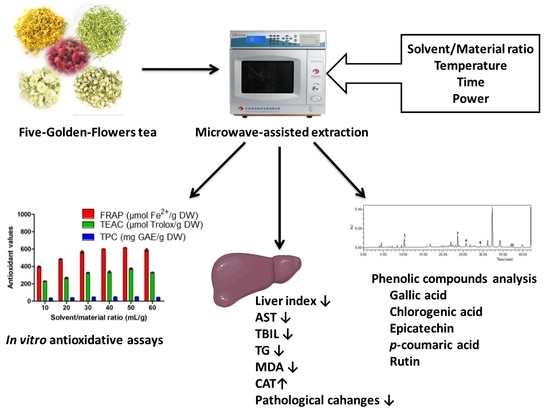
Five-Golden-Flowers Tea: Green Extraction and Hepatoprotective Effect against Oxidative Damage
Abstract
1. Introduction
2. Results and Discussion
2.1. Results of Single-Factor Tests
2.2. Results of Response Surface Methodology Tests
2.2.1. Results of Central Composite Design
2.2.2. Model Fitting
2.2.3. Graphical Analysis
2.2.4. Optimal Extraction Parameters and Responses
2.3. Comparison of Different Extraction Methods
2.4. Correlations between FRAP, TEAC, and TPC
2.5. Results of Animals Study
2.5.1. Hepatoprotective Effects of the Herbal Tea against Alcohol-Induced Injury
2.5.2. The In Vivo Antioxidant Activity of the Herbal Tea
2.5.3. Histopathological Evaluation
2.6. Analysis of Phenolic Compounds by HPLC
3. Materials and Methods
3.1. Sample Preparation
3.2. Chemicals
3.3. Microwave-Assisted Extraction
3.4. Decocting Extraction
3.5. Soxhlet Extraction
3.6. Measurement of Antioxidant Capacities and Total Phenolic Contents
3.7. Optimization of Extraction Parameters
3.8. Animal Study
3.9. HPLC Assay
3.10. Data Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zhang, Y.J.; Gan, R.Y.; Li, S.; Zhou, Y.; Li, A.N.; Xu, D.P.; Li, H.B. Antioxidant phytochemicals for the prevention and treatment of chronic diseases. Molecules 2015, 20, 21138–21156. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.N.; Meng, X.; Li, Y.; Li, S.; Liu, Q.; Tang, G.Y.; Li, H.B. Fruits for prevention and treatment of cardiovascular diseases. Nutrients 2017, 9, 598. [Google Scholar] [CrossRef] [PubMed]
- Fu, L.; Xu, B.T.; Gan, R.Y.; Zhang, Y.; Xu, X.R.; Xia, E.Q.; Li, H.B. Total phenolic contents and antioxidant capacities of herbal and tea infusions. Int. J. Mol. Sci. 2011, 12, 2112–2124. [Google Scholar] [CrossRef] [PubMed]
- Li, A.N.; Li, S.; Li, H.B.; Xu, D.P.; Xu, X.R.; Chen, F. Total phenolic contents and antioxidant capacities of 51 edible and wild flowers. J. Funct. Food. 2014, 6, 319–330. [Google Scholar] [CrossRef]
- Jin, L.; Li, X.; Tian, D.; Fang, X.; Yu, Y.; Zhu, H.; Ge, Y.; Ma, G.; Wang, W.; Xiao, W.; et al. Antioxidant properties and color parameters of herbal teas in China. Ind. Crop. Prod. 2016, 87, 198–209. [Google Scholar] [CrossRef]
- Fu, Y.; Yang, J.; Cunningham, A.B.; Towns, A.M.; Zhang, Y.; Yang, H.; Li, J.; Yang, X. A billion cups: The diversity, traditional uses, safety issues and potential of Chinese herbal teas. J. Ethnopharmacol. 2018, 222, 217–228. [Google Scholar] [CrossRef] [PubMed]
- Fu, L.; Xu, B.T.; Xu, X.R.; Gan, R.Y.; Zhang, Y.; Xia, E.Q.; Li, H.B. Antioxidant capacities and total phenolic contents of 62 fruits. Food Chem. 2011, 129, 345–350. [Google Scholar] [CrossRef]
- Wong, S.; Leong, L.; Williamkoh, J. Antioxidant activities of aqueous extracts of selected plants. Food Chem. 2006, 99, 775–783. [Google Scholar] [CrossRef]
- Oroian, M.; Escriche, I. Antioxidants: Characterization, natural sources, extraction and analysis. Food Res. Int. 2015, 74, 10–36. [Google Scholar] [CrossRef] [PubMed]
- Routray, W.; Orsat, V. Microwave-assisted extraction of flavonoids: A review. Food Bioprocess Technol. 2012, 5, 409–424. [Google Scholar] [CrossRef]
- Altemimi, A.; Choudhary, R.; Watson, D.G.; Lightfoot, D.A. Effects of ultrasonic treatments on the polyphenol and antioxidant content of spinach extracts. Ultrason. Sonochem. 2015, 24, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.J.; Li, Y.; Lin, S.J.; Li, H.B. Green extraction of natural antioxidants from the Sterculia nobilis fruit waste and analysis of phenolic profile. Molecules 2018, 23, 1059. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Xie, M.; Gong, X. Microwave-assisted extraction used for the isolation of total triterpenoid saponins from Ganoderma atrum. J. Food Eng. 2007, 81, 162–170. [Google Scholar] [CrossRef]
- Kala, H.K.; Mehta, R.; Sen, K.K.; Tandey, R.; Mandal, V. Critical analysis of research trends and issues in microwave assisted extraction of phenolics: Have we really done enough. TrAC-Trends Anal. Chem. 2016, 85, 140–152. [Google Scholar] [CrossRef]
- Florez, N.; Conde, E.; Dominguez, H. Microwave assisted water extraction of plant compounds. J. Chem. Technol. Biotechnol. 2015, 90, 590–607. [Google Scholar] [CrossRef]
- Bouras, M.; Chadni, M.; Barba, F.J.; Grimi, N.; Bals, O.; Vorobiev, E. Optimization of microwave-assisted extraction of polyphenols from Quercus bark. Ind. Crop. Prod. 2015, 77, 590–601. [Google Scholar] [CrossRef]
- Dahmoune, F.; Nayak, B.; Moussi, K.; Remini, H.; Madani, K. Optimization of microwave-assisted extraction of polyphenols from Myrtus communis L. leaves. Food Chem. 2015, 166, 585–595. [Google Scholar] [CrossRef] [PubMed]
- Bhuyan, D.J.; Quan, V.V.; Chalmers, A.C.; van Altena, I.A.; Bowyer, M.C.; Scarlett, C.J. Microwave-assisted extraction of Eucalyptus robusta leaf for the optimal yield of total phenolic compounds. Ind. Crop. Prod. 2015, 69, 290–299. [Google Scholar] [CrossRef]
- Li, Y.; Li, S.; Lin, S.J.; Zhang, J.J.; Zhao, C.N.; Li, H.B. Microwave-assisted extraction of natural antioxidants from the exotic Gordonia axillaris fruit: Optimization and identification of phenolic compounds. Molecules 2017, 22, 1481. [Google Scholar] [CrossRef] [PubMed]
- Altemimi, A.; Lightfoot, D.A.; Kinsel, M.; Watson, D.G. Employing response surface methodology for the optimization of ultrasound assisted extraction of lutein and β-Carotene from Spinach. Molecules 2015, 20, 6611–6625. [Google Scholar] [CrossRef] [PubMed]
- Yu, F.; Li, H.; Meng, Y.; Yang, D. Extraction optimization of Angelica sinensis polysaccharides and its antioxidant activity in vivo. Carbohydr. Polym. 2013, 94, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.J.; Zhou, T.; Wang, F.; Zhou, Y.; Li, Y.; Zhang, J.J.; Zheng, J.; Xu, D.P.; Li, H.B. The effects of Syzygium samarangense, Passiflora edulis and Solanum muricatum on alcohol-induced liver injury. Int. J. Mol. Sci. 2016, 17, 1616. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Zhang, N.; Shen, X.; Mei, W.; He, Y.; Ge, W. Preparation of total flavonoids from loquat flower and its protective effect on acute alcohol-induced liver injury in mice. J. Food Drug Anal. 2015, 23, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.; Zhang, Y.J.; Xu, D.P.; Wang, F.; Zhou, Y.; Zheng, J.; Li, Y.; Zhang, J.J.; Li, H.B. Protective effects of lemon juice on alcohol-induced liver injury in mice. Biomed. Res. Int. 2017, 2017, 7463571. [Google Scholar] [CrossRef] [PubMed]
- Granato, D.; Shahidi, F.; Wrolstad, R.; Kilmartin, P.; Melton, L.D.; Hidalgo, F.J.; Miyashita, K.; Camp, J.V.; Alasalvar, C.; Ismail, A.B.; et al. Antioxidant activity, total phenolics and flavonoids contents: Should we ban in vitro screening methods? Food Chem. 2018, 264, 471–475. [Google Scholar] [CrossRef] [PubMed]
- Cederbaum, A.I.; Lu, Y.; Wu, D. Role of oxidative stress in alcohol-induced liver injury. Arch. Toxicol. 2009, 83, 519–548. [Google Scholar] [CrossRef] [PubMed]
- Massey, V.L.; Arteel, G.E. Acute alcohol-induced liver injury. Front. Physiol. 2012. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Li, Y.; Li, S.; Gan, R.Y.; Li, H.B. Natural products for prevention and treatment of chemical-induced liver injuries. Compr. Rev. Food. Sci. Food Saf. 2018, 17, 472–495. [Google Scholar] [CrossRef]
- Li, W.; Qu, X.N.; Han, Y.; Zheng, S.W.; Wang, J.; Wang, Y.P. Ameliorative effects of 5-hydroxymethyl-2-furfural (5-HMF) from Schisandra chinensis on alcoholic liver oxidative injury in mice. Int. J. Mol. Sci. 2015, 16, 2446–2457. [Google Scholar] [CrossRef] [PubMed]
- Huo, X.W.; Liu, C.Q.; Gao, L.; Xu, X.D.; Zhu, N.L.; Cao, L. Hepatoprotective effect of aqueous extract from the seeds of Orychophragmus violaceus against liver injury in mice and HepG2 cells. Int. J. Mol. Sci. 2017, 18, 1197. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Guo, J.; Yuan, J. In vitro antioxidant properties of rutin. LWT-Food Sci. Technol. 2008, 41, 1060–1066. [Google Scholar] [CrossRef]
- Gao, Z.H.; Xu, H.B.; Huang, K.X. Effects of rutin supplementation on antioxidant status and iron, copper, and zinc contents in mouse liver and brain. Biol. Trace Elem. Res. 2002, 88, 271–279. [Google Scholar] [PubMed]
- Kim, H.; Pan, J.H.; Kim, S.H.; Lee, J.H.; Park, J. Chlorogenic acid ameliorates alcohol-induced liver injuries through scavenging reactive oxygen species. Biochimie 2018, 150, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Li, A.N.; Li, S.; Zhang, Y.J.; Xu, X.R.; Chen, Y.M.; Li, H.B. Resources and biological activities of natural polyphenols. Nutrients 2014, 6, 6020–6047. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Luo, Q.; Sun, M.; Corke, H. Antioxidant activity and phenolic compounds of 112 traditional Chinese medicinal plants associated with anticancer. Life Sci. 2004, 74, 2157–2184. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of Five-Golden-Flowers tea and several phenolic standards are available from the authors. |






| Run | S/M Ratio (X1, mL/g) | Temperature (X2, °C) | Time (X3, min) | FRAP (Y1, µmol Fe2+/g DW) | TEAC (Y2, µmol Trolox/g DW) | TPC (Y3, mg GAE/g DW) | |||
|---|---|---|---|---|---|---|---|---|---|
| Actual | Predicted | Actual | Predicted | Actual | Predicted | ||||
| 1 | 33.18 (−1.68) | 60 (0) | 30 (0) | 730.34 | 739.28 | 400.82 | 408.20 | 55.49 | 55.93 |
| 2 | 40 (−1) | 50 (−1) | 40 (1) | 717.07 | 695.95 | 356.78 | 351.29 | 50.95 | 50.97 |
| 3 | 60 (1) | 50 (−1) | 20 (−1) | 718.60 | 707.21 | 384.12 | 382.34 | 55.08 | 55.29 |
| 4 | 60 (1) | 70 (1) | 20 (−1) | 769.67 | 787.98 | 426.78 | 430.07 | 59.66 | 59.55 |
| 5 | 50 (0) | 60 (0) | 13.18 (−1.68) | 726.33 | 723.71 | 395.54 | 398.51 | 56.75 | 56.96 |
| 6 | 50 (0) | 60 (0) | 30 (0) | 837.24 | 840.61 | 459.39 | 461.70 | 63.69 | 64.40 |
| 7 | 50 (0) | 60 (0) | 30 (0) | 843.60 | 840.61 | 445.18 | 461.70 | 64.19 | 64.40 |
| 8 | 50 (0) | 60 (0) | 30 (0) | 860.73 | 840.61 | 466.76 | 461.70 | 65.04 | 64.40 |
| 9 | 50 (0) | 76.82 (1.68) | 30 (0) | 822.17 | 812.19 | 434.81 | 443.16 | 60.41 | 60.85 |
| 10 | 50 (0) | 60 (0) | 46.82 (1.68) | 739.25 | 745.84 | 396.43 | 396.57 | 57.51 | 57.43 |
| 11 | 60 (1) | 50 (−1) | 40 (1) | 716.60 | 720.97 | 376.63 | 386.82 | 56.28 | 56.62 |
| 12 | 50 (0) | 43.18 (−1.68) | 30 (0) | 677.58 | 691.54 | 347.78 | 342.54 | 50.84 | 50.52 |
| 13 | 50 (0) | 60 (0) | 30 (0) | 814.25 | 840.61 | 476.77 | 461.70 | 64.97 | 64.40 |
| 14 | 66.82 (1.68) | 60 (0) | 30 (0) | 800.50 | 795.53 | 442.40 | 438.13 | 60.81 | 60.50 |
| 15 | 60 (1) | 70 (1) | 40 (1) | 839.67 | 834.47 | 446.86 | 440.56 | 60.28 | 60.29 |
| 16 | 40 (−1) | 70 (1) | 20 (−1) | 753.28 | 746.10 | 442.40 | 430.01 | 60.19 | 59.77 |
| 17 | 40 (−1) | 50 (−1) | 20 (−1) | 713.73 | 716.12 | 359.99 | 364.09 | 51.25 | 51.15 |
| 18 | 50 (0) | 60 (0) | 30 (0) | 853.40 | 840.61 | 466.76 | 461.70 | 63.87 | 64.40 |
| 19 | 50 (0) | 60 (0) | 30 (0) | 835.10 | 840.61 | 455.90 | 461.70 | 64.69 | 64.40 |
| 20 | 40 (−1) | 70 (1) | 40 (1) | 750.08 | 758.66 | 423.65 | 423.23 | 59.30 | 59.00 |
| Source | FRAP | TEAC | TPC | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sum of Squares | df | Mean Square | F value | p Value | Sum of Squares | df | Mean Square | F Value | p Value | Sum of Squares | df | Mean Square | F Value | p Value | |
| Model | 61,370.96 | 9 | 6819.00 | 23.92 | <0.0001 | 29,448.79 | 9 | 3272.09 | 29.03 | <0.0001 | 417.00 | 9 | 46.33 | 169.39 | <0.0001 |
| X1 | 3818.96 | 1 | 3818.96 | 13.39 | 0.0044 | 1080.92 | 1 | 1080.92 | 9.59 | 0.0113 | 25.22 | 1 | 25.22 | 92.19 | <0.0001 |
| X2 | 17,571.62 | 1 | 17571.62 | 61.63 | <0.0001 | 12,221.12 | 1 | 12,221.12 | 108.43 | <0.0001 | 128.95 | 1 | 128.95 | 471.43 | <0.0001 |
| X3 | 591.38 | 1 | 591.38 | 2.07 | 0.1804 | 4.54 | 1 | 4.54 | 0.04 | 0.8450 | 0.27 | 1 | 0.27 | 0.97 | 0.3468 |
| X1X2 | 1289.81 | 1 | 1289.81 | 4.52 | 0.0593 | 165.53 | 1 | 165.53 | 1.47 | 0.2534 | 9.48 | 1 | 9.48 | 34.67 | 0.0002 |
| X1X3 | 575.62 | 1 | 575.62 | 2.02 | 0.1858 | 149.21 | 1 | 149.21 | 1.32 | 0.2767 | 1.13 | 1 | 1.13 | 4.14 | 0.0692 |
| X2X3 | 535.63 | 1 | 535.63 | 1.88 | 0.2005 | 18.09 | 1 | 18.09 | 0.16 | 0.6971 | 0.17 | 1 | 0.17 | 0.63 | 0.4473 |
| X12 | 9652.51 | 1 | 9652.51 | 33.85 | 0.0002 | 2675.42 | 1 | 2675.42 | 23.74 | 0.0006 | 69.13 | 1 | 69.13 | 252.72 | <0.0001 |
| X22 | 14,187.47 | 1 | 14,187.47 | 49.76 | <0.0001 | 8540.02 | 1 | 8540.02 | 75.77 | <0.0001 | 136.96 | 1 | 136.96 | 500.73 | <0.0001 |
| X32 | 20,175.95 | 1 | 20,175.95 | 70.76 | <0.0001 | 7416.22 | 1 | 7416.22 | 65.80 | <0.0001 | 93.77 | 1 | 93.77 | 342.80 | <0.0001 |
| Residual | 2851.23 | 10 | 285.12 | 1127.15 | 10 | 112.71 | 2.74 | 10 | 0.27 | ||||||
| Lack of fit | 1537.40 | 5 | 307.48 | 1.17 | 0.4336 | 537.00 | 5 | 107.40 | 0.91 | 0.5400 | 1.09 | 5 | 0.22 | 0.66 | 0.6699 |
| Pure error | 1313.83 | 5 | 262.77 | 590.15 | 5 | 118.03 | 1.65 | 5 | 0.33 | ||||||
| Cor total | 64,222.19 | 19 | 30,575.93 | 19 | 419.73 | 19 | |||||||||
| R2 | 0.9556 | 0.9631 | 0.9935 | ||||||||||||
| Adjusted R2 | 0.9156 | 0.9300 | 0.9876 | ||||||||||||
| Extraction Methods | Solvent | Temperature (°C) | Time | FRAP (μmol Fe2+/g DW) | TEAC (μmol Trolox/g DW) | TPC (mg GAE/g DW) |
|---|---|---|---|---|---|---|
| Decocting method | Distilled water | 65.52 | 30.89 min | 726.16 ± 1.25 | 372.61 ± 1.33 | 55.65 ± 0.39 |
| Soxhlet extraction | 50% ethanol | 95 | 4 h | 847.17 ± 2.36 | 479.15 ± 1.26 | 68.55 ± 0.32 |
| MAE | Distilled water | 65.52 | 30.89 min | 862.90 ± 2.44 | 474.37 ± 1.92 | 65.50 ± 1.26 |
| Group | Liver Index (%) | AST (U/L) | ALT (U/L) | ALP (U/L) | TBIL (μmol/L) | Serum TG (mmol/L) | Liver TG (mmol/gprot) |
|---|---|---|---|---|---|---|---|
| Control | 4.11 ± 0.13 | 115.25 ± 9.99 | 30.10 ± 6.50 | 318.67 ± 39.39 | 1.96 ± 0.40 | 0.88 ± 0.23 | 0.25 ± 0.05 |
| Model | 4.58 ± 0.32 ## | 153.72 ± 22.18 ## | 40.68 ± 7.82 # | 303.33 ± 49.79 | 2.38 ± 0.32 # | 1.28 ± 0.33 # | 0.27 ± 0.04 |
| 200 mg/kg | 4.43 ± 0.15 | 128.62 ± 16.73 * | 32.77 ± 5.63 | 305.67 ± 68.56 | 1.70 ± 0.39 ** | 0.99 ± 0.38 | 0.31 ± 0.04 |
| 400 mg/kg | 4.51 ± 0.21 | 125.50 ± 17.81 * | 38.68 ± 6.58 | 327.83 ± 69.15 | 1.98 ± 0.12 * | 1.01 ± 0.32 | 0.31 ± 0.05 |
| 800 mg/kg | 4.25 ± 0.22 * | 126.92 ± 12.61 * | 35.38 ± 6.10 | 338.33 ± 54.40 | 1.83 ± 0.23 ** | 0.91 ± 0.05 * | 0.30 ± 0.04 |
| Group | MDA (nmol/mgprot) | GSH (µmol/gprot) | SOD (U/mgprot) | CAT (U/mgprot) |
|---|---|---|---|---|
| Control | 1.08 ± 0.42 | 8.39 ± 0.91 | 271.03 ± 15.79 | 62.46 ± 2.74 |
| Model | 1.96 ± 0.70 ## | 5.88 ± 1.01 | 255.54 ± 13.71 # | 56.04 ± 8.33 # |
| 200 mg/kg | 1.00 ± 0.25 ** | 5.99 ±1.09 | 264.78 ± 4.90 | 51.19 ± 2.25 |
| 400 mg/kg | 0.87 ± 0.12 ** | 8.63 ± 0.64 | 256.33 ± 8.78 | 57.49 ± 4.58 |
| 800 mg/kg | 0.82 ± 0.17 ** | 10.05 ± 0.69 | 262.06 ± 3.94 | 62.57 ± 4.18 * |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, C.-N.; Tang, G.-Y.; Liu, Q.; Xu, X.-Y.; Cao, S.-Y.; Gan, R.-Y.; Zhang, K.-Y.; Meng, S.-L.; Li, H.-B. Five-Golden-Flowers Tea: Green Extraction and Hepatoprotective Effect against Oxidative Damage. Molecules 2018, 23, 2216. https://doi.org/10.3390/molecules23092216
Zhao C-N, Tang G-Y, Liu Q, Xu X-Y, Cao S-Y, Gan R-Y, Zhang K-Y, Meng S-L, Li H-B. Five-Golden-Flowers Tea: Green Extraction and Hepatoprotective Effect against Oxidative Damage. Molecules. 2018; 23(9):2216. https://doi.org/10.3390/molecules23092216
Chicago/Turabian StyleZhao, Cai-Ning, Guo-Yi Tang, Qing Liu, Xiao-Yu Xu, Shi-Yu Cao, Ren-You Gan, Ke-Yi Zhang, Shuang-Li Meng, and Hua-Bin Li. 2018. "Five-Golden-Flowers Tea: Green Extraction and Hepatoprotective Effect against Oxidative Damage" Molecules 23, no. 9: 2216. https://doi.org/10.3390/molecules23092216
APA StyleZhao, C.-N., Tang, G.-Y., Liu, Q., Xu, X.-Y., Cao, S.-Y., Gan, R.-Y., Zhang, K.-Y., Meng, S.-L., & Li, H.-B. (2018). Five-Golden-Flowers Tea: Green Extraction and Hepatoprotective Effect against Oxidative Damage. Molecules, 23(9), 2216. https://doi.org/10.3390/molecules23092216