Alkene Difunctionalization Using Hypervalent Iodine Reagents: Progress and Developments in the Past Ten Years
Abstract
1. Introduction
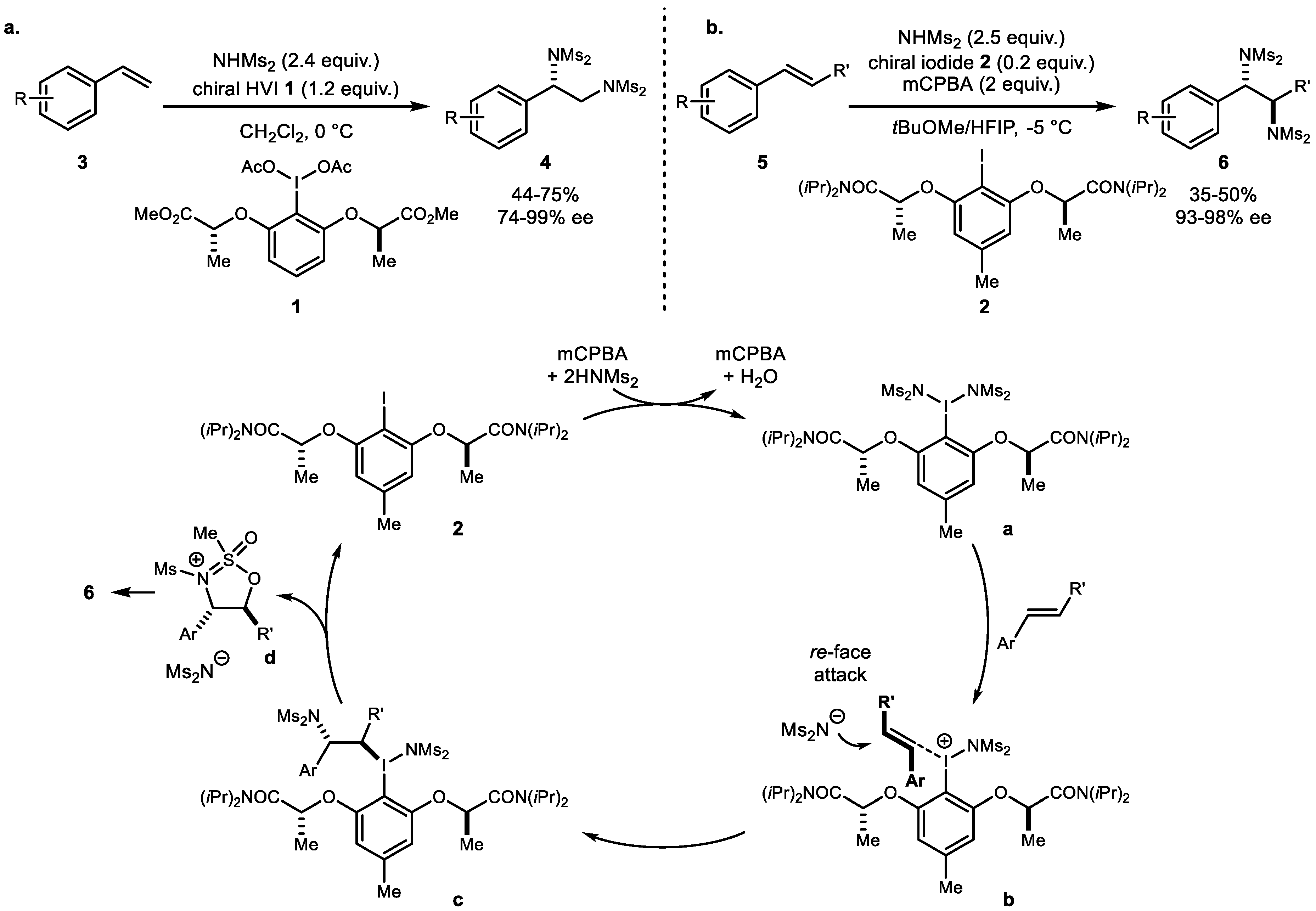
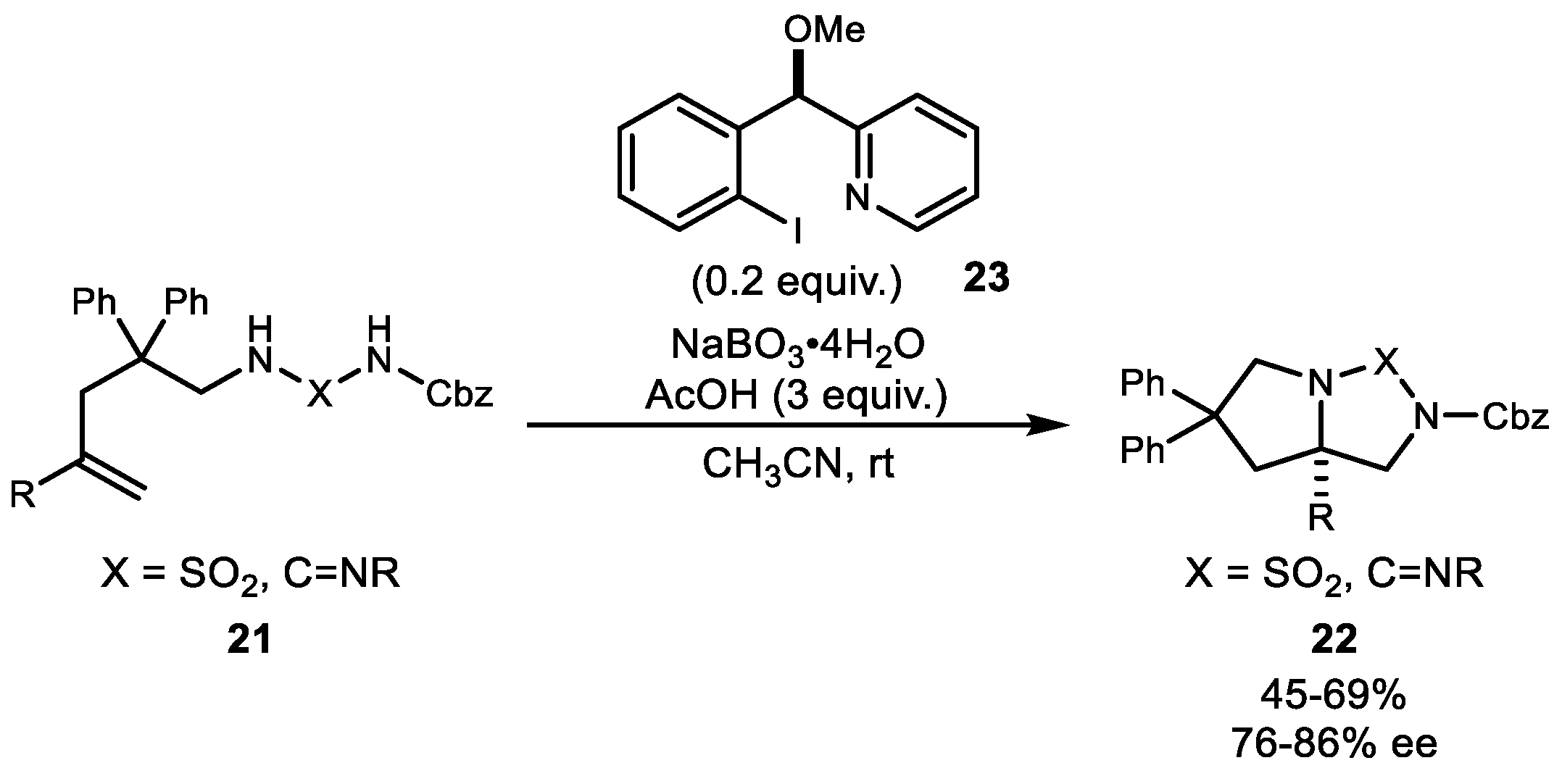
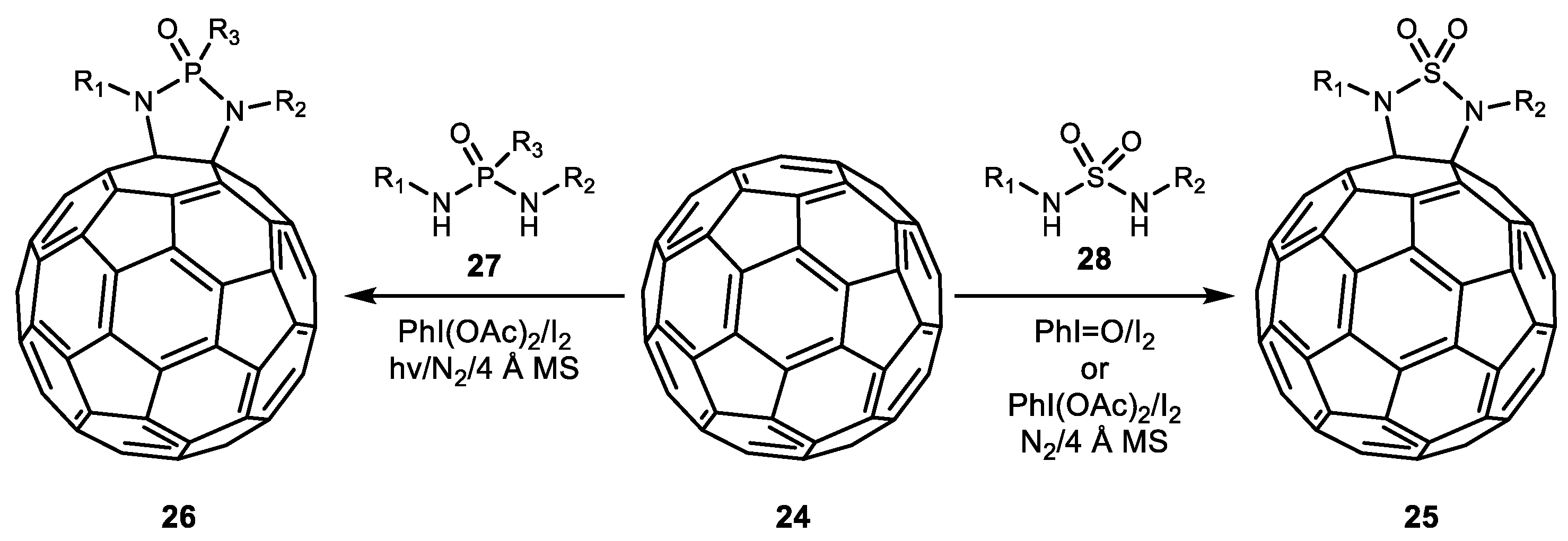
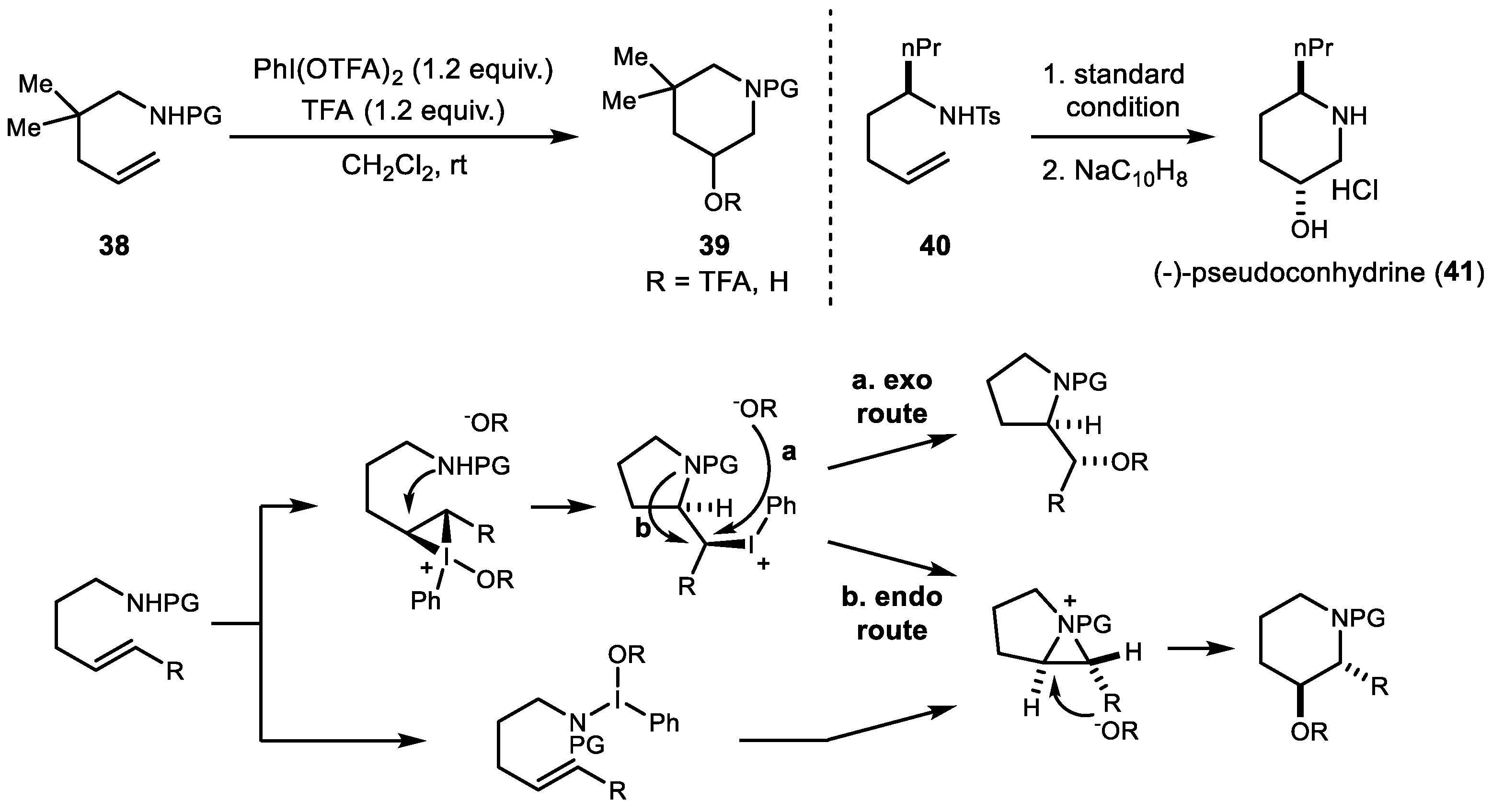
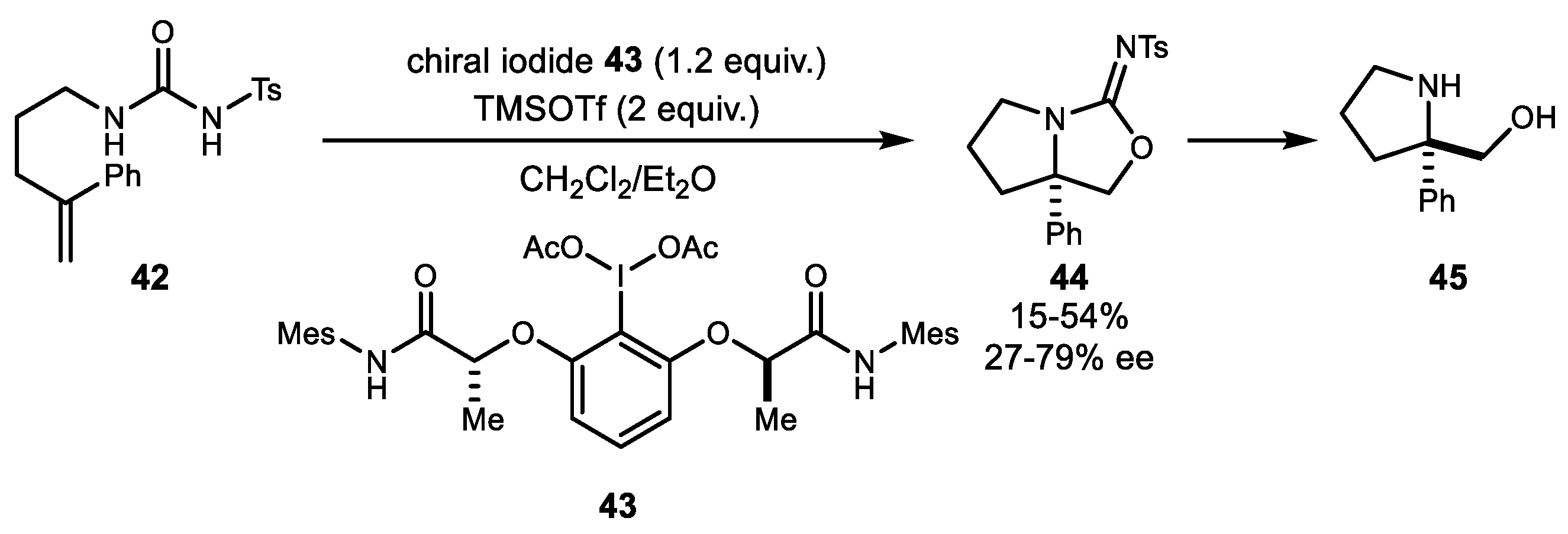
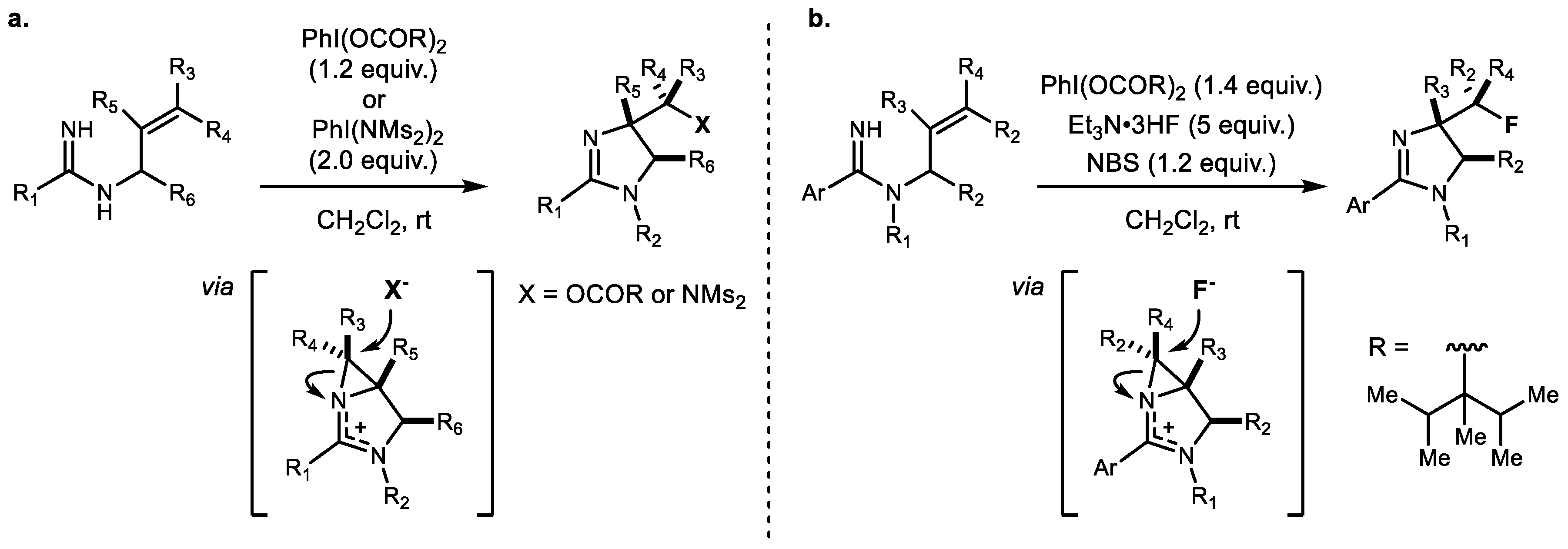
2. Alkene Diamination
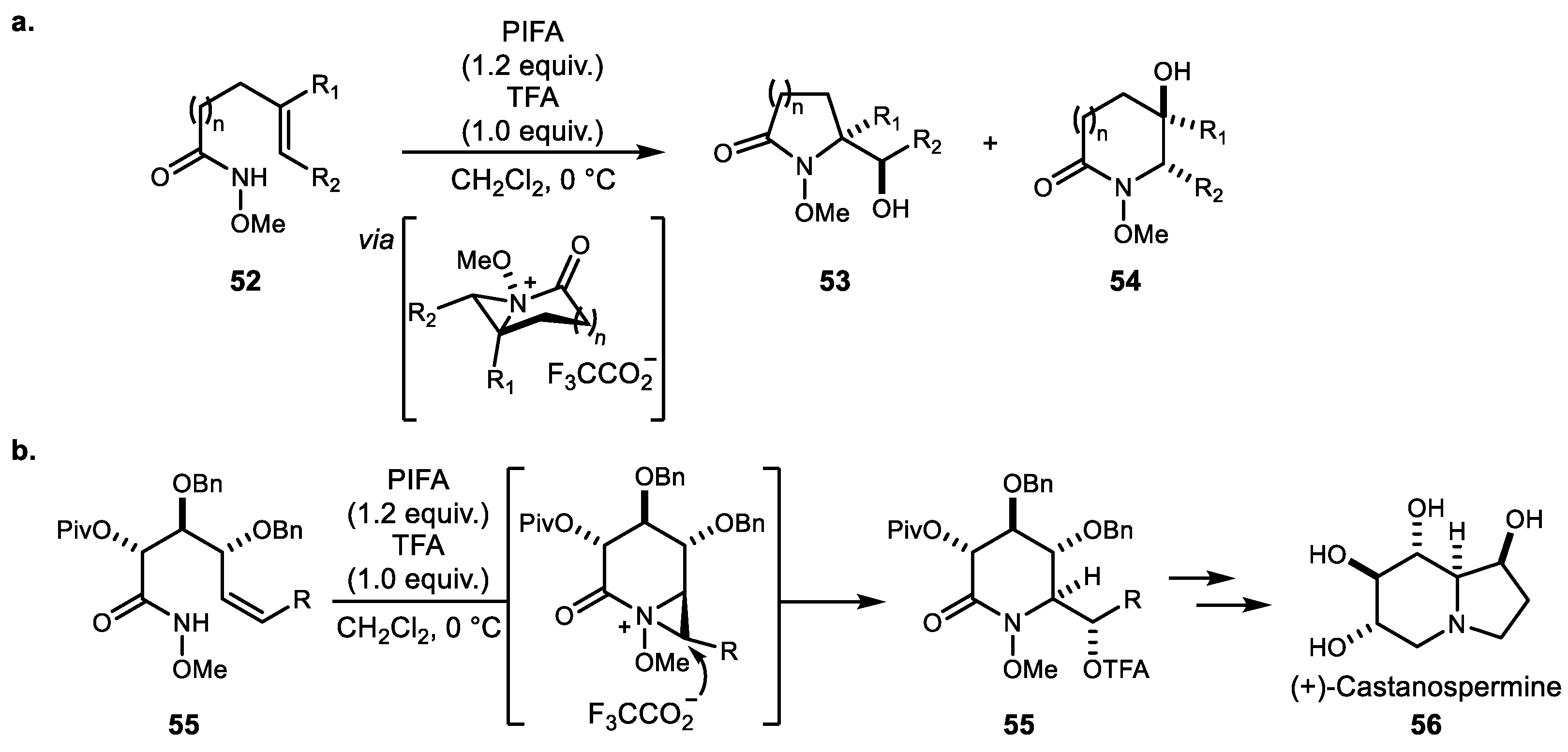
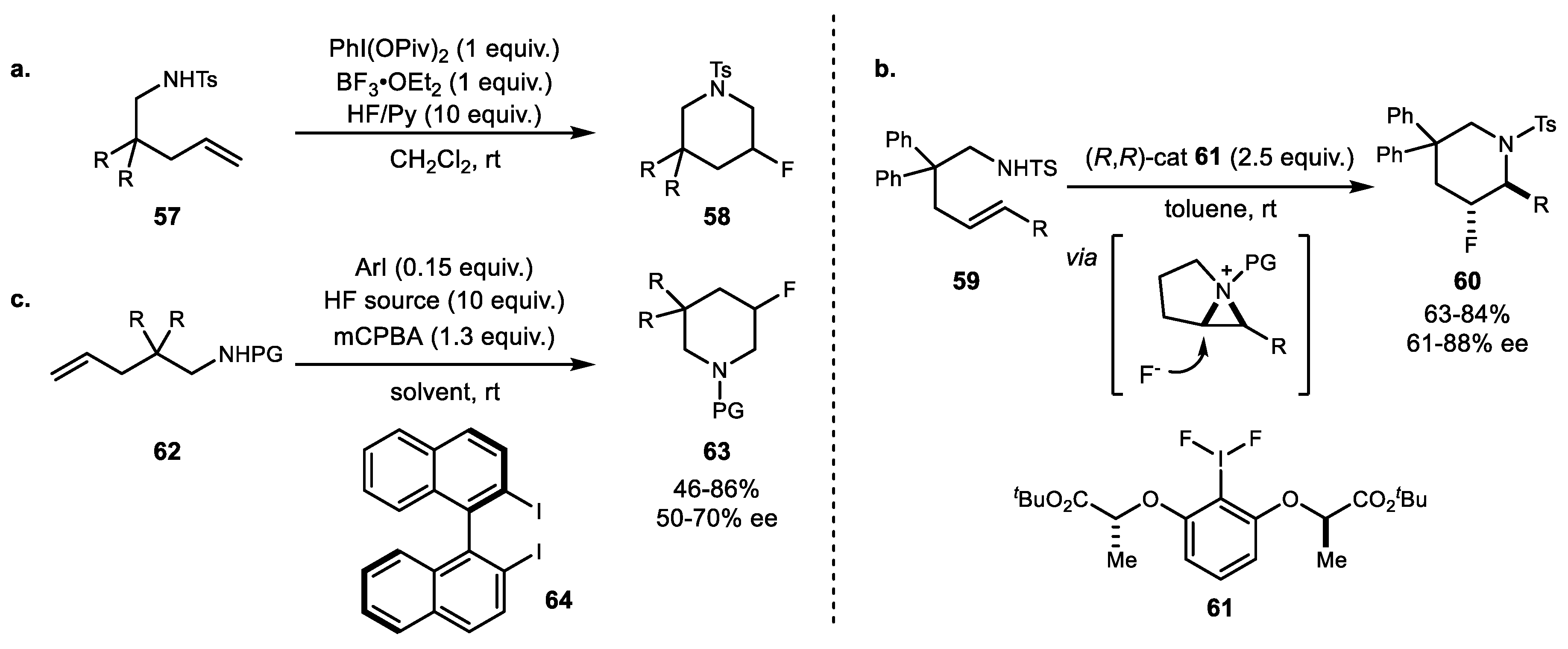
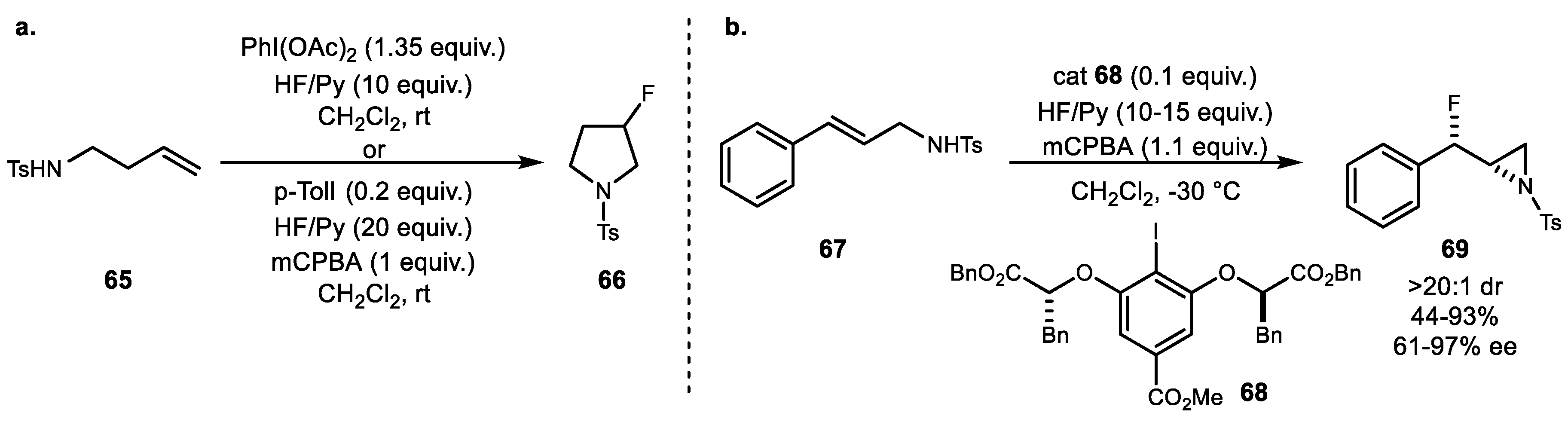
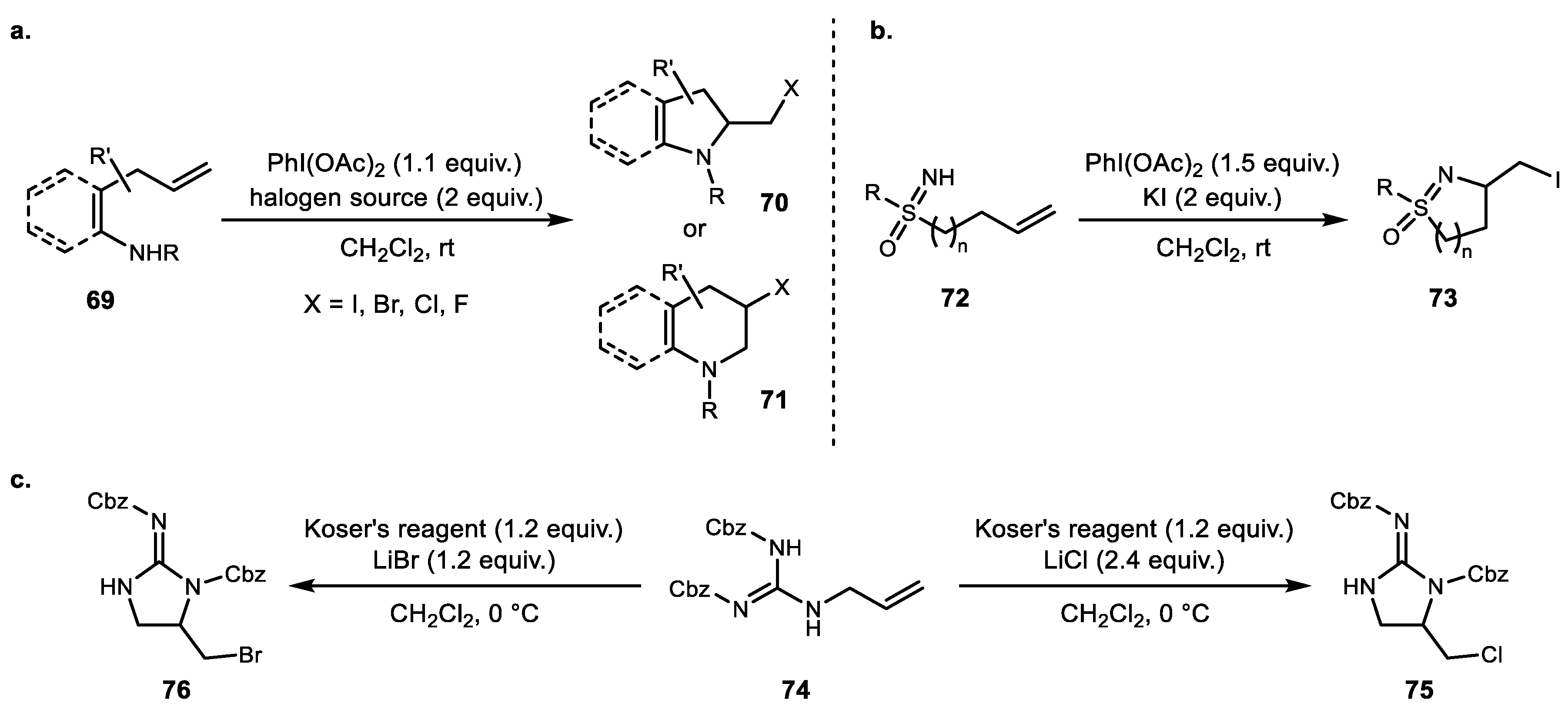
3. Alkene Aminofunctionalization
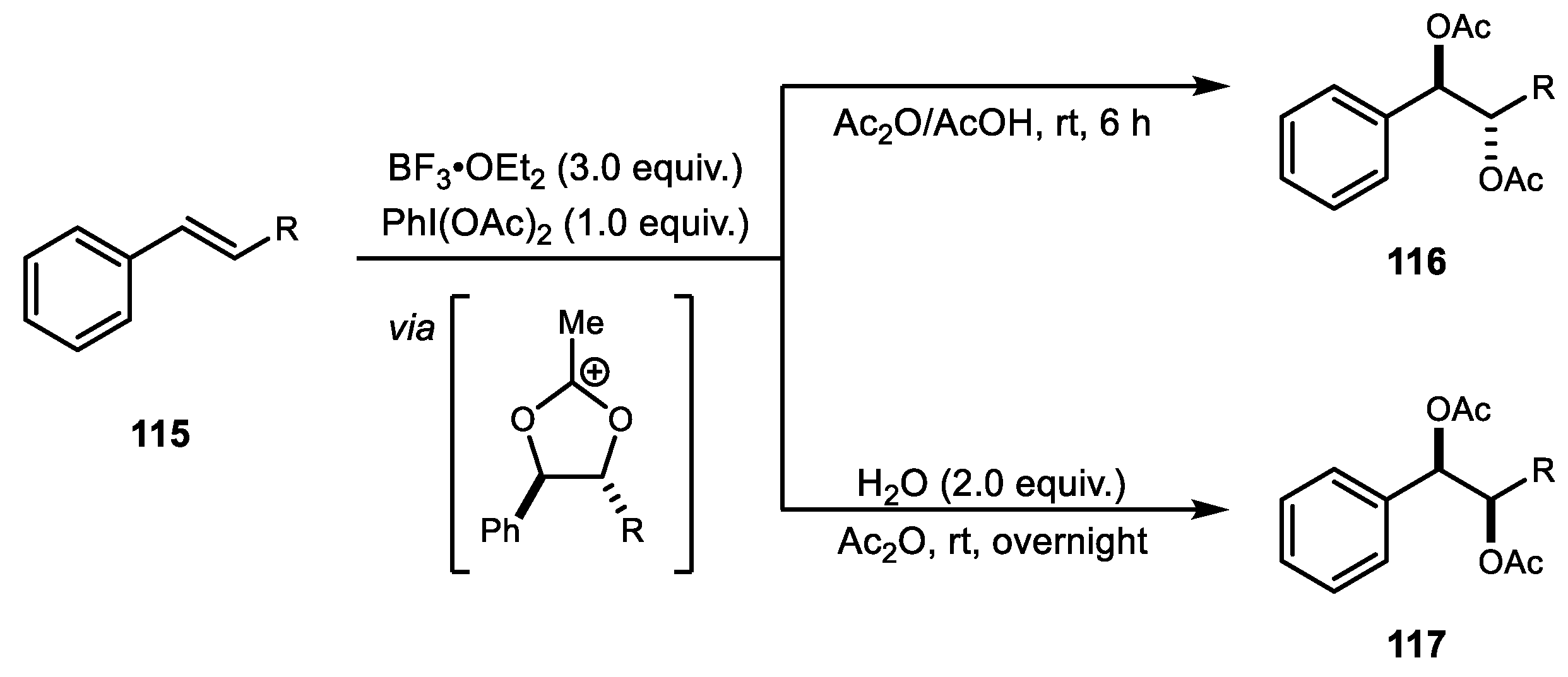
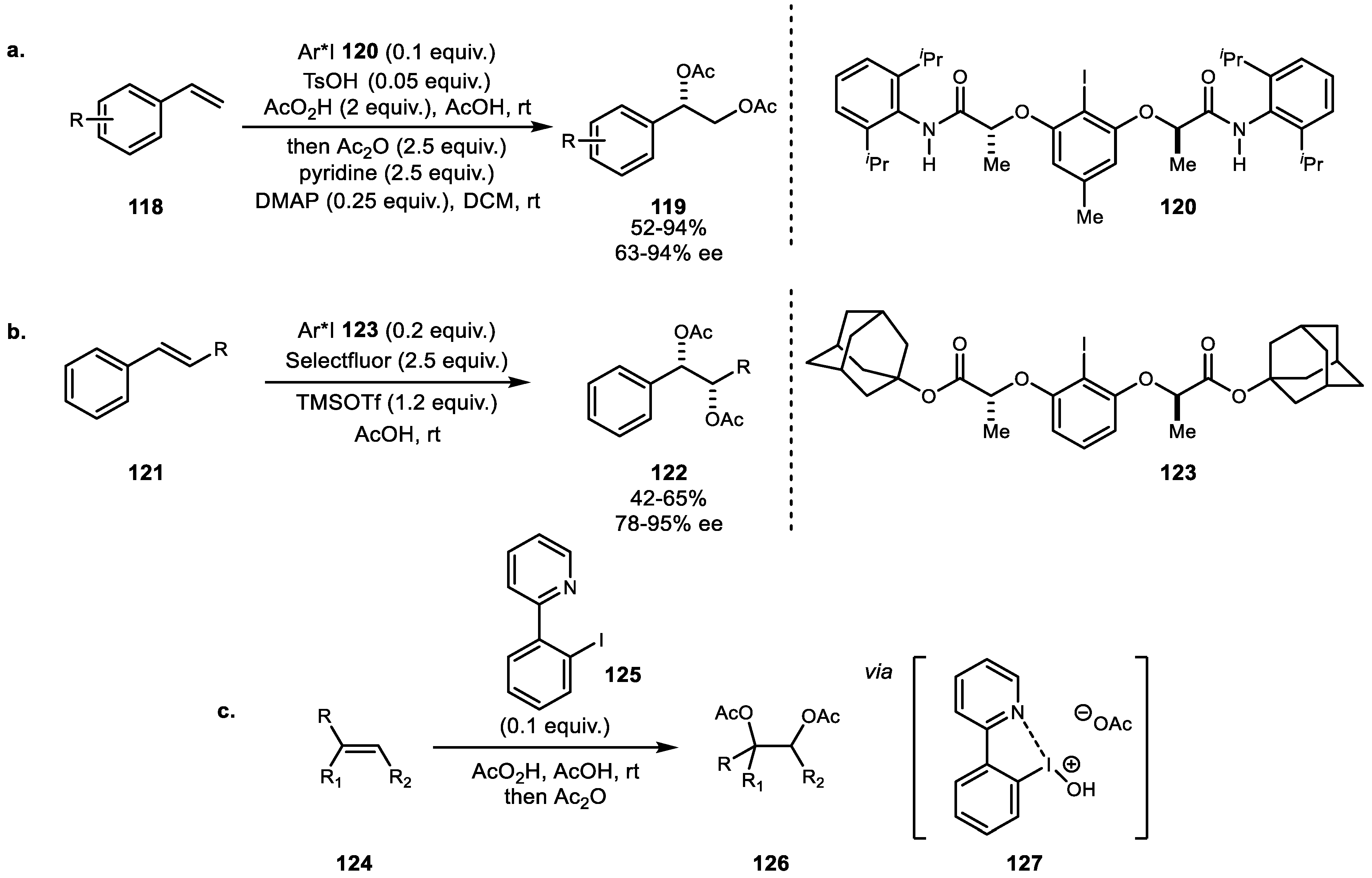
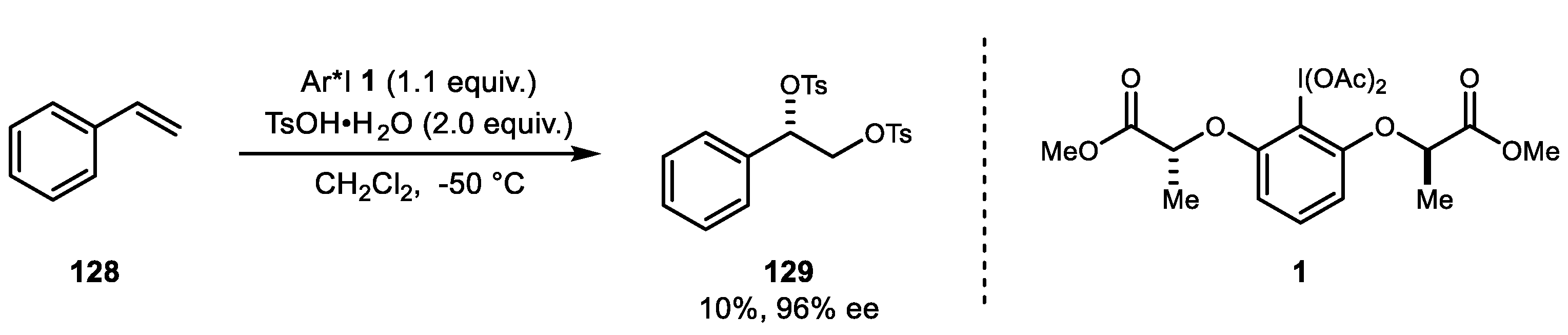
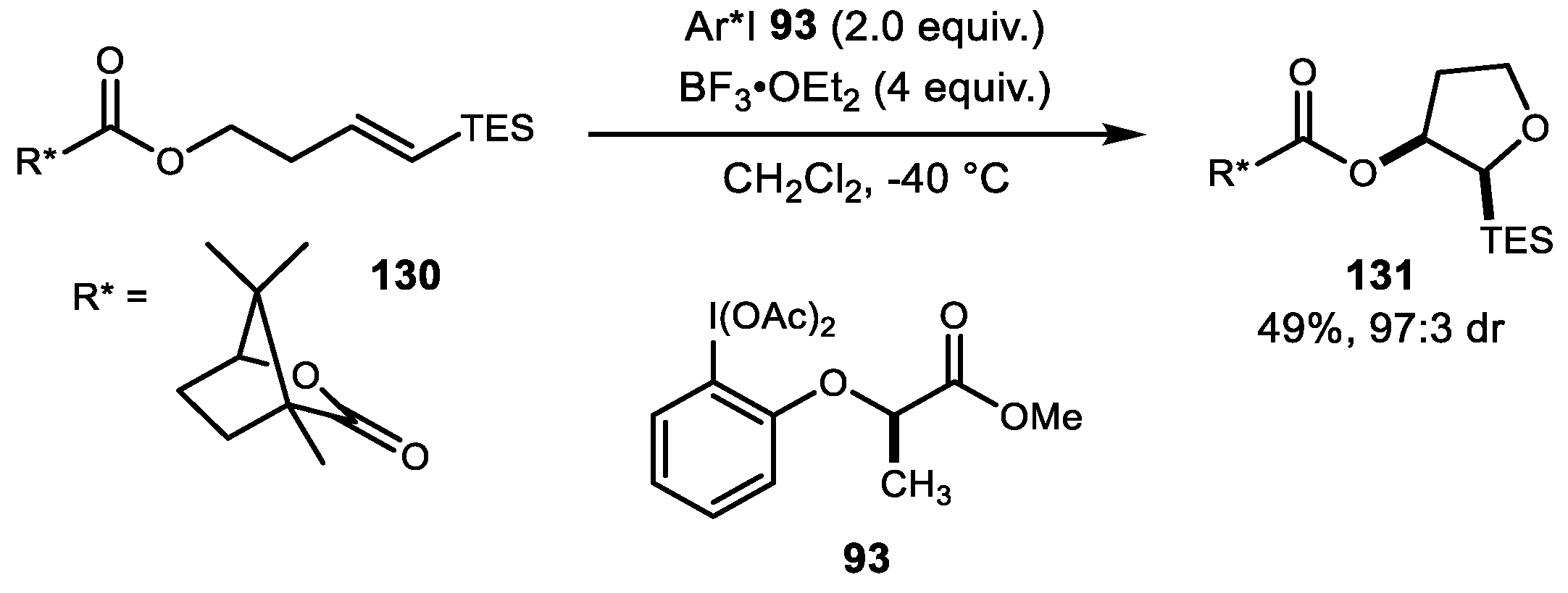
4. Alkene Diacetoxylation
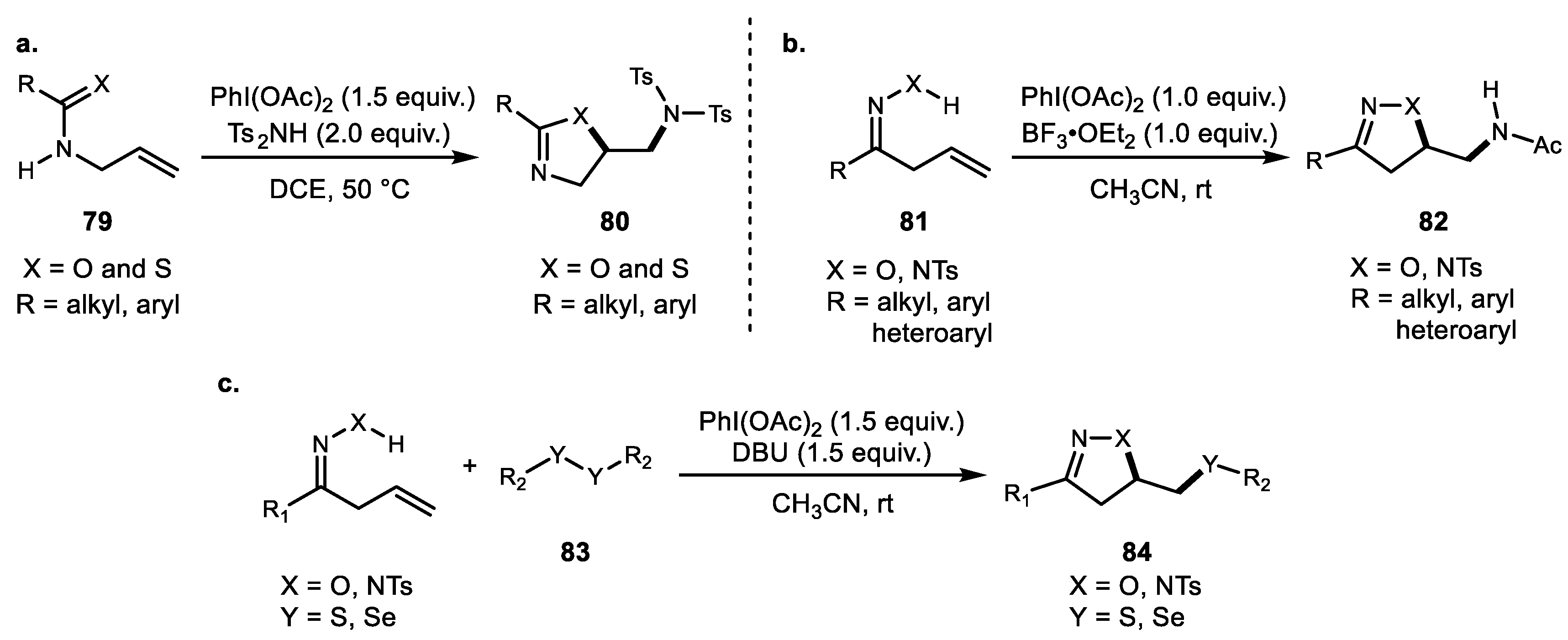
5. Alkene Oxyfunctionalization
6. Alkene Dihalogenation
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Wirth, T.; Hirt, U.H. Hypervalent iodine compounds. Recent advances in synthetic applications. Synthesis 1999, 1999, 1271–1287. [Google Scholar] [CrossRef]
- Wirth, T. Hypervalent iodine chemistry in synthesis: Scope and new directions. Angew. Chem. Int. Ed. 2005, 44, 3656–3665. [Google Scholar] [CrossRef] [PubMed]
- Ladziata, U.; Zhdankin, V.V. Hypervalent iodine(V) reagents in organic synthesis. Arkivoc 2006, 9, 26–58. [Google Scholar] [CrossRef]
- Zhdankin, V.V.; Stang, P.J. Chemistry of Polyvalent Iodine. Chem. Rev. 2008, 108, 5299–5358. [Google Scholar] [CrossRef] [PubMed]
- Dohi, T.; Kita, Y. Hypervalent iodine reagents as a new entrance to organocatalysts. Chem. Commun. 2009, 2073–2085. [Google Scholar] [CrossRef]
- Zhdankin, V.V. Hypervalent iodine(III) reagents in organic synthesis. Arkivoc 2009, 1–62. [Google Scholar]
- Farid, U.; Wirth, T. Stereoselective Synthesis with Hypervalent Iodine Reagents; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2012; pp. 197–203. [Google Scholar]
- Brown, M.; Farid, U.; Wirth, T. Hypervalent iodine reagents as powerful electrophiles. Synlett 2013, 24, 424–431. [Google Scholar]
- Singh, F.V.; Wirth, T. Oxidative Functionalization with Hypervalent Halides; Elsevier B.V.: Amsterdam, The Netherlands, 2014; pp. 880–933. [Google Scholar]
- Yoshimura, A.; Zhdankin, V.V. Advances in Synthetic Applications of Hypervalent Iodine Compounds. Chem. Rev. 2016, 116, 3328–3435. [Google Scholar] [CrossRef]
- Quideau, S. Hypervalent iodine chemistry: Recent advances and applications; Elsevier: Amsterdam, The Netherlands, 2010. [Google Scholar]
- Zhdankin, V.V. Hypervalent Iodine Chemistry Preparation, Structure and Synthetic Applications of Polyvalent Iodine Compounds; John Wiley & Sons: Chichester/West Sussex, UK, 2014. [Google Scholar]
- Wirth, T. Hypervalent Iodine Chemistry; Springer: Berlin/Heidelberg, Germany, 2018. [Google Scholar]
- Moriarty, R.M.; Prakash, O. Oxidation of phenolic compounds with organohypervalent iodine reagents. Org. React. 2001, 57, 327–415. [Google Scholar]
- Rodriguez, S.; Wipf, P. Oxidative spiroacetalizations and spirolactonizations of arenes. Synthesis 2004, 2767–2783. [Google Scholar] [CrossRef]
- Zhdankin, V.V. Benziodoxole-based hypervalent iodine reagents in organic synthesis. Curr. Org. Synth. 2005, 2, 121–145. [Google Scholar] [CrossRef]
- Ciufolini, M.A.; Braun, N.A.; Canesi, S.; Ousmer, M.; Chang, J.; Chai, D. Oxidative amidation of phenols through the use of hypervalent iodine reagents. Development and applications. Synthesis 2007, 3759–3772. [Google Scholar] [CrossRef]
- Pouysegu, L.; Deffieux, D.; Quideau, S. Hypervalent iodine-mediated phenol dearomatization in natural product synthesis. Tetrahedron 2010, 66, 2235–2261. [Google Scholar] [CrossRef]
- Fernandez Gonzalez, D.; Benfatti, F.; Waser, J. Asymmetric Organocatalysis Meets Hypervalent Iodine Chemistry for the α-Functionalization of Carbonyl Compounds. ChemCatChem 2012, 4, 955–958. [Google Scholar] [CrossRef]
- Parra, A.; Reboredo, S. Chiral Hypervalent Iodine Reagents: Synthesis and Reactivity. Chem. Eur. J. 2013, 19, 17244–17260. [Google Scholar] [CrossRef] [PubMed]
- Singh, F.V.; Wirth, T. Oxidative rearrangements with hypervalent iodine reagents. Synthesis 2013, 45, 2499–2511. [Google Scholar]
- Romero, R.M.; Woeste, T.H.; Muniz, K. Vicinal Difunctionalization of Alkenes with Iodine (III) Reagents and Catalysts. Chem. Asian J. 2014, 9, 972–983. [Google Scholar] [CrossRef] [PubMed]
- Charpentier, J.; Fruh, N.; Togni, A. Electrophilic Trifluoromethylation by Use of Hypervalent Iodine Reagents. Chem. Rev. 2015, 115, 650–682. [Google Scholar] [CrossRef]
- Kumar, R.; Wirth, T. Asymmetric synthesis with hypervalent iodine reagents. Top. Curr. Chem. 2016, 373, 243–262. [Google Scholar]
- Muniz, K. Aminations with hypervalent iodine. Top. Curr. Chem. 2016, 373, 105–134. [Google Scholar]
- Waser, J. Alkynylation with hypervalent iodine reagents. Top. Curr. Chem. 2016, 373, 187–222. [Google Scholar] [PubMed]
- Boelke, A.; Finkbeiner, P.; Nachtsheim, B.J. Atom-economical group-transfer reactions with hypervalent iodine compounds. Beilstein J. Org. Chem. 2018, 14, 1263–1280. [Google Scholar] [CrossRef] [PubMed]
- Reddy Kandimalla, S.; Prathima Parvathaneni, S.; Sabitha, G.; Subba Reddy, B.V. Recent Advances in Intramolecular Metal-Free Oxidative C-H Bond Aminations Using Hypervalent Iodine (III) Reagents. Eur. J. Org. Chem. 2019, 2019, 1687–1714. [Google Scholar] [CrossRef]
- Xing, L.; Zhang, Y.; Du, Y. Hypervalent Iodine-Mediated Synthesis of Spiroheterocycles via Oxidative Cyclization. Curr. Org. Chem. 2019, 23, 14–37. [Google Scholar] [CrossRef]
- Röben, C.; Souto, J.A.; González, Y.; Lishchynskyi, A.; Muñiz, K. Enantioselective Metal-Free Diamination of Styrenes. Angew. Chem. Int. Ed. 2011, 50, 9478–9482. [Google Scholar] [CrossRef] [PubMed]
- Muñiz, K.; Barreiro, L.; Romero, R.M.; Martínez, C. Catalytic Asymmetric Diamination of Styrenes. J. Am. Chem. Soc. 2017, 139, 4354–4357. [Google Scholar] [CrossRef] [PubMed]
- Souto, J.A.; González, Y.; Iglesias, A.; Zian, D.; Lishchynskyi, A.; Muñiz, K. Iodine (III)-Promoted Intermolecular Diamination of Alkenes. Chem. Asian J. 2012, 7, 1103–1111. [Google Scholar] [CrossRef] [PubMed]
- Souto, J.A.; Martínez, C.; Velilla, I.; Muñiz, K. Defined Hypervalent Iodine (III) Reagents Incorporating Transferable Nitrogen Groups: Nucleophilic Amination through Electrophilic Activation. Angew. Chem. Int. Ed. 2013, 52, 1324–1328. [Google Scholar] [CrossRef]
- Romero, R.M.; Souto, J.A.; Muñiz, K. Substitution Effects of Hypervalent Iodine (III) Reagents in the Diamination of Styrene. J. Org. Chem. 2016, 81, 6118–6122. [Google Scholar] [CrossRef]
- Röben, C.; Souto, J.A.; Escudero-Adán, E.C.; Muñiz, K. Oxidative Diamination Promoted by Dinuclear Iodine (III) Reagents. Org. Lett. 2013, 15, 1008–1011. [Google Scholar] [CrossRef]
- Kong, A.; Blakey, S.B. Intramolecular Olefin Diamination for the Stereoselective Synthesis of 3-Aminopiperidines. Synthesis 2012, 44, 1190–1198. [Google Scholar]
- Kim, H.J.; Cho, S.H.; Chang, S. Intramolecular Oxidative Diamination and Aminohydroxylation of Olefins under Metal-Free Conditions. Org. Lett. 2012, 14, 1424–1427. [Google Scholar] [CrossRef] [PubMed]
- Muñiz, K. Advancing Palladium-Catalyzed C–N Bond Formation: Bisindoline Construction from Successive Amide Transfer to Internal Alkenes. J. Am. Chem. Soc. 2007, 129, 14542–14543. [Google Scholar] [CrossRef] [PubMed]
- Mizar, P.; Laverny, A.; El-Sherbini, M.; Farid, U.; Brown, M.; Malmedy, F.; Wirth, T. Enantioselective Diamination with Novel Chiral Hypervalent Iodine Catalysts. Chem. Eur. J. 2014, 20, 9910–9913. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.-T.; Lu, X.-W.; Xing, M.-L.; Sun, X.-Q.; Miao, C.-B. Hypervalent Iodine Reagent Mediated Diamination of [60] Fullerene with Sulfamides or Phosphoryl Diamides. Org. Lett. 2014, 16, 5882–5885. [Google Scholar] [CrossRef] [PubMed]
- Hong, K.B.; Johnston, J.N. Alkene Diamination Using Electron-Rich Amines: Hypervalent Iodine-Promoted Inter-/Intramolecular C–N Bond Formation. Org. Lett. 2014, 16, 3804–3807. [Google Scholar] [CrossRef]
- Danneman, M.W.; Hong, K.B.; Johnston, J.N. Oxidative Inter-/Intermolecular Alkene Diamination of Hydroxy Styrenes with Electron-Rich Amines. Org. Lett. 2015, 17, 2558–2561. [Google Scholar] [CrossRef]
- Danneman, M.W.; Hong, K.B.; Johnston, J.N. A Unified Approach to the Four Azaindoline Families by Inter-/Intramolecular Annulative Diamination of Vinylpyridines. Org. Lett. 2015, 17, 3806–3809. [Google Scholar] [CrossRef]
- Wu, X.-L.; Wang, G.-W. Hypervalent iodine-mediated aminobromination of olefins in water. Tetrahedron 2009, 65, 8802–8807. [Google Scholar] [CrossRef]
- Lovick, H.M.; Michael, F.E. Metal-Free Highly Regioselective Aminotrifluoroacetoxylation of Alkenes. J. Am. Chem. Soc. 2010, 132, 1249–1251. [Google Scholar] [CrossRef]
- Farid, U.; Wirth, T. Highly Stereoselective Metal-Free Oxyaminations Using Chiral Hypervalent Iodine Reagents. Angew. Chem. Int. Ed. 2012, 51, 3462–3465. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Kaga, A.; Chiba, S. Diastereoselective Aminooxygenation and Diamination of Alkenes with Amidines by Hypervalent Iodine (III) Reagents. Org. Lett. 2014, 16, 6136–6139. [Google Scholar] [CrossRef] [PubMed]
- Sanjaya, S.; Chiba, S. Copper-Catalyzed Aminooxygenation of N-Allylamidines with PhI(OAc)2. Org. Lett. 2012, 14, 5342–5345. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-F.; Zhu, X.; Chiba, S. Copper-Catalyzed Aerobic [3 + 2]-Annulation of N-Alkenyl Amidines. J. Am. Chem. Soc. 2012, 134, 3679–3682. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Kaga, A.; Chiba, S. Anti-Selective aminofluorination of alkenes with amidines mediated by hypervalent iodine (iii) reagents. Org. Biomol. Chem. 2016, 14, 5481–5485. [Google Scholar] [CrossRef] [PubMed]
- Wardrop, D.J.; Bowen, E.G.; Forslund, R.E.; Sussman, A.D.; Weerasekera, S.L. Intramolecular Oxamidation of Unsaturated O-Alkyl Hydroxamates: A Remarkably Versatile Entry to Hydroxy Lactams. J. Am. Chem. Soc. 2010, 132, 1188–1189. [Google Scholar] [CrossRef] [PubMed]
- Bowen, E.G.; Wardrop, D.J. Diastereoselective Nitrenium Ion-Mediated Cyclofunctionalization: Total Synthesis of (+)-Castanospermine. Org. Lett. 2010, 12, 5330–5333. [Google Scholar] [CrossRef] [PubMed]
- Wardrop, D.J.; Bowen, E.G. Nitrenium Ion-Mediated Alkene Bis-Cyclofunctionalization: Total Synthesis of (−)-Swainsonine. Org. Lett. 2011, 13, 2376–2379. [Google Scholar] [CrossRef]
- Bhattacharjee, A.; Gerasimov, M.V.; DeJong, S.; Wardrop, D.J. Oxamidation of Unsaturated O-Alkyl Hydroxamates: Synthesis of the Madangamine Diazatricylic (ABC Rings) Skeleton. Org. Lett. 2017, 19, 6570–6573. [Google Scholar] [CrossRef]
- Wang, Q.; Zhong, W.; Wei, X.; Ning, M.; Meng, X.; Li, Z. Metal-free intramolecular aminofluorination of alkenes mediated by PhI(OPiv)2/hydrogen fluoride–pyridine system. Org. Biomol. Chem. 2012, 10, 8566–8569. [Google Scholar] [CrossRef]
- Kong, W.; Feige, P.; de Haro, T.; Nevado, C. Regio- and Enantioselective Aminofluorination of Alkenes. Angew. Chem. Int. Ed. 2013, 52, 2469–2473. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, S.; Kamo, T.; Fukushi, K.; Hiramatsu, T.; Tokunaga, E.; Dohi, T.; Kita, Y.; Shibata, N. Iodoarene-catalyzed fluorination and aminofluorination by an Ar-I/HF·pyridine/mCPBA system. Chem. Sci. 2014, 5, 2754–2760. [Google Scholar] [CrossRef]
- Cui, J.; Jia, Q.; Feng, R.-Z.; Liu, S.-S.; He, T.; Zhang, C. Boron Trifluoride Etherate Functioning as a Fluorine Source in an Iodosobenzene-Mediated Intramolecular Aminofluorination of Homoallylic Amines. Org. Lett. 2014, 16, 1442–1445. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, T.; Miyake, A.; Muta, K.; Oyamada, A.J. Hypervalent Iodine/HF Reagents for the Synthesis of 3-Fluoropyrrolidines. J. Org. Chem. 2017, 82, 11721–11726. [Google Scholar] [CrossRef] [PubMed]
- Mennie, K.M.; Banik, S.M.; Reichert, E.C.; Jacobsen, E.N. Catalytic Diastereo- and Enantioselective Fluoroamination of Alkenes. J. Am. Chem. Soc. 2018, 140, 4797–4802. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.-Q.; Li, Y.-M. Regioselective (Diacetoxyiodo)benzene-Promoted Halocyclization of Unfunctionalized Olefins. J. Org. Chem. 2014, 79, 10094–10109. [Google Scholar] [CrossRef]
- Wang, H.; Frings, M.; Bolm, C. Halocyclizations of Unsaturated Sulfoximines. Org. Lett. 2016, 18, 2431–2434. [Google Scholar] [CrossRef]
- Daniel, M.; Blanchard, F.; Nocquet-Thibault, S.; Cariou, K.; Dodd, R.H. Halocyclization of Unsaturated Guanidines Mediated by Koser’s Reagent and Lithium Halides. J. Org. Chem. 2015, 80, 10624–10633. [Google Scholar] [CrossRef]
- Mizar, P.; Niebuhr, R.; Hutchings, M.; Farooq, U.; Wirth, T. Thioamination of Alkenes with Hypervalent Iodine Reagents. Chem. Eur. J. 2016, 22, 1614–1617. [Google Scholar] [CrossRef]
- Jeon, H.; Kim, D.; Lee, J.H.; Song, J.; Lee, W.S.; Kang, D.W.; Kang, S.; Lee, S.B.; Choi, S.; Hong, K.B. Hypervalent Iodine-Mediated Alkene Functionalization: Oxazoline and Thiazoline Synthesis via Inter-/Intramolecular Aminohydroxylation and Thioamination. Adv. Synth. Catal. 2018, 360, 779–783. [Google Scholar] [CrossRef]
- Park, S.W.; Kim, S.-H.; Song, J.; Park, G.Y.; Kim, D.; Nam, T.-G.; Hong, K.B. Hypervalent iodine-mediated Ritter-type amidation of terminal alkenes: The synthesis of isoxazoline and pyrazoline cores. Beilstein J. Org. Chem. 2018, 14, 1028–1033. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.-M.; Cai, C. Iodine(iii)-mediated intramolecular sulfeno- and selenofunctionalization of β,γ-unsaturated tosyl hydrazones and oximes. Org. Biomol. Chem. 2018, 16, 490–498. [Google Scholar] [CrossRef] [PubMed]
- Fujita, M.; Yoshida, Y.; Miyata, K.; Wakisaka, A.; Sugimura, T. Enantiodifferentiating endo-Selective Oxylactonization of ortho-Alk-1-enylbenzoate with a Lactate-Derived Aryl-λ3-Iodane. Angew. Chem. Int. Ed. 2010, 49, 7068–7071. [Google Scholar] [CrossRef] [PubMed]
- Fujita, M.; Wakita, M.; Sugimura, T. Enantioselective Prévost and Woodward reactions using chiral hypervalent iodine(iii): Switchover of stereochemical course of an optically active 1,3-dioxolan-2-yl cation. Chem. Commun. 2011, 47, 3983–3985. [Google Scholar] [CrossRef] [PubMed]
- Fujita, M.; Mori, K.; Shimogaki, M.; Sugimura, T. Asymmetric Synthesis of 4,8-Dihydroxyisochroman-1-one Polyketide Metabolites Using Chiral Hypervalent Iodine(III). Org. Lett. 2012, 14, 1294–1297. [Google Scholar] [CrossRef] [PubMed]
- Fujita, M.; Mori, K.; Shimogaki, M.; Sugimura, T. Total synthesis of (12R)- and (12S)-12-hydroxymonocerins: Stereoselective oxylactonization using a chiral hypervalent iodine(iii) species. RSC Adv. 2013, 3, 17717–17725. [Google Scholar] [CrossRef]
- Shimogaki, M.; Fujita, M.; Sugimura, T. Enantioselective Oxidation of Alkenylbenzoates Catalyzed by Chiral Hypervalent Iodine(III) To Yield 4-Hydroxyisochroman-1-ones. Eur. J. Org. Chem. 2013, 2013, 7128–7138. [Google Scholar] [CrossRef]
- Takesue, T.; Fujita, M.; Sugimura, T.; Akutsu, H. A Series of Two Oxidation Reactions of ortho-Alkenylbenzamide with Hypervalent Iodine(III): A Concise Entry into (3R,4R)-4-Hydroxymellein and (3R,4R)-4-Hydroxy-6-methoxymellein. Org. Lett. 2014, 16, 4634–4637. [Google Scholar] [CrossRef]
- Shimogaki, M.; Fujita, M.; Sugimura, T. Stereoselective Formation of Substituted 1,3-Dioxolanes through a Three-Component Assembly during the Oxidation of Alkenes with Hypervalent Iodine(III). Molecules 2015, 20, 17041. [Google Scholar] [CrossRef]
- Alhalib, A.; Kamouka, S.; Moran, W.J. Iodoarene-Catalyzed Cyclizations of Unsaturated Amides. Org. Lett. 2015, 17, 1453–1456. [Google Scholar] [CrossRef]
- Moon, N.G.; Harned, A.M. Iodine(III)-promoted synthesis of oxazolines from N-allylamides. Tetrahedron Lett. 2013, 54, 2960–2963. [Google Scholar] [CrossRef]
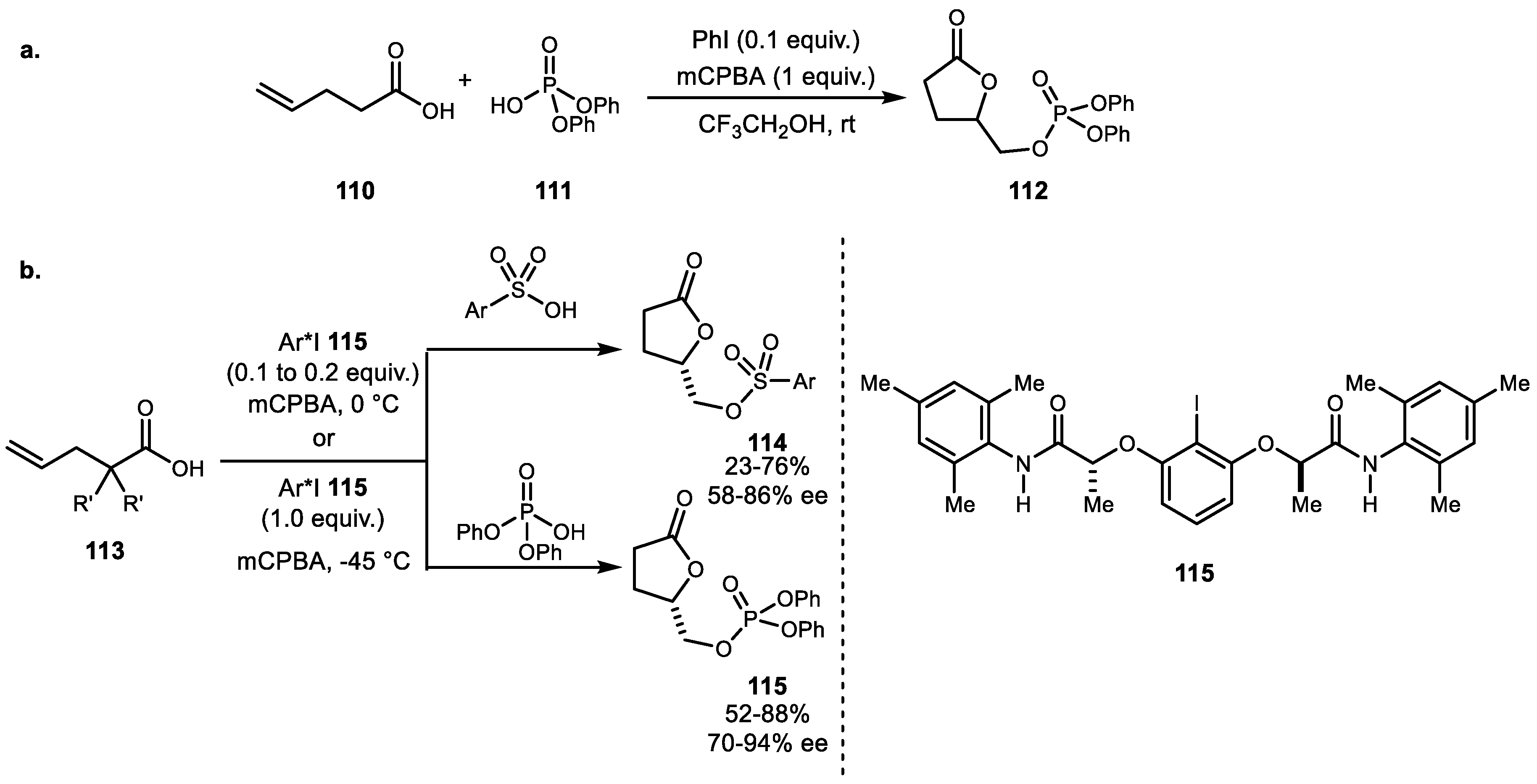
- Koser, G.F.; Lodaya, J.S.; Ray, D.G.; Kokil, P.B. Direct. alpha.-phosphoryloxylation of ketones and (phosphoryloxy) lactonization of pentenoic acids with [hydroxy[(bis(phenyloxy)phosphoryl)oxy]iodo]benzene. J. Am. Chem. Soc. 1988, 110, 2987–2988. [Google Scholar] [CrossRef]
- Zhou, Z.-S.; He, X.-H. A convenient phosphoryloxylactonization of pentenoic acids with catalytic hypervalent iodine(III) reagent. Tetrahedron Lett. 2010, 51, 2480–2482. [Google Scholar] [CrossRef]
- Gelis, C.; Dumoulin, A.; Bekkaye, M.; Neuville, L.; Masson, G. Chiral Hypervalent Iodine(III) Catalyst Promotes Highly Enantioselective Sulfonyl- and Phosphoryl-oxylactonizations. Org. Lett. 2017, 19, 278–281. [Google Scholar] [CrossRef] [PubMed]
- Zhong, W.; Yang, J.; Meng, X.; Li, Z. BF3·OEt2-Promoted Diastereoselective Diacetoxylation of Alkenes by PhI(OAc)2. J. Org. Chem. 2011, 76, 9997–10004. [Google Scholar] [CrossRef] [PubMed]
- Zhong, W.; Liu, S.; Yang, J.; Meng, X.; Li, Z. Metal-Free, Organocatalytic Syn Diacetoxylation of Alkenes. Org. Lett. 2012, 14, 3336–3339. [Google Scholar] [CrossRef]
- Haubenreisser, S.; Wöste, T.H.; Martínez, C.; Ishihara, K.; Muñiz, K. Structurally Defined Molecular Hypervalent Iodine Catalysts for Intermolecular Enantioselective Reactions. Angew. Chem. Int. Ed. 2016, 55, 413–417. [Google Scholar] [CrossRef]
- Wöste, T.H.; Muñiz, K. Enantioselective Vicinal Diacetoxylation of Alkenes under Chiral Iodine(III) Catalysis. Synthesis 2016, 48, 816–827. [Google Scholar]
- Aertker, K.; Rama, R.J.; Opalach, J.; Muñiz, K. Vicinal Difunctionalization of Alkenes under Iodine(III) Catalysis involving Lewis Base Adducts. Adv. Synth. Catal. 2017, 359, 1290–1294. [Google Scholar] [CrossRef]
- Fujita, M.; Miura, K.; Sugimura, T. Enantioselective dioxytosylation of styrenes using lactate-based chiral hypervalent iodine(III). Beilstein J. Org. Chem. 2018, 14, 659–663. [Google Scholar] [CrossRef]
- Fujita, M.; Ookubo, Y.; Sugimura, T. Asymmetric cycloetherification based on a chiral auxiliary for 4-acyloxy-1-butene substrates during oxidation with iodosylbenzene via a 1,3-dioxan-2-yl cation. Tetrahedron Lett. 2009, 50, 1298–1300. [Google Scholar] [CrossRef]
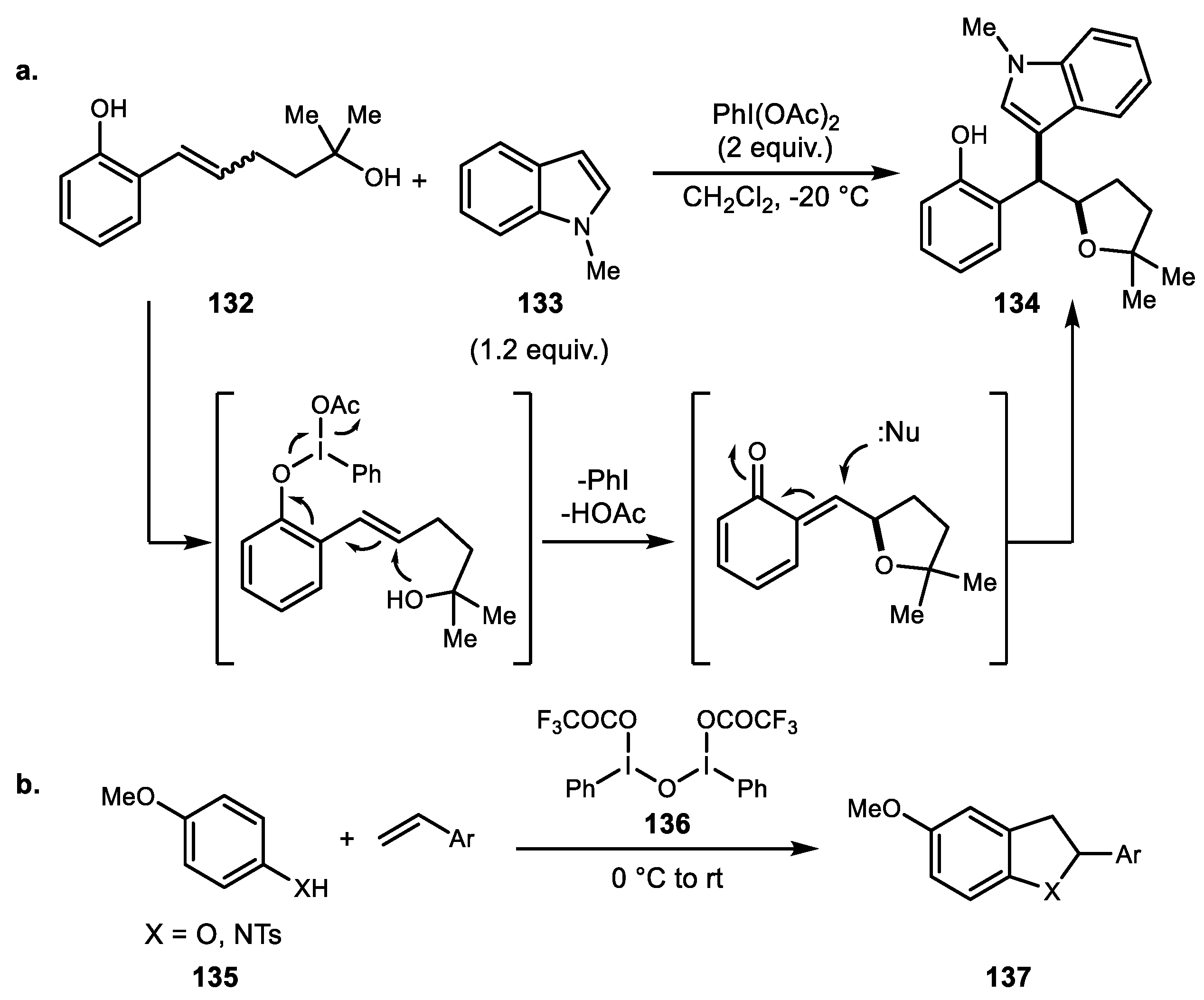
- Zhang, K.; Wang, H.; Zheng, J.; Yu, L.; Ding, H. Hypervalent iodine mediated alkene difunctionalization of vinylphenols: Diastereoselective synthesis of substituted indoles and indolizines. Chem. Commun. 2015, 51, 6399–6402. [Google Scholar] [CrossRef] [PubMed]
- Jensen, K.H.; Pathak, T.P.; Zhang, Y.; Sigman, M.S. Palladium-Catalyzed Enantioselective Addition of Two Distinct Nucleophiles across Alkenes Capable of Quinone Methide Formation. J. Am. Chem. Soc. 2009, 131, 17074–17075. [Google Scholar] [CrossRef] [PubMed]
- Jensen, K.H.; Webb, J.D.; Sigman, M.S. Advancing the Mechanistic Understanding of an Enantioselective Palladium-Catalyzed Alkene Difunctionalization Reaction. J. Am. Chem. Soc. 2010, 132, 17471–17482. [Google Scholar] [CrossRef] [PubMed]
- Dohi, T.; Toyoda, Y.; Nakae, T.; Koseki, D.; Kubo, H.; Kamitanaka, T.; Kita, Y. Phenol and aniline oxidative coupling with alkenes by using hypervalent iodine dimer for the rapid access to dihydrobenzofurans and indolines. Heterocylces 2015, 90, 631–644. [Google Scholar]
- Yoshimura, A.; Middleton, K.R.; Todora, A.D.; Kastern, B.J.; Koski, S.R.; Maskaev, A.V.; Zhdankin, V.V. Hypervalent Iodine Catalyzed Generation of Nitrile Oxides from Oximes and their Cycloaddition with Alkenes or Alkynes. Org. Lett. 2013, 15, 4010–4013. [Google Scholar] [CrossRef] [PubMed]
- Xiang, C.; Li, T.; Yan, J. Hypervalent Iodine–Catalyzed Cycloaddition of Nitrile Oxides to Alkenes. Synth. Commun. 2014, 44, 682–688. [Google Scholar] [CrossRef]
- Kumar, R.; Kumar, M.; Prakash, O. A Simple and Efficient Method for the Synthesis of Novel Pyrazolyl Isoxazoline Derivatives Using Hypervalent Iodine (III) Reagent. Heteroat. Chem. 2016, 27, 228–234. [Google Scholar] [CrossRef]
- Nocquet-Thibault, S.; Retailleau, P.; Cariou, K.; Dodd, R.H. Iodine(III)-Mediated Umpolung of Bromide Salts for the Ethoxybromination of Enamides. Org. Lett. 2013, 15, 1842–1845. [Google Scholar] [CrossRef]
- Nocquet-Thibault, S.; Minard, C.; Retailleau, P.; Cariou, K.; Dodd, R.H. Iodine(III)-mediated ethoxychlorination of enamides with iron(III) chloride. Tetrahedron 2014, 70, 6769–6775. [Google Scholar] [CrossRef]
- Nocquet-Thibault, S.; Rayar, A.; Retailleau, P.; Cariou, K.; Dodd, R.H. Iodine(III)-Mediated Diazidation and Azido-oxyamination of Enamides. Chem. Eur. J. 2015, 21, 14205–14210. [Google Scholar] [CrossRef] [PubMed]
- Shimogaki, M.; Fujita, M.; Sugimura, T. Metal-Free Enantioselective Oxidative Arylation of Alkenes: Hypervalent-Iodine-Promoted Oxidative C–C Bond Formation. Angew. Chem. Int. Ed. 2016, 55, 15797–15801. [Google Scholar] [CrossRef] [PubMed]
- Shimogaki, M.; Fujita, M.; Sugimura, T. Enantioselective C-C Bond Formation during the Oxidation of 5-Phenylpent-2-enyl Carboxylates with Hypervalent Iodine(III). J. Org. Chem. 2017, 82, 11836–11840. [Google Scholar] [CrossRef] [PubMed]
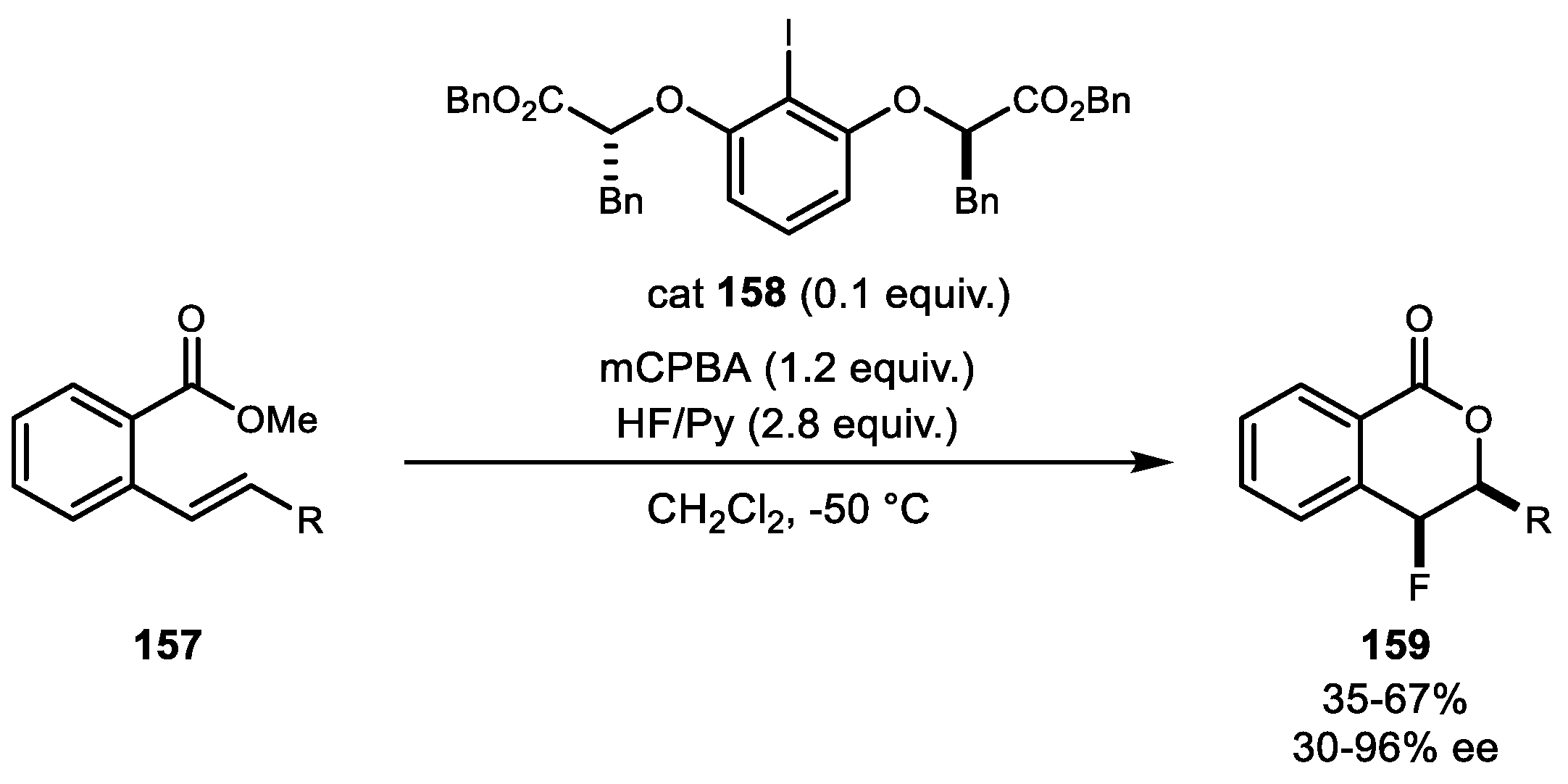
- Woerly, E.M.; Banik, S.M.; Jacobsen, E.N. Enantioselective, Catalytic Fluorolactonization Reactions with a Nucleophilic Fluoride Source. J. Am. Chem. Soc. 2016, 138, 13858–13861. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wu, S.; Yan, J. PhI Catalyzed Acetoxyselenylation and Formyloxyselenylation of Alkenes. Helv. Chim. Acta 2017, 100, e1600306. [Google Scholar] [CrossRef]
- Ngatimin, M.; Gartshore, C.J.; Kindler, J.P.; Naidu, S.; Lupton, D.W. The α-halogenation of α,β-unsaturated carbonyls and dihalogenation of alkenes using bisacetoxyiodobenzene/pyridine hydrohalides. Tetrahedron Lett. 2009, 50, 6008–6011. [Google Scholar] [CrossRef]
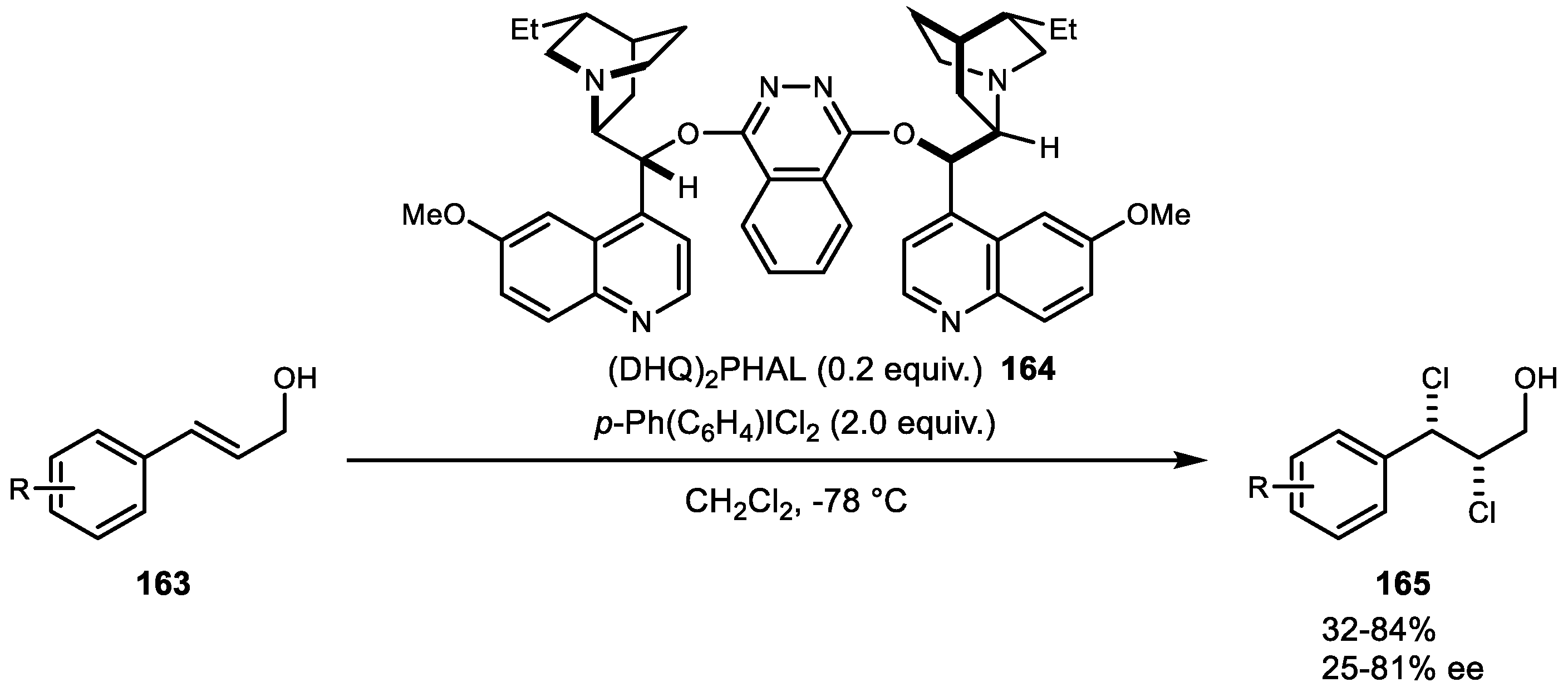
- Nicolaou, K.C.; Simmons, N.L.; Ying, Y.; Heretsch, P.M.; Chen, J.S. Enantioselective Dichlorination of Allylic Alcohols. J. Am. Chem. Soc. 2011, 133, 8134–8137. [Google Scholar] [CrossRef]
- Hara, S.; Nakahigashi, J.; Ishi-i, K.; Sawaguchi, M.; Sakai, H.; Fukuhara, T.; Yoneda, N. Difluorination of Alkenes with Iodotoluene Difluoride. Synlett 1998, 1998, 495–496. [Google Scholar] [CrossRef]
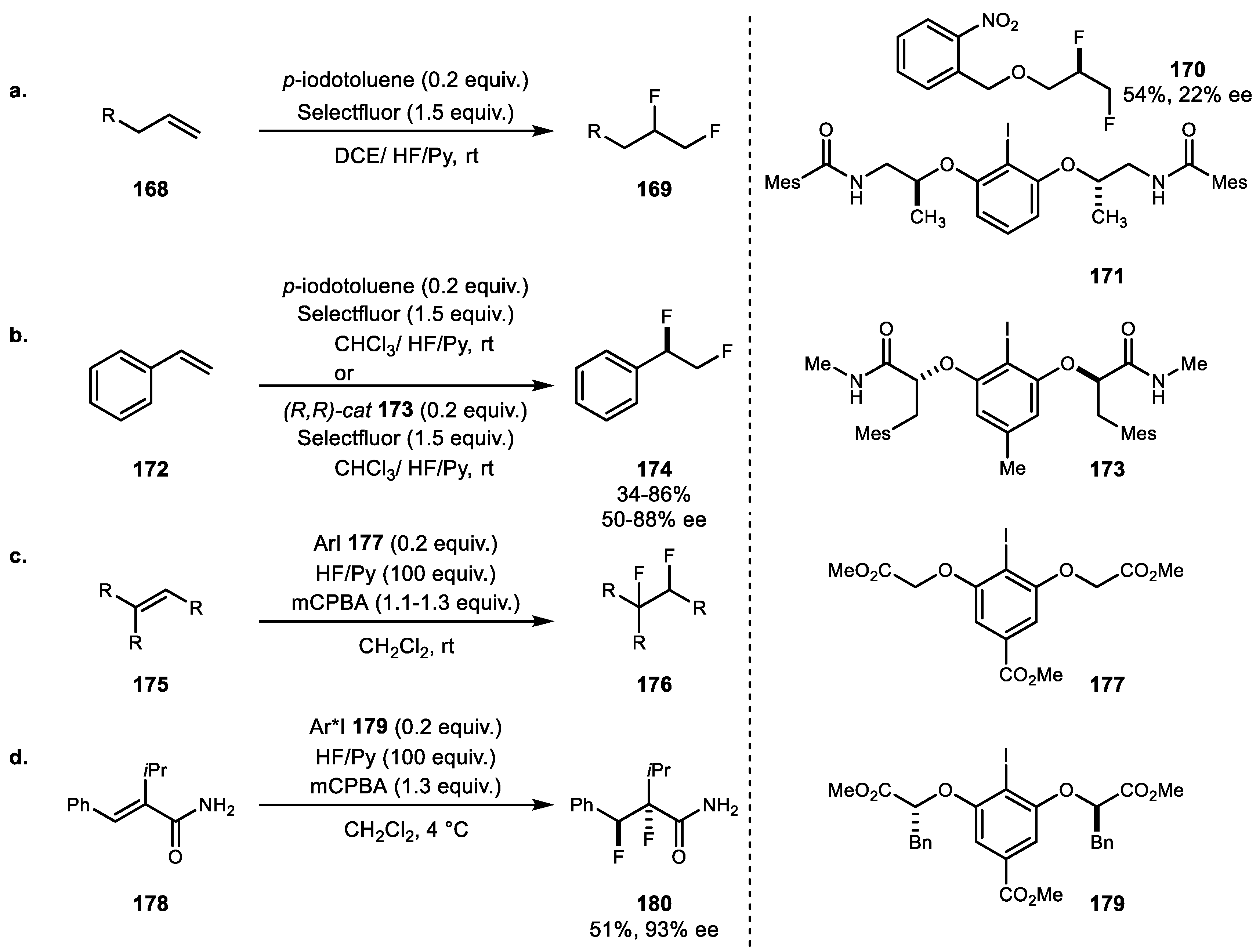
- Molnár, I.G.; Gilmour, R. Catalytic Difluorination of Olefins. J. Am. Chem. Soc. 2016, 138, 5004–5007. [Google Scholar] [CrossRef]
- Scheidt, F.; Schäfer, M.; Sarie, J.C.; Daniliuc, C.G.; Molloy, J.J.; Gilmour, R. Enantioselective, Catalytic Vicinal Difluorination of Alkenes. Angew. Chem. Int. Ed. 2018, 57, 16431–16435. [Google Scholar] [CrossRef]
- Banik, S.M.; Medley, J.W.; Jacobsen, E.N. Catalytic, Diastereoselective 1,2-Difluorination of Alkenes. J. Am. Chem. Soc. 2016, 138, 5000–5003. [Google Scholar] [CrossRef] [PubMed]
- Grayfer, T.D.; Retailleau, P.; Dodd, R.H.; Dubois, J.; Cariou, K. Chemodivergent, Tunable, and Selective Iodine(III)-Mediated Bromo-Functionalizations of Polyprenoids. Org. Lett. 2017, 19, 4766–4769. [Google Scholar] [CrossRef] [PubMed]
- Peilleron, L.; Grayfer, T.D.; Dubois, J.; Dodd, R.H.; Cariou, K. Iodine(III)-mediated halogenations of acyclic monoterpenoids. Beilstein J. Org. Chem. 2018, 14, 1103–1111. [Google Scholar] [CrossRef] [PubMed]



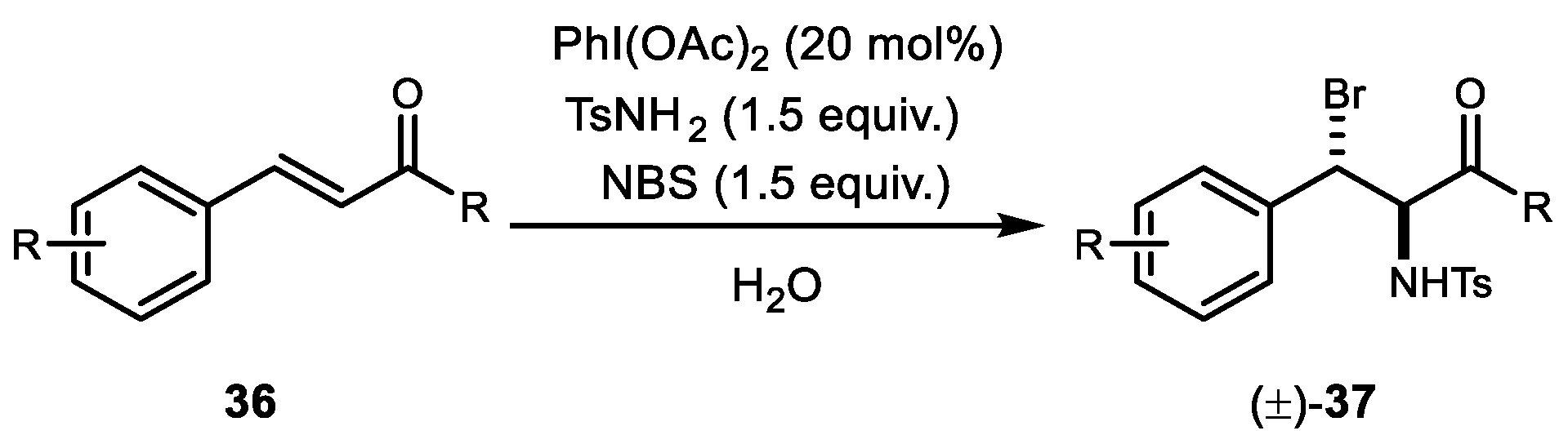






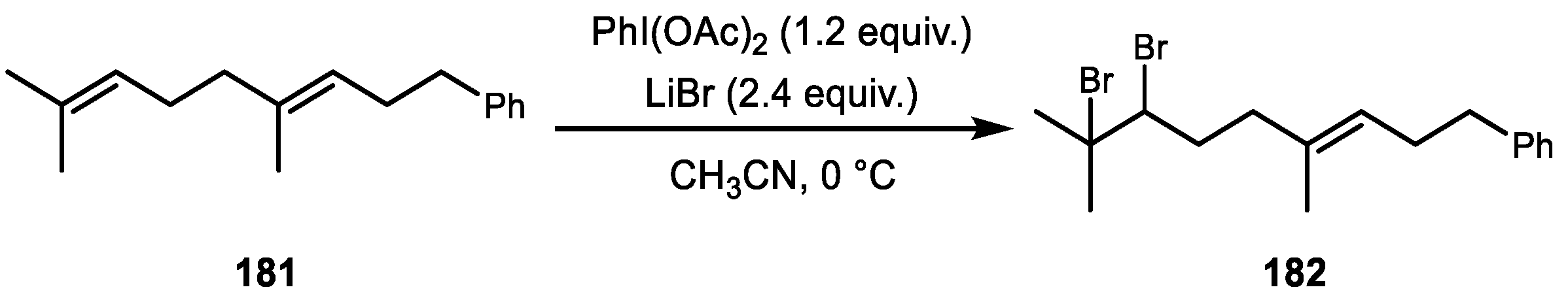
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, J.H.; Choi, S.; Hong, K.B. Alkene Difunctionalization Using Hypervalent Iodine Reagents: Progress and Developments in the Past Ten Years. Molecules 2019, 24, 2634. https://doi.org/10.3390/molecules24142634
Lee JH, Choi S, Hong KB. Alkene Difunctionalization Using Hypervalent Iodine Reagents: Progress and Developments in the Past Ten Years. Molecules. 2019; 24(14):2634. https://doi.org/10.3390/molecules24142634
Chicago/Turabian StyleLee, Ji Hoon, Sungwook Choi, and Ki Bum Hong. 2019. "Alkene Difunctionalization Using Hypervalent Iodine Reagents: Progress and Developments in the Past Ten Years" Molecules 24, no. 14: 2634. https://doi.org/10.3390/molecules24142634
APA StyleLee, J. H., Choi, S., & Hong, K. B. (2019). Alkene Difunctionalization Using Hypervalent Iodine Reagents: Progress and Developments in the Past Ten Years. Molecules, 24(14), 2634. https://doi.org/10.3390/molecules24142634




