LC-MS Analysis of Serum for the Metabolomic Investigation of the Effects of Pulchinenoside b4 Administration in Monosodium Urate Crystal-Induced Gouty Arthritis Rat Model
Abstract
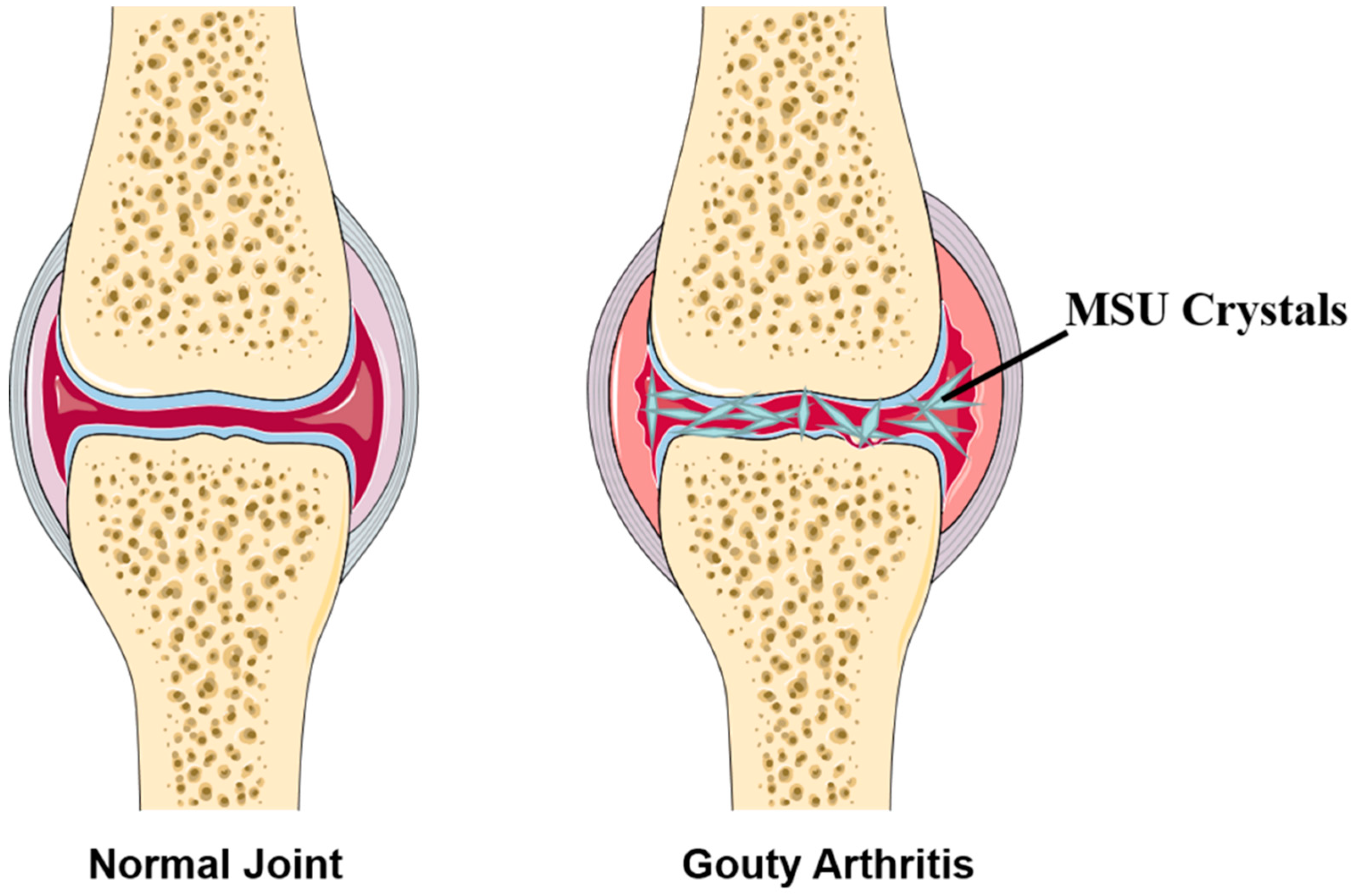
:1. Introduction
2. Results
2.1. The Major Pharmacodynamics Investigations (Anti-GA Ability) of P-b4
2.1.1. The Effect of Each Group on Toes/Ankle Swelling
2.1.2. The Effect of Each Group on Pain Threshold
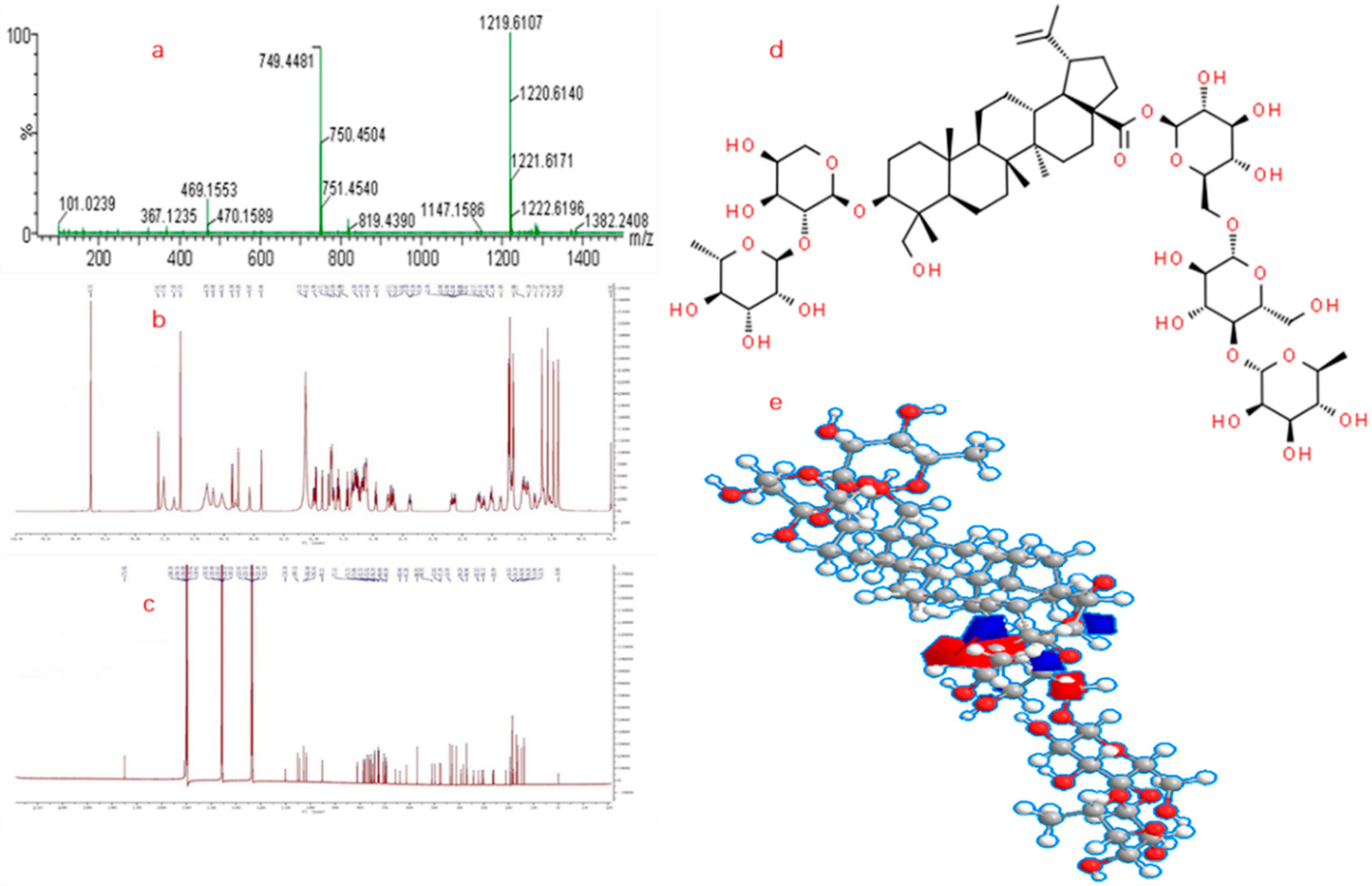
2.2. Validation of Analytical Method
2.3. Changes in Serum Metabolites of the MSU Crystal-Induced GA Rats after Being Treated by P-b4
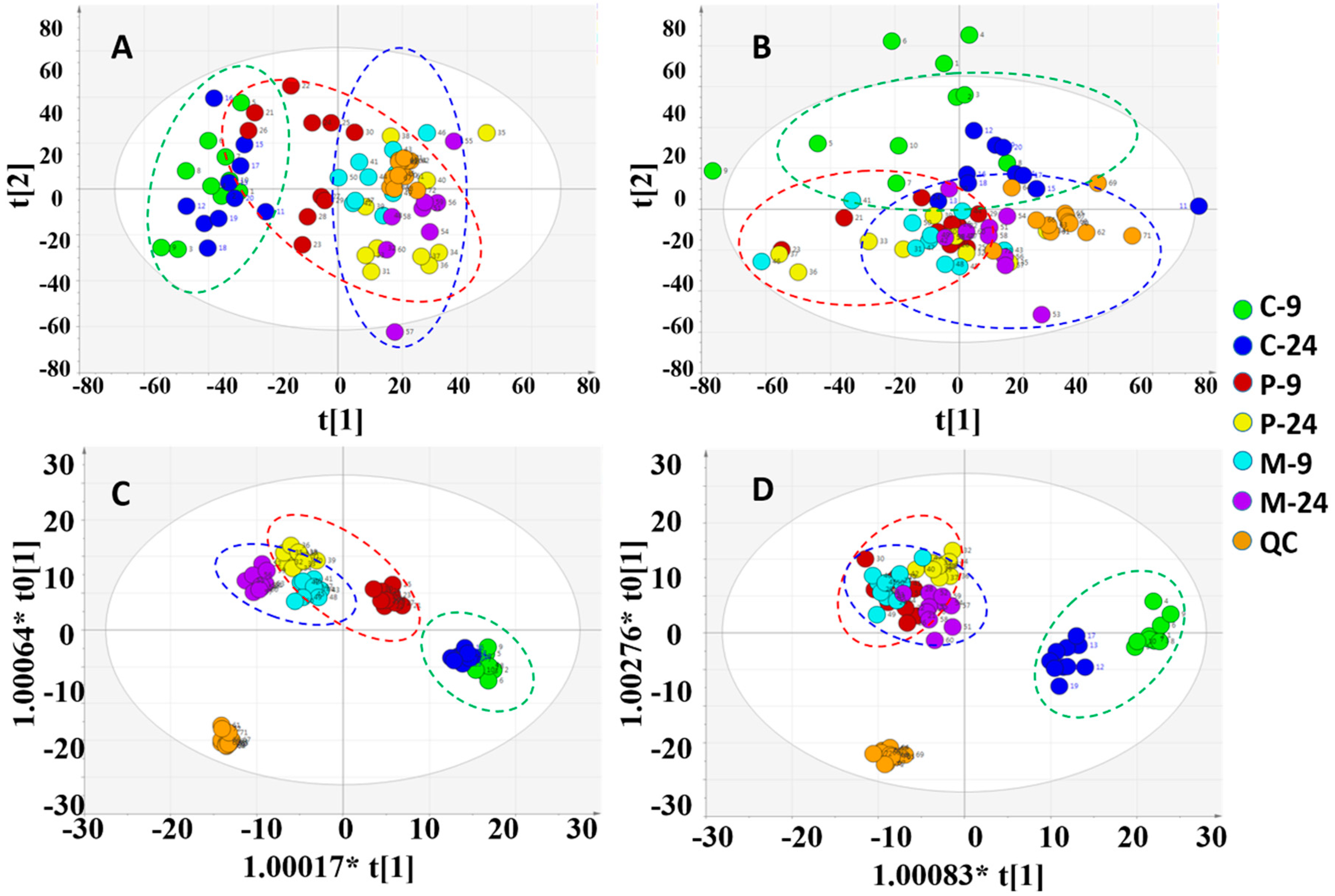
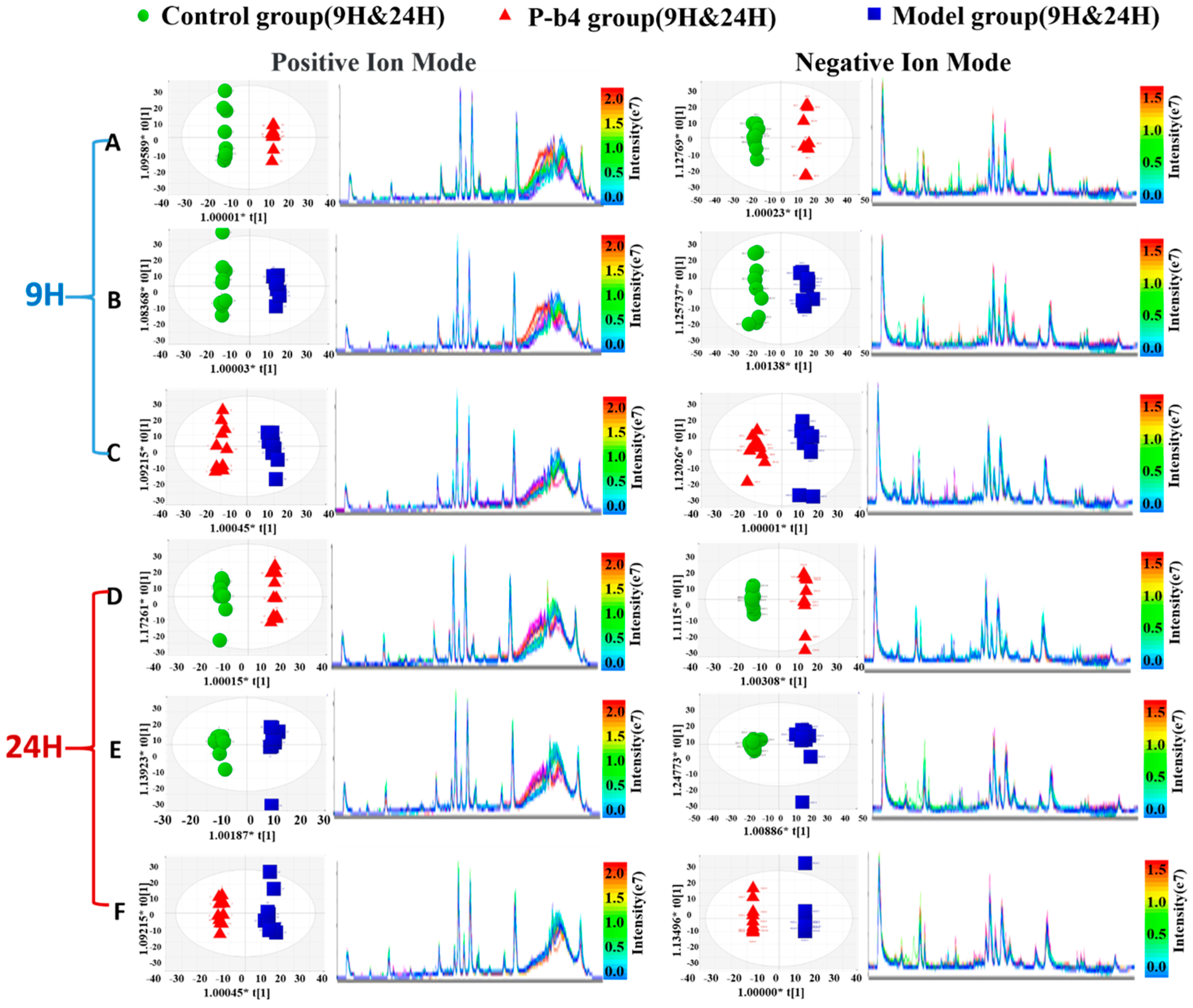
2.3.1. Multivariate Analysis of Rats Serum Metabolic Profile
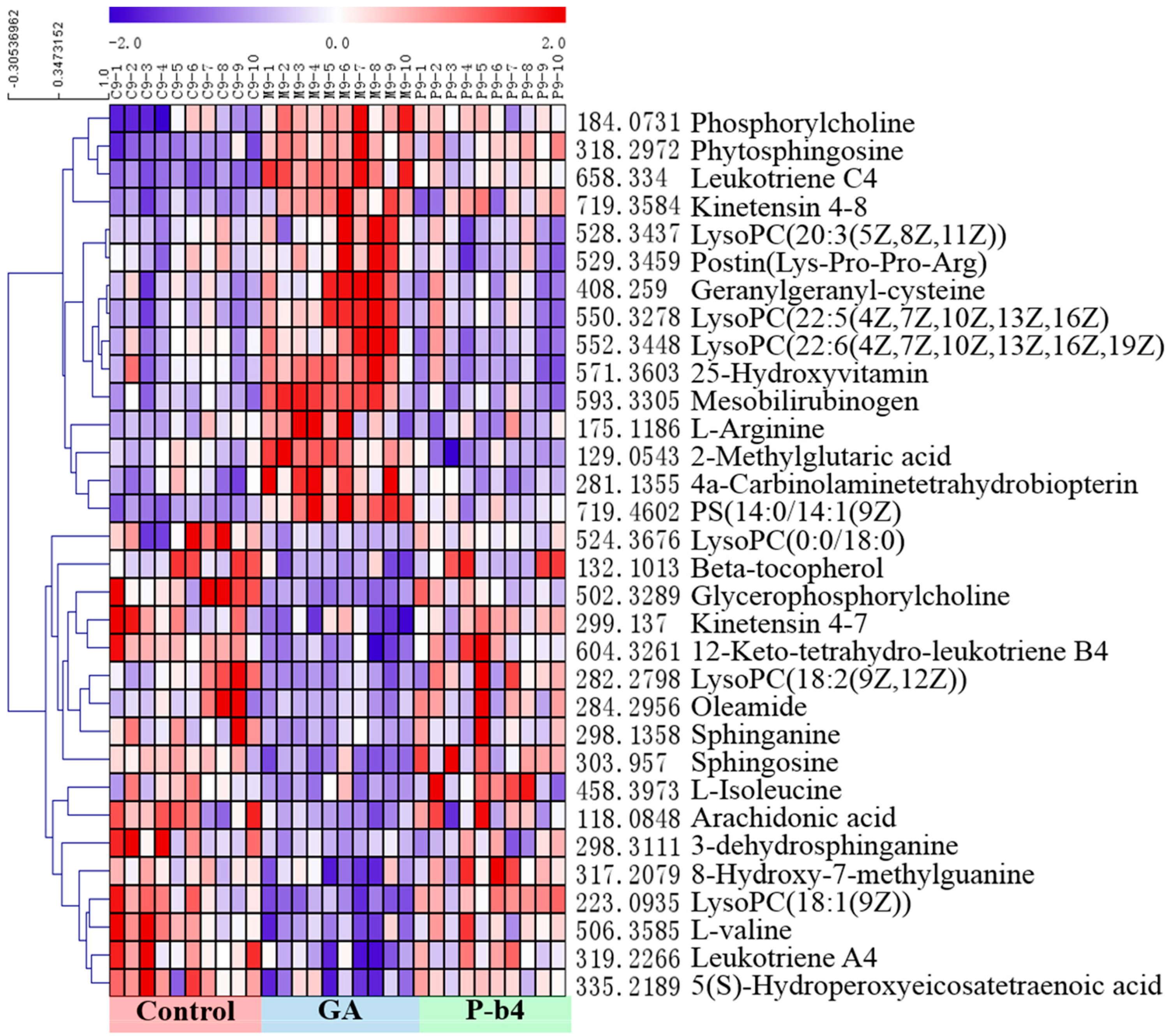
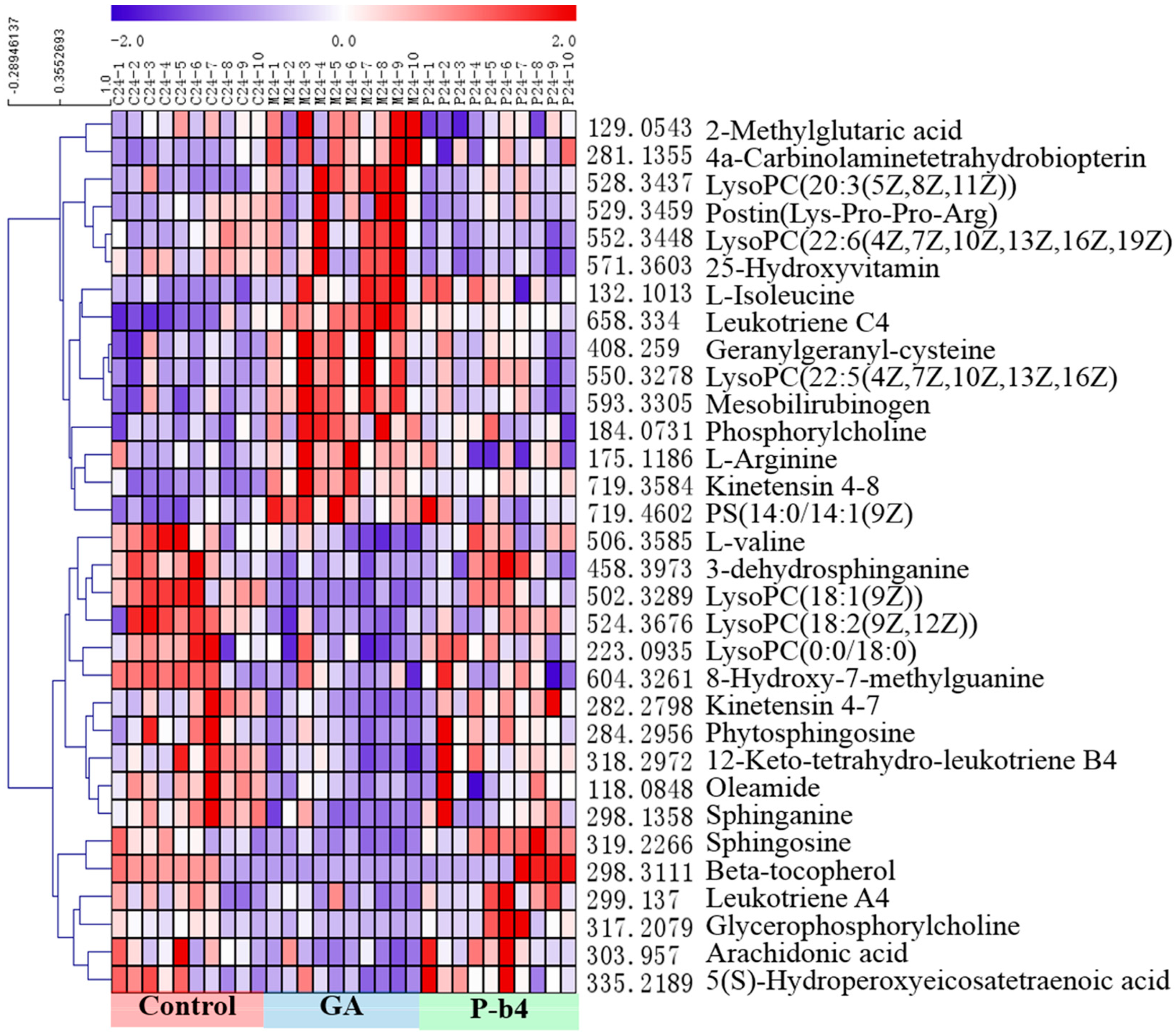
2.3.2. Identification of Potential Biomarkers for the Therapeutic Effect of p-b4 on MSU Crystal-Induced GA Rats
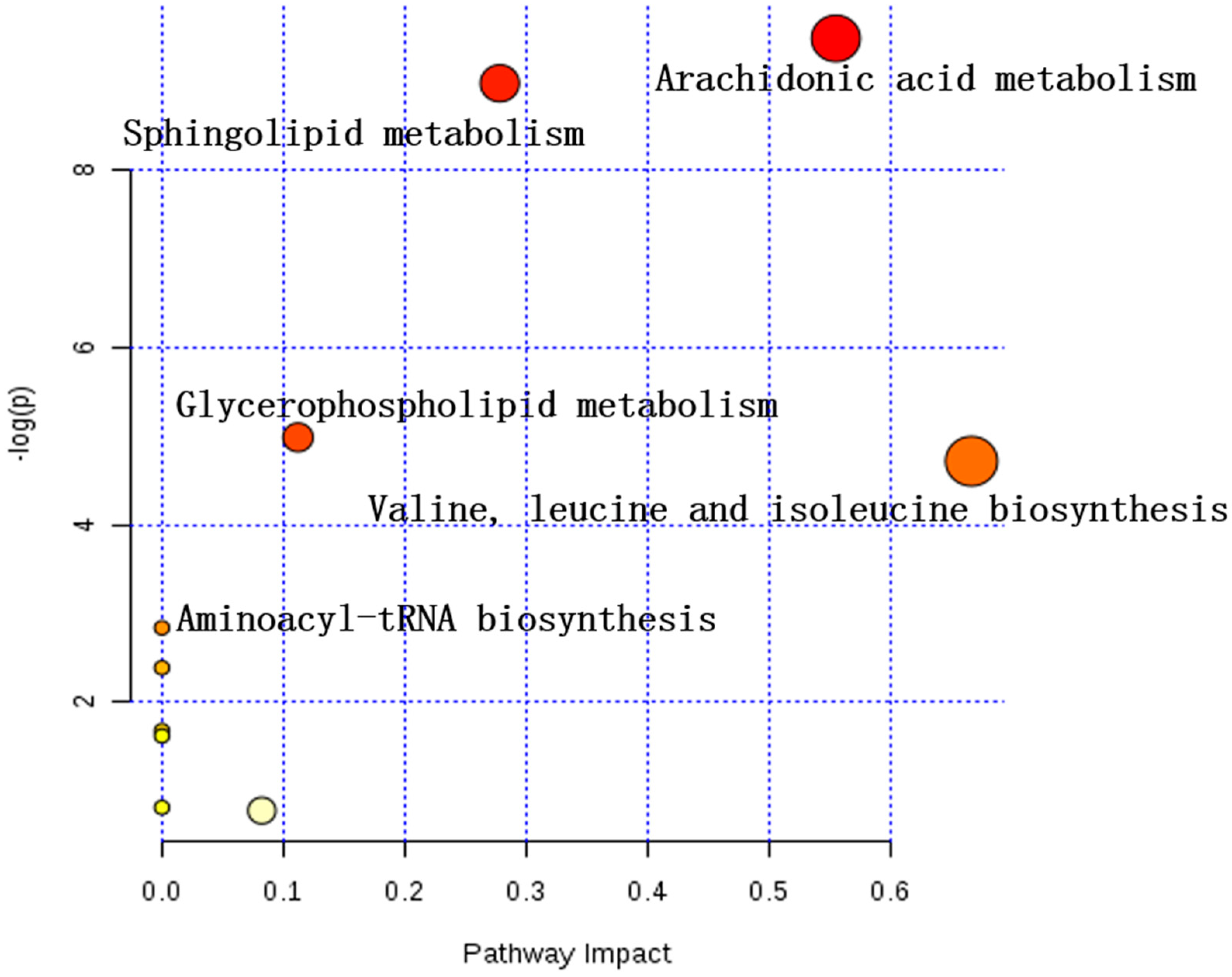
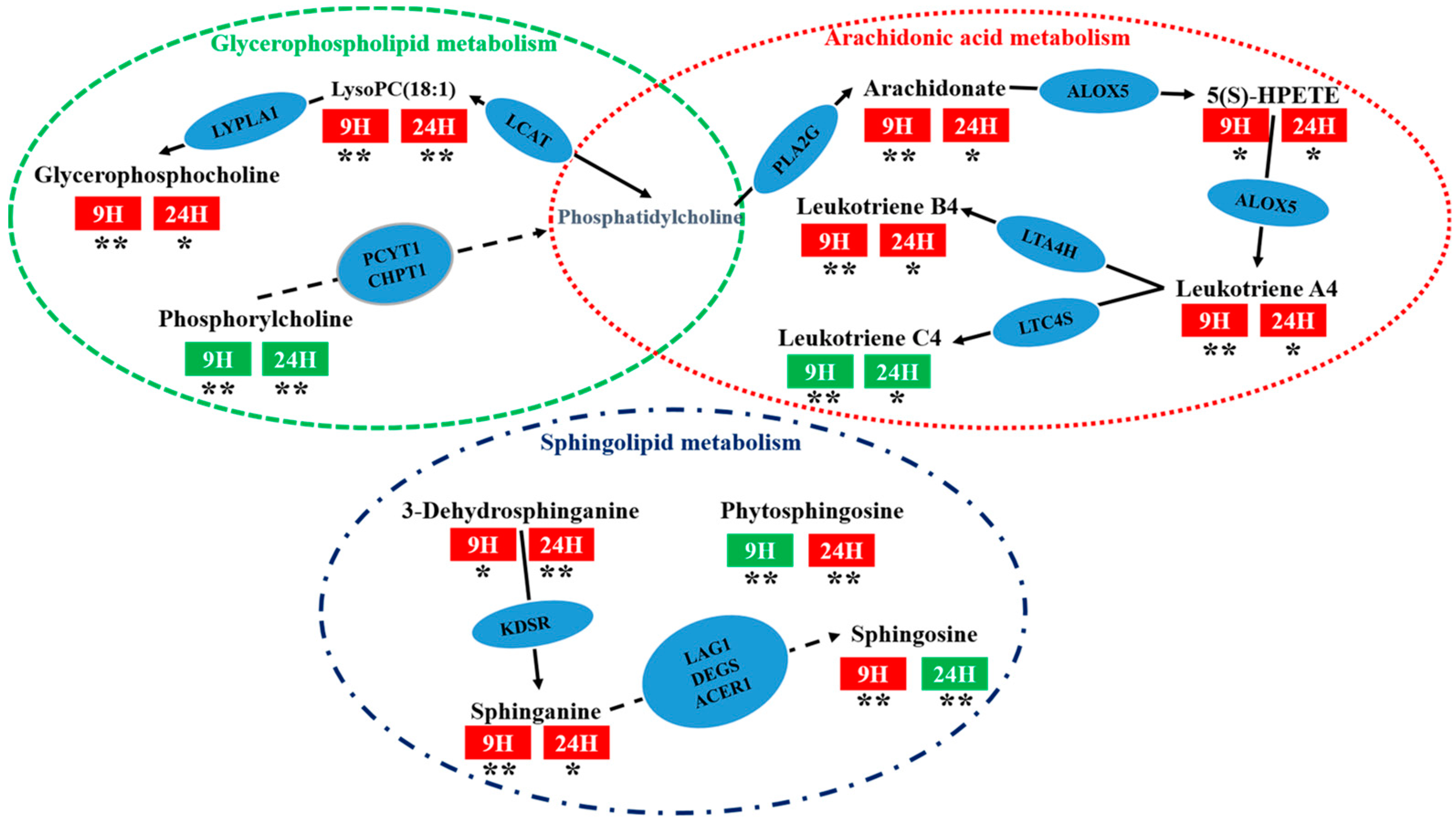
2.3.3. Analysis of Metabolic Pathways
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Monosodium Urate Crystal Preparation
4.3. Animal Handling for MSU Crystal-Induced GA Rats Model
4.4. Rat Serum Sample Collection and Pretreatment
4.5. LC-MS Conditions
4.6. Data Processing and Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Kuo, C.F.; Grainge, M.J.; Zhang, W.; Doherty, M. Global epidemiology of gout: Prevalence, incidence and risk factors. Nat. Rev. Rheumatol. 2015, 11, 649–662. [Google Scholar] [CrossRef]
- Neogi, T. Gout. N Engl. J. Med. 2011, 364, 443–452. [Google Scholar] [CrossRef]
- Richette, P.; Bardin, T. Gout. Lancet 2010, 375, 318–328. [Google Scholar] [CrossRef]
- Dalbeth, N.; Merriman, T.R.; Stamp, L.K. Gout. Lancet 2016, 388, 2039–2052. [Google Scholar] [CrossRef]
- Roddy, E.; Menon, A.; Hal, A.; Datta, P.; Packham, J. Polyarticular sonographic assessment of gout: A hospital-based cross-sectional study. Joint Bone Spine 2013, 80, 295–300. [Google Scholar] [CrossRef]
- He, M.Y.; Ouyang, H.; He, M.Z.; Tan, T.; Li, J.M.; Zhang, X.Y.; Jia, J.; Feng, Y.L.; Yang, S.L. Application of a liquid chromatography-tandem mass spectrometry method to the pharmacokinetics, tissue distribution and excretion in the study of anemoside B4, a novel antiviral agent candidate, in rats. Biomed. Chromatogr. 2017, 31, e3914. [Google Scholar] [CrossRef]
- Hu, Y.; Chen, X.; Duan, H.; Hu, Y.; Mu, X. Chinese herbal medicinal ingredients inhibit secretion of IL-6, IL-8, E-selectin and TXB(2) in LPS-induced rat intestinal microvascular endothelial cells. Immunopharm. Immunot. 2009, 31, 550–555. [Google Scholar] [CrossRef]
- Hu, Y.; He, K.; Wang, X. Role of Chinese herbal medicinal ingredients in secretion of cytokines by PCV2-induced endothelial cells. J. Immunot. 2016, 13, 141–147. [Google Scholar] [CrossRef]
- Liu, M.; Zhao, X.Z.; Xiao, L.; Liu, G.; Liu, H.Z.; Wang, X.Y.; Feng, X.; Lin, X.K. Cytotoxicity of the compounds isolated from Pulsatilla chinensis saponins and apoptosis induced by 23-hydroxybetulinic acid. Pharm. Bio. 2015, 53, 1–9. [Google Scholar] [CrossRef]
- So, A.K.; Martinon, F. Inflammation in gout: Mechanisms and therapeutic targets. Nat. Rev. Rheumatol. 2017, 13, 639–647. [Google Scholar] [CrossRef]
- Broz, P.; Dixit, V.M. Inflammasomes: Mechanism of assembly, regulation and signalling. Nat. Rev. Immunol. 2016, 16, 407–420. [Google Scholar] [CrossRef]
- Liu, Y.Z.; Duan, C.F.; Chen, H.L.; Wang, C.L.; Liu, X.X.; Qiu, M.; Tang, H.L.; Zhang, F.; Zhou, X.Y.; Yang, J. Inhibition of COX-2/mPGES-1 and 5-LOX in macrophages by leonurine ameliorates monosodium urate crystal-induced inflammation. Toxicolo Appl. Pharmacol. 2018, 351, 1–11. [Google Scholar] [CrossRef]
- Thottam, G.E.; Krasnokutsky, S.; Pillinger, M.H. Gout and Metabolic Syndrome: A Tangled Web. Curr. Rheumatol. Rep. 2017, 19. [Google Scholar] [CrossRef]
- Eva, M.L.; Ian, D.W. Analytical strategies in metabonomics. J. Proteome Res. 2007, 1, 443. [Google Scholar]
- Lyu, S.; Yang, S.L.; Rao, Y.; Feng, Y.L. Application of metabolomics and related technologies in the R&D field of traditional Chinese medicine. China J. Chin. Mater. Med. 2018, 43, 4182–4191. [Google Scholar]
- Fu, H.Y.; Guo, X.M.; Zhang, Y.M.; Song, J.J.; Zheng, Q.X.; Liu, P.P.; Lu, P.; Chen, Q.S.; Yu, Y.J.; She, Y.B. AntDAS: Automatic Data Analysis Strategy for UPLC-QTOF-Based Nontargeted Metabolic Profiling Analysis. Anal. Chem. 2017, 89, 11083–11090. [Google Scholar] [CrossRef]
- Zhang, X.Y.; Li, J.M.; Xie, B.; Wu, B.; Lei, S.; Yao, Y.; He, M.Z.; Ouyang, H.; Feng, Y.L.; Xu, W.; et al. Comparative Metabolomics Analysis of Cervicitis in Human Patients and a Phenol Mucilage-Induced Rat Model Using Liquid Chromatography Tandem Mass Spectrometry. Front. Pharmacol. 2018, 9. [Google Scholar] [CrossRef] [Green Version]
- Akkaya-Ulum, Y.Z.; Balci-Peynircioglu, B.; Karadag, O.; Eroglu, F.K.; Kalyoncu, U.; Kiraz, S.; Ertenli, A.I.; Özen, S.; Yilmaz, E. Alteration of the microRNA expression profile in familial Mediterranean fever patients. Clin. Exp. Rheumatol. 2017, 35, S90–S94. [Google Scholar]
- Chong, J.; Soufan, O.; Li, C.; Caraus, I.; Li, S.; Bourque, G.; Wishart, D.S.; Xia, J. MetaboAnalyst 4.0: Towards more transparent and integrative metabolomics analysis. Nucl. Acids Res. 2018, 46, W486–W494. [Google Scholar] [CrossRef]
- Muangchan, C.; Bhurihirun, T. An investigation of the independent risk factors that differentiate gout from pseudogout in patients with crystal-induced acute arthritis: A cross-sectional study. Rheumatol. Int. 2017, 38, 89–95. [Google Scholar] [CrossRef]
- Finkenstaedt, T.; Manoliou, A.; Toniolo, M.; Higashigaito, K.; Andreisek, G.; Guggenberger, R.; Michel, B.; Alkadhi, H. Gouty arthritis: The diagnostic and therapeutic impact of dual-energy CT. Eur. Radiol. 2016, 26, 3989–3999. [Google Scholar] [CrossRef]
- Simon Taylor, R. BET 1: Prednisolone for the treatment of acute gouty arthritis. Emerg. Med. J. 2017, 34, 687–689. [Google Scholar] [CrossRef]
- Van Walsem, A.; Pandhi, S.; Nixon, R.M.; Guyot, P.; Karabis, A.; Moore, R.A. Relative benefit-risk comparing diclofenac to other traditional non-steroidal anti-inflammatory drugs and cyclooxygenase-2 inhibitors in patients with osteoarthritis or rheumatoid arthritis: A network meta-analysis. Arthritis Res. Ther. 2015, 17. [Google Scholar] [CrossRef]
- Nonaka, F.; Migita, K.; Haramura, T.; Sumiyoshi, R.; Kawakami, A.; Eguchi, K. Colchicine-responsive protracted gouty arthritis with systemic inflammatory reactions. Mod. Rheumatol. 2014, 24, 540–543. [Google Scholar] [CrossRef]
- Chen, Q.; Zhang, X.T.; Xiong, Y.M.; Chen, C.; Lv, S.M. The CD25+/CD4+ T cell ratio and levels of CII, CIX and CXI antibodies in serum may serve as biomarkers of pristane-induced arthritis in rats and Rheumatoid Arthritis in humans. Comp. Biochem. Phys. C 2019, 217, 25–31. [Google Scholar] [CrossRef]
- Otsubo, H.; Tsuneyoshi, Y.; Nakamura, T.; Matsuda, T.; Komiya, S.; Matsuyama, T. Serum-soluble folate receptor β as a biomarker for the activity of rheumatoid arthritis synovitis and the response to anti-TNF agents. Clin. Rheumatol. 2018. [Google Scholar] [CrossRef]
- Kamiya, Y.; Kawada, J.; Kawano, Y.; Torii, Y.; Kawabe, S.; Iwata, N.; Ito, Y. Serum microRNAs as Potential Biomarkers of Juvenile Idiopathic Arthritis. Clin. Rheumatol. 2015, 34, 1705–1712. [Google Scholar] [CrossRef]
- Daniela, C.; Kun, L.; Punit, S.; Ihor, B.; Eleftherios, P.D.; Vinod, C. Quantitative tandem mass-spectrometry of skin tissue reveals putative psoriatic arthritis biomarkers. Clin. Proteom. 2015, 12. [Google Scholar] [CrossRef]
- Couderc, M.; Peyrode, C.; Pereira, B.; Miot-Noirault, E.; Mathieu, S.; Soubrier, M.; Dubost, J. Comparison of several biomarkers (MMP-2, MMP-9, the MMP-9 inhibitor TIMP-1, CTX-II, calprotectin, and COMP) in the synovial fluid and serum of patients with and without septic arthritis. Joint Bone Spine 2019, 86, 261–262. [Google Scholar] [CrossRef]
- Imagama, T.; Tokushige, A.; Seki, K.; Seki, T.; Nakashima, D.; Ogasa, H.; Sakai, T.; Taguchi, T. Early diagnosis of septic arthritis using synovial fluid presepsin: A preliminary study. J. Infect. Chemother. 2019, 25, 170–174. [Google Scholar] [CrossRef]
- Wang, W.; Yang, G.J.; Zhang, J.; Chen, C.; Jia, Z.Y.; Li, J.; Xu, W.D. Plasma, urine and ligament tissue metabolite profiling reveals potential biomarkers of ankylosing spondylitis using NMR-based metabolic profiles. Arthritis Res. Ther. 2016, 18. [Google Scholar] [CrossRef]
- Stefano, A.; Dario, B.; Barbara, T.; Laura, B.; Luca, P.; Maria, R.; Domenico, B.; Anna, L.; Giusy, P.; Francesco, F.; et al. Differential synovial tissue biomarkers among psoriatic arthritis and rheumatoid factor/anti-citrulline antibody-negative rheumatoid arthritis. Arthritis Res. Ther. 2019, 21. [Google Scholar] [CrossRef]
- Meng, H.; Liu, Y.; Lai, L.H. Diverse ways of perturbing the human arachidonic acid metabolic network to control inflammation. Accounts Chem. Res. 2015, 48, 2242–2250. [Google Scholar] [CrossRef]
- Jung, S.; Kim, M.; Ryu, H.J.; Chae, J.S.; Lee, S.H.; Lee, J.H. Age-related increase in LDL-cholesterol is associated with enhanced oxidative stress and disturbed sphingolipid metabolism. Metabolomics 2014, 11, 40–49. [Google Scholar] [CrossRef]
- Gika, H.G.; Theodoridis, G.A.; Wingate, J.E.; Wilson, I.D. Within-day reproducibility of an HPLC-MS-based method for metabonomic analysis: Application to human urine. J. Proteome Res. 2007, 6, 3291–3303. [Google Scholar] [CrossRef]
- Zou, L.; Zhang, Y.; Li, W.; Zhang, J.M.; Wang, D.; Fu, J.; Wang, P. Comparison of Chemical Profiles, Anti-Inflammatory Activity, and UPLC-Q-TOF/MS-Based Metabolomics in Endotoxic Fever Rats between Synthetic Borneol and Natural Borneol. Molecules 2017, 22, 1446. [Google Scholar] [CrossRef]
- Zheng, T.T.; Su, S.L.; Dai, X.X.; Zhang, L.W.; Duan, J.M.; OuYang, Z. Metabolomic Analysis of Biochemical Changes in the Serum and Urine of Freund’s Adjuvant-Induced Arthritis in Rats after Treatment with Silkworm Excrement. Molecules 2018, 23, 1490. [Google Scholar] [CrossRef]
- Hang, H.; Robinson, P.N.; Wang, K. Phenolyzer: Phenotype-based prioritization of candidate genes for human diseases. Nat. Methods 2015, 12, 841–843. [Google Scholar]
Sample Availability: Samples of the compounds P-b4 is available from the authors. |
| Groups | Toes and Ankle Swelling (mL) | ||||
| 3 h | 6 h | 9 h | 12 h | 24 h | |
| Control | −0.13 ± 0.12 ** | −0.16 ± 0.10 ** | −0.16 ± 0.09 ** | −0.16 ± 0.10 ** | −0.18 ± 0.14 ** |
| GA Model | 0.16 ± 0.11 | 0.51 ± 0.22 | 0.95 ± 0.25 | 0.99 ± 0.22 | 0.47 ± 0.16 |
| Diclofenac Sodium | 0.10 ± 0.14 | 0.32 ± 0.14 * | 0.51 ± 0.15 ** | 0.44 ± 0.17 ** | 0.08 ± 0.19 ** |
| P-b4 (hypodermic, 20 mg × 4) | 0.13 ± 0.11 | 0.44 ± 0.13 | 0.83 ± 0.20 | 0.84 ± 0.23 | 0.29 ± 0.15 * |
| P-b4 (hypodermic, 10 mg × 4) | 0.11 ± 0.14 | 0.37 ± 0.18 | 0.69 ± 0.24 * | 0.73 ± 0.27 * | 0.46 ± 0.23 |
| P-b4 (hypodermic, 5 mg × 4) | 0.09 ± 0.12 | 0.41 ± 0.17 | 0.76 ± 0.29 | 0.79 ± 0.29 * | 0.39 ± 0.12 |
| P-b4 (intravenous, 1.2 5mg × 4) | 0.11 ± 0.13 | 0.42 ± 0.17 | 0.68 ± 0.21 * | 0.70 ± 0.14 ** | 0.27 ± 0.16 ** |
| Groups | Pain Threshold (s) | ||||
| 3 h | 6 h | 9 h | 12 h | 24 h | |
| Control | 10.16 ± 3.05 | 9.00 ± 1.80 * | 8.56 ± 1.49 | 8.08 ± 0.91 * | 9.12 ± 2.27 |
| GA Model | 9.27 ± 2.25 | 7.17 ± 1.95 | 7.93 ± 1.95 | 6.86 ± 1.17 | 9.56 ± 1.32 |
| Diclofenac Sodium | 9.96 ± 1.58 | 7.52 ± 2.71 | 8.18 ± 1.63 | 8.11 ± 2.46 | 8.87 ± 1.64 |
| P-b4 (hypodermic, 20 mg × 4) | 10.10 ± 1.81 | 7.07 ± 1.09 | 6.80 ± 1.82 | 8.32 ± 2.44 * | 10.27 ± 2.38 |
| P-b4 (hypodermic, 10 mg × 4) | 9.36 ± 1.90 | 8.22 ± 2.01 | 8.24 ± 2.35 | 8.08 ± 1.92 * | 11.01 ± 2.44 * |
| P-b4 (hypodermic, 5 mg × 4) | 11.57 ± 1.94 * | 8.02 ± 2.62 | 8.70 ± 2.07 | 7.19 ± 1.51 | 9.63 ± 2.38 |
| P-b4 (intravenous, 1.25 mg × 4) | 10.73 ± 2.09 | 8.21 ± 1.75 | 10.13 ± 2.78 * | 7.16 ± 0.97 | 9.81 ± 2.24 |
| NO. | Metabolites | RT | M.F | p-Value (9 h) | p-Value (24 h) | Trend | |||
|---|---|---|---|---|---|---|---|---|---|
| M-9 VS P-9 | C-9 VS M-9 | M-24 VS P-24 | C-24 VS M-24 | ||||||
| 1 | 2-Methylglutaric acid | 9.62 | C6H10O4 | 1.95 × 10−2 | 3.43 × 10−3 | ↓ | ↑ | ↓ | ↑ |
| 2 | l-Isoleucine | 5.29 | C6H13NO2 | 1.62 × 10−3 | 3.40 × 10−3 | ↑ | ↓ | ↓ | ↑ |
| 3 | l-Arginine | 11.90 | C6H14N4O2 | 3.97 × 10−3 | 4.65 × 10−3 | ↓ | ↑ | ↓ | ↑ |
| 4 | Phosphorylcholine | 12.55 | C5H15NO4P | 2.38 × 10−5 | 3.27 × 10−4 | ↓ | ↑ | ↓ | ↑ |
| 5 | 8-Hydroxy-7-methylguanine | 11.98 | C6H7N5O2 | 1.95 × 10−2 | 2.88 × 10−2 | ↑ | ↓ | ↑ | ↓ |
| 6 | 4a-Carbinolaminetetrahydrobiopterin | 9.62 | C9H13N5O3 | 3.11 × 10−4 | 2.14 × 10−3 | ↓ | ↑ | ↓ | ↑ |
| 7 | Oleamide | 12.44 | C18H35NO | 1.18 × 10−2 | 2.18 × 10−3 | ↑ | ↓ | ↑ | ↓ |
| 8 | Sphinganine | 12.78 | C18H39NO2 | 5.74 × 10−3 | 2.24 × 10−2 | ↑ | ↓ | ↑ | ↓ |
| 9 | Glycerophosphorylcholine | 22.46 | C8H20NO6P | 9.67 × 10−3 | 4.06 × 10−2 | ↑ | ↓ | ↑ | ↓ |
| 10 | Phytosphingosine | 9.73 | C18H39NO3 | 1.79 × 10−4 | 3.26 × 10−3 | ↓ | ↑ | ↑ | ↓ |
| 11 | Geranylgeranyl-cysteine | 11.04 | C23H37NO3S | 3.96 × 10−6 | 5.79 × 10−4 | ↓ | ↑ | ↓ | ↑ |
| 12 | Beta-tocopherol | 20.28 | C28H48O2 | 1.50 × 10−2 | 8.25 × 10−3 | ↑ | ↓ | ↑ | ↓ |
| 13 | LysoPC(18:2(9Z,12Z)) | 11.43 | C26H50NO7P | 9.71 × 10−12 | 1.68 × 10−7 | ↑ | ↓ | ↑ | ↓ |
| 14 | LysoPC(18:1(9Z)) | 16.79 | C26H52NO6P | 2.33 × 10−5 | 1.85 × 10−3 | ↑ | ↓ | ↑ | ↓ |
| 15 | LysoPC(0:0/18:0) | 16.81 | C26H46NO7P | 8.62 × 10−4 | 2.67 × 10−4 | ↑ | ↓ | ↑ | ↓ |
| 16 | LysoPC(20:3(5Z,8Z,11Z)) | 16.79 | C28H50NO7P | 5.53 × 10−8 | 2.95 × 10−4 | ↓ | ↑ | ↓ | ↑ |
| 17 | Postin(Lys-Pro-Pro-Arg) | 12.42 | C22H40N8O5 | 8.69 × 10−4 | 4.03 × 10−2 | ↓ | ↑ | ↓ | ↑ |
| 18 | LysoPC(22:5(4Z,7Z,10Z,13Z,16Z) | 11.98 | C30H52NO7P | 6.29 × 10−6 | 8.63 × 10−4 | ↓ | ↑ | ↓ | ↑ |
| 19 | LysoPC(22:6(4Z,7Z,10Z,13Z,16Z,19Z) | 11.34 | C30H50NO7P | 9.13 × 10−7 | 1.43 × 10−3 | ↓ | ↑ | ↓ | ↑ |
| 20 | 25-Hydroxyvitamin | 11.98 | C34H52O8 | 3.63 × 10−5 | 1.74 × 10−3 | ↓ | ↑ | ↓ | ↑ |
| 21 | Mesobilirubinogen | 11.98 | C33H44N4O6 | 6.44 × 10−7 | 3.91 × 10−7 | ↓ | ↑ | ↓ | ↑ |
| 22 | Kinetensin 4–7 | 4.82 | C26H37N9O6 | 5.52 × 10−3 | 1.88 × 10−5 | ↑ | ↓ | ↑ | ↓ |
| 23 | Leukotriene C4 | 4.82 | C30H47N3O9S | 2.05 × 10−12 | 3.87 × 10−2 | ↓ | ↑ | ↓ | ↑ |
| 24 | Kinetensin 4–8 | 4.89 | C35H46N10O7 | 2.05 × 10−5 | 4.71 × 10−5 | ↓ | ↑ | ↓ | ↑ |
| 25 | PS(14:0/14:1(9Z) | 20.85 | C34H64NO10P | 3.67 × 10−9 | 7.77 × 10−4 | ↓ | ↑ | ↓ | ↑ |
| 26 | l-valine | 9.94 | C5H11NO2 | 3.49 × 10−2 | 1.15 × 10−2 | ↑ | ↓ | ↑ | ↓ |
| 27 | Sphingosine | 5.61 | C18H37NO2 | 6.97 × 10−3 | 4.00 × 10−5 | ↑ | ↓ | ↓ | ↑ |
| 28 | 12-Keto-tetrahydro-leukotriene B4 | 12.93 | C20H34O4 | 3.00 × 10−4 | 1.10 × 10−2 | ↑ | ↓ | ↑ | ↓ |
| 29 | 3-dehydrosphinganine | 18.69 | C18H37NO2 | 4.08 × 10−2 | 1.44 × 10−3 | ↑ | ↓ | ↑ | ↓ |
| 30 | Arachidonic acid | 19.99 | C20H32O2 | 9.44 × 10−4 | 1.51 × 10−2 | ↑ | ↓ | ↑ | ↓ |
| 31 | Leukotriene A4 | 13.40 | C20H30O3 | 5.12 × 10−3 | 1.90 × 10−2 | ↑ | ↓ | ↑ | ↓ |
| 32 | 5(S)-Hydroperoxyeicosatetraenoic acid | 10.93 | C20H32O4 | 3.85 × 10−2 | 3.48 × 10−2 | ↑ | ↓ | ↑ | ↓ |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lyu, S.; Ding, R.; Liu, P.; OuYang, H.; Feng, Y.; Rao, Y.; Yang, S. LC-MS Analysis of Serum for the Metabolomic Investigation of the Effects of Pulchinenoside b4 Administration in Monosodium Urate Crystal-Induced Gouty Arthritis Rat Model. Molecules 2019, 24, 3161. https://doi.org/10.3390/molecules24173161
Lyu S, Ding R, Liu P, OuYang H, Feng Y, Rao Y, Yang S. LC-MS Analysis of Serum for the Metabolomic Investigation of the Effects of Pulchinenoside b4 Administration in Monosodium Urate Crystal-Induced Gouty Arthritis Rat Model. Molecules. 2019; 24(17):3161. https://doi.org/10.3390/molecules24173161
Chicago/Turabian StyleLyu, Shang, Ruowen Ding, Peng Liu, Hui OuYang, Yulin Feng, Yi Rao, and Shilin Yang. 2019. "LC-MS Analysis of Serum for the Metabolomic Investigation of the Effects of Pulchinenoside b4 Administration in Monosodium Urate Crystal-Induced Gouty Arthritis Rat Model" Molecules 24, no. 17: 3161. https://doi.org/10.3390/molecules24173161




