Theoretical Studies on the Electronic Structure Parameters and Reactive Activity of Neu5Gc and Neu5Ac under Food Processing Solvent Environment
Abstract
:1. Introduction
2. Computational Details
2.1. Molecular Structure Parameters Calculation
2.2. Antioxidant Mechanism
3. Results and Discussion
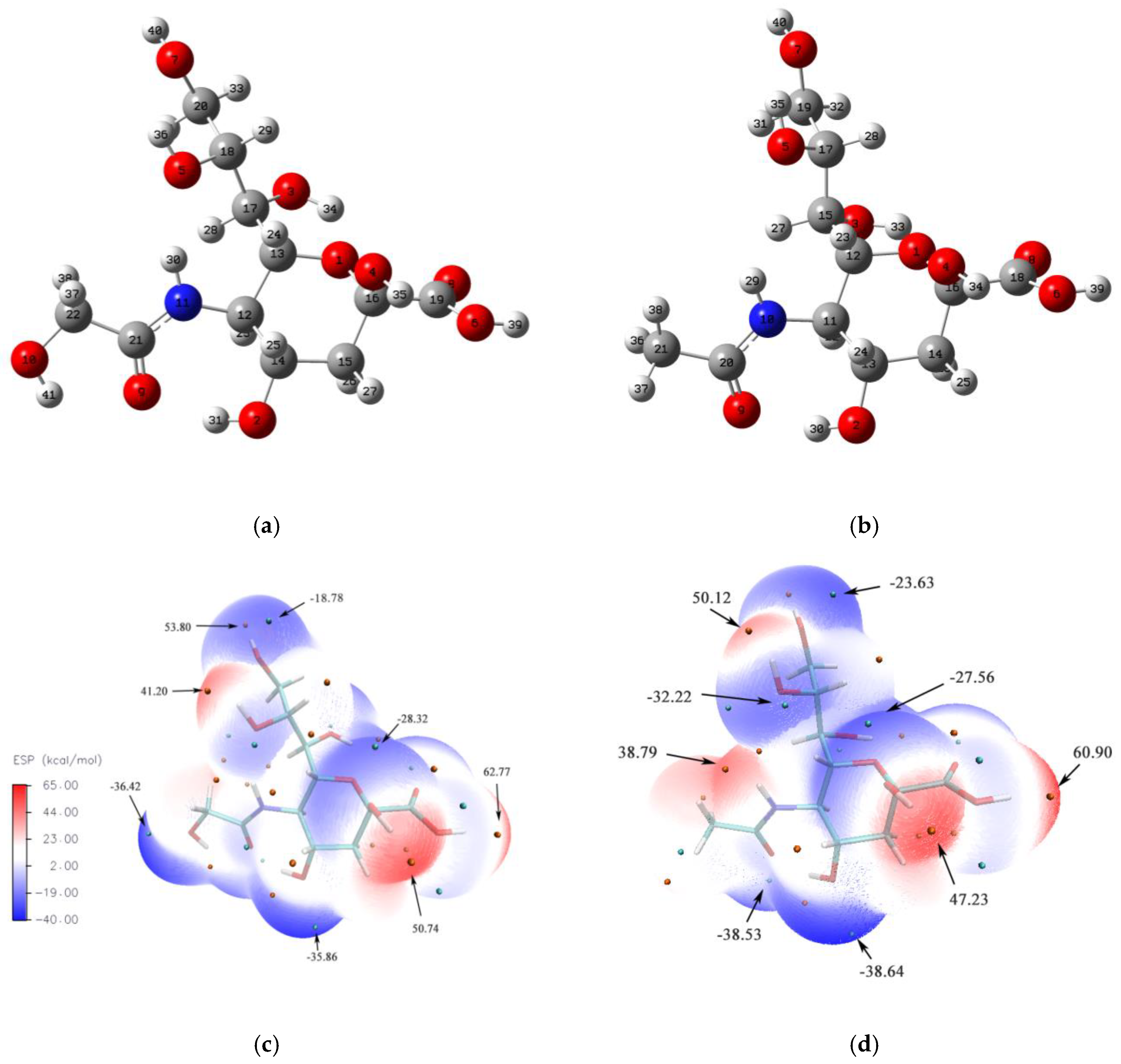
3.1. Molecular Structure and Electrostatic Potential
3.2. Frontier Molecular Orbital and Natural Bond Orbital
3.3. Intramolecular Hydrogen Bonds
3.4. IGM Analysis of Intramolecular Hydrogen Bonds
3.5. Molecular Reactivity Index
3.6. Molecular Condensed Fukui Function
3.7. Antioxidation Mechanism of Neu5Ac and Neu5Gc
3.7.1. Hydrogen Atom Transfer Mechanism (HAT)
3.7.2. Single Electron Transfer Mechanism (SET)
3.7.3. Single-electron Transfer Followed by Proton Transfer Mechanism (SET-PT)
3.7.4. Sequential Proton Loss Electron Transfer Mechanism (SPLET)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Angata, T.; Varki, A. Chemical diversity in the sialic acids and related α-keto acids: An evolutionary perspective. Chem. Rev. 2002, 102, 439–470. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Brand-Miller, J. The role and potential of sialic acid in human nutrition. Eur. J. Clin. Nutr. 2003, 57, 1351. [Google Scholar] [CrossRef] [PubMed]
- Hedlund, M.; Padler-Karavani, V.; Varki, N.M.; Varki, A. Evidence for a human-specific mechanism for diet and antibody-mediated inflammation in carcinoma progression. Proc. Natl. Acad. Sci. USA 2008, 105, 18936–18941. [Google Scholar] [CrossRef] [Green Version]
- Samraj, A.N.; Pearce, O.M.; Läubli, H.; Crittenden, A.N.; Bergfeld, A.K.; Banda, K.; Gregg, C.J.; Bingman, A.E.; Secrest, P.; Diaz, S.L. A red meat-derived glycan promotes inflammation and cancer progression. Proc. Natl. Acad. Sci. USA 2015, 112, 542–547. [Google Scholar] [CrossRef] [PubMed]
- Tangvoranuntakul, P.; Gagneux, P.; Diaz, S.; Bardor, M.; Varki, N.; Varki, A.; Muchmore, E. Human uptake and incorporation of an immunogenic nonhuman dietary sialic acid. Proc. Natl. Acad. Sci. USA 2003, 100, 12045–12050. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santarelli, R.L.; Pierre, F.; Corpet, D.E. Processed meat and colorectal cancer: a review of epidemiologic and experimental evidence. Nutr. Cancer 2008, 60, 131–144. [Google Scholar] [CrossRef]
- Djeradi, H.; Rahmouni, A.; Cheriti, A. Antioxidant activity of flavonoids: A QSAR modeling using Fukui indices descriptors. J. Mol. Model. 2014, 20, 2476. [Google Scholar] [CrossRef]
- Veluraja, K.; Rao, V. Studies on the conformations of sialyloligosaccharides and implications. J. Biosci. 1984, 6, 625–634. [Google Scholar] [CrossRef]
- Van Lenthe, J.H.; den Boer, D.H.; Havenith, R.W.; Schauer, R.; Siebert, H.-C. Ab initio calculations on various sialic acids provide valuable information about sialic acid-specific enzymes. J. Mol. Struc. THEOCHEM 2004, 677, 29–37. [Google Scholar] [CrossRef] [Green Version]
- Priyadarzini, T.R.; Subashini, B.; Selvin, J.F.; Veluraja, K. Molecular dynamics simulation and quantum mechanical calculations on α-dN-acetylneuraminic acid. Carbohydr. Res. 2012, 351, 93–97. [Google Scholar] [CrossRef]
- Sawada, T.; Hashimoto, T.; Nakano, H.; Shigematsu, M.; Ishida, H.; Kiso, M. Conformational Study of α-N-Acetyl-d-Neuraminic Acid by Density Functional Theory. J. Carbohydr. Chem. 2006, 25, 387–405. [Google Scholar] [CrossRef]
- Blessy, J.J.; Sharmila, D.J.S. Molecular simulation of N-acetylneuraminic acid analogs and molecular dynamics studies of cholera toxin-Neu5Gc complex. J. Biomol. Struct. Dyn. 2015, 33, 1126–1139. [Google Scholar] [CrossRef] [PubMed]
- Reuter, S.; Gupta, S.C.; Chaturvedi, M.M.; Aggarwal, B.B. Oxidative stress, inflammation, and cancer: How are they linked? Free Radic. Biol. Med. 2010, 49, 1603–1616. [Google Scholar] [CrossRef] [Green Version]
- Wijewickreme, A.; Krejpcio, Z.; Kitts, D. Hydroxyl Scavenging Activity of Glucose, Fructose, and Ribose-Lysine Model Maillard Products. J. Food Sci. 1999, 64, 457–461. [Google Scholar] [CrossRef]
- Morelli, R.; Russo-Volpe, S.; Bruno, N.; Lo Scalzo, R. Fenton-dependent damage to carbohydrates: Free radical scavenging activity of some simple sugars. J. Agric. Food Chem. 2003, 51, 7418–7425. [Google Scholar] [CrossRef] [PubMed]
- Iijima, R.; Takahashi, H.; Namme, R.; Ikegami, S.; Yamazaki, M. Novel biological function of sialic acid (N-acetylneuraminic acid) as a hydrogen peroxide scavenger. FEBS Lett. 2004, 561, 163–166. [Google Scholar] [CrossRef]
- Iijima, R.; Takahashi, H.; Ikegami, S.; Yamazaki, M. Characterization of the reaction between sialic acid (N-acetylneuraminic acid) and hydrogen peroxide. Biol. Pharm. Bull. 2007, 30, 580–582. [Google Scholar] [CrossRef] [PubMed]
- Serdar, Z.; Yeşilbursa, D.; Dirican, M.; Sarandöl, E.; Serdar, A. Sialic acid and oxidizability of lipid and proteins and antioxidant status in patients with coronary artery disease. Cell Biochem. Funct. 2007, 25, 655–664. [Google Scholar] [CrossRef] [PubMed]
- Schnaar, R.L.; Gerardy-Schahn, R.; Hildebrandt, H. Sialic acids in the brain: gangliosides and polysialic acid in nervous system development, stability, disease, and regeneration. Physiol. Rev. 2014, 94, 461–518. [Google Scholar] [CrossRef]
- Reuter, G.; Schauer, R.; Szeiki, C.; Kamerling, J.P.; Vliegenthart, J.F. A detailed study of the periodate oxidation of sialic acids in glycoproteins. Glycoconjugate 1989, 6, 35–44. [Google Scholar] [CrossRef] [Green Version]
- Ogasawara, Y.; Namai, T.; Yoshino, F.; Ishii, K. Sialic acid is an essential moiety of mucin as a hydroxyl radical scavenger. FEBS Lett. 2007, 581, 2473–2477. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, S.; Thiessen, P.A.; Bolton, E.E.; Chen, J.; Fu, G.; Gindulyte, A.; Han, L.Y.; He, J.N.; He, S.Q.; Benjamin, A. PubChem substance and compound databases. Nucleic Acids Res. 2015, 44, D1202–D1213. [Google Scholar] [CrossRef] [PubMed]
- Ho, J.; Klamt, A.; Coote, M.L. Comment on the correct use of continuum solvent models. J. Chem. Phys. A 2010, 114, 13442–13444. [Google Scholar] [CrossRef] [PubMed]
- Geerlings, P.; De Proft, F.; Langenaeker, W. Conceptual density functional theory. Chem. Rev. 2003, 103, 1793–1874. [Google Scholar] [CrossRef] [PubMed]
- Bultinck, P.; Fias, S.; Van Alsenoy, C.; Ayers, P.W.; Carbó-Dorca, R. Critical thoughts on computing atom condensed Fukui functions. J. Chem. Phys. 2007, 127, 034102. [Google Scholar] [CrossRef] [Green Version]
- Oliveira, B.; Pereira, F.; de Araújo, R.; Ramos, M. The hydrogen bond strength: New proposals to evaluate the intermolecular interaction using DFT calculations and the AIM theory. Chem. Phys. Lett. 2006, 427, 181–184. [Google Scholar] [CrossRef]
- Klein, E.; Lukeš, V.; Ilčin, M. DFT/B3LYP study of tocopherols and chromans antioxidant action energetics. Chem. Phys. 2007, 336, 51–57. [Google Scholar] [CrossRef]
- Klein, E.; Lukeš, V. DFT/B3LYP study of the substituent effect on the reaction enthalpies of the individual steps of single electron transfer-proton transfer and sequential proton loss electron transfer mechanisms of phenols antioxidant action. J. Chem. Phys. A 2006, 110, 12312–12320. [Google Scholar] [CrossRef]
- Marković, Z.; Tošović, J.; Milenković, D.; Marković, S. Revisiting the solvation enthalpies and free energies of the proton and electron in various solvents. Comput. Theor. Chem. 2016, 1077, 11–17. [Google Scholar] [CrossRef]
- Rimarčík, J.; Lukeš, V.; Klein, E.; Ilčin, M. Study of the solvent effect on the enthalpies of homolytic and heterolytic N–H bond cleavage in p-phenylenediamine and tetracyano-p-phenylenediamine. J. Mol. Struct. THEOCHEM 2010, 952, 25–30. [Google Scholar] [CrossRef]
- Alecu, I.; Zheng, J.; Zhao, Y.; Truhlar, D.G. Computational thermochemistry: scale factor databases and scale factors for vibrational frequencies obtained from electronic model chemistries. J. Chem. Theory Comput. 2010, 6, 2872–2887. [Google Scholar] [CrossRef]
- Frisch, M.; Trucks, G.; Schlegel, H.; Scuseria, G.; Robb, M.; Cheeseman, J.; Scalmani, G.; Barone, V.; Petersson, G.; Nakatsuji, H. Gaussian 16, revision A. 03; Gaussian Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Lu, T.; Chen, F. Multiwfn: A multifunctional wavefunction analyzer. J. Comput. Chem. 2012, 33, 580–592. [Google Scholar] [CrossRef]
- Lu, T.; Manzetti, S. Wavefunction and reactivity study of benzo [a] pyrene diol epoxide and its enantiomeric forms. Struct. Chem. 2014, 25, 1521–1533. [Google Scholar] [CrossRef]
- Humphrey, W.; Dalke, A.; Schulten, K. VMD: Visual Molecular Dynamics. J. Mol. Graph. 1996, 14, 33–38. [Google Scholar] [CrossRef]
- Arroyo-Currás, N.; Rosas-García, V.M.; Videa, M. Substituent inductive effects on the electrochemical oxidation of flavonoids studied by square wave voltammetry and Ab initio calculations. Molecules 2016, 21, 1422. [Google Scholar] [CrossRef]
- Tao, Y.; Han, L.; Li, X.; Han, Y.; Liu, Z. Molecular structure, spectroscopy (FT-IR, FT-Raman), thermodynamic parameters, molecular electrostatic potential and HOMO-LUMO analysis of 2,6-dichlorobenzamide. J. Mol. Struct. 2016, 1108, 307–314. [Google Scholar] [CrossRef]
- Garzόn, A.; Bravo, I.; Barbero, A.J.; Albaladejo, J. Mechanistic and kinetic study on the reactions of coumaric acids with reactive oxygen species: A DFT approach. J. Agric. Food Chem. 2014, 62, 9705–9710. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; Zhong, H.; Qiu, X.; Wang, S.; Gao, Y.; Dai, Z.; Huang, J.; Liu, G. The DFT study of cyclohexyl hydroxamic acid as a collector in scheelite flotation. Miner. Eng. 2013, 49, 54–60. [Google Scholar] [CrossRef]
- Glendening, E.D.; Landis, C.R.; Weinhold, F. Natural bond orbital methods. Wires Comput. Mol. Sci. 2012, 2, 1–42. [Google Scholar] [CrossRef]
- Lipkowski, P.; Grabowski, S.J.; Robinson, T.L.; Leszczynski, J. Properties of the C-H…H Dihydrogen Bond: An ab Initio and Topological Analysis. J. Chem. Phys. A 2004, 108, 10865–10872. [Google Scholar] [CrossRef]
- Rozas, I.; Alkorta, I.; Elguero, J. Behavior of ylides containing N, O, and C atoms as hydrogen bond acceptors. J. Am. Chem. Soc. 2000, 122, 11154–11161. [Google Scholar] [CrossRef]
- Lefebvre, C.; Rubez, G.; Khartabil, H.; Boisson, J.C.; Contreras-García, J.; Hénon, E. Accurately extracting the signature of intermolecular interactions present in the NCI plot of the reduced density gradient versus electron density. Phys. Chem. Chem. Phys. 2017, 19, 17928–17936. [Google Scholar] [CrossRef]
- Marcus, Y. The properties of organic liquids that are relevant to their use as solvating solvents. Chem. Soc. Rev. 1993, 22, 409–416. [Google Scholar] [CrossRef]
- Chen, Y.; Pan, L.; Liu, N.; Troy, F.A.; Wang, B. LC-MS/MS quantification of N-acetylneuraminic acid, N-glycolylneuraminic acid and ketodeoxynonulosonic acid levels in the urine and potential relationship with dietary sialic acid intake and disease in 3-to 5-year-old children. Br. J. Nutr. 2014, 111, 332–341. [Google Scholar] [CrossRef] [PubMed]
- Stachowicz-Kuśnierz, A.; Korchowiec, J. Nucleophilic properties of purine bases: Inherent reactivity versus reaction conditions. Struct. Chem. 2016, 27, 543–555. [Google Scholar] [CrossRef]
- Galano, A.; Mazzone, G.; Alvarez-Diduk, R.; Marino, T.; Alvarez-Idaboy, J.R.; Russo, N. Food antioxidants: chemical insights at the molecular level. Annu. Rev. Food Sci. Technol. 2016, 7, 335–352. [Google Scholar] [CrossRef] [PubMed]
- Diwathe, M.C.; Gogate, P.R. Ultrasound assisted intensified synthesis of 1-benzyloxy-4-nitrobenzene in the presence of phase transfer catalyst. Chem. Eng. J. 2018, 346, 438–446. [Google Scholar] [CrossRef]
- Litwinienko, G.; Ingold, K. Solvent effects on the rates and mechanisms of reaction of phenols with free radicals. Acc. Chem. Res. 2007, 40, 222–230. [Google Scholar] [CrossRef]
- Wang, G.; Xue, Y.; An, L.; Zheng, Y.; Dou, Y.; Zhang, L.; Liu, Y. Theoretical study on the structural and antioxidant properties of some recently synthesised 2, 4, 5-trimethoxy chalcones. Food Chem. 2015, 171, 89–97. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |


| Neu5Gc | Bond | ρ | ▽2ρ | V | G | HBCP | EHB | Length | Angle |
|---|---|---|---|---|---|---|---|---|---|
| Gas | O2–H31...O9 | 0.02953 | 0.1126 | −0.02557 | 0.02687 | 0.001301 | −33.56 | 2.794 | 4.804 |
| O3–H34...O1 | 0.02885 | 0.1327 | –0.02784 | 0.03050 | 0.002673 | −36.55 | 2.524 | 6.080 | |
| O10–H41...O9 | 0.02385 | 0.1051 | –0.02086 | 0.02357 | 0.002710 | –27.39 | 2.646 | 13.65 | |
| N11–H30...O5 | 0.02255 | 0.09263 | –0.01774 | 0.02045 | 0.002705 | –23.29 | 2.921 | 2.811 | |
| Acetic acid | O10–H41...O9 | 0.2266 | 0.1019 | –0.01975 | 0.2262 | 0.002869 | –25.94 | 2.653 | 4.951 |
| Benzene | O10–H41...O9 | 0.02319 | 0.1038 | –0.02031 | 0.02313 | 0.002817 | –26.67 | 2.652 | 5.516 |
| Gas | O3–H33...O1 | 0.02262 | 0.1058 | –0.02053 | 0.02349 | 0.002964 | –26.95 | 2.586 | 8.897 |
| O2–H30...O9 | 0.02907 | 0.1101 | –0.02485 | 0.02619 | 0.001343 | –32.62 | 2.800 | 4.906 | |
| Acetic acid | O2–H30...O9 | 0.02361 | 0.08990 | –0.01891 | 0.02069 | 0.001782 | –24.82 | 2.850 | 5.233 |
| Benzene | O3–H33...O1 | 0.02027 | 0.09740 | –0.01829 | 0.02132 | 0.003026 | –24.02 | 2.625 | 11.40 |
| O2–H30...O9 | 0.02818 | 0.1064 | –0.02373 | 0.02518 | 0.001443 | –31.16 | 2.816 | 4.511 |
| Neu5Gc | ||||||||
|---|---|---|---|---|---|---|---|---|
| Media | HOMO | LUMO | Egap | η | s | χ | μ | ω |
| Gas | −8.900 | 0.753 | 9.653 | 4.826 | 0.1036 | 4.074 | −4.074 | 1.719 |
| Benzene | −8.867 | 0.955 | 9.822 | 4.911 | 0.1018 | 3.955 | −3.955 | 1.593 |
| Ethanol | −8.660 | 1.039 | 9.699 | 4.849 | 0.1030 | 3.810 | −3.810 | 1.496 |
| Acetic acid | −8.728 | 0.995 | 9.723 | 4.861 | 0.1028 | 3.866 | −3.866 | 1.537 |
| Lactic acid | −8.715 | 1.125 | 9.840 | 4.920 | 0.1016 | 3.795 | −3.795 | 1.463 |
| Formic acid | −8.720 | 1.013 | 9.733 | 4.866 | 0.1027 | 3.853 | −3.853 | 1.525 |
| Water | −8.731 | 0.988 | 9.720 | 4.860 | 0.1028 | 3.871 | −3.871 | 1.542 |
| Neu5Ac | ||||||||
| Gas | −8.766 | 0.878 | 9.644 | 4.822 | 0.1036 | 3.944 | −3.944 | 1.613 |
| Benzene | −8.674 | 1.027 | 9.702 | 4.851 | 0.1030 | 3.823 | −3.823 | 1.506 |
| Ethanol | −8.594 | 1.068 | 9.662 | 4.831 | 0.1034 | 3.762 | −3.762 | 1.465 |
| Acetic acid | −8.701 | 1.021 | 9.722 | 4.860 | 0.1028 | 3.840 | −3.840 | 1.516 |
| Lactic acid | −8.578 | 1.141 | 9.719 | 4.859 | 0.1029 | 3.719 | −3.718 | 1.423 |
| Formic acid | −8.548 | 1.030 | 9.578 | 4.789 | 0.1043 | 3.759 | −3.759 | 1.475 |
| Water | −8.718 | 1.007 | 9.726 | 4.863 | 0.1028 | 3.855 | −3.855 | 1.528 |
| f − | Electrophilic Activity of Neu5Gc | ||||||
|---|---|---|---|---|---|---|---|
| Media | O2 | O3 | O4 | O5 | O6 | O7 | O10 |
| Gas | 0.1237 | 0.02612 | 0.01854 | −0.003166 | 0.01067 | 0.009959 | 0.05184 |
| Benzene | 0.08462 | 0.01581 | 0.01468 | 0.001390 | 0.008207 | 0.007863 | 0.04109 |
| Acetic acid | 0.06119 | 0.01006 | 0.008661 | 0.002425 | 0.005481 | 0.004638 | 0.03003 |
| Ethanol | 0.03166 | 0.01348 | 0.008483 | 0.004389 | 0.004433 | 0.003873 | 0.02739 |
| Formic acid | 0.02883 | 0.01327 | 0.008223 | 0.004598 | 0.003941 | 0.003317 | 0.02415 |
| Lactic acid | 0.05031 | 0.009583 | 0.007909 | 0.002491 | 0.004599 | 0.003322 | 0.03325 |
| Water | 0.03077 | 0.01305 | 0.008335 | 0.004534 | 0.003866 | 0.003205 | 0.02450 |
| f + | Nucleophilic Activity of Neu5Gc | ||||||
| Gas | 0.02046 | 0.01737 | 0.04551 | 0.001062 | 0.0694 | 0.01091 | 0.01983 |
| Benzene | 0.01696 | 0.01141 | 0.05685 | 0.002364 | 0.09615 | 0.003067 | 0.007161 |
| Acetic acid | 0.01556 | 0.006019 | 0.04722 | 0.001904 | 0.1224 | 0.001077 | 0.002703 |
| Ethanol | 0.01396 | 0.004336 | 0.04283 | 0.001304 | 0.1240 | 0.0006920 | 0.001186 |
| Formic acid | 0.01279 | 0.003578 | 0.04548 | 0.001167 | 0.1269 | 0.0005470 | 0.0008410 |
| Lactic acid | 0.01419 | 0.007517 | 0.04970 | 0.001725 | 0.1086 | 0.001256 | 0.001962 |
| Water | 0.01274 | 0.003522 | 0.04491 | 0.001134 | 0.1270 | 0.0005410 | 0.0007780 |
| f − | Electrophilic Activity of Neu5Ac | |||||
|---|---|---|---|---|---|---|
| Media | O2 | O3 | O4 | O5 | O6 | O7 |
| Gas | 0.1140 | 0.02236 | 0.01776 | 0.0001650 | 0.01092 | 0.01185 |
| Benzene | 0.06632 | 0.01300 | 0.009534 | 0.0005200 | 0.007312 | 0.007243 |
| Acetic acid | 0.05073 | 0.009047 | 0.008116 | 0.001855 | 0.005053 | 0.004259 |
| Ethanol | 0.02826 | 0.01184 | 0.007726 | 0.003867 | 0.004107 | 0.003490 |
| Formic acid | 0.05069 | 0.008504 | 0.007130 | 0.003015 | 0.003647 | 0.002429 |
| Lactic acid | 0.05070 | 0.009064 | 0.007035 | 0.002215 | 0.004367 | 0.003040 |
| Water | 0.04718 | 0.008686 | 0.007317 | 0.003156 | 0.003574 | 0.002371 |
| f + | Nucleophilic Activity of Neu5Ac | |||||
| Gas | 0.02053 | 0.01515 | 0.05226 | 0.001968 | 0.08011 | 0.006588 |
| Benzene | 0.01705 | 0.01137 | 0.05745 | 0.002491 | 0.09739 | 0.003006 |
| Acetic acid | 0.01432 | 0.006090 | 0.04783 | 0.002124 | 0.1225 | 0.001194 |
| Ethanol | 0.01363 | 0.004200 | 0.04485 | 0.001290 | 0.1236 | 0.0006360 |
| Formic acid | 0.01236 | 0.003406 | 0.04676 | 0.001115 | 0.1266 | 0.0005200 |
| Lactic acid | 0.01399 | 0.007559 | 0.05077 | 0.001706 | 0.1076 | 0.001251 |
| Water | 0.01237 | 0.003225 | 0.04587 | 0.001147 | 0.1267 | 0.0005170 |
| HAT | BDE of Neu5Gc | ||||||
|---|---|---|---|---|---|---|---|
| Bond | O2-H | O3-H | O4-H | O5-H | O6-H | O7-H | O10-H |
| Gas | 104.78 | 108.41 | 104.14 | 108.24 | 114.22 | 103.51 | 108.98 |
| Benzene | 104.78 | 109.22 | 105.93 | 108.81 | 115.21 | 104.79 | 109.30 |
| Acetic acid | 108.56 | 109.89 | 108.24 | 109.72 | 118.61 | 106.76 | 109.51 |
| Ethanol | 107.86 | 108.75 | 108.10 | 108.36 | 117.94 | 106.04 | 107.98 |
| Formic acid | 109.37 | 109.86 | 110.17 | 110.40 | 116.21 | 108.08 | 109.40 |
| Lactic acid | 106.73 | 107.93 | 105.98 | 107.60 | 114.60 | 104.55 | 107.64 |
| Water | 105.77 | 106.59 | 106.16 | 106.09 | 118.10 | 104.03 | 105.25 |
| PDE of Neu5Gc | |||||||
| Bond | O2-H | O3-H | O4-H | O5-H | O6-H | O7-H | O10-H |
| Gas | 218.48 | 222.10 | 217.84 | 221.93 | 227.91 | 217.21 | 222.67 |
| Benzene | 24.55 | 28.98 | 25.70 | 28.58 | 34.98 | 24.56 | 29.06 |
| Acetic acid | 9.60 | 10.94 | 9.29 | 10.76 | 19.65 | 7.80 | 10.55 |
| Ethanol | 7.47 | 8.35 | 7.70 | 7.96 | 17.55 | 5.65 | 7.59 |
| Formic acid | 13.01 | 13.51 | 13.82 | 14.05 | 19.86 | 11.73 | 13.05 |
| Lactic acid | 18.35 | 19.55 | 17.60 | 19.22 | 26.22 | 16.17 | 19.26 |
| Water | 9.78 | 10.60 | 10.17 | 10.10 | 22.11 | 8.04 | 9.26 |
| PA of Neu5Gc | |||||||
| Bond | O2-H | O3-H | O4-H | O5-H | O6-H | O7-H | O10-H |
| Gas | 349.69 | 335.18 | 337.41 | 321.84 | 317.18 | 322.18 | 366.36 |
| Benzene | 115.46 | 119.74 | 101.89 | 95.90 | 80.55 | 90.27 | 124.17 |
| Acetic acid | 63.65 | 67.18 | 53.90 | 54.12 | 34.16 | 62.87 | 66.19 |
| Ethanol | 49.60 | 51.33 | 39.32 | 52.78 | 20.34 | 50.54 | 54.36 |
| Formic acid | 50.97 | 52.44 | 42.04 | 54.02 | 24.23 | 53.13 | 52.79 |
| Lactic acid | 75.79 | 77.98 | 63.25 | 59.65 | 40.95 | 71.48 | 79.43 |
| Water | 47.40 | 48.75 | 37.94 | 49.74 | 23.80 | 49.78 | 48.86 |
| ETE of Neu5Gc | |||||||
| Bond | O2-H | O3-H | O4-H | O5-H | O6-H | O7-H | O10-H |
| Gas | 59.60 | 87.74 | 81.24 | 100.90 | 111.54 | 95.84 | 57.12 |
| Benzene | 81.85 | 91.99 | 96.56 | 105.43 | 127.18 | 107.04 | 77.65 |
| Acetic acid | 83.46 | 81.26 | 92.89 | 94.15 | 123.00 | 82.44 | 79.87 |
| Ethanol | 97.84 | 97.00 | 108.36 | 95.15 | 137.17 | 95.08 | 93.19 |
| Formic acid | 96.61 | 95.64 | 106.35 | 94.60 | 130.20 | 93.17 | 94.83 |
| Lactic acid | 82.39 | 81.41 | 94.19 | 99.41 | 125.11 | 84.53 | 79.67 |
| Water | 96.37 | 95.84 | 106.22 | 94.35 | 132.30 | 92.25 | 94.39 |
| HAT | BDE of Neu5Ac | |||||
|---|---|---|---|---|---|---|
| Bond | O2-H | O3-H | O4-H | O5-H | O6-H | O7-H |
| Gas | 108.40 | 108.26 | 104.24 | 108.14 | 113.67 | 103.38 |
| Benzene | 109.13 | 109.20 | 105.84 | 108.85 | 114.97 | 104.85 |
| Acetic acid | 109.41 | 109.24 | 108.42 | 110.01 | 118.39 | 107.14 |
| Ethanol | 107.93 | 108.57 | 108.09 | 108.45 | 118.04 | 106.18 |
| Formic acid | 108.52 | 109.09 | 109.32 | 109.74 | 114.48 | 107.42 |
| Lactic acid | 107.70 | 107.86 | 105.88 | 107.66 | 114.56 | 104.57 |
| Water | 105.33 | 105.92 | 105.65 | 105.38 | 112.17 | 107.94 |
| PDE of Neu5Ac | ||||||
| Bond | O2-H | O3-H | O4-H | O5-H | O6-H | O7-H |
| Gas | 224.53 | 224.39 | 220.37 | 224.27 | 229.80 | 219.51 |
| Benzene | 33.07 | 33.13 | 29.78 | 32.78 | 38.90 | 28.79 |
| Acetic acid | 12.24 | 12.06 | 11.25 | 12.84 | 21.22 | 9.97 |
| Ethanol | 9.10 | 9.74 | 9.25 | 9.62 | 19.21 | 7.35 |
| Formic acid | 14.68 | 15.25 | 15.48 | 15.89 | 20.63 | 13.57 |
| Lactic acid | 22.60 | 22.76 | 20.78 | 22.56 | 29.45 | 19.47 |
| Water | 11.71 | 12.29 | 12.02 | 11.76 | 18.30 | 14.32 |
| PA of Neu5Ac | ||||||
| Bond | O2-H | O3-H | O4-H | O5-H | O6-H | O7-H |
| Gas | 354.54 | 342.11 | 340.61 | 327.11 | 319.68 | 327.11 |
| benzene | 117.94 | 121.75 | 103.89 | 100.18 | 81.95 | 94.44 |
| Acetic acid | 65.75 | 68.15 | 53.78 | 69.30 | 35.28 | 65.96 |
| Ethanol | 50.07 | 51.18 | 39.54 | 52.67 | 20.76 | 50.83 |
| Formic acid | 50.67 | 51.98 | 41.52 | 53.51 | 23.54 | 52.55 |
| Lactic acid | 77.73 | 78.55 | 64.91 | 63.05 | 48.85 | 73.14 |
| water | 49.79 | 48.11 | 38.00 | 49.49 | 23.82 | 48.58 |
| ETE of Neu5Ac | ||||||
| Bond | O2-H | O3-H | O4-H | O5-H | O6-H | O7-H |
| Gas | 68.37 | 80.65 | 78.13 | 95.53 | 108.49 | 90.78 |
| Benzene | 83.71 | 79.96 | 94.46 | 101.18 | 125.53 | 102.93 |
| Acetic acid | 82.21 | 79.63 | 93.19 | 79.26 | 121.66 | 79.73 |
| Ethanol | 97.44 | 96.96 | 108.12 | 95.35 | 137.09 | 94.93 |
| Formic acid | 96.06 | 95.32 | 106.01 | 96.44 | 129.15 | 93.08 |
| Lactic acid | 81.43 | 80.77 | 92.43 | 96.06 | 117.16 | 82.88 |
| Water | 93.54 | 95.80 | 105.65 | 93.89 | 126.11 | 97.36 |
| IP | Gas | Benzene | Acetic Acid | Ethanol | Formic Acid | Lactic Acid | Water |
|---|---|---|---|---|---|---|---|
| Neu5Ac | 198.37 | 168.58 | 135.72 | 138.41 | 132.06 | 136.56 | 131.63 |
| Neu5Gc | 200.81 | 172.75 | 137.51 | 139.97 | 134.57 | 139.84 | 133.99 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, R.; Yang, B.; Zhu, Q.-J. Theoretical Studies on the Electronic Structure Parameters and Reactive Activity of Neu5Gc and Neu5Ac under Food Processing Solvent Environment. Molecules 2019, 24, 313. https://doi.org/10.3390/molecules24020313
Chang R, Yang B, Zhu Q-J. Theoretical Studies on the Electronic Structure Parameters and Reactive Activity of Neu5Gc and Neu5Ac under Food Processing Solvent Environment. Molecules. 2019; 24(2):313. https://doi.org/10.3390/molecules24020313
Chicago/Turabian StyleChang, Rui, Bowen Yang, and Qiu-Jin Zhu. 2019. "Theoretical Studies on the Electronic Structure Parameters and Reactive Activity of Neu5Gc and Neu5Ac under Food Processing Solvent Environment" Molecules 24, no. 2: 313. https://doi.org/10.3390/molecules24020313






