A New Series of EDOT Based Co-Sensitizers for Enhanced Efficiency of Cocktail DSSC: A Comparative Study of Two Different Anchoring Groups
Abstract
:1. Introduction
2. Experimental Section
2.1. General
2.2. Solar Cell Fabrication and Photovoltaic Characterization
2.3. Procedures for the Synthesis of CSGR Dyes
3. Results and Discussion
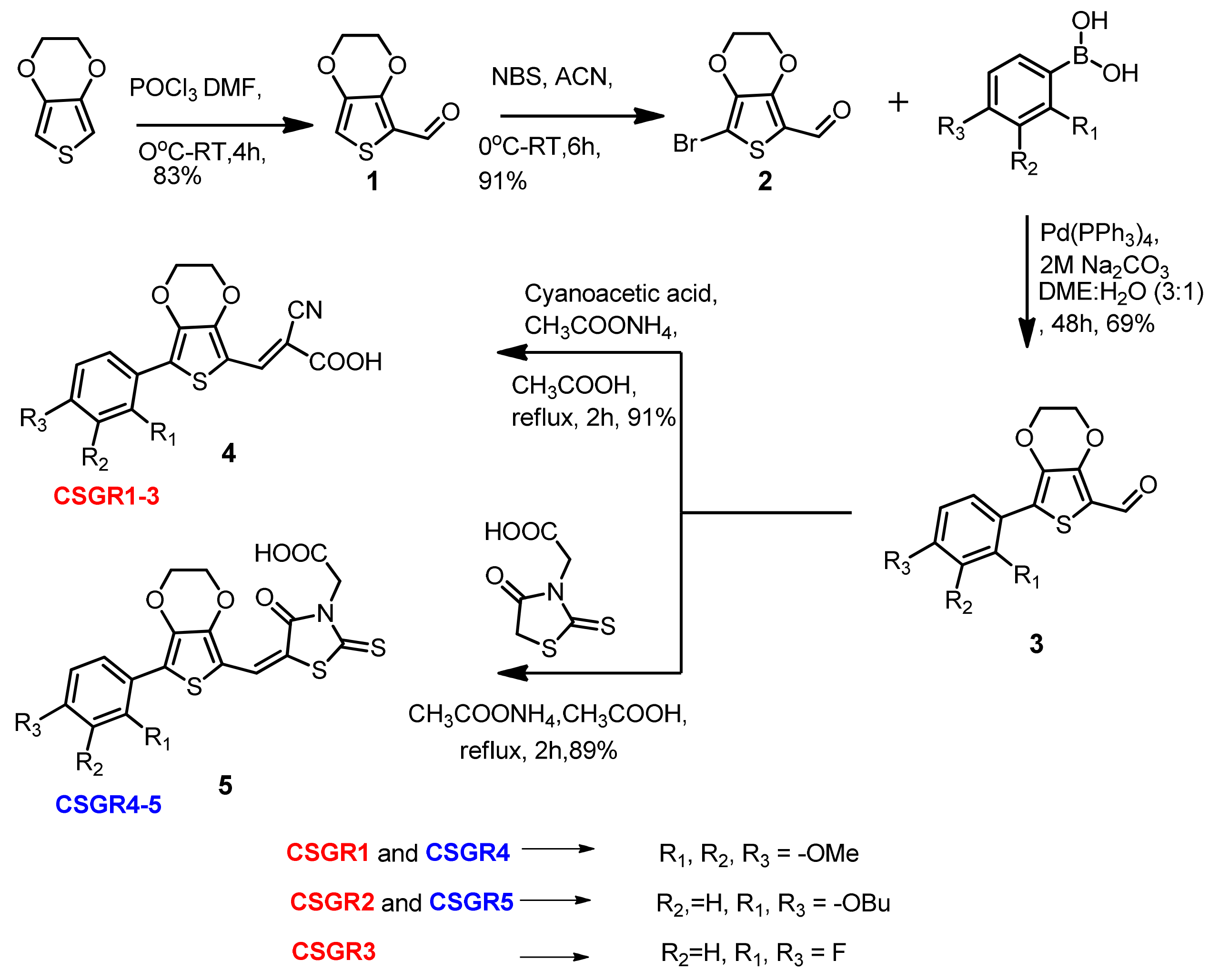
3.1. Synthesis
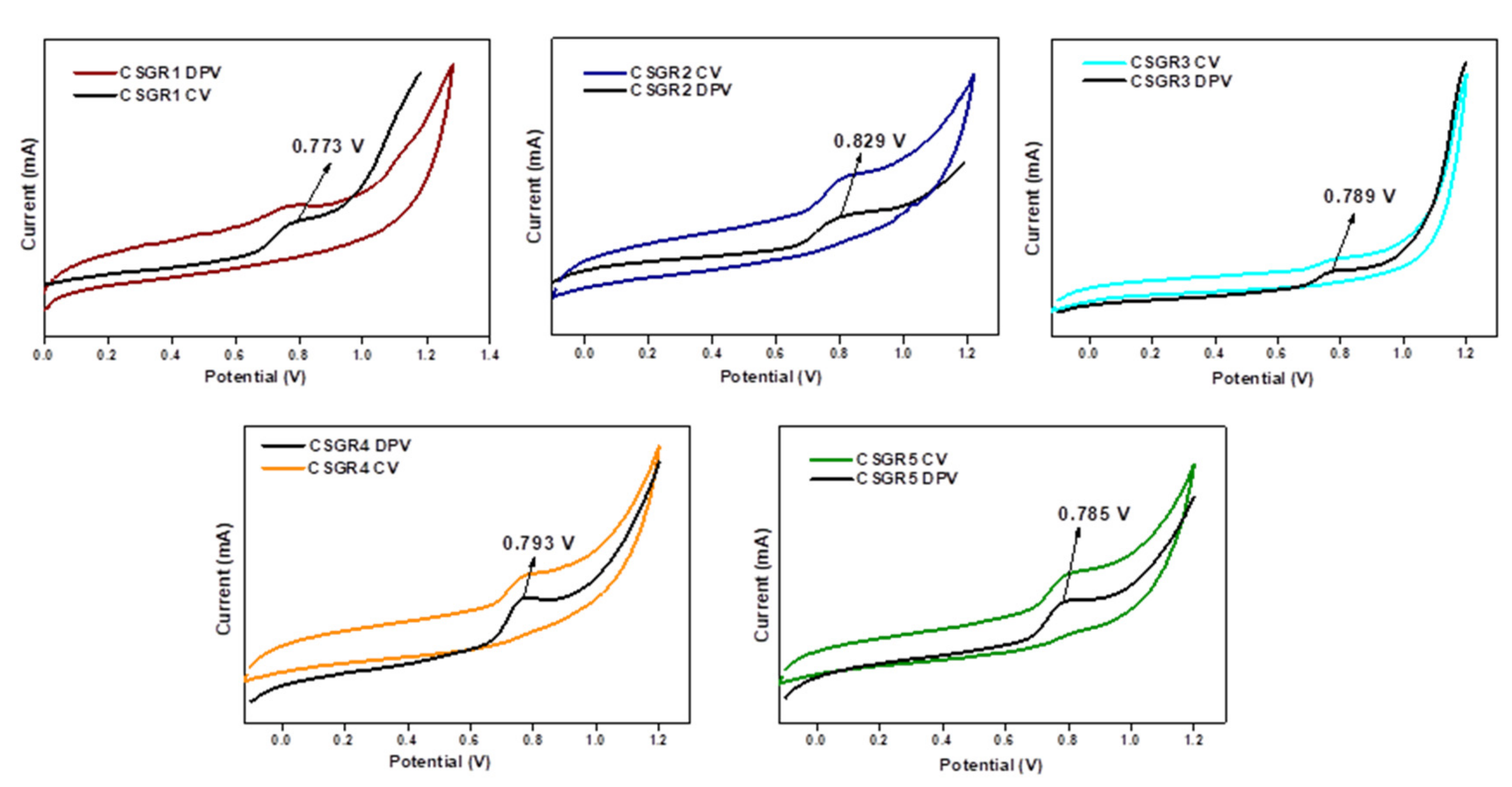
3.2. Optical and Electrochemical Measurements
3.3. Computational Studies
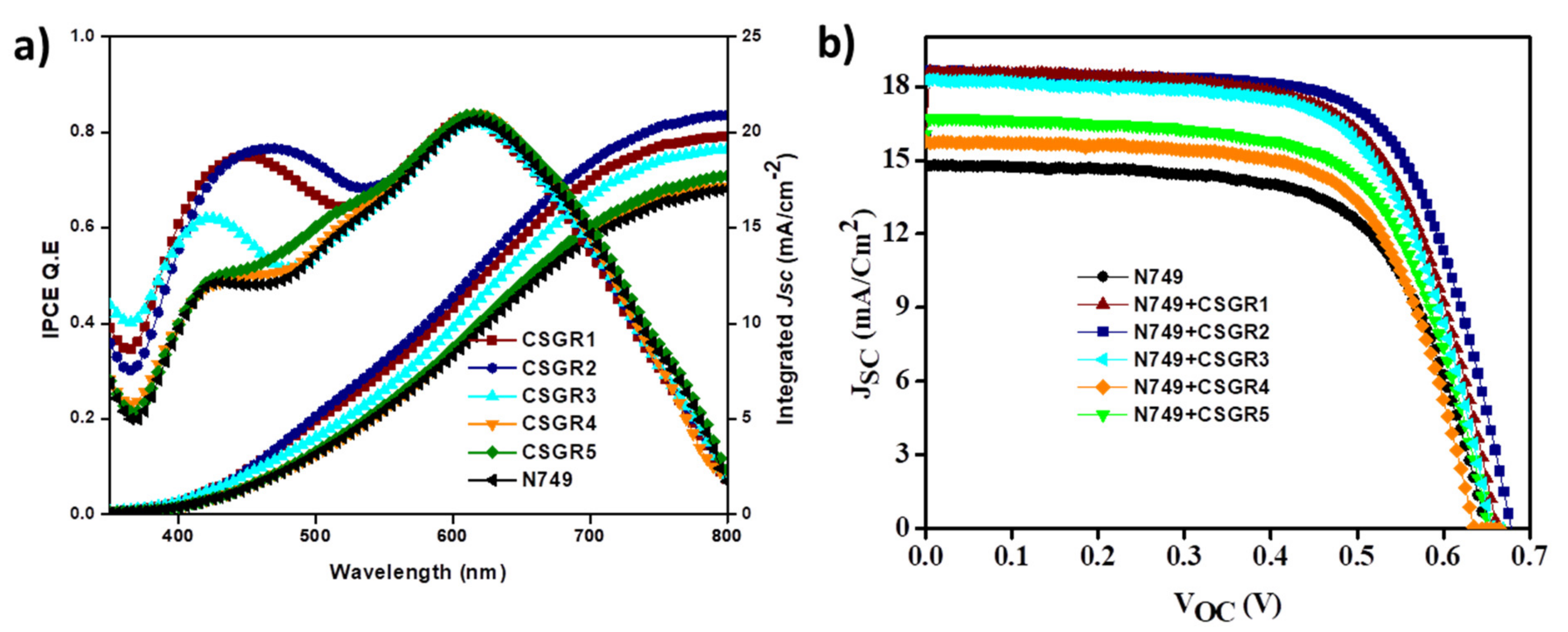
3.4. Photovoltaic Properties
3.5. Dye Loading Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Badawy, W.A. A review on solar cells from Si-single crystals to porous materials and quantum dots. J. Adv. Res. 2015, 6, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Matthew, S.M.; Niva, A.R.; Guillermo, C.B.; Richard, H.F. Understanding energy loss in organic solar cells: Toward a new efficiency regime. Joule 2018, 2, 25–35. [Google Scholar]
- Antonio, C.; Borbone, F.; Roberto, C. Research progress on photosensitizers for DSSC. Front. Chem. 2018, 6, 481. [Google Scholar]
- Green, M.A.; Ho-Baillie, A. Perovskite solar cells: The birth of a new era in photovoltaics. ACS Energy Lett. 2017, 2, 822–830. [Google Scholar] [CrossRef]
- Ravindiran, M.; Praveenkumar, C. Status review and the future prospects of CZTS based solar cell—A novel approach on the device structure and material modeling for CZTS based photovoltaic device. Renew. Sustain. Energy Rev. 2018, 94, 317–329. [Google Scholar] [CrossRef]
- Mehmood, U.; Rahman, S.U.; Harrabi, K.; Hussein, I.A.; Reddy, B.V.S. Recent advances in dye sensitized solar cells. Adv. Mater. Sci. Eng. 2014, 2014, 974782. [Google Scholar] [CrossRef]
- Luceño-Sánchez, J.A.; Díez-Pascual, A.M.; Capilla, R.P. Materials for photovoltaics: State of art and recent developments. Int. J. Mol. Sci. 2019, 20, 976. [Google Scholar] [CrossRef] [PubMed]
- O’Regan, B.; Grätzel, M. High-efficiency solar cell based on dye-sensitized colloidal TiO2 films. Nature 1991, 353, 737–740. [Google Scholar] [CrossRef]
- Yella, A.; Lee, H.W.; Tsao, H.N.; Yi, C.; Chandiran, A.K.; Nazeeruddin, M.K. Porphyrin-sensitized solar cells with Cobalt (II/III)–based redox electrolyte exceed 12 percent efficiency. Science 2011, 334, 629–634. [Google Scholar] [CrossRef]
- Mathew, S.; Yella, A.; Gao, P.; Humphry-Baker, R.; Curchod, B.F.; Ashari-Astani, N. Dye-sensitized solar cells with 13% efficiency achieved through the molecular engineering of porphyrin sensitizers. Nat. Chem. 2014, 6, 242–247. [Google Scholar] [CrossRef] [Green Version]
- Kakiage, K.; Aoyama, Y.; Yano, T.; Oya, K.; Fujisawa, J.; Hanaya, M. Highly-efficient dye-sensitized solar cells with collaborative sensitization by silyl-anchor and carboxy-anchor dyes. Chem. Commun. 2015, 51, 15894–15897. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, S.C.; Hagfeldt, A.; Soman, S. Resurgence of DSC with copper electrolyte: A detailed investigation of interfacial charge dynamics with cobalt and iodine based electrolytes. J. Mater. Chem. A 2018, 6, 22204–22214. [Google Scholar] [CrossRef]
- Zhang, L.; Cole, J.M. Dye aggregation in dye-sensitized solar cells. J. Mater. Chem. A 2017, 5, 19541–19559. [Google Scholar] [CrossRef] [Green Version]
- Naik, P.; Su, R.; Babu, D.D.; Ei-Shafei, A.; Adhkari, A.V. Structurally simple D-A-type organic sensitizers for dye-sensitized solar cells: Effect of anchoring moieties on the cell performance. J. Iran. Chem. Soc. 2017, 14, 2457–2466. [Google Scholar] [CrossRef]
- Aghazad, S.; Nazeeruddin, M.K. Ruthenium complexes as sensitizers in dye-sensitized solar cells. Inorganics 2018, 6, 52. [Google Scholar] [CrossRef]
- Yum, J.H.; Baranoff, E.; Wenger, S.; Nazeeruddin, M.K.; Gratzel, M. Panchromatic engineering for dye-sensitized solar cells. Energy Environ. Sci. 2011, 4, 842–857. [Google Scholar] [CrossRef]
- Ganesh, K.; Pavan, K.C.H.; Paolo, S.; Gabriele, M.; Maria, G.L.; Olivia, B.; Filippo, D.A.; Chandrasekharam, M. New terpyridine-based ruthenium complexes for dye sensitized solar cells applications. Inorg. Chim. Acta 2016, 442, 158–166. [Google Scholar]
- Clifford, J.N.; Martinez-Ferrero, E.; Viterisi, A.; Palomares, E. Sensitizer molecular structure-device efficiency relationship in dye sensitized solar cells. Chem. Soc. Rev. 2011, 40, 1635–1646. [Google Scholar] [CrossRef] [PubMed]
- Salvatori, P.; Agrawal, S.; Barreddi, C.; Chandrasekharam, M.; de Borniol, M.; Angelis, F.D. Stability of ruthenium/organic dye co-sensitized solar cells: A joint experimental and computational investigation. RSC Adv. 2014, 4, 57620–57628. [Google Scholar] [CrossRef]
- Timofey, N.C.; Ekaterina, A.K.; Ellie, T.; Vadim, V.P.; Ludmila, V.M.; Neil, R.; Oleg, A.R. [1,2,5]Thiadiazolo[3,4-d]Pyridazine as an internal acceptor in the D-A-π-A organic sensitizers for dye-sensitized solar cells. Molecules 2019, 24, 1588. [Google Scholar]
- Connell, A.; Holliman, P.J.; Davies, M.L.; Gwenin, C.D.; Weiss, S.; Pitak, M.B.; Horton, P.N.; Coles, S.J.; Cooke, G. A study of dye anchoring points in half-squarylium dyes for dye-sensitized solar cells. J. Mater. Chem. A 2014, 2, 4055–4066. [Google Scholar] [CrossRef] [Green Version]
- Connell, A.; Holliman, P.J.; Jones, E.W.; Furnell, L.; Kershaw, C.; Davies, M.L.; Gwenin, C.D.; Pitak, M.B.; Coles, S.J.; Cooke, G. Multiple linker half-squarylium dyes for dye-sensitized solar cells; are two linkers better than one? J. Mater. Chem. A 2015, 3, 2883–2894. [Google Scholar] [CrossRef]
- Burke, A.; Schmidt-Mende, L.; Ito, S.; Gratzel, M. A novel blue dye for near-IR ‘dye-sensitised’ solar cell applications. Chem. Commun. 2007, 3, 234–236. [Google Scholar] [CrossRef] [PubMed]
- Geiger, T.; Kuster, S.; Yum, J.-H.; Moon, S.-J.; Nazeeruddin, M.K.; Gratzel, M.; Nuesch, F. Molecular design of unsymmetrical squaraine dyes for high efficiency conversion of low energy photons into electrons using TiO2 nanocrystalline films. Adv. Funct. Mater. 2009, 19, 2720–2727. [Google Scholar] [CrossRef]
- Yen, Y.-S.; Chou, H.-H.; Chen, Y.-C.; Hsu, C.-Y.; Lin, J.T. Recent developments in molecule-based organic materials for dye-sensitized solar cells. J. Mater. Chem. 2012, 22, 8734–8747. [Google Scholar] [CrossRef]
- Son, C.H.; Suresh, T.; Chitumalla, R.K.; Koyyada, G.; Cheruku, R.; Jang, J.; Jung, J.H.; Kim, J.H. Synthesis and investigation of anchoring unit effect in blue-colored isoindigo-based D-A-π-A organic dyes for dye-sensitized solar cells. Jpn. J. Appl. Phys. 2018, 57, 122302. [Google Scholar] [CrossRef]
- Bao, L.Q.; Thogiti, S.; Koyyada, G.; Kim, J.H. Synthesis and photovoltaic performance of novel ullazine-based organic dyes for dye-sensitized solar cells. Jpn. J. Appl. Phys. 2019, 58, 012011. [Google Scholar] [CrossRef]
- Lee, C.P.; Li, C.T.; Ho, K.C. Use of organic materials in dye-sensitized solar cells. Mater. Today 2017, 20, 267–283. [Google Scholar] [CrossRef]
- Adewale, O.A.; Peter, A.A. Towards the development of functionalized polypyridine ligands for Ru(II) complexes as photosensitizers in dye-sensitized solar cells (DSSCs). Molecules 2014, 19, 12421–12460. [Google Scholar]
- Abdalhadi, S.M.; Connell, A.; Zhang, X.; Wiles, A.A.; Davies, M.L.; Holliman, P.J.; Cooke, G. Convenient synthesis of EDOT-based dyes by C-H activation and their application as dyes in dye sensitized solar cells. J. Mater. Chem. A 2016, 4, 15655–15661. [Google Scholar] [CrossRef]
- Al-Eid, M.; Lim, S.H.; Park, K.-W.; Fitzpatrick, B.; Han, C.-H.; Kwak, K.; Hong, J.; Cooke, G. Facile synthesis of metal-free organic dyes featuring a thienylethynyl spacer for dye sensitized solar cells. Dyes Pigments 2014, 104, 197–204. [Google Scholar] [CrossRef]
- Bella, F.; Gerbaldi, C.; Barolo, C.; Gratzel, M. Aqueous dye-sensitized solar cells. Chem. Soc. Rev. 2015, 44, 3431–3473. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kurotobi, K.; Toude, Y.; Kawamoto, K.; Fujimori, Y.; Ito, S.; Chabera, P. Highly asymmetrical porphyrins with enhanced push-pull character for dye-sensitized solar cells. Chem. A Eur. J. 2013, 19, 17075–17081. [Google Scholar] [CrossRef] [PubMed]
- Han, L.; Islam, A.; Chen, H.; Malapaka, C.; Zhang, S.; Yang, X.; Yanagida, M.; Chiranjeevi, B. High-efficiency dye-sensitized solar cell with a novel co-adsorbent. Energy Environ. Sci. 2012, 5, 6057–6060. [Google Scholar] [CrossRef]
- Luo, J.; Wan, Z.; Jia, C.; Wang, Y.; Wu, X. A co-sensitized approach to efficiently fill the absorption valley, avoid dye aggregation and reduce the charge recombination. Electrochim. Acta 2016, 215, 506–514. [Google Scholar] [CrossRef]
- Naik, P.; Abdellah, I.M.; Shakour, M.A.; Su, R.; Keremane, K.S.; El-Shafei, A.; Adhikaria, A.V. Improvement in performance of N3 sensitized DSSCs with structurally simple aniline based organic co-sensitizers. Sol. Energy 2018, 174, 999–1007. [Google Scholar] [CrossRef]
- Chandrasekharam, M.; Chiranjeevi, B.; Gupta, K.S.V.; Singh, S.P.; Islam, A.; Han, L.; Kantam, M.L. Simple metal-free organic D-A dyes with alkoxy- or fluorine substitutions: Application in dye sensitized solar cells. J. Nanosci. Nanotechnol. 2012, 12, 4489–4494. [Google Scholar] [CrossRef] [PubMed]
- Ganesh, K.; Shome, S.; Chandrasekharam, M.; Sharma, G.D.; Singh, S.P. High performance dye-sensitized solar cell from a cocktail solution of a ruthenium dye and metal free organic dye. RSC Adv. 2016, 6, 41151–41155. [Google Scholar]
- Wang, Q.; Chen, B.; Wu, W.J.; Li, X.; Zhu, W.H.; Tian, H.; Xie, Y.S. Efficient solar cells sensitized by porphyrins with an extended conjugation framework and a carbazole donor: From molecular design to cosensitization. Angew. Chem. 2014, 126, 10955–10959. [Google Scholar] [CrossRef]
- Nguyen, L.H.; Hemant, K.M.; Dharani, S.; Sneha, A.K.; Sudip, K.B.; Kazuteru, N.; Gratzel, M.; Subodh, G.M. A selective co-sensitization approach to increase photon conversion efficiency and electron lifetime in dye-sensitized solar cells. Phys. Chem. Chem. Phys. 2012, 14, 16182–16186. [Google Scholar] [CrossRef]
- Patil, D.; Jadhav, M.; Avhad, K.; Chowdhury, T.H.; Islam, A.; Bedjac, I.; Sekar, N. A new class of triphenylamine-based novel sensitizers for DSSCs: A comparative study of three different anchoring groups. New J. Chem. 2018, 42, 11555–11564. [Google Scholar] [CrossRef]
- Chen, B.-S.; Chen, D.-Y.; Chen, C.-L.; Hsu, C.-W.; Hsu, H.-C.; Wu, K.-L.; Liu, S.-H.; Chou, P.-T.; Chi, Y. Donor-acceptor dyes with fluorine substituted phenylene spacer for dye-sensitized solar cells. J. Mater. Chem. 2011, 21, 1937–1945. [Google Scholar] [CrossRef]
- Ganesh, K.; Singh, S.P.; Bhanuprakash, K.; Han, L.; Bedja, I.M.; Gupta, R.K.; Islam, A.; Chandrasekharam, M. Study of donor-acceptor-π-acceptor architecture sensitizers with benzothiazole acceptor for dye-sensitized solar cells. Energy Technol. 2016, 4, 458–468. [Google Scholar]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian 09, Revision D.01; Gaussian, Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Becke, A.D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 1993, 98, 5648–5652. [Google Scholar] [CrossRef]
- Becke, A.D. Density-functional thermochemistry. IV. A new dynamical correlation functional and implications for exact-exchange mixing. J. Chem. Phys. 1996, 104, 1040–1046. [Google Scholar] [CrossRef]
- Miertus, S.; Scrocco, E.; Tomasi, J. Electrostatic interaction of a solute with a continuum. A direct utilizaion of AB initio molecular potentials for the prevision of solvent effects. Chem. Phys. 1981, 55, 117–129. [Google Scholar] [CrossRef]
- Cossi, M.; Barone, V.; Cammi, R.; Tomasi, J. Ab initio study of solvated molecules: A new implementation of the polarizable continuum model. Chem. Phys. Lett. 1996, 255, 327–335. [Google Scholar] [CrossRef]
- Liu, B.; Li, W.; Wang, B.; Li, X.; Liu, Q.; Naruta, Y.; Zhu, W. Influence of different anchoring groups in indoline dyes for dye-sensitized solar cells: Electron injection, impedance and charge recombination. J. Power Sources 2013, 234, 139–146. [Google Scholar] [CrossRef]
- Babu, D.D.; Su, R.; El-Shafei, A.; Adhikari, A.V. New indole based co-sensitizers for dye sensitized solar cells exceeding 10% efficiency. RSC Adv. 2016, 6, 30205–30216. [Google Scholar] [CrossRef]
- Bonomo, M.; Barbero, N.; Naponiello, G.; Giordano, M.; Dini, D.; Barolo, C. Sodium hydroxide pretreatment as an effective approach to reduce the dye/holes recombination reaction in P-type DSCs. Front. Chem. 2019, 7, 99. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are available upon request |
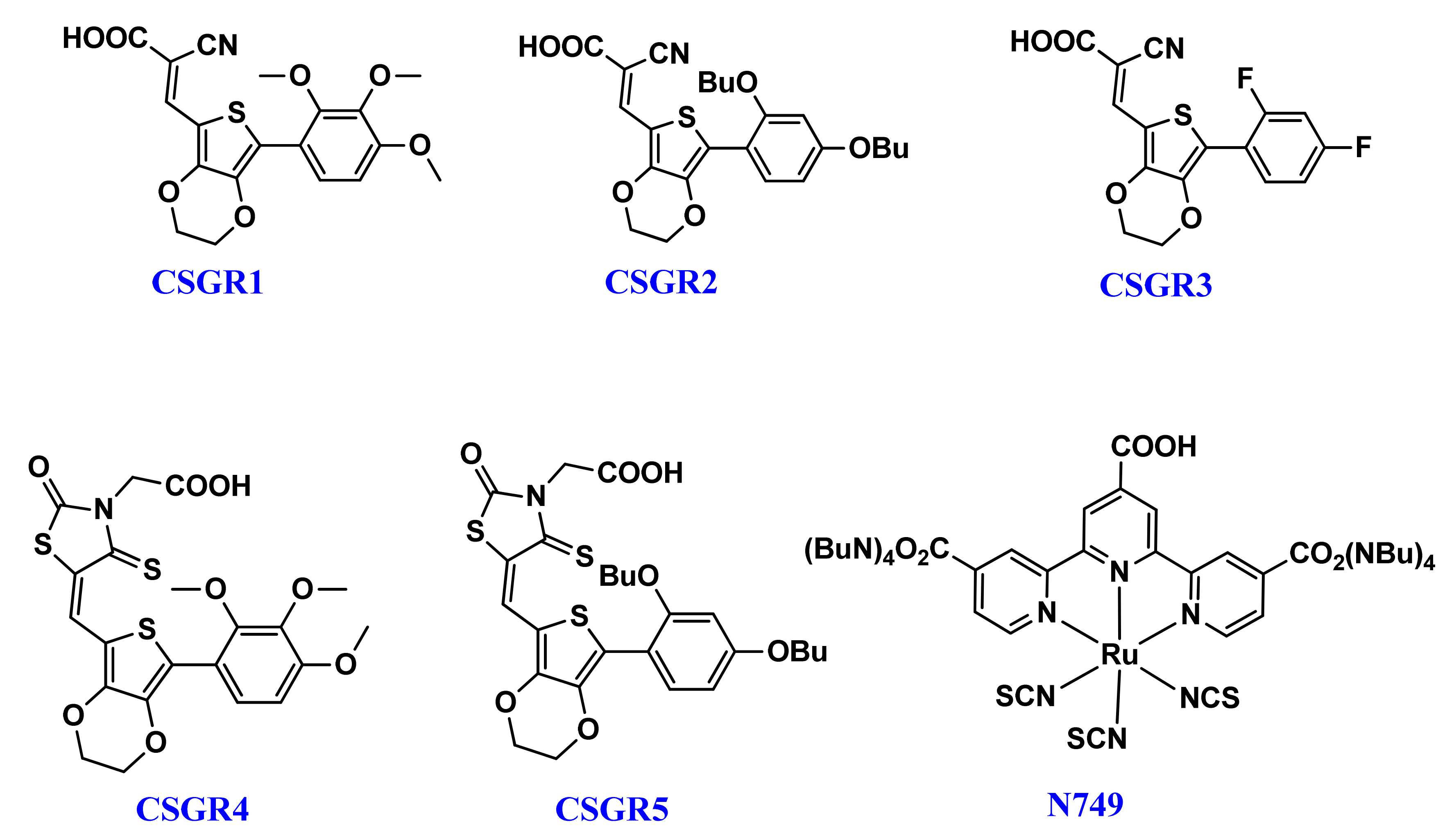
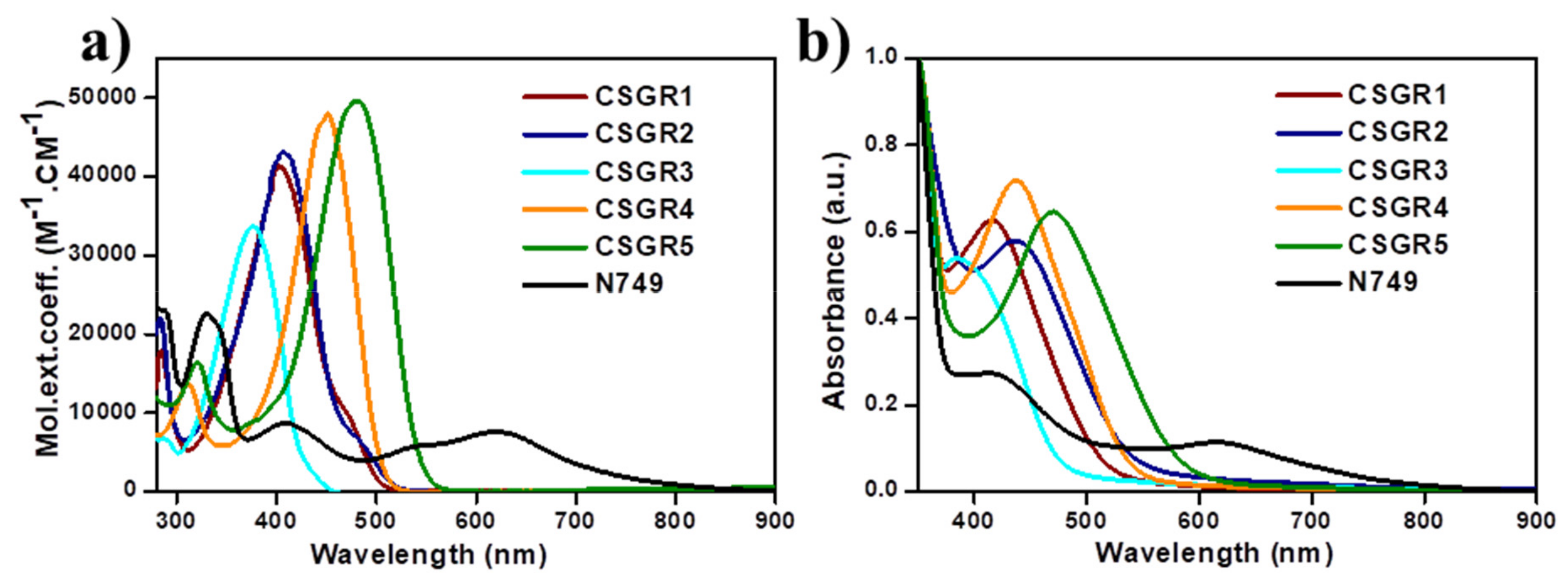


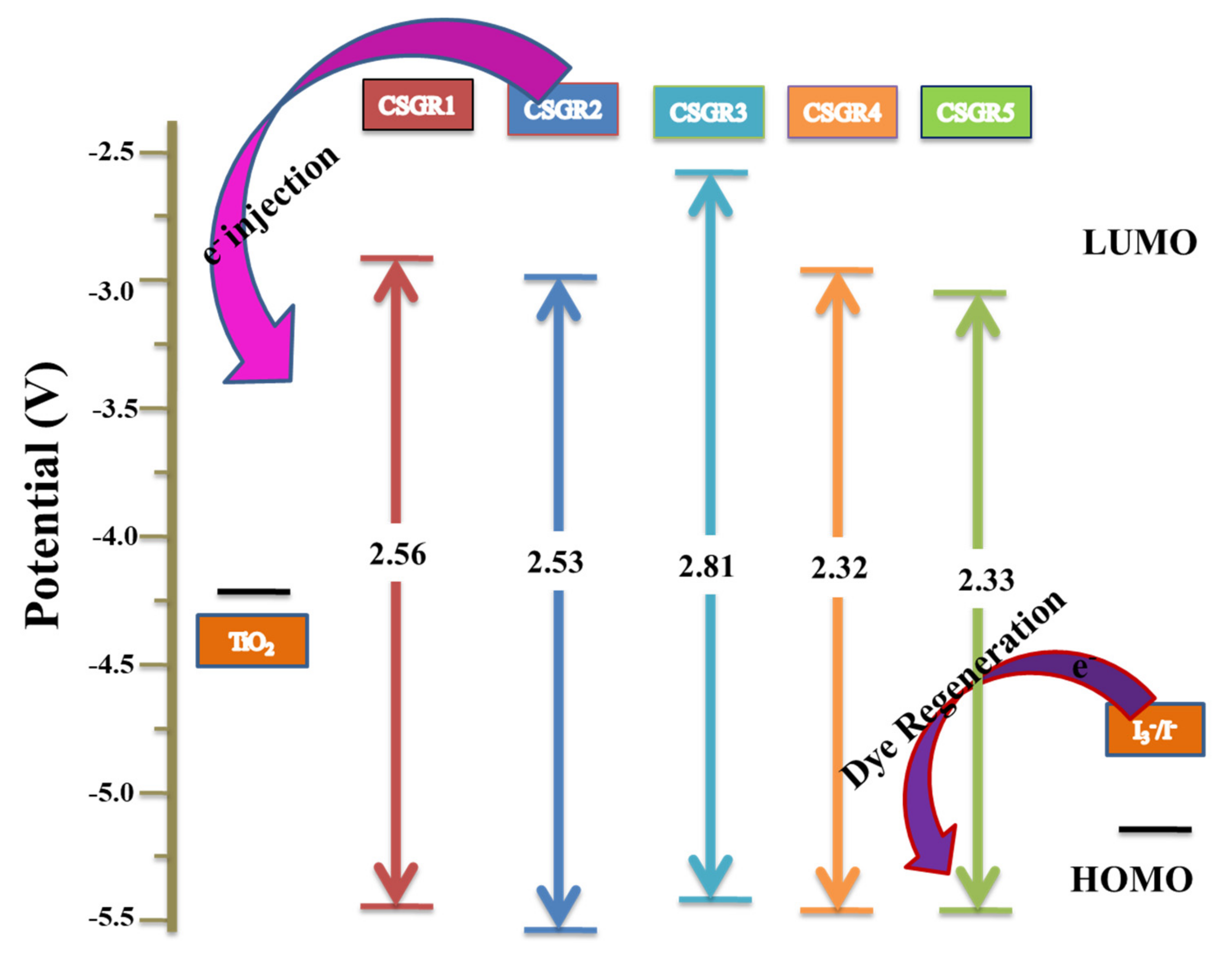

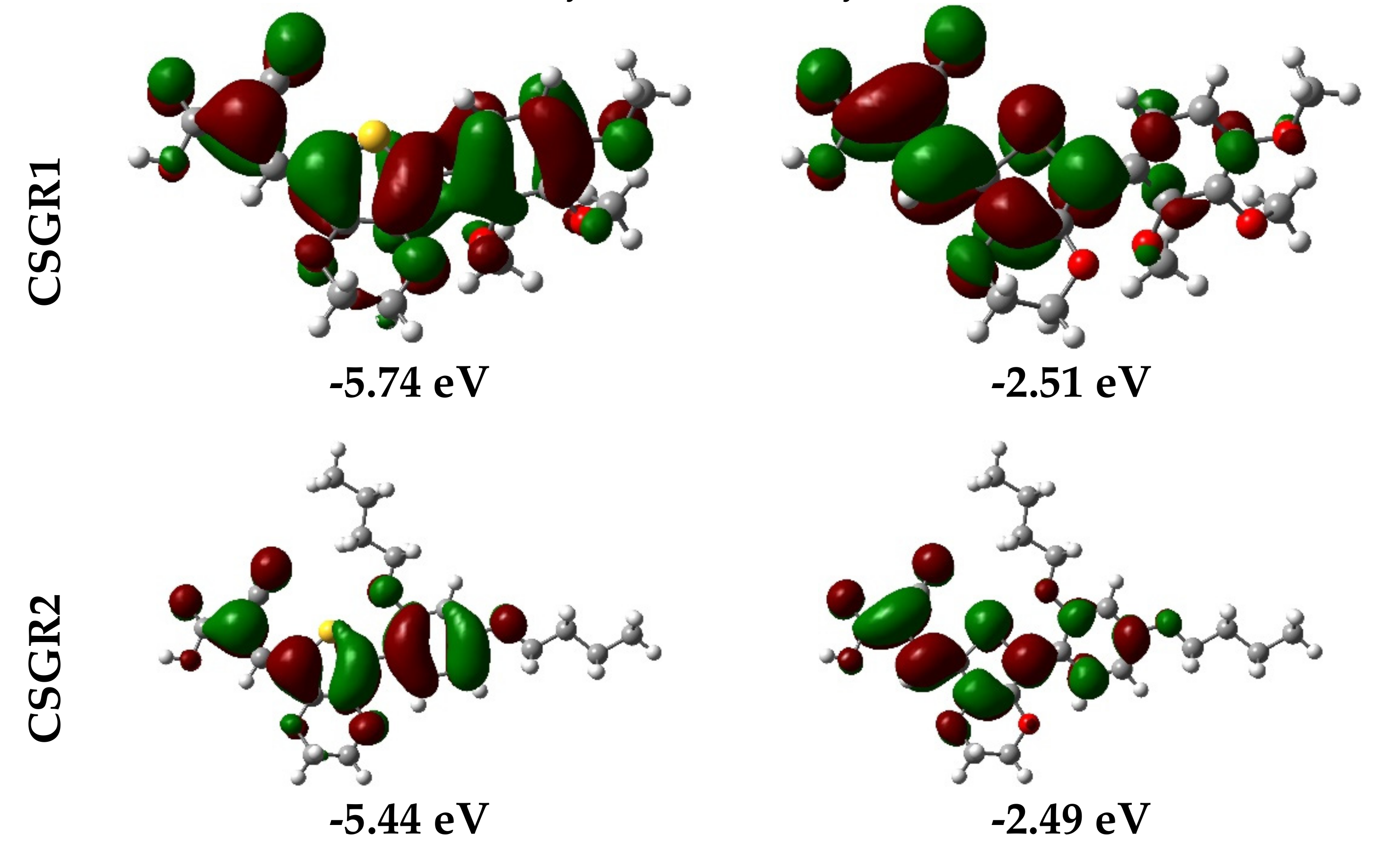


| Dye | λmax (Solution) (nm) a | Ε (M−1 Cm−1) | λmax (TiO2) (nm) b | λem (nm) c | Stokes Shif (nm) | E0X (V) d | E0-0 (V) e | E0X* (V) f | HOMO (eV) g | LUMO (eV) h |
|---|---|---|---|---|---|---|---|---|---|---|
| CSGR1 | 402 | 41182 | 415 | 526 | 101 | 0.773 | 2.56 | −1.787 | 5.473 | −2.913 |
| CSGR2 | 408 | 43904 | 437 | 533 | 114 | 0.829 | 2.53 | −1.701 | 5.529 | −2.993 |
| CSGR3 | 376 | 33557 | 389 | 478 | 85 | 0.789 | 2.81 | −2.021 | 5.489 | −2.679 |
| CSGR4 | 450 | 47688 | 436 | 528 | 92 | 0.793 | 2.55 | −1.757 | 5.493 | −2.943 |
| CSGR5 | 482 | 49522 | 471 | 574 | 78 | 0.785 | 2.33 | −1.545 | 5.485 | |
| N749 | 623 | 8725 | 618 |
| Dye | JSC (mA cm−2) | VOC (V) | FF | η (%) |
|---|---|---|---|---|
| N749 | 14.73 | 0.648 | 64.74 | 6.18 |
| 14.32 + 0.41 | 0.637 + 0.008 | 64.08 + 0.55 | 5.84 + 0.13 | |
| N749 + CSGR1 | 18.51 | 0.659 | 65.18 | 7.95 |
| 17.84 + 0.58 | 0.641 + 0.010 | 66.802 + 1.24 | 7.62 + 0.19 | |
| N749 + CSGR2 | 18.55 | 0.674 | 67.08 | 8.40 |
| 18.34 + 0.44 | 0.659 + 0.009 | 66.90 + 0,74 | 8.07 + 0.12 | |
| N749 + CSGR3 | 18.15 | 0.658 | 65.42 | 7.81 |
| 17.51 + 0.41 | 0.648 + 0.008 | 65.27 + 1.04 | 7.39 + 0.13 | |
| N749 + CSGR4 | 15.72 | 0.634 | 65.83 | 6.56 |
| 15.10 + 0.45 | 0.632 + 0.005 | 65.95 + 0.90 | 6.29 + 0.22 | |
| N749 + CSGR5 | 16.59 | 0.651 | 64.68 | 6.99 |
| 15.99 + 0.53 | 0.647 + 0.010 | 64.18 + 1.05 | 6.66 + 0.22 |
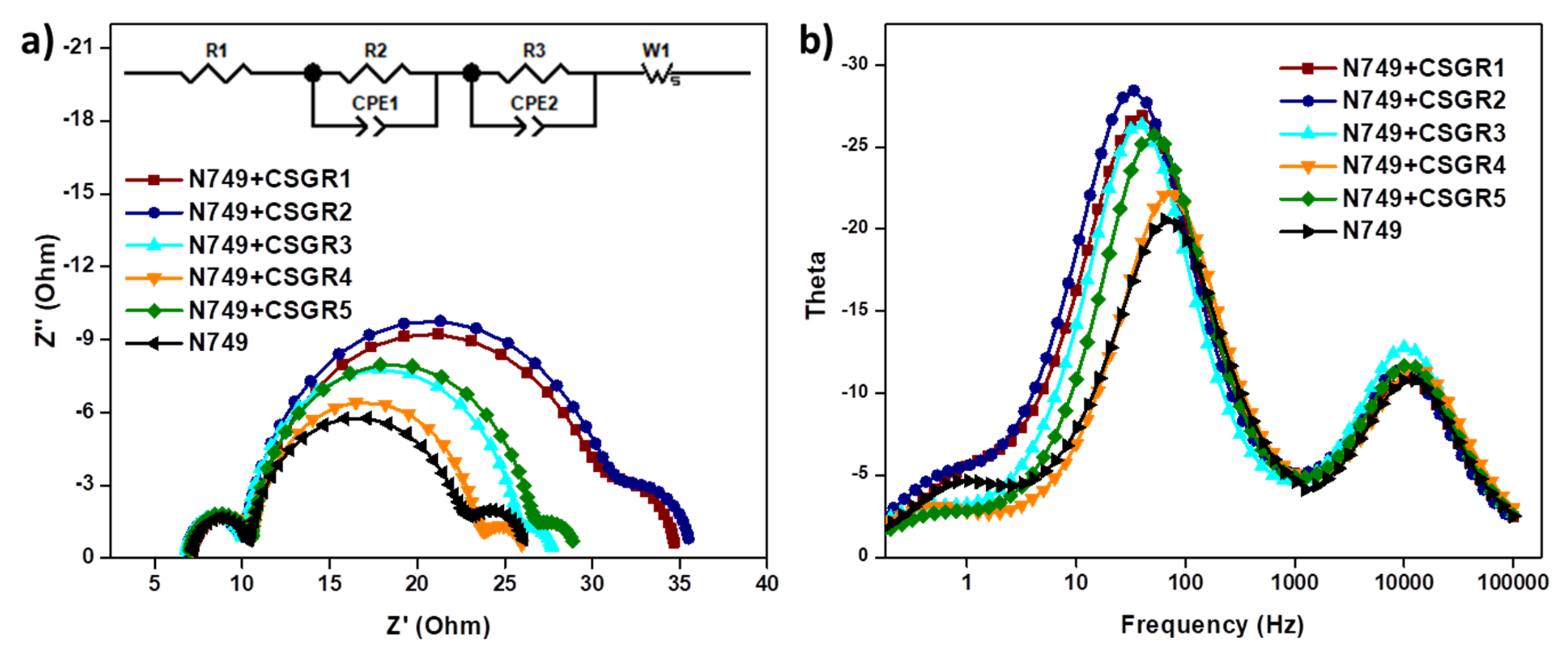
| Dye | R1 | R2 | R3 | Ʈe (ms) |
|---|---|---|---|---|
| N749 + CSGR1 | 7.138 | 3.165 | 19.38 | 4.46 |
| N749 + CSGR2 | 6.785 | 3.197 | 20.37 | 4.75 |
| N749 + CSGR3 | 6.763 | 3.328 | 15.28 | 3.89 |
| N749 + CSGR4 | 7.124 | 3.439 | 12.6 | 2.43 |
| N749 + CSGR5 | 6.393 | 3.538 | 15.64 | 3.08 |
| N749 | 7.139 | 3.206 | 11.72 | 2.17 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koyyada, G.; Kumar Chitumalla, R.; Thogiti, S.; Kim, J.H.; Jang, J.; Chandrasekharam, M.; Jung, J.H. A New Series of EDOT Based Co-Sensitizers for Enhanced Efficiency of Cocktail DSSC: A Comparative Study of Two Different Anchoring Groups. Molecules 2019, 24, 3554. https://doi.org/10.3390/molecules24193554
Koyyada G, Kumar Chitumalla R, Thogiti S, Kim JH, Jang J, Chandrasekharam M, Jung JH. A New Series of EDOT Based Co-Sensitizers for Enhanced Efficiency of Cocktail DSSC: A Comparative Study of Two Different Anchoring Groups. Molecules. 2019; 24(19):3554. https://doi.org/10.3390/molecules24193554
Chicago/Turabian StyleKoyyada, Ganesh, Ramesh Kumar Chitumalla, Suresh Thogiti, Jae Hong Kim, Joonkyung Jang, Malapaka Chandrasekharam, and Jae Hak Jung. 2019. "A New Series of EDOT Based Co-Sensitizers for Enhanced Efficiency of Cocktail DSSC: A Comparative Study of Two Different Anchoring Groups" Molecules 24, no. 19: 3554. https://doi.org/10.3390/molecules24193554







