Esterification of Aryl/Alkyl Acids Catalysed by N-bromosuccinimide under Mild Reaction Conditions
Abstract
:1. Introduction
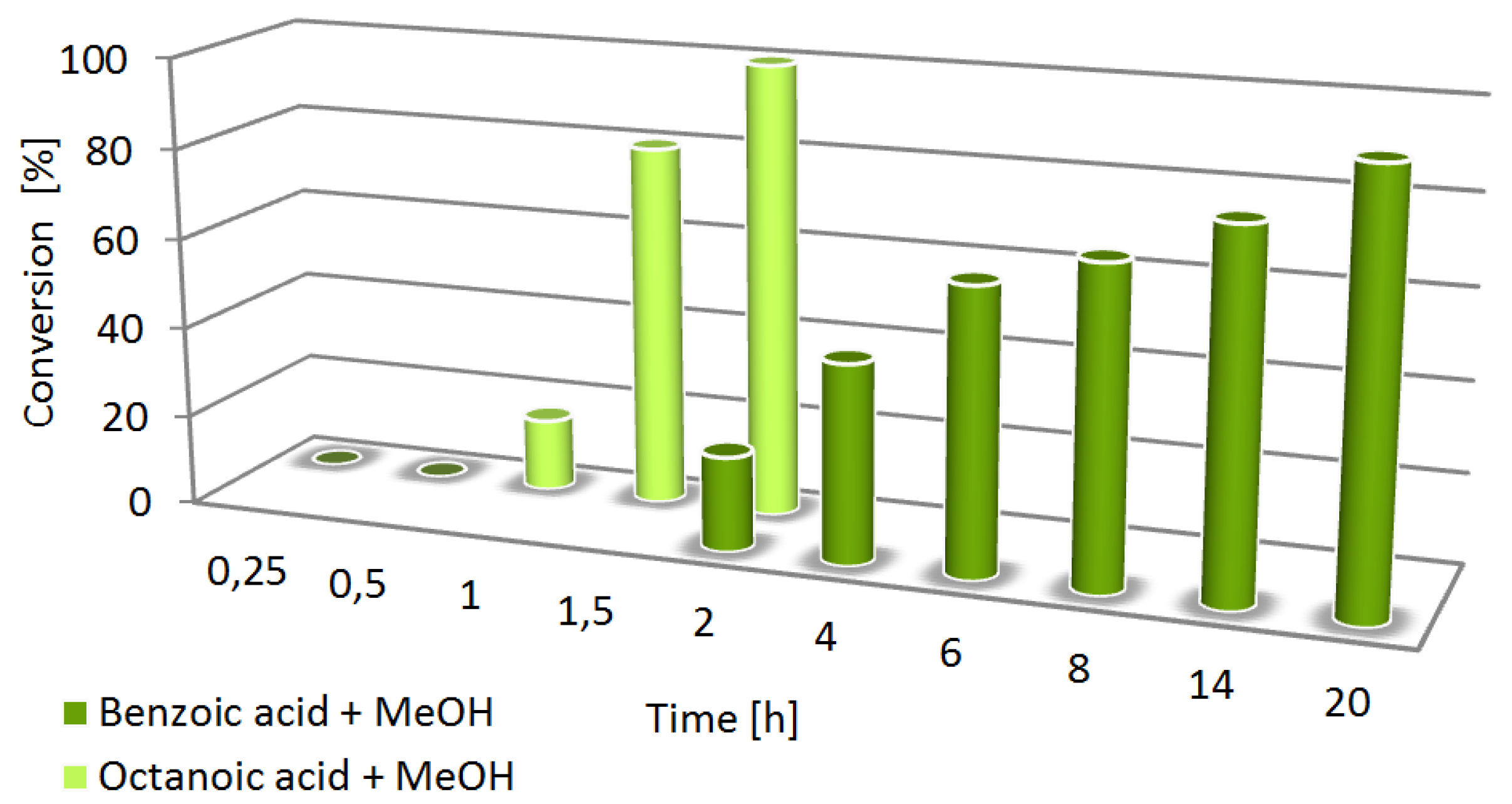
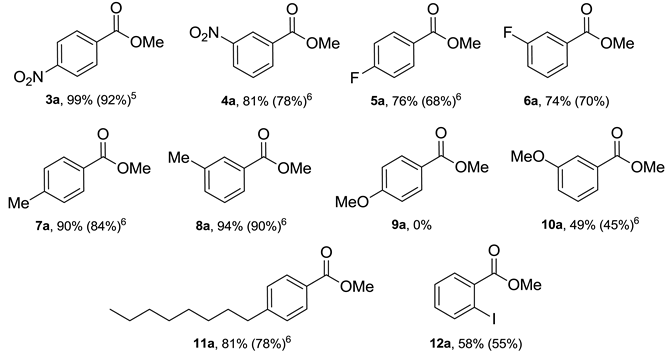
2. Results and Discussion
3. Materials and Methods
3.1. General Information
3.2. Experimental Procedures
3.2.1. General Procedure for the Esterification between Carboxylic Acids and Alcohols
3.2.2. Scale-Up Procedure for Preparation of Methyl Benzoate (1a) and Isolation of Succinimide
- Yield (methyl benzoate): 4.60 g, 85%.
- Yield (succinimide [57]): 272 mg, 98%.
- 1H NMR (300 MHz, CDCl3) δ 10.04 (s, 1H), 2.72 (s, 4H).
- 13C NMR (76 MHz, CDCl3) δ 178.8, 29.4.
3.2.3. Scale-Up Procedure for Preparation of Trimethyl Citrate (16a)
- Yield: 2.55 g, 99%.
3.2.4. Scale-Up Procedure for Preparation of Methyl Stearate (13a)
- Yield: 2.99 g, 100%.
3.2.5. Procedure for Recycling of N-bromosuccinimide (NBS) from Waste Succinimide
3.2.6. Detailed Procedures for the Preparation of Synthesized Compounds
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Otera, J. Esterification. Esterification; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2004; pp. 3–144. ISBN 9783527601844. [Google Scholar]
- Baskar, G.; Aiswarya, R. Trends in catalytic production of biodiesel from various feedstocks. Renew. Sustain. Energy Rev. 2016, 57, 496–504. [Google Scholar] [CrossRef]
- Talebian-Kiakalaieh, A.; Amin, N.A.S.; Mazaheri, H. A review on novel processes of biodiesel production from waste cooking oil. Appl. Energy 2013, 104, 683–710. [Google Scholar] [CrossRef]
- Hosseini-Sarvari, M.; Sodagar, E. Esterification of free fatty acids (Biodiesel) using nano sulfated-titania as catalyst in solvent-free conditions. C R. Chim. 2013, 16, 229–238. [Google Scholar] [CrossRef]
- Mallesham, B.; Govinda Rao, B.; Reddy, B.M. Production of biofuel additives by esterification and acetalization of bioglycerol. C R. Chim. 2016, 19, 1194–1202. [Google Scholar] [CrossRef]
- Tsakos, M.; Schaffert, E.S.; Clement, L.L.; Villadsen, N.L.; Poulsen, T.B. Ester coupling reactions-An enduring challenge in the chemical synthesis of bioactive natural products. Nat. Prod. Rep. 2015, 32, 605–632. [Google Scholar] [CrossRef] [PubMed]
- Larock, R.C.; Dubrovskiy, A.V.; Markina, N.A.; Pletnev, A.A.; Kesharwani, T.; Raminelli, C.; Yao, T.; Zeni, G.; Zhang, L.; Rozhkov, R. Comprehensive Organic Transformations, 4 Volume Set: A Guide to Functional Group Preparations, 3rd ed.; Larock, R.C., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2018; Volume 1, pp. 3753–3764. ISBN 9780470927953. [Google Scholar]
- Ishihara, K. Dehydrative condensation catalyses. Tetrahedron 2009, 65, 1085–1109. [Google Scholar] [CrossRef]
- Sakakura, A.; Koshikari, Y.; Ishihara, K. Open-air and solvent-free ester condensation catalyzed by sulfonic acids. Tetrahedron Lett. 2008, 49, 5017–5020. [Google Scholar] [CrossRef]
- Huang, Y.-B.; Yang, T.; Cai, B.; Chang, X.; Pan, H. Highly efficient metal salt catalyst for the esterification of biomass derived levulinic acid under microwave irradiation. RSC Adv. 2016, 6, 2106–2111. [Google Scholar] [CrossRef]
- Jeschke, J.; Korb, M.; Rüffer, T.; Gäbler, C.; Lang, H. Atom economic ruthenium-catalyzed synthesis of bulky β-Oxo esters. Adv. Synth. Catal. 2015, 357, 4069–4081. [Google Scholar] [CrossRef]
- Da Silva, M.J.; Liberto, N.A.; De Andrade Leles, L.C.; Pereira, U.A. Fe4(SiW12O40)3-catalyzed glycerol acetylation: Synthesis of bioadditives by using highly active Lewis acid catalyst. J. Mol. Catal. A Chem. 2016, 422, 69–83. [Google Scholar] [CrossRef]
- Kato, C.N.; Ogasawara, T.; Kondo, A.; Kato, D. Heterogeneous esterification of fatty acids with methanol catalyzed by Lewis acidic organozirconium complexes with Keggin-type mono-aluminum-substituted polyoxotungstates. Catal. Commun. 2017, 96, 41–45. [Google Scholar] [CrossRef]
- Minakawa, M.; Baek, H.; Yamada, Y.M.A.; Han, J.W.; Uozumi, Y. Direct dehydrative esterification of alcohols and carboxylic acids with a macroporous polymeric acid catalyst. Org. Lett. 2013, 15, 5798–5801. [Google Scholar] [CrossRef] [PubMed]
- Dell’Anna, M.M.; Capodiferro, V.F.; Mali, M.; Mastrorilli, P. Esterification, transesterification and hydrogenation reactions of polyunsaturated compounds catalyzed by a recyclable polymer supported palladium catalyst. J. Organomet. Chem. 2016, 818, 106–114. [Google Scholar] [CrossRef]
- Furuta, A.; Fukuyama, T.; Ryu, I. Efficient flow fischer esterification of carboxylic acids with alcohols using sulfonic acid-functionalized silica as supported catalyst. Bull. Chem. Soc. Jpn. 2017, 90, 607–612. [Google Scholar] [CrossRef]
- Chen, Z.; Wen, Y.; Fu, Y.; Chen, H.; Ye, M.; Luo, G. Graphene oxide: An efficient acid catalyst for the construction of esters from acids and alcohols. Synlett 2017, 28, 981–985. [Google Scholar] [CrossRef]
- Han, X.-X.; Du, H.; Hung, C.-T.; Liu, L.-L.; Wu, P.-H.; Ren, D.-H.; Huang, S.-J.; Liu, S.-B. Syntheses of novel halogen-free Bronsted-Lewis acidic ionic liquid catalysts and their applications for synthesis of methyl caprylate. Green Chem. 2015, 17, 499–508. [Google Scholar] [CrossRef]
- Dong, B.; Song, H.; Zhang, W.; He, A.; Yao, S. Ionic liquids as heterogeneous and homogeneous catalysts for condensation and esterification reactions. Curr. Org. Chem. 2016, 20, 2894–2910. [Google Scholar] [CrossRef]
- Phakhodee, W.; Duangkamol, C.; Pattarawarapan, M. Ph3P-I2 mediated aryl esterification with a mechanistic insight. Tetrahedron Lett. 2016, 57, 2087–2089. [Google Scholar] [CrossRef]
- Yeh, W.K.; Yang, H.C.; McCarthy, J.R. Enzyme Technologies: Metagenomics, Evolution, Biocatalysis and Biosynthesis; Wiley: Hoboken, NJ, USA, 2011; pp. 125–250. ISBN 9781118125038. [Google Scholar]
- Bezbradica, D.; Crovic, M.; Tanaskovic, S.J.; Lukovic, N.; Carevic, M.; Milivojevic, A.; Knezevic-Jugovic, Z. Enzymatic Syntheses of Esters-Green Chemistry for Valuable Food, Fuel and Fine Chemicals. Curr. Org. Chem. 2017, 21, 104–138. [Google Scholar] [CrossRef]
- Gokulakrishnan, N.; Pandurangan, A.; Sinha, P.K. Esterification of acetic acid with propanol isomers under autogeneous pressure: A catalytic activity study of Al-MCM-41 molecular sieves. J. Mol. Catal. A Chem. 2007, 263, 55–61. [Google Scholar] [CrossRef]
- Chung, K.-H.; Park, B.-G. Esterification of oleic acid in soybean oil on zeolite catalysts with different acidity. J. Ind. Eng. Chem. 2009, 15, 388–392. [Google Scholar] [CrossRef]
- Kolvari, E.; Ghorbani-Choghamarani, A.; Salehi, P.; Shirini, F.; Zolfigol, M.A. Application of N-halo reagents in organic synthesis. J. Iran. Chem. Soc. 2007, 4, 126–174. [Google Scholar] [CrossRef]
- Koval, I.V. N-Halo Reagents. N-Halosuccinimides in organic synthesis and in chemistry of natural compounds. Russ. J. Org. Chem. 2002, 38, 301–337. [Google Scholar] [CrossRef]
- Barton, D.; Ollis, W.D. Comprehensive Organic Chemistry: The Synthesis and Reactions of Organic Compounds, 1st ed.; Pergamon Press: Oxford, UK; New York, NY, USA, 1979; Volume 2. [Google Scholar]
- Kadam, S.T.; Kim, S.S. N-Iodosuccinimide (NIS) a novel and effective catalyst for the cyanosilylation of aldehydes under mild reaction conditions. Catal. Commun. 2008, 9, 1342–1345. [Google Scholar] [CrossRef]
- Nagarajappa Giridhar, B.; Pandey Krishna, K.; Shinde Aniket, S.; Vagdevi Hosadu, M. N-Bromosuccinimide (NBS)–An efficient catalyst for acetylation of wood. Holzforschung 2016, 70, 421–427. [Google Scholar] [CrossRef]
- Maleki, B.; Sedigh Ashrafi, S. N-Bromosuccinimide catalyzed three component one-pot efficient synthesis of 2,4,5-Triaryl-1H-imidazoles from aldehyde, ammonium acetate, and 1,2-Diketone or ±-Hydroxyketone. J. Mex. Chem. Soc. 2014, 58, 76–81. [Google Scholar] [CrossRef]
- Karimi, B.; Zamani, A.; Zareyee, D. N-Iodosuccinimide (NIS) as a mild and highly chemoselective catalyst for deprotection of tert-butyldimethylsilyl ethers. Tetrahedron Lett. 2004, 45, 9139–9141. [Google Scholar] [CrossRef]
- Saikia, I.; Borah, A.J.; Phukan, P. Use of bromine and bromo-organic compounds in organic synthesis. Chem. Rev. 2016, 116, 6837–7042. [Google Scholar] [CrossRef] [PubMed]
- Stavber, G.; Iskra, J.; Zupan, M.; Stavber, S. Aerobic oxidative iodination of organic compounds with iodide catalyzed by sodium nitrite. Adv. Synth. Catal. 2008, 350, 2921–2929. [Google Scholar] [CrossRef]
- Stavber, G.; Iskra, J.; Zupan, M.; Stavber, S. Aerobic oxidative iodination of ketones catalysed by sodium nitrite "on water" or in a micelle-based aqueous system. Green Chem. 2009, 11, 1262–1267. [Google Scholar] [CrossRef]
- Stavber, G.; Stavber, S. Towards Greener Fluorine Organic Chemistry: Direct Electrophilic Fluorination of Carbonyl Compounds in Water and Under Solvent-Free Reaction Conditions. Adv. Synth. Catal. 2010, 352, 2838–2846. [Google Scholar] [CrossRef]
- Prebil, R.; Stavber, G.; Stavber, S. Aerobic oxidation of alcohols by using a completely metal-free catalytic system. Eur. J. Org. Chem. 2014, 2014, 395–402. [Google Scholar] [CrossRef]
- Ajvazi, N.; Stavber, S. Direct halogenation of alcohols with halosilanes under catalyst- and organic solvent-free reaction conditions. Tetrahedron Lett. 2016, 57, 2430–2433. [Google Scholar] [CrossRef]
- Ajvazi, N.; Stavber, S. Transformation of tertiary benzyl alcohols into the vicinal halo-substituted derivatives using N-Halosuccinimides. Molecules 2016, 21, 1325. [Google Scholar] [CrossRef] [PubMed]
- Vražič, D.; Jereb, M.; Laali, K.; Stavber, S. Brønsted acidic ionic liquid accelerated halogenation of organic compounds with N-Halosuccinimides (NXS). Molecules 2013, 18, 74–96. [Google Scholar] [CrossRef] [PubMed]
- Podgoršek, A.; Stavber, S.; Zupan, M.; Iskra, J. Environmentally benign electrophilic and radical bromination ‘on water’: H2O2–HBr system versus N-bromosuccinimide. Tetrahedron 2009, 65, 4429–4439. [Google Scholar] [CrossRef]
- Jereb, M.; Zupan, M.; Stavber, S. Visible-light-promoted wohl–ziegler functionalization of organic molecules with N-Bromosuccinimide under solvent-free reaction conditions. Helv. Chim. Acta 2009, 92, 555–566. [Google Scholar] [CrossRef]
- Pravst, I.; Zupan, M.; Stavber, S. Halogenation of ketones with N-halosuccinimides under solvent-free reaction conditions. Tetrahedron 2008, 64, 5191–5199. [Google Scholar] [CrossRef]
- Pravst, I.; Zupan, M.; Stavber, S. Directed regioselectivity of bromination of ketones with NBS: Solvent-free conditions versus water. Tetrahedron Lett. 2006, 47, 4707–4710. [Google Scholar] [CrossRef]
- Podgoršek, A.; Stavber, S.; Zupan, M.; Iskra, J. Visible light induced ‘on water’ benzylic bromination with N-bromosuccinimide. Tetrahedron Lett. 2006, 47, 1097–1099. [Google Scholar] [CrossRef]
- Pravst, I.; Zupan, M.; Stavber, S. Solvent-free bromination of 1,3-diketones and β-keto esters with NBS. Green Chem. 2006, 8, 1001–1005. [Google Scholar] [CrossRef]
- Bandgar, B.P.; Uppalla, L.S.; Sadavarte, V.S. Chemoselective transesterification of β-Keto esters under neutral conditions using NBS as a catalyst. Synlett 2001, 2001, 1715–1718. [Google Scholar] [CrossRef]
- Karimi, B.; Seradj, H. N-Bromosuccinimide (NBS), a novel and highly effective catalyst for acetylation of alcohols under mild reaction conditions. Synlett 2001, 2001, 519–520. [Google Scholar] [CrossRef]
- Sucheta, K.; Reddy, G.S.R.; Ravi, D.; Rama Rao, N. A novel general route to the synthesis of carboxylic acid esters and thiolesters. Tetrahedron Lett. 1994, 35, 4415–4416. [Google Scholar] [CrossRef]
- Ramalinga, K.; Vijayalakshmi, P.; Kaimal, T.N.B. A mild and efficient method for esterification and transesterification catalyzed by iodine. Tetrahedron Lett. 2002, 43, 879–882. [Google Scholar] [CrossRef]
- Jereb, M.; Vražič, D.; Zupan, M. Dual behavior of alcohols in iodine-catalyzed esterification under solvent-free reaction conditions. Tetrahedron Lett. 2009, 50, 2347–2352. [Google Scholar] [CrossRef]
- Jereb, M.; Vražič, D.; Zupan, M. Iodine-catalyzed transformation of molecules containing oxygen functional groups. Tetrahedron 2011, 67, 1355–1387. [Google Scholar] [CrossRef]
- Vairamani, M.; Rao, G.K.V. Use of bromine in methanol - preparation of methyl-esters. Indian J. Chem. Sect B 1985, 24, 691. [Google Scholar]
- Bowman, P.T.; Ko, E.I.; Sides, P.J. A potential hazard in preparing bromine-methanol solutions. J. Electrochem. Soc. 1990, 137, 1309–1311. [Google Scholar] [CrossRef]
- Virtanen, E.; Kolehmainen, E. Use of bile acids in pharmacological and supramolecular applications. Eur. J. Org. Chem. 2004, 2004, 3385–3399. [Google Scholar] [CrossRef]
- Cravotto, G.; Binello, A.; Boffa, L.; Rosati, O.; Boccalini, M.; Chimichi, S. Regio- and stereoselective reductions of dehydrocholic acid. Steroids 2006, 71, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Bose, A.; Mal, P. Electrophilic aryl-halogenation using N-halosuccinimides under ball-milling. Tetrahedron Lett. 2014, 55, 2154–2156. [Google Scholar] [CrossRef]
- Annese, C.; D'Accolti, L.; Fusco, C.; Licini, G.; Zonta, C. Heterolytic (2 e) vs Homolytic (1 e) Oxidation reactivity: N−H versus C−H switch in the oxidation of lactams by Dioxirans. Chem. Eur. J. 2017, 23, 259–262. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.V.; Lyons, D.J.M. A novel aromatic carbocation-based coupling reagent for esterification and amidation reactions. Chem. Commun. 2015, 51, 3131–3134. [Google Scholar] [CrossRef] [PubMed]
- Shen, G.; Zhao, L.; Liu, W.; Huang, X.; Song, H.; Zhang, T. Convenient, metal-free ipso-nitration of arylboronic acids using nitric acid and trifluoroacetic acid. Synth. Commun. 2017, 47, 10–14. [Google Scholar] [CrossRef]
- Jia, J.; Jiang, Q.; Zhao, A.; Xu, B.; Liu, Q.; Luo, W.-P.; Guo, C.-C. Copper-catalyzed O-methylation of carboxylic acids using DMSO as a methyl source. Synthesis 2016, 48, 421–428. [Google Scholar] [CrossRef]
- Powell, A.B.; Stahl, S.S. Aerobic Oxidation of diverse primary alcohols to methyl esters with a readily accessible heterogeneous Pd/Bi/Te catalyst. Org. Lett. 2013, 15, 5072–5075. [Google Scholar] [CrossRef] [PubMed]
- Cheung, C.W.; Buchwald, S.L. Mild and General palladium-catalyzed synthesis of methyl aryl ethers enabled by the use of a palladacycle precatalyst. Org. Lett. 2013, 15, 3998–4001. [Google Scholar] [CrossRef] [PubMed]
- Stephens, M.D.; Yodsanit, N.; Melander, C. Potentiation of the fosmidomycin analogue FR 900098 with substituted 2-oxazolines against Francisella novicida. MedChemComm 2016, 7, 1952–1956. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Offermann, D.A.; McKendrick, J.E.; Sejberg, J.J.P.; Mo, B.; Holdom, M.D.; Helm, B.A.; Leatherbarrow, R.J.; Beavil, A.J.; Sutton, B.J.; Spivey, A.C. Synthesis and incorporation into cyclic peptides of tolan amino acids and their hydrogenated congeners: construction of an array of A–B-loop mimetics of the Cε3 domain of human IgE. J. Org. Chem. 2012, 77, 3197–3214. [Google Scholar] [CrossRef] [PubMed]
- Strazzolini, P.; Gambi, A.G.; Giumanini, A.; Vancik, H. The reaction between ethanedioyl (oxalyl) dihalides and Ag2C2O4: A route to Staudinger's elusive ethanedioic (oxalic) acid anhydride. J. Chem. Soc. Perkin Trans. 1998, 1, 2553–2558. [Google Scholar] [CrossRef]
- Sun, H.-B.; Hua, R.; Yin, Y. ZrOCl2·8H2O: An efficient, cheap and reusable catalyst for the esterification of acrylic acid and other carboxylic acids with equimolar amounts of alcohols. Molecules 2006, 11, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Maslov, M.A.; Morozova, N.G.; Solomatina, T.V.; Shaforostova, N.G.; Serebrennikova, G.A. Synthesis of amino analogues of cholic acid. Russ. J. Bioorg. Chem. 2011, 37, 507–515. [Google Scholar] [CrossRef]
- Rohacova, J.; Marin, M.L.; Martinez-Romero, A.; O'Connor, J.-E.; Gomez-Lechon, M.J.; Donato, M.T.; Castell, J.V.; Miranda, M.A. Synthesis of new, UV-photoactive dansyl derivatives for flow cytometric studies on bile acid uptake. Org. Biomol. Chem. 2009, 7, 4973–4980. [Google Scholar] [CrossRef] [PubMed]
- Hutchby, M.; Houlden, C.E.; Haddow, M.F.; Tyler, S.N.G.; Lloyd-Jones, G.C.; Booker-Milburn, K.I. Switching pathways: room-temperature neutral solvolysis and substitution of amides. Angew. Chem. Int. Ed. 2012, 51, 548–551. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Zatolochnaya, O.V.; Wang, X.-J.; Rodríguez, S.; Qu, B.; Desrosiers, J.-N.; Mangunuru, H.P.R.; Biswas, S.; Rivalti, D.; Karyakarte, S.D.; Sieber, J.D.; Grinberg, N.; Wu, L.; Lee, H.; Haddad, N.; Fandrick, D.R.; Yee, N.K.; Song, J.J.; Senanayake, C.H. BABIPhos family of biaryl dihydrobenzooxaphosphole ligands for asymmetric hydrogenation. Org. Lett. 2018, 20, 1725–1729. [Google Scholar] [CrossRef] [PubMed]
- Dethe, D.H.; Erande, R.D.; Ranjan, A. Biomimetic total syntheses of borreverine and flinderole alkaloids. J. Org. Chem. 2013, 78, 10106–10120. [Google Scholar] [CrossRef] [PubMed]
- Lai, L.; Li, A.N.; Zhou, J.; Guo, Y.; Lin, L.; Chen, W.; Wang, R. Mg(OMe)2 promoted allylic isomerization of γ-hydroxy-α,β-alkenoic esters to synthesize γ-ketone esters. Org. Biomol. Chem. 2017, 15, 2185–2190. [Google Scholar] [CrossRef] [PubMed]
- Suwada, M.; Fukuhara, T.; Hara, S. Selective mono-fluorination of diols via a cyclic acetal of N,N-diethyl-4-methoxybenzamide. J. Fluorine Chem. 2007, 128, 1121–1125. [Google Scholar] [CrossRef]
- Jiang, Q.; Zhao, A.; Xu, B.; Jia, J.; Liu, X.; Guo, C. PIFA-Mediated Esterification Reaction of Alkynes with Alcohols via Oxidative Cleavage of Carbon Triple Bonds. J. Org. Chem. 2014, 79, 2709–2715. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, S.; Fujita, T.; Sakamoto, M.; Kuramochi, T.; Kitazume, T. Reactions of monoesters of ethylene glycol with N,N-diethyl-1,1,2,3,3,3-hexafluoropropylamine. J. Fluorine Chem. 1987, 36, 361–372. [Google Scholar] [CrossRef]
- Umeda, R.; Nishimura, T.; Kaiba, K.; Tanaka, T.; Takahashi, Y.; Nishiyama, Y. Rhenium complex-catalyzed acylative cleavage of ethers with acyl chlorides. Tetrahedron 2011, 67, 7217–7221. [Google Scholar] [CrossRef]
- Whittaker, A.M.; Dong, V.M. Nickel-catalyzed dehydrogenative cross-coupling: direct transformation of aldehydes into esters and amides. Angew. Chem. Int. Ed. 2015, 54, 1312–1315. [Google Scholar] [CrossRef] [PubMed]
- Gianetti, T.L.; Annen, S.P.; Santiso-Quinones, G.; Reiher, M.; Driess, M.; Grützmacher, H. Nitrous Oxide as a Hydrogen Acceptor for the Dehydrogenative Coupling of Alcohols. Angew. Chem. Int. Ed. 2016, 55, 1854–1858. [Google Scholar] [CrossRef] [PubMed]
- Kaul, S.; Kumar, A.; Sain, B.; Gupta, A.K. A simple and convenient one-pot synthesis of fatty acid esters from hindered alcohols using N,N-dimethylchloro-sulfitemethaniminium chloride as dehydrating agent. Synth. Commun. 2002, 32, 2885–2891. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |


| Entry | NXS | Loading [mol%] | Temperature [°C] | Conversion of Benzoic Acid to Methyl Benzoate [%] 2 |
|---|---|---|---|---|
| 1 | / | / | 70 | 0 |
| 2 | NCS | 15 | 70 | 17 |
| 3 | NBS | 15 | 70 | 94 |
| 4 | NIS | 15 | 70 | 30 |
| 5 | NBS | 10 | 70 | 94 |
| 6 | NBS | 7 | 70 | 94 |
| 7 | NBS | 3 | 70 | 76 |
| 8 3 | NBS | 7 | 70 | <10 |
| 9 | NBS | 7 | 30 | 0 |
| 10 | NBS | 7 | 50 | 75 |
| 11 | NBS | 7 | 100 | 94 |
| 12 | HCl | 7 | 70 | 93 |
| 13 | HBr | 7 | 70 | 84 |
| 14 | HI | 7 | 70 | 68 |
| 15 | Br2 | 7 | 70 | 94 |

| R2OH | R1 = Ph | R1 = Heptyl |
|---|---|---|
 | ||

| Methyl Benzoates |
 |
 |
| Methyl Alkyl Esters and Methyl Esters of Cholic Acid Derivatives |
 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Čebular, K.; Božić, B.Đ.; Stavber, S. Esterification of Aryl/Alkyl Acids Catalysed by N-bromosuccinimide under Mild Reaction Conditions. Molecules 2018, 23, 2235. https://doi.org/10.3390/molecules23092235
Čebular K, Božić BĐ, Stavber S. Esterification of Aryl/Alkyl Acids Catalysed by N-bromosuccinimide under Mild Reaction Conditions. Molecules. 2018; 23(9):2235. https://doi.org/10.3390/molecules23092235
Chicago/Turabian StyleČebular, Klara, Bojan Đ. Božić, and Stojan Stavber. 2018. "Esterification of Aryl/Alkyl Acids Catalysed by N-bromosuccinimide under Mild Reaction Conditions" Molecules 23, no. 9: 2235. https://doi.org/10.3390/molecules23092235







