Immunomodulatory Effects of Phosphorylated Radix Cyathulae officinalis Polysaccharides in Immunosuppressed Mice
Abstract
:1. Introduction
2. Results and Discussion
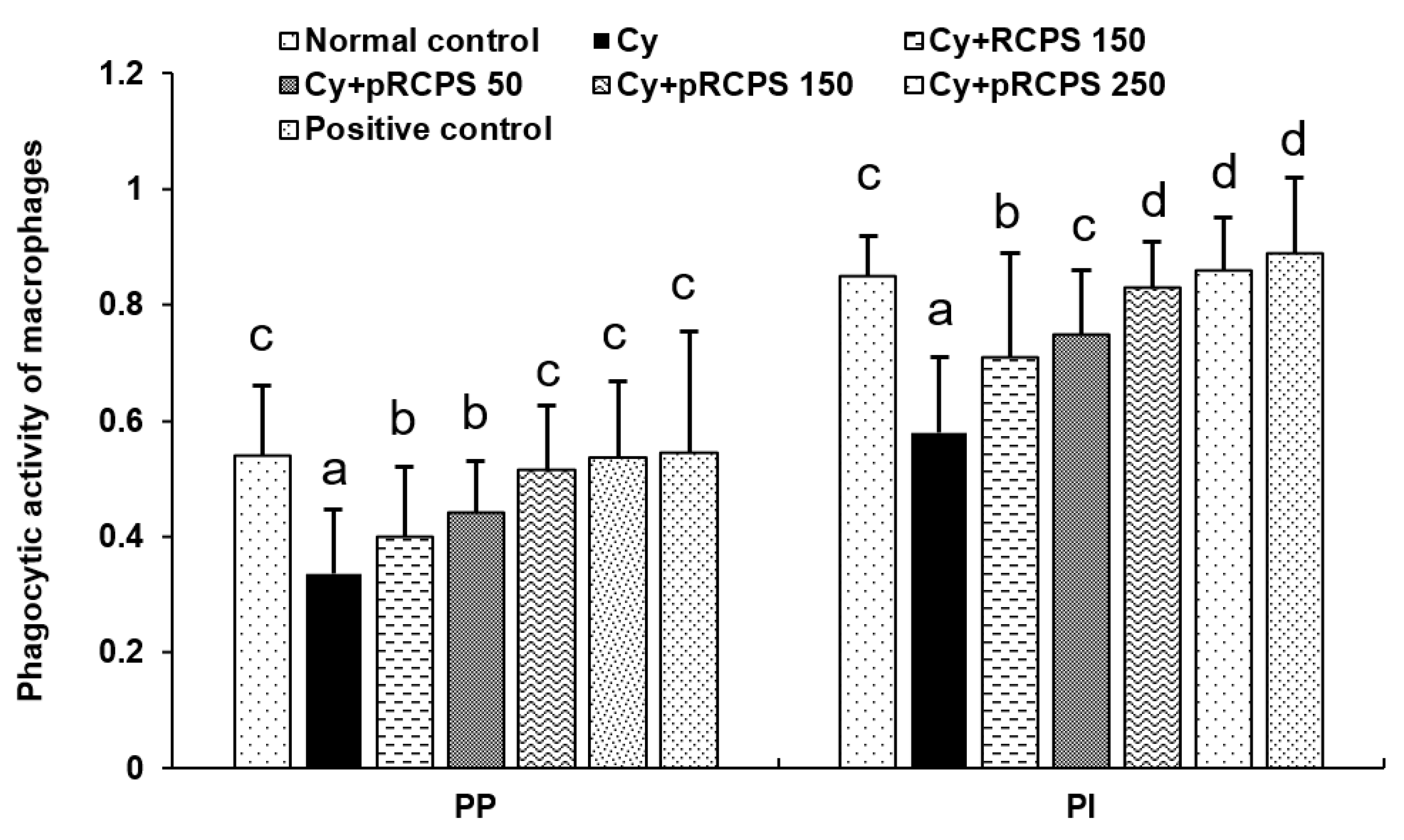
2.1. pRCPS and the PM Phagocytic Function
2.2. pRCPS Affects Body Weight and Thymus and Spleen Indices
2.3. pRCPS and Proliferation of Splenocyte
2.4. Effects of pRCPS on the Proportion of Peripheral Blood T-Lymphocyte Subsets in Immunosuppressed Mice
2.5. pRCPS Effects on Cytokine Serum Concentrations in Immunosuppressed Mice
2.6. Effects of pRCPS on Serum IgM, IgA, and IgG Concentrations in Immunosuppressed Mice
2.7. pRCPS Effects on Cytokine Secretion of Splenocytes in Immunosuppressed Mice
3. Experimental Section
3.1. Kits and Reagents
3.2. RCPS and pRCPS Preparation
3.3. Experimental Animals and Design
3.4. Assay for Splenocyte Proliferation
3.5. Phagocytosis Assay
3.6. Analysis of the Concentration of Serum Cytokines
3.7. Serum Ig concentrations
3.8. T-lymphocyte Phenotyping by Flow Cytometry
3.9. IL-4 and IFN-γ Production by Splenocytes In Vitro
3.10. Statistical Data Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| RCPS | Radix Cyathulae officinalis Kuan polysaccharides |
| FACS | fluorescence activated cell sorting |
| FITC | Fluorescein isothiocyanate |
| IL-2 | interleukin-2 |
| IL-4 | interleukin-4 |
| IL-5 | interleukin-5 |
| IL-6 | interleukin-6 |
| IL-10 | interleukin-2 |
| IFN-γ | interferon-γ |
| LPS | lipopolysaccharide |
| ConA | Concanavalin A |
| IR | infrared |
| PM | peritoneal macrophages |
References
- Datz, C.A. Noninfectious Causes of Immunosuppression in Dogs and Cats. Veterinary Clinics of North America. Small Anim. Pract. 2010, 40, 459–467. [Google Scholar] [CrossRef]
- Tizard, I.R. Veterinary Immunology; Saunders Elsevier: St. Louis, MO, USA, 2009; pp. 473–479. [Google Scholar]
- Singleton, H.; Graham, S.P.; Frossard, J.P.; Bodman-Smithm, K.B.; Steinbach, F. Infection.of monocytes with European porcine reproductive and respiratory syndrome virus (PRRSV-1) strain Lena is significantly enhanced by dexamethasone and IL-10. Virology 2018, 517, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.L.; Zhang, W.L.; Xiang, L.R.; Leng, C.L.; Tian, Z.J.; Tang, Y.D.; Cai, X.H. Emergence of novel porcine reproductive and respiratory syndrome viruses (ORF5 RFLP 1-7-4 viruses) in China. Vet. Microbiol. 2018, 222, 105–108. [Google Scholar] [CrossRef] [PubMed]
- Nedumpun, T.; Sirisereewan, C.; Thanmuan, C.; Techapongtada, P.; Puntarotairung, R.; Naraprasertkul, S.; Thanawongnuwech, R.; Suradhat, S. Induction of porcine reproductive and respiratory syndrome virus (PRRSV)-specific regulatory T lymphocytes (Treg) in the lungs and tracheobronchial lymph nodes of PRRSV-infected pigs. Vet. Microbiol. 2018, 216, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Neumann, E.J.; Kliebenstein, J.B.; Johnson, C.D.; Mabry, J.W.; Bush, E.J.; Seitzinger, A.H.; Green, A.L.; Zimmerman, J.J. Assessment of the economic impact of porcine reproductive and respiratory syndrome on swine production in the United States. J. Am. Vet. Med. Assoc. 2005, 227, 385–392. [Google Scholar] [CrossRef]
- Li, B.; Wei, K.; Yang, S.F.; Yang, Y.; Zhang, Y.B.; Zhu, F.J.; Wang, D.; Zhu, R. Immunomodulatory effects of Taishan Pinus massoniana pollen polysaccharide and propolis on immunosuppressed chickens. Microb. Pathog. 2015, 78, 7–13. [Google Scholar] [CrossRef]
- Wang, M.; Meng, X.Y.; Yang, L.R.; Qin, T.; Wang, X.Y.; Zhang, Y.K.; Fei, C.Z.; Li, Y.; Hu, Y.L.; Cue, F.Q. Cordyceps militaris polysaccharides can enhance the immunity and antioxidation activity in immunosuppressed mice. Carbohydr. Polym. 2012, 89, 461–466. [Google Scholar] [CrossRef]
- Huang, F.; Zhang, R.F.; Liu, Y.; Xiao, J.; Liu, L.; Wei, Z.C.; Yi, Y.; Zhang, M.; Liu, D. Dietary litchi pulp polysaccharides could enhance immunomodulatory and antioxidant effects in mice. Int. J. Biol. Macromol. 2016, 92, 1067–1073. [Google Scholar] [CrossRef]
- Zhao, L.M.; Jia, Y.L.; Ma, M.; Duan, Y.Q.; Liu, L.H. Prevention effects of Schisandra polysaccharide.on radiation-induced immune system dysfunction. Int. J. Biol. Macromol. 2015, 76, 63–69. [Google Scholar] [CrossRef]
- Parish, C.R. Cancer immunotherapy: The past, the present and the future. Immunol. Cell Biol. 2003, 81, 106–113. [Google Scholar] [CrossRef]
- Feng, H.B.; Du, X.G.; Liu, J.; Han, X.F.; Cao, X.H.; Zeng, X.Y. Novel polysaccharide from Radix Cyathulae officinalis Kuan can improve immune response to ovalbumin in mice. Int. J. Biol. Macromol. 2014, 65, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Feng, H.B.; Du, X.G.; Tang, J.; Cao, X.H.; Han, X.F.; Chen, Z.Y.; Chen, Y.G.; Zeng, X.Y. Enhancement of the immune responses to foot-and-mouth disease vaccination in mice by oral administration of a Novel polysaccharide from the roots of Radix Cyathulae officinalis Kuan (RC). Cell. Immunol. 2013, 281, 111–121. [Google Scholar] [CrossRef] [PubMed]
- Feng, H.B.; Fan, J.; Du, X.G.; Song, Z.H.; Wang, Z.H.; Han, X.F. Sulfated Radix Cyathulae officinalis polysaccharides act as adjuvant via promoting the dendritic cell maturation and suppressing treg frequency. Immunol. Investig. 2015, 4, 1–21. [Google Scholar]
- Cassado Ados, A.; D’Império Lima, M.R.; Bortoluci, K.R. Revisiting mouse peritoneal macrophages: Heterogeneity, development, and function. Front. Immunol. 2015, 19, 6225. [Google Scholar] [CrossRef]
- Hirayama, D.; Iida, T.; Nakase, H. The Phagocytic Function of Macrophage-Enforcing Innate Immunity and Tissue Homeostasis. Int. J. Mol. Sci. 2017, 19, 92. [Google Scholar] [CrossRef]
- Bain, C.C.; Jenkins, S.J. The biology of serous cavity macrophages. Cell. Immunol. 2018, 330, 126–135. [Google Scholar] [CrossRef]
- Li, J.E.; Nie, S.P.; Xie, M.Y.; Huang, D.F.; Wang, Y.T.; Li, C. Chemical composition and antioxidant activities in immumosuppressed mice of polysaccharides isolated from Mosla chinensis Maxim cv. Jiangxiangru. Int. Immunopharmacol. 2013, 17, 267–274. [Google Scholar] [CrossRef]
- Quakyi, E.; Carter, P.; Tsai, C.M.; Marti, G. Immunizationwith meningococcal membrane-bound lipooligosaccharideaccelerates granulocyte recovery and enhances lymphocyteproliferation in myelosuppressed mice. Pathobiology 1997, 65, 26–38. [Google Scholar] [CrossRef]
- Zhu, X.L.; Chen, A.F.; Lin, Z.B. Ganoderma lucidum polysaccharides enhance the function of immunological effector cells in immunosuppressed mice. J. Ethnopharmacol. 2007, 111, 219–226. [Google Scholar] [CrossRef]
- Nijkamp, F.P.; Parnham, M.J. T cell subsets and T cell-mediated immunity. In Principles of Immunopharmacology; Birkhäuser Press: Basel, Switzerland, 2011; pp. 15–27. [Google Scholar]
- Yan, Z.F.; Liu, N.X.; Mao, X.X.; Li, Y.; Li, C.T. Activation Effects of Polysaccharides of Flammulina velutipes Mycorrhizae on the T Lymphocyte Immune Function. J. Immunol. Res. 2014, 2014, 1–7. [Google Scholar] [CrossRef]
- Takahashi, A.; Watanabe, T.; Mondal, A.; Suzuki, K.; Kurusu-Kanno, M.; Li, Z.; Yamazaki, T.; Fujiki, H.; Suganuma, M. Mechanism-based inhibition of cancer metastasis with (-) epigallocatechin gallate. Biochem. Biophys. Res. Commun. 2014, 443, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, J.; Huang, J. CD8+ T cells and NK cells: Parallel and complementary soldiers of immunotherapy. Curr. Opin. Chem. Eng. 2018, 19, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Chiacchio, T.; Petruccioli, E.; Vanini, V.; Cuzzi, G.; Goletti, D. Impact of antiretroviral and tuberculosis therapies on CD4+ and CD8+ HIV/M. tuberculosis-specific T-cell in co-infected subjects, Immunol. Lett. 2018, 198, 33–43. [Google Scholar] [PubMed]
- Koeck, S.; Zwierzin, M.; Gamerith, G.; Lorenz, Z.H.; Kern, J.; Amann, A. The influence of stromal cells on CD3+ CD8+ tumor infiltrating lymphocyte subpopulations in cancer microtissues. Eur. J. Cancer 2018, 92, S12. [Google Scholar]
- Shang, X.Y.; Chao, Y.; Zhang, Y.; Lu, C.Y.; Xu, C.L.; Niu, W.N. Immunomodulatory and. Antioxidant Effects of Polysaccharides from Gynostemma pentaphyllum Makino in Immunosuppressed Mice. Molecules 2016, 21, 1085. [Google Scholar] [CrossRef] [PubMed]
- Millán, O.; Brunet, M. Cytokine-based immune monitoring. Clin. Biochem. 2016, 49, 338–346. [Google Scholar] [CrossRef] [PubMed]
- Dillinger, B.; Ahmadi-Erber, S.; Laua, M.; Hoelzl, M.A.; Erhart, F.; Juergens, B.; Fuchs, D.; Heitger, A.; Ladisch, S.M.; Dohnal, A. IFN-γ and tumor gangliosides: Implications for the tumor microenvironment. Cell. Immunol. 2018, 325, 33–40. [Google Scholar] [CrossRef]
- Stewart, D.; Nichol, A. Inflammation, immunity and allergy. Anaesth. Intensive Care Med. 2018, 19, 534–539. [Google Scholar] [CrossRef]
- Redpath, S.A.; Heieis, G.; Perona-Wright, G. Spatial regulation of IL-4 signalling In Vivo. Cytokine 2015, 75, 51–56. [Google Scholar] [CrossRef]
- Greenspan, N.S.; Cavacini, L.A. 15: Immunoglobulin Function. In Clinical Immunology, 5th ed.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 223–233. [Google Scholar]
- Hsu, E. Immune System Receptors in Vertebrates: Immunoglobulins. Ref. Modul. Life Sci. 2018. [Google Scholar] [CrossRef]
- Huang, Y.; Jiang, C.M.; Hu, Y.L.; Zhao, X.J.; Shi, C.; Yu, Y.; Liu, C.; Tao, Y.; Pan, H.R.; Feng, Y.B.; et al. Immunoenhancement effect of rehmannia glutinosa polysaccharide on lymphocyte proliferation and dendritic cell. Carbohydr. Polym. 2013, 96, 516–521. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.S.; Wu, Y.W.; Xu, S.F.; Sun, H.X.; Chen, F.Y.; Yao, L. Antitumor and immunomodulatory activity of polysaccharides from the roots of Actinidia eriantha. J. Ethnopharmacol. 2009, 125, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Feng, H.B.; Fan, J.; Yang, S.P.; Zhao, X.L.; Yi, X. Antivira activity of phosphorylated Radix Cyathulae officinalis polysaccharide against Canine Parvovirus In Vitro. Int. J. Biol. Macromol. 2017, 2, 511–518. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Qiu, S.Y.; Mo, L.P.; Wang, W.P.; Wu, Y.G. Process optimization of phosphorylation of Lentinan. Sci. Technol. Food Ind. 2008, 29, 185–188. [Google Scholar]
- Feng, H.B.; McDonough, S.P.; Fan, J.; Yang, S.P.; Zhao, X.L.; Lu, Y.; Zhao, X.L.; Lu, Y.; Gan, Y.; Yi, X.; et al. Phosphorylated Radix Cyathulae officinalis Polysaccharides Act as Adjuvant via Promoting Dendritic Cell Maturation. Molecules 2017, 22, 106. [Google Scholar] [CrossRef] [Green Version]
- Zhang, N.W.; Li, J.F.; Cheng, G.L.; Zhu, X.Y.; Liu, F.Q.; Zhang, Y.J.; Liu, Z.J.; Xu, J.Q. Effects of astragalus polysaccharide on the immune response to foot-and-mouth disease vaccine in mice. Carbohydr. Polym. 2010, 82, 680–686. [Google Scholar] [CrossRef]
- Zhou, X.; Liu, Z.J.; Long, T.T.; Zhou, L.J.; Bao, Y.X. Immunomodulatory effects of herbal formula of astragalus polysaccharide (APS) and polysaccharopeptide (PSP) in mice with lung cancer. Int. J. Biol. Macromol. 2018, 106, 596–601. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are available from the authors. |






| Group | Cy or Saline | Compounds |
|---|---|---|
| Normal control | Physiological saline (0.9%) | Physiological saline (0.9%) |
| Cy | Cy (80 mg/kg/d) | Physiological saline (0.9%) |
| RCPS+Cy | Cy (80 mg/kg/d) | 150 mg/kg B.W. RCPS |
| pRCPS 50+Cy | Cy (80 mg/kg/d) | 50 mg/kg B.W. pRCPS |
| pRCPS 150+Cy | Cy (80 mg/kg/d) | 150 mg/kg B.W. pRCPS |
| pRCPS 250+Cy | Cy (80 mg/kg/d) | 250 mg/kg B.W. pRCPS |
| Positive control | Cy (80 mg/kg/d) | 10 mg/kg B.W. Levamisole hydrochloride |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Feng, H.; Fan, J.; Lin, L.; Liu, Y.; Chai, D.; Yang, J. Immunomodulatory Effects of Phosphorylated Radix Cyathulae officinalis Polysaccharides in Immunosuppressed Mice. Molecules 2019, 24, 4150. https://doi.org/10.3390/molecules24224150
Feng H, Fan J, Lin L, Liu Y, Chai D, Yang J. Immunomodulatory Effects of Phosphorylated Radix Cyathulae officinalis Polysaccharides in Immunosuppressed Mice. Molecules. 2019; 24(22):4150. https://doi.org/10.3390/molecules24224150
Chicago/Turabian StyleFeng, Haibo, Jing Fan, Lang Lin, Yunjie Liu, Dongkun Chai, and Jie Yang. 2019. "Immunomodulatory Effects of Phosphorylated Radix Cyathulae officinalis Polysaccharides in Immunosuppressed Mice" Molecules 24, no. 22: 4150. https://doi.org/10.3390/molecules24224150






