), 63.28 (d, 2J(COP) = 6.4 Hz, CH2OP), 62.41 (d, 2J(COP) = 6.7 Hz, CH2OP), 47.21 (CH3N), 38.01 (C4), 29.69 (CH3CH2) 16.56 (d, 3J(CCOP) = 5.5 Hz, CH3CH2OP), 16.50 (d, 3J(CCOP) = 6.2 Hz, CH3CH2OP), 14.29 (CH3CH2). 31P-NMR (243 MHz, CDCl3): δ = 22.08. Anal. Calcd. for C18H25BrN3O5P: C, 45.58; H, 5.31; N, 8.86. Found: C, 45.37; H, 5.31; N, 8.54.