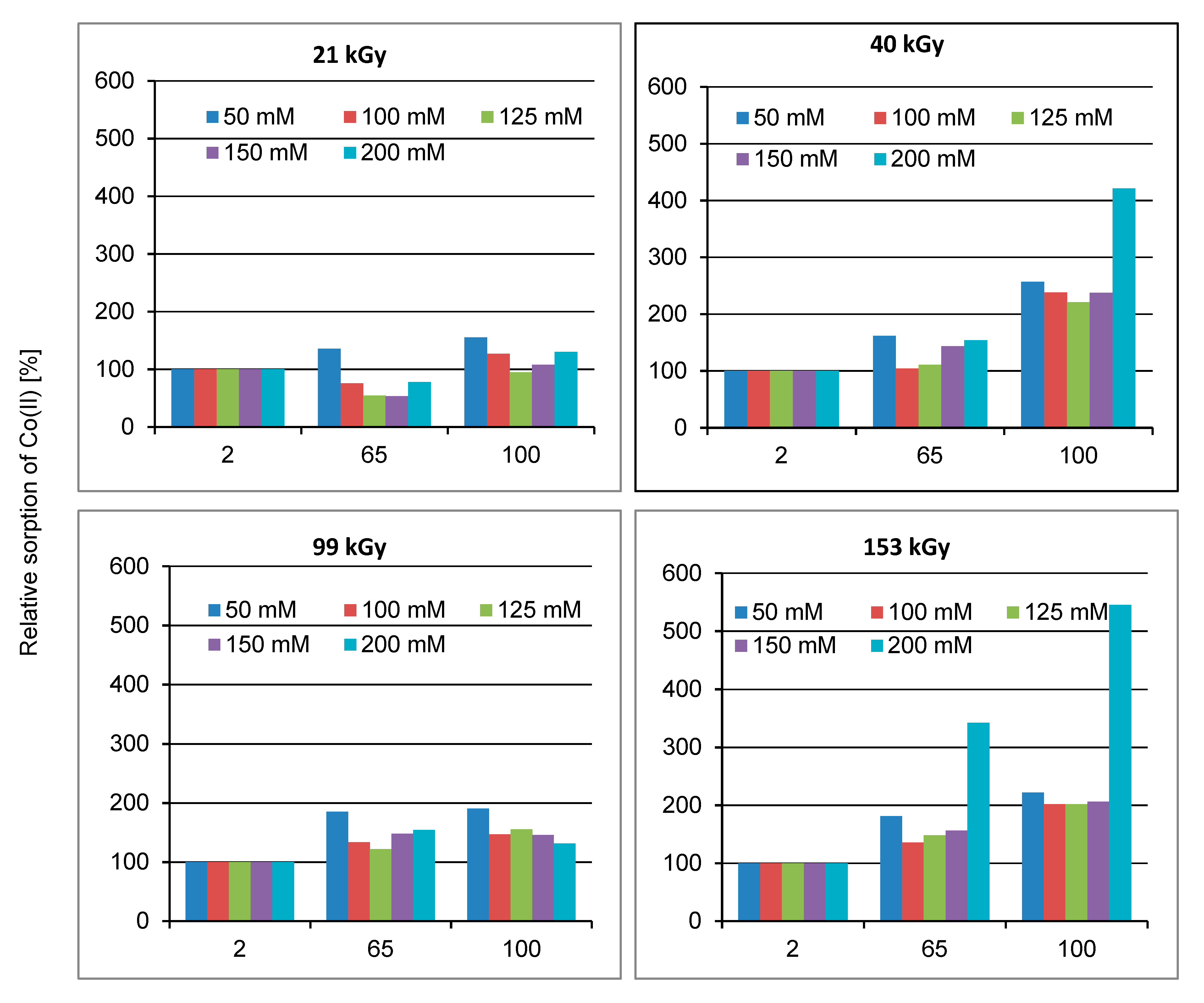
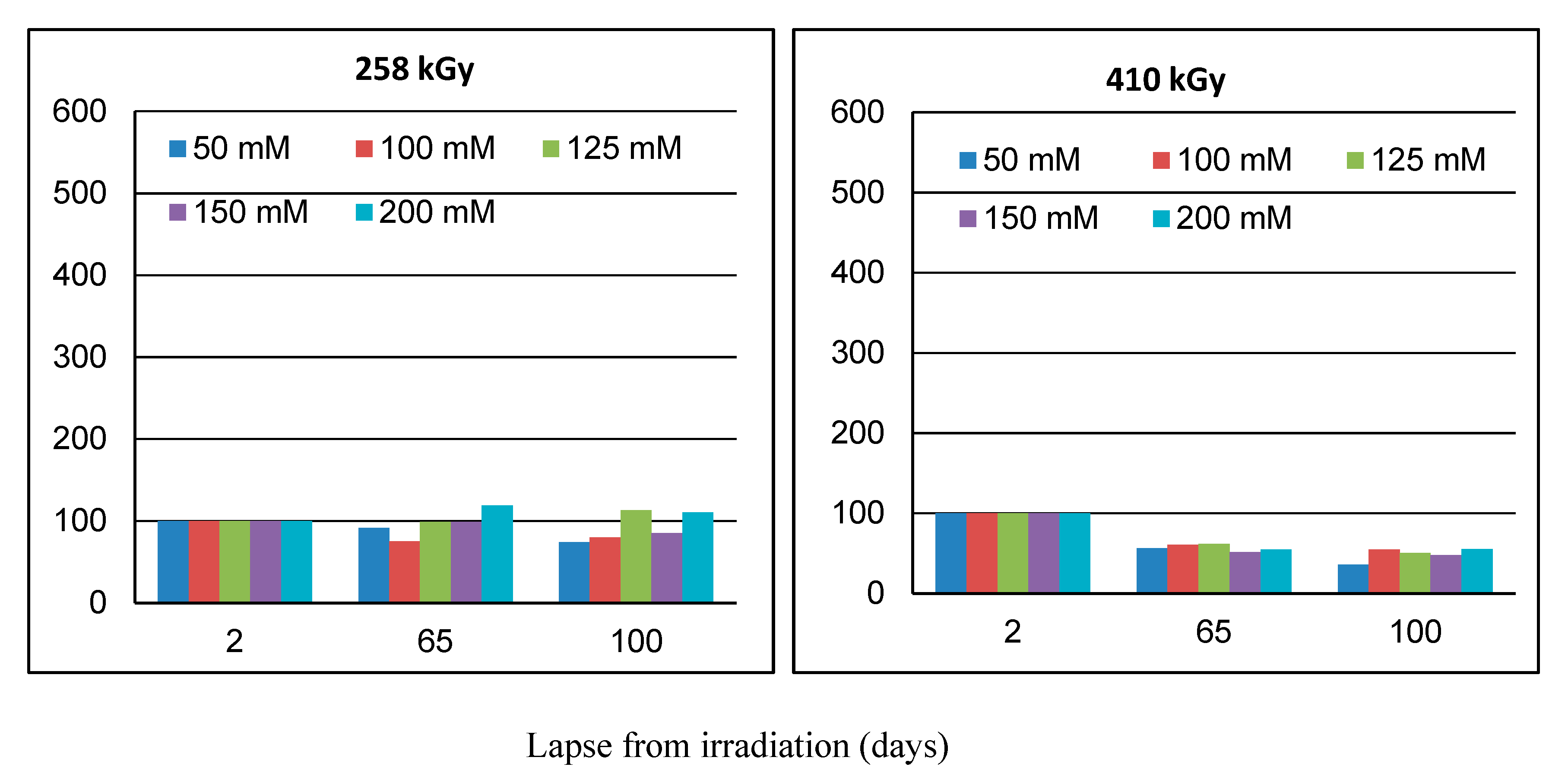
2.1. Effect of Lapse from Wool Exposure on Co(II)-Sorption
Electron beam irradiation of sheep wool splits disulphide bonds of keratin with following oxidation of radicals generated and consecutive transformation of the transitive species for up to cysteic acid [
18,
19]. In this way, in the exposed wool, additional acidic groups are added capable of forming Co-salts and subsequently complexes with available ligands. As found, primary but also secondary structure of the radiation-modified wool varies with time [
19]. It is a gradual complex transformation of conformations in the secondary structure initiated by the absorbed dose of energy and linked with chemical changes. Since both factors play an important role in Co(II) sorption, also sorption potential of the wool should be time-dependent following the dynamics of these changes. To confirm this assumption, we performed sorption experiments at various time intervals of 2–65–100 days from the irradiation with the initial Co(II) concentrations in the (50–200) mmol·dm
−3 range (
Figure 1,
Figure 2 and
Figure 3).
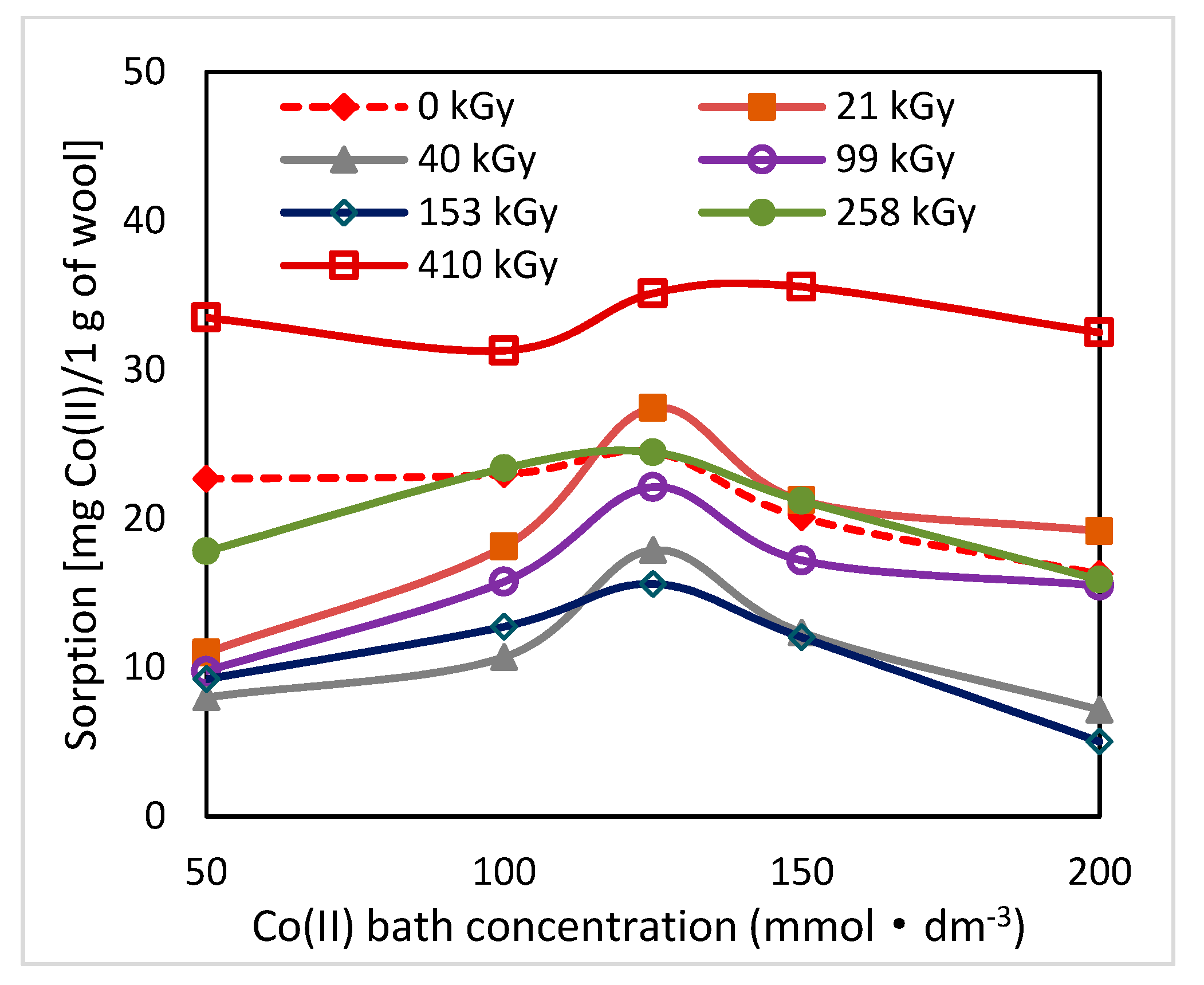
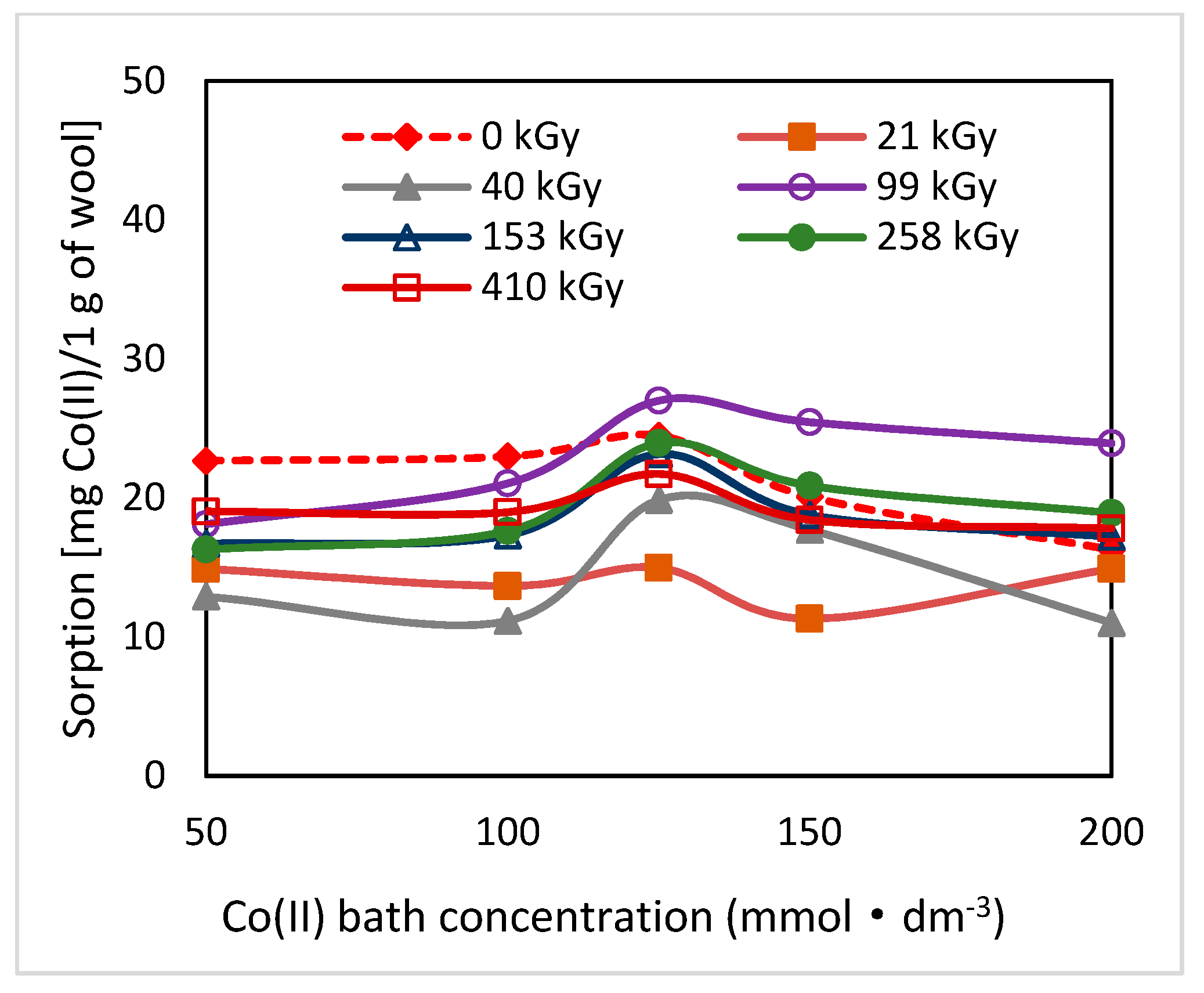
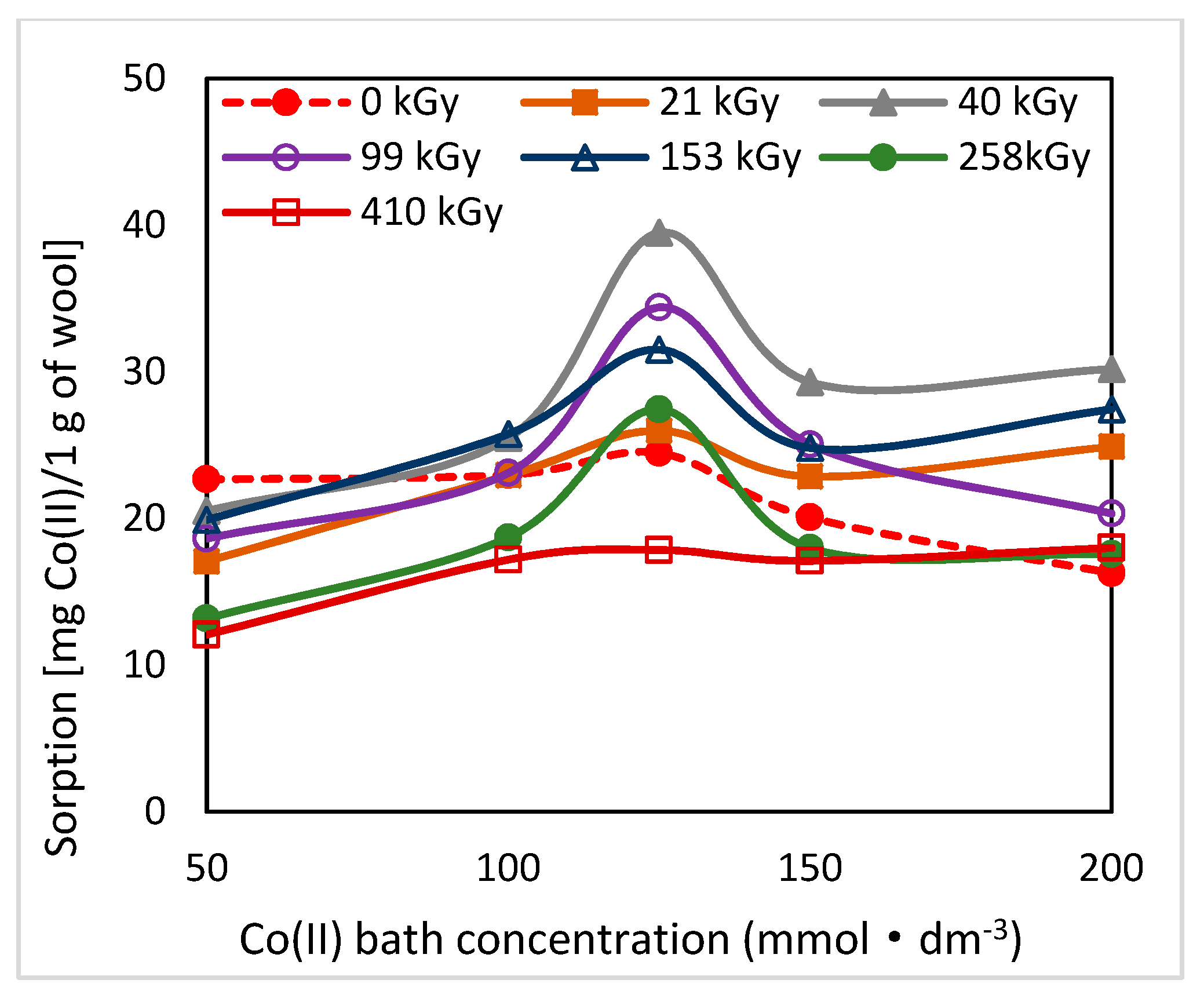
As can be seen from
Figure 1,
Figure 2 and
Figure 3, the related sorption isotherms and their mutual positions were indeed varied with time. The most variable sorption was measured for 2 days after the wool exposure. These data are consistent with content of the S-oxidized groups observed in irradiated wool at a 4-day interval from the exposure [
19]. The greatest change in the position of the sorption isotherms can be seen for doses of 410 and 40 kGy (
Figure 1,
Figure 2 and
Figure 3). While, after two days after the exposure the 410 kGy isotherm showed the highest sorption (
Figure 1), this gradually decreased over time (
Figure 2) and, after 100 days, its corresponding sorption was the lowest (
Figure 3). A considerable jump is also observed for the 40 kGy isotherm; from the lowest position after 2 days, it reached maximum after 100 days. The overall trend in the development of sorption by the irradiated samples suggests an initial lower sorption capacity than the non-irradiated wool excepting 410 kGy sample. The sorption of the exposed samples gradually increased and, at the end of the observation time for concentrations over 125 mmol·dm
−3, exceeded the sorption of the non-irradiated sample (
Figure 3). After 100 days, a maximum for concentration of 125 mmol·dm
−3 is visible on all isotherms (
Figure 1,
Figure 2 and
Figure 3), the least pronounced for 410 kGy sample. We believe that at this Co(II) concentration, extremely favorable conditions have been developed for keratin-Co(II) interaction. Since Co(II) as a weak Lewis acid tends to form complex forms, the amine and hydroxyl groups of keratin can act as the ligands for Co-carboxylate or Co-cysteinate complexes. Whereas the extreme occurs at all absorbed doses, including 0 kGy, it should not be a result of the absorbed dose but the concentration of Co(II), as the amount of the used wool sorbent was constant. To verify our hypothesis, we used Arginine to be a simplified model of complexing partner for Co (II) cation, since Arginine (
Figure 4) is abundantly present in wool keratin in amount around 10% [
20].
Arginine contains acidic group -COOH where Co(II) as the central ion replaces H+, but also basic ending guanine-group (-NH-C(=NH)-NH2), capable to provide non-binding electrons for coordination. Thus, Arginine with Co(II) may create an intrinsically complex salt, a chelatonate.
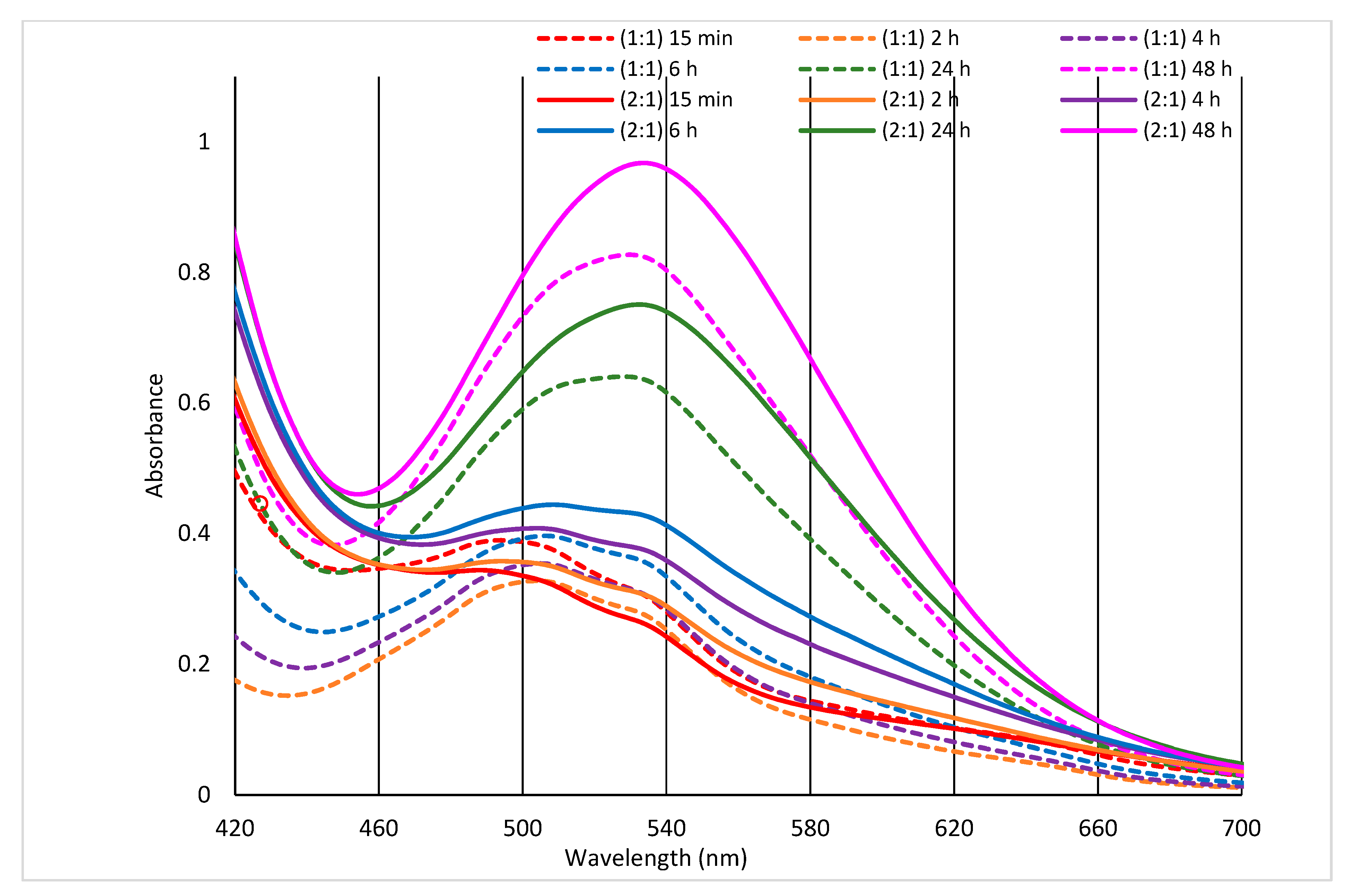
Arginine and Co(II) in mixed solution with 1:1 or 2:1 molar ratio were allowed to react, and time development of the reaction was monitored by VIS spectrometry. The time-dependent VIS spectrum is displayed in
Figure 5.
While the CoCl
2 spectrum itself is simple and smooth with λ
max = 512 nm (not displayed), the spectra of the mixed solutions are mildly rugged up to the interval of 6 h from the mixture (
Figure 5) indicating generation of transitive structures. Excepting the first 15 min, the absorbance of the 2:1 sample around 500 nm is always higher compared to the solution 1:1, although the Co(II) portion in the sample 2:1 is lower and, alone prevailing Arginine does not absorb in visible region at all. The shape variations of the spectra and the λ
max shifting characterize progressive building of a complex. Here, compared to the CoCl
2 spectrum, soft batochromic and hypsochromic but, namely hyperchromic shifts are observable and, the solution 2:1 of Arginine prevalence shows larger variations.
In principle the model reaction of Co(II) with Arginine can be a certain simplified simulation for the interaction of Co(II) with acid- and amine-groups in wool keratin when are positioned favorably. However, if in (non-)irradiated wool owing to any reasons, mainly spatial or structural ones, some ligand coming from other keratin chain occurs within force field of Co(II) carboxylate or cysteinate, the ligand can become constituent of the complex. In such case such crosslinking involves two or even more macro-chains. Similar situation arises when Co(II) reacts with two acid groups bound to different keratin chains by substitution mechanism. Chemical nodes shaped in such way obstruct diffusion of next sorbate into the fibers. Based on FTIR spectra the same fact has been documented for Cu(II) sorption on wool, too [
17]. In addition, as mentioned by Zhang et al. [
21], depending on the different coordination ability of the amino acids towards Co(II) based on different pH of medium, formation of the complexes can be various. As regards to adsorption studies, those reasons have been mentioned only rarely, if any. The aim is mostly to find optimal conditions enhancing sorption capacity without a deeper reasoning.
The decrease in sorption beyond the maximum for 125 mmol·dm
−3 and a further slight rise/fall demonstrates the dynamics of transformation of the intermediates corresponding to the individual absorbed doses. However, the main reason for the observed course (
Figure 3) is the formation of complex/complexes at higher Co(II) concentrations, which is not possible at insufficient concentration. The formation of complexes already on the fiber surfaces means hindered access of other Co(II)-ions inside the keratin fibers due to the mentioned cross-linking of the macro-chains. The Co(II)-ions can overcome such barrier for diffusion only due to increasing concentration. In comparison with Co(II), in the study of Cu(II) sorption, which is highly prone to form complexes, several sorption extremes on the irradiated wool were observed within the concentration of (12–80) mmol·dm
−3 [
17]. This points to the different coordination properties of Cu(II) and Co(II). As reported in papers [
22,
23,
24], the spatial arrangement of Cu-complexes is variable according to environmental conditions. Therefore, several maxima on the sorption dependence may correspond to several types of polyhedron. Similar variability for Co(II) was not observed in our conditions.
If the formation of the Co(II)-Arginine complex is time-dependent (
Figure 5), even more complicated course of the interaction must be in the wool. The changing process of Co(II)-sorption illustrates the relative time developing of the sorption compared to the sorption 2 days after the exposure (
Figure 6).
While sorption on the non-irradiated wool (0 kGy) did not change over time, the exposed wool showed time variations depending on both dose and Co(II) concentration. The sorption for samples with doses of (21–153) kGy achieved a higher sorption after 100 days than after 2 days, respectively, and the same could be said – except for 21 kGy sample – on 65-day´s lapse, too. Another situation appears for the samples with doses of 258 and 410 kGy; the corresponding sorption is smaller than after 2 days, with a lower level for 410 kGy sample. Based on our above mentioned observations, the wool with lapse of 100 days after the exposure and with absorbed dose of (40–153) kGy appears to be suitable for Co(II) sorption from bath with concentration above 50 mmol·dm−3.
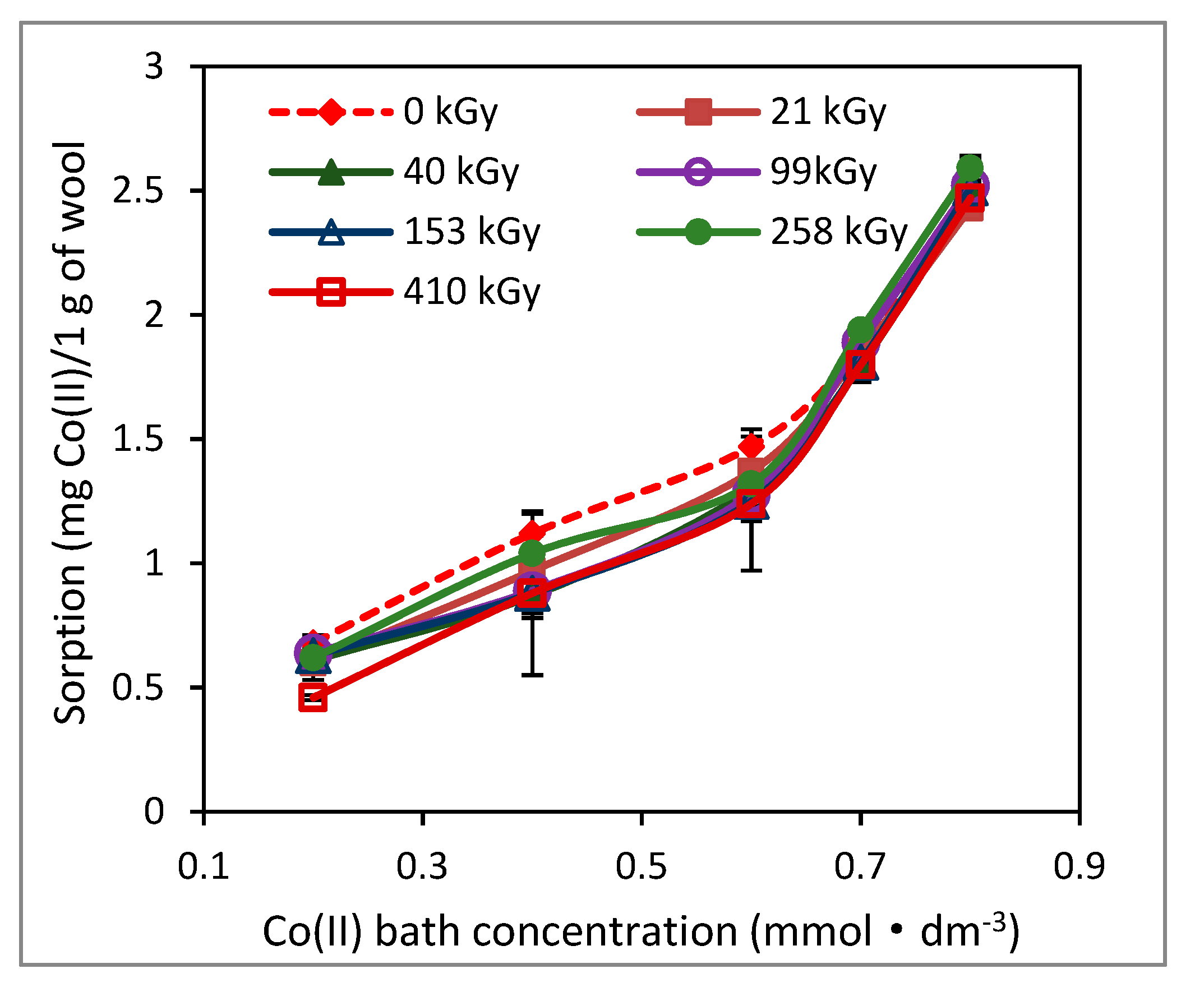
Most authors, if not all, examined (ad)sorption of Co(II) only for low (ad)sorbate concentrations. We also tested the sorption capacity of the irradiated wool for low Co(II) concentrations in the range (0.2–0.8) mmol·dm
−3 after 100 days (
Figure 7).
Of course, lower order Co (II) bath concentrations showed a correspondingly lower sorption. The isotherms for both the non-irradiated wool and the wool with different absorbed doses are virtually identical for such low concentrations. Our explanation is that a small amount of Co(II) is not enough to form Co(II) complexes and, the original carboxyl groups of keratin are sufficient even without the participation of cysteine forms in the irradiated samples. Therefore, no significant differences in the sorption are observed. Similar results for sorption of Pb(II), Cr(III) and Cd(II) on both irradiated and non-irradiated wools for concentrations up to 0.4 mmol·dm
−3 were also obtained by Hanzliková et al. [
16].
2.2. Effect of Post-Exposure Wool Heating on Co(II)-Sorption
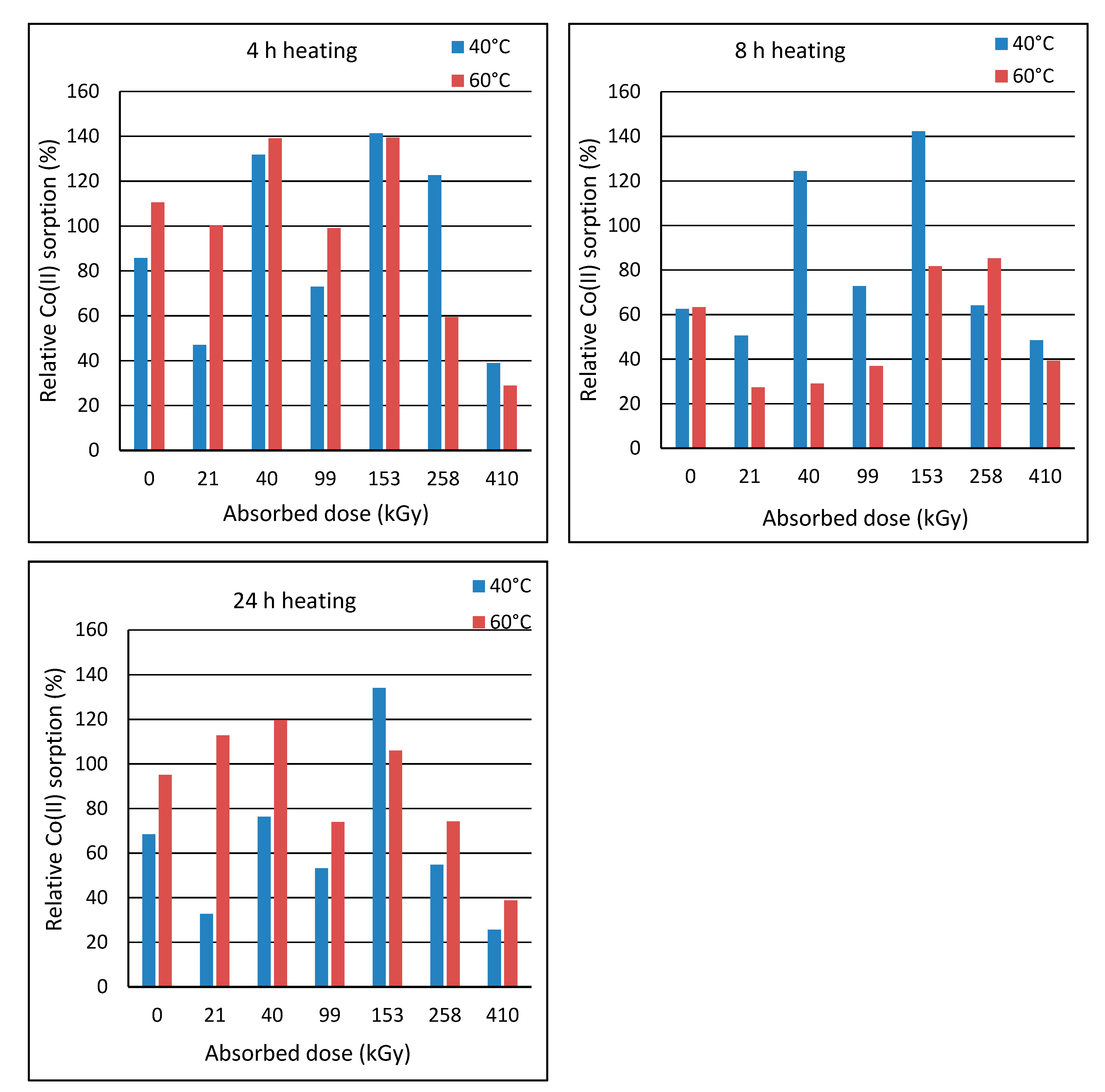
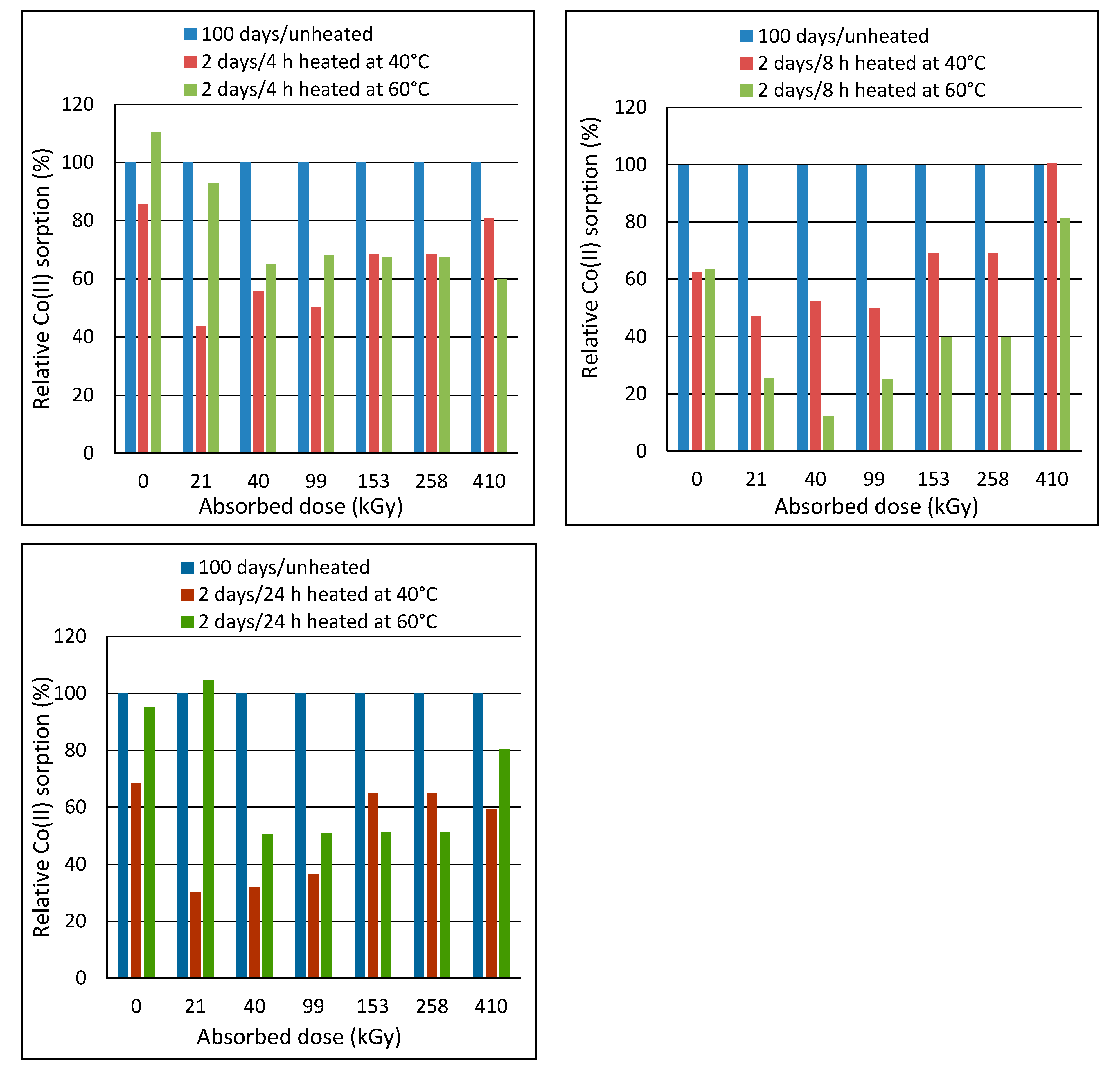
Post-exposure heating of the wool prior to sorption was motivated by an assumption that the heating could accelerate transformation of the intermediates to final cysteine acid. The wool with the least stable structure, i.e., of 2-day´s lapse from the irradiation, was heated to 40 °C or 60 °C in laboratory oven for 4 h, 8 h and 24 h. Then, these samples were subjected to the sorption experiments with the initial bath concentration of 150 mmol·dm
−3. The sorption results were compared with the sorption corresponding to the unheated sample (
Figure 8).
Figure 8 shows that the heating effect depends on the absorbed dose. Heating at 60 °C for 4 h was more effective for doses up to 153 kGy, and the heating at 40 °C was more effective for higher doses. The practical importance of the heating appears only for doses of 40 and 153 kGy with almost the same effect for both temperatures. The heating at 40 °C for 8 h improved sorption only for the wool with doses of 40 and 153 kGy. The other samples showed impaired sorption for 40 °C. All samples had worse sorption when heated to 60 °C, which is attributed to a gradual wool denaturation. The heating at 40 °C for 24 h showed a positive effect only on 153 kGy wool and the sorption improvement was practically the same as the heating at 40 °C for 4 h. Milder improvement in the sorption after the heating to 60 °C were observed only for 21, 40 and 153 kGy.
Period of 100 days was preliminary assessed as sufficient to transform the intermediates into a final form. To obtain information to what measure the post-exposure heating wool with 2-day´s lapse is equivalent to 100-day´s lapse, we compared Co(II) sorption on the unheated wool with 100-day´s lapse with the sorption on the wool heated 2 days after the exposure (
Figure 9).
The results of the comparison showed that the sorption of none from the heated exposed samples reached the sorption level of the unheated wool with 100-day´s lapse. The only, but only slight improvement was achieved by heating the native wool at 60 °C for 4 h. Only 410 kGy sample showed an equalizing the effect of the 8 h heating at 40 °C with 100-day´s lapse from the exposure. All other heat treatments worsened the sorption. The 24 h-heating at 60 °C resulted in a slight sorption improvement only for 21 kGy sample. None of the heat treatments achieved the effect of 100-day´s simple storage of the samples. Thus, the heating of the wool did not accelerate the transformation processes as expected.
Corresponding Co(II) sorption is an indirect summary indicator of processes passed involving, besides the chemical transformation of S-oxidized groups, changes in the configuration of keratin chains. The finding that conditioning wool at higher temperatures did not bring any significant improvement in sorption capacity is beneficial for potential practical applications, as simple storage without handling will simplify the manipulation and will not require energy consumption.
2.3. Effect of Lapse from Wool Exposure on pH of Aqueous Extract
The pH of medium has a considerable effect on the Co(II) sorption but, not only of this cation [
25]. In general, the pH value of protein environment is important factor because it determines value of isoelectric point varying around the pH~4 [
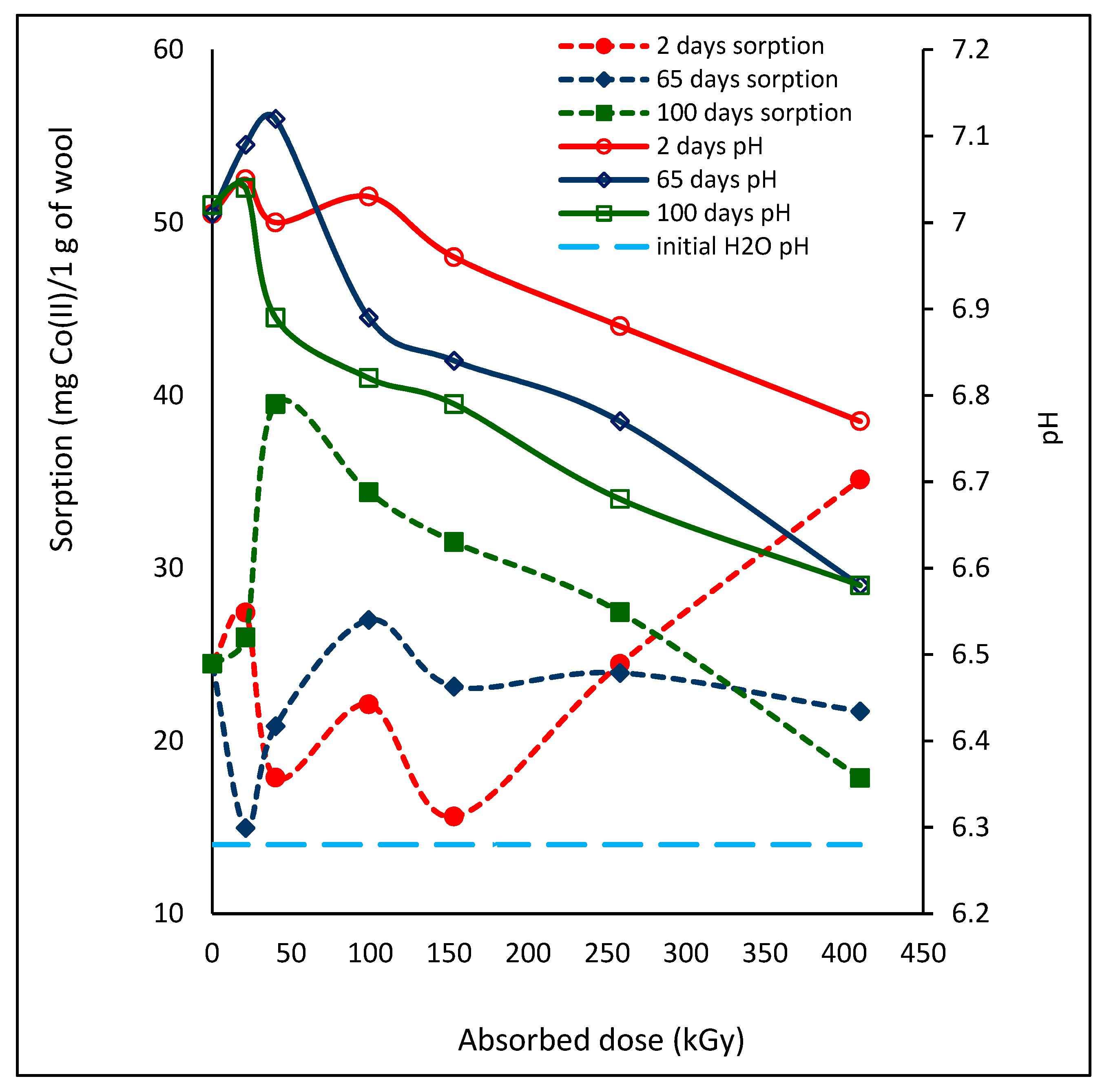
26]. To sorb any cation effectively, the process has to be conducted at pH above the wool isoelectric point. In such case the positively charged cation is not repelled by the positively charged surface but readily interacts with the negatively charged fiber surface. In order to better understand the Co(II) sorption process on the irradiated wool, we measured pH value of the aqueous extract of the wool samples after 24 h wool contact with deionized water at time intervals consistent with the measured Co(II) sorption. In our case, the pH of deionized water with initial value of 6.28 was increasing after contact with all wool samples and at each time interval from the exposure, although not monotonously (
Figure 10).
The largest changes in the pH of the aqueous extract showed wool samples at 65 days from the exposure, with one peak for 21 kGy and total change of 0.54 pH units. One extreme was also observed for 100 day samples, but for 40 kGy, with total span of 0.46 pH units. The samples with 2-day´s lapse showed qualitatively different pH course; although the overall pH change was the smallest, only 0.28 units, the curve had two extremes, for 21 and 99 kGy. Two peaks for the same doses are also shown by the corresponding sorption after 2 days and both dependencies go quasi in parallel up to 99 kGy and, reversely from 153 kGy. The 65-day´s lapse with one peak showed qualitatively mirror image for both dependencies up to 258 kGy dose. At the 100-day´s lapse is observed a small mutual shift in the maximum for both pH and sorption parameters (see 21 and 40 kGy), but only in this case, a reverse course did not occur; the both dependencies indicate a quasi-parallel development. Thus, with increasing lapse, the differences in character of the pH aqueous extracts and the Co(II) sorption are reduced. For the 100-day´s lapse it can be already said that the lower the pH, the lower the sorption. This fact is consistent with accepted view that the higher pH of isoelectric point, the more negative surface charge of the wool and the higher attraction for cations. Also, Wen et al. [
25] presented higher Co(II) adsorption on wool at pH~10 than at pH~8. However, when increasing the pH, the limiting factor for the sorption is possible precipitation of related cation from the solution.
The increasing pH in low dose wool extracts compared to the pH of deionized water means increasing concentration of OH
- and decreasing H
+, and vice versa. We attribute the decrease of H
+ in the extract to its binding by negatively charged groups of wool such as cystine oxides (cystine monoxide -SO-S- and cystine dioxide –SO
2-S-) and S-sulfonate (R-S-SO
3−), which corresponds to low absorbed doses being precursors of cysteine acid R- SO
3H [
18,
19]. As reported by Oae and Doi [
27], cystine oxides are strong hydrogen acceptors, reducing the presence of free H
+ in solution. In this case, the amount of OH
- must be predominant in the solution to keep the ionic product of water. Vice versa, acidification of the media is due to release of H
+ as a result of dissociation of the acidic groups initially present (R-COOH) as well as resulting from running transformation processes (R-SO
3H). Summarized, the development of the pH of the extract (
Figure 10) is a function of the corresponding absorbed dose and the wool chemical structure at a given time.