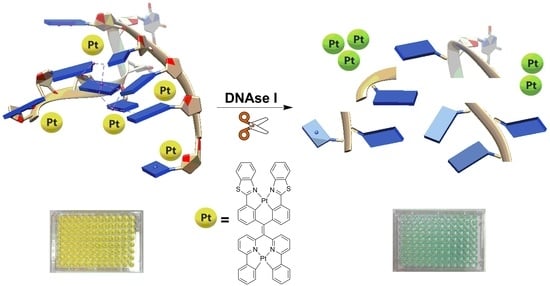
Expanding the Toolbox for Label-Free Enzyme Assays: A Dinuclear Platinum(II) Complex/DNA Ensemble with Switchable Near-IR Emission
Abstract
:1. Introduction
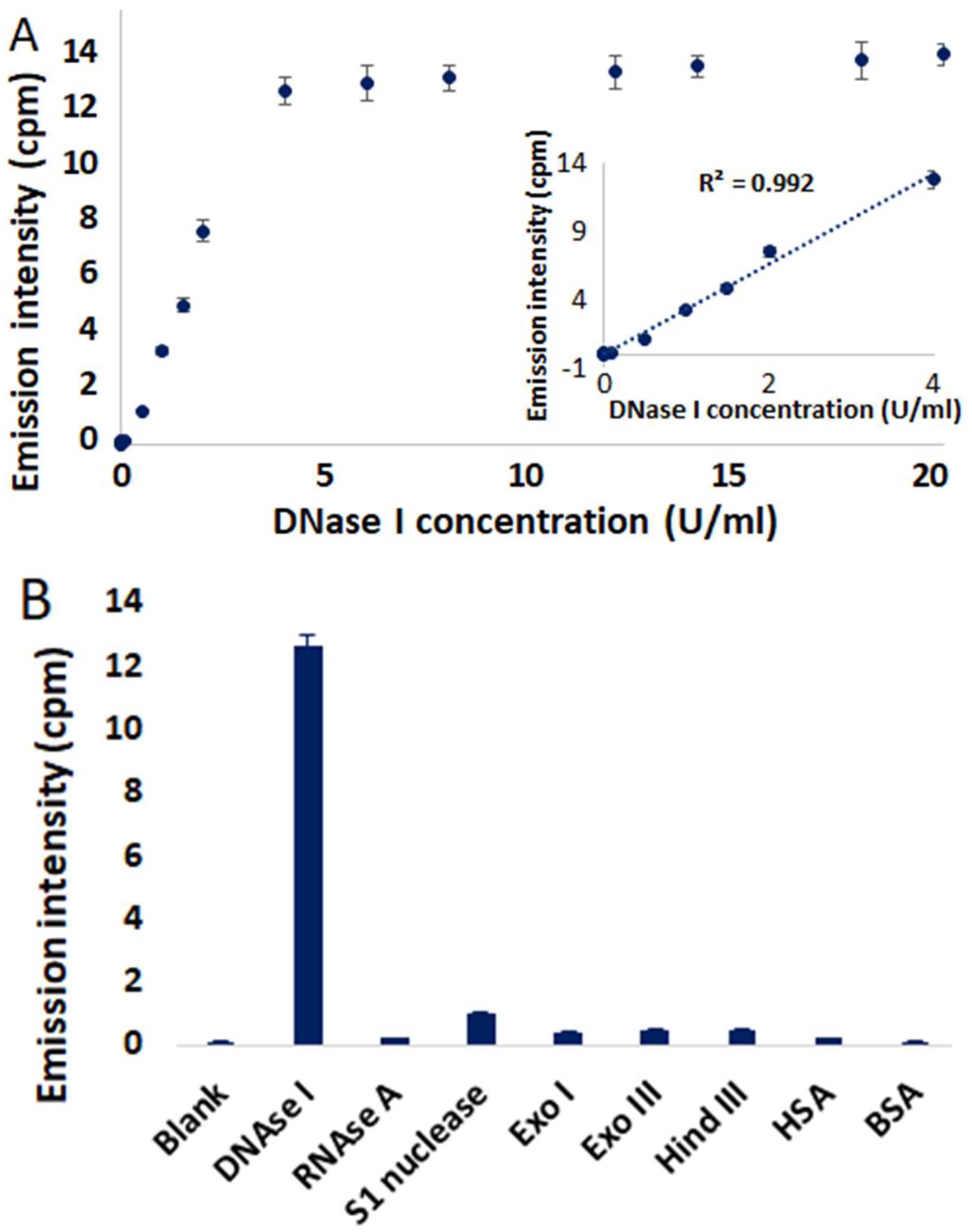
2. Results and Discussion
3. Materials and Methods
3.1. UV–Visible Spectroscopy
3.2. Fluorescence Spectroscopy
3.3. General Procedure for Multiwell DNAse I Assay
3.4. Lineweaver-Burk Plot
3.5. Detection of DNAse I in Human Serum
3.6. Determination of IC50 Values of DNAse I Inhibitors
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Lavis, L.D.; Raines, R.T. Bright ideas for chemical biology. ACS Chem. Biol. 2008, 3, 142–155. [Google Scholar] [CrossRef]
- Grimm, J.B.; Heckman, L.M.; Lavis, L.D. The chemistry of small-molecule fluorogenic probes. Prog. Mol. Biol. Transl. Sci. 2013, 113, 1–34. [Google Scholar]
- Zheng, Q.; Lavis, L.D. Development of photostable fluorophores for molecular imaging. Curr. Opin. Chem. Biol. 2017, 39, 32–38. [Google Scholar] [CrossRef]
- Garland, M.; Yim, J.J.; Bogyo, M. A bright future for precision medicine: Advances in fluorescent chemical probe design and their clinical application. Cell Chem. Biol. 2016, 23, 122–136. [Google Scholar] [CrossRef]
- Yao, J.; Yang, M.; Duan, Y. Chemistry, biology, and medicine of fluorescent nanomaterials and related systems: New insights into biosensing, bioimaging, genomics, diagnostics, and therapy. Chem. Rev. 2014, 114, 6130–6178. [Google Scholar] [CrossRef]
- Chyan, W.; Raines, R.T. Enzyme-activated fluorogenic probes for live-cell and in vivo imaging. ACS Chem. Biol. 2018, 13, 1810–1823. [Google Scholar] [CrossRef]
- Singh, K.; Rotaru, A.M.; Beharry, A.A. Fluorescent chemosensors as future tools for cancer biology. ACS Chem. Biol. 2018, 13, 1785–1798. [Google Scholar] [CrossRef]
- Shi, J.; Li, Y.; Li, Q.; Li, Z. Enzyme-responsive bioprobes based on the mechanism of aggregation-induced emission. ACS Appl. Mater. Interfaces 2018, 10, 12278–12294. [Google Scholar] [CrossRef] [PubMed]
- Baranovskii, A.G.; Buneva, V.N.; Nevinsky, G.A. Mechanism of action of DNA-hydrolyzing antibodies to DNA from blood of patients with systemic lupus erythematosus. Biochemistry (Moscow) 2004, 69, 587–601. [Google Scholar] [CrossRef] [PubMed]
- Cherepanova, A.; Tamkovich, S.; Pyshnyi, D.; Kharkova, M.; Vlassov, V.; Laktionov, P. Immunochemical assay for deoxyribonuclease activity in body fluids. J. Immunol. Methods 2007, 325, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Basnakian, A.G.; Apostolov, E.O.; Yin, X.; Napirei, M.; Mannherz, H.G.; Shah, S.V. Cisplatin nephrotoxicity is mediated by deoxyribonuclease I. J. Am. Soc. Nephrol. 2005, 16, 697–702. [Google Scholar] [CrossRef] [PubMed]
- Keyel, P.A. DNAses in health and disease. Dev. Biol. 2017, 429, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Samejima, K.; Earnshaw, W.C. Trashing the genome: The role of nucleases during apoptosis. Nat. Rev. Mol. Cell. Biol. 2005, 6, 677–688. [Google Scholar] [CrossRef] [PubMed]
- Cherepanova, A.V.; Tamkovich, S.N.; Bryzgunova, O.E.; Vlassov, V.V.; Laktionov, P.P. Deoxyribonuclease activity and circulating DNA concentration in blood plasma of patients with prostate tumors. Ann. N. Y. Acad. Sci. 2008, 1137, 218–221. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, H.L.; Horvei, K.D.; Thiyagarajan, D.; Norby, G.E.; Seredkina, N.; Moroni, G.; Eilertsen, G.; Holdaas, H.; Strom, E.H.; Bakland, G.; et al. Lupus nephritis: Low urinary DNAse I levels reflect loss of renal DNAse I and may be utilized as a biomarker of disease progression. J. Pathol. Clin. Res. 2018, 4, 193–203. [Google Scholar] [CrossRef]
- Morikawa, N.; Kawai, Y.; Arakawa, K.; Kumamoto, T.; Miyamori, I.; Akao, H.; Kitayama, M.; Kajinami, K.; Lee, J.D.; Takeshita, H.; et al. Serum deoxyribonuclease I activity can be used as a novel marker of transient myocardial ischaemia: Results in vasospastic angina pectoris induced by provocation test. Eur. Heart J. 2007, 28, 2992–2997. [Google Scholar] [CrossRef] [PubMed]
- Skiljevic, D.; Jeremic, I.; Nikolic, M.; Andrejevic, S.; Sefik-Bukilica, M.; Stojimirovic, B.; Bonaci-Nikolic, B. Serum DNAse I activity in systemic lupus erythematosus: Correlation with immunoserological markers, the disease activity and organ involvement. Clin. Chem. Lab. Med. 2013, 51, 1083–1091. [Google Scholar] [CrossRef]
- Hernandez, L.I.; Ozalp, V.C.; Hernandez, F.J. Nuclease activity as a specific biomarker for breast cancer. Chem. Commun. 2016, 52, 12346–12349. [Google Scholar] [CrossRef]
- Nadano, D.; Yasuda, T.; Kishi, K. Measurement of deoxyribonuclease I activity in human tissues and body fluids by a single radial enzyme-diffusion method. Clin. Chem. 1993, 39, 448–452. [Google Scholar]
- Fojta, M.; Kubicarova, T.; Palecek, E. Cleavage of supercoiled DNA by deoxyribonuclease I in solution and at the electrode surface. Electroanalysis 1999, 11, 1005–1012. [Google Scholar] [CrossRef]
- Jang, D.S.; Penthala, N.R.; Apostolov, E.O.; Wang, X.; Fahmi, T.; Crooks, P.A.; Basnakian, A.G. Novel high-throughput deoxyribonuclease I assay. J. Biomol. Screen. 2015, 20, 202–211. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Chen, Y.; Fang, J.; Fan, J.; Tong, C.; Liu, X.; Liu, B.; Wang, W. DNAse-targeted natural product screening based on a sensitive and selective DNAse I detecting system. RSC Adv. 2017, 7, 30911–30918. [Google Scholar] [CrossRef]
- Lee, C.Y.; Park, K.S.; Jung, Y.K.; Park, H.G. A label-free fluorescent assay for deoxyribonuclease I activity based on DNA-templated silver nanocluster/graphene oxide nanocomposite. Biosens. Bioelectron. 2017, 93, 293–297. [Google Scholar] [CrossRef] [PubMed]
- Sato, S.; Takenaka, S. Highly sensitive nuclease assays based on chemically modified DNA or RNA. Sensors 2014, 14, 12437–12450. [Google Scholar] [CrossRef]
- Mozioğlu, E.; Akgoz, M.; Kocagöz, T.; Tamerler, C. Detection of nuclease activity using a simple fluorescence-based biosensor. Anal. Methods 2016, 8, 4017–4021. [Google Scholar] [CrossRef]
- Zhang, Q.; Li, J.; Zhang, Y.; Hu, Z.; Yu, C. One-pot synthesis of quencher labeled hairpin DNA-CdTe QDs conjugate for target DNA and deoxyribonuclease I detection. Anal. Sci. 2016, 32, 1035–1037. [Google Scholar] [CrossRef]
- Jung, Y.L.; Lee, C.Y.; Park, J.H.; Park, K.S.; Park, H.G. A signal-on, colorimetric determination of deoxyribonuclease I activity utilizing the photoinduced synthesis of gold nanoparticles. Nanoscale 2018, 10, 4339–4343. [Google Scholar] [CrossRef]
- Sun, S.-K.; Wang, B.-B.; Yan, X.-P. A label-free near-infrared fluorescent assay for the determination of deoxyribonuclease I activity based on malachite green/G-quadruplexes. Analyst 2013, 138, 2592–2597. [Google Scholar] [CrossRef]
- Dou, Y.; Yang, X. Novel high-sensitive fluorescent detection of deoxyribonuclease I based on DNA-templated gold/silver nanoclusters. Anal. Chim. Acta 2013, 784, 53–58. [Google Scholar] [CrossRef]
- Hao, C.; Kuang, H.; Xu, L.; Liu, L.; Ma, W.; Wang, L.; Xu, C. Chiral supernanostructures for ultrasensitive endonuclease analysis. J. Mater. Chem. B 2013, 1, 5539–5542. [Google Scholar] [CrossRef]
- Choi, S.-J.; Szoka, F.C. Fluorometric determination of deoxyribonuclease I activity with PicoGreen. Anal. Biochem. 2000, 281, 95–97. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Zhu, J.; Zhang, L.; Du, Y.; Dong, S.; Wang, E. G-quadruplex-based fluorescent assay of S1 nuclease activity and K+. Anal. Chem. 2013, 85, 2431–2435. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; Chan, K.H.-Y.; Wong, K.M.-C.; Yam, V.W.-W. Nucleic acid-induced self-assembly of a platinum(II) terpyridyl complex: Detection of a G-quadruplex formation and nuclease activity. Chem. Commun. 2009, 3756–3758. [Google Scholar] [CrossRef] [PubMed]
- Yam, V.W.-W.; Au, V.K.-M.; Leung, S.Y.-L. Light-emitting self-assembled materials based on d8 and d10 transition metal complexes. Chem. Rev. 2015, 115, 7589–7728. [Google Scholar] [CrossRef] [PubMed]
- Mauro, M.; Aliprandi, A.; Cebrian, C.; Wang, D.; Kubel, C.; De Cola, L. Self-assembly of a neutral platinum(II) complex into highly emitting microcrystalline fibers through metallophilic interactions. Chem. Commun. 2014, 50, 7269–7272. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuang, S.; Hu, Z.; Zhang, H.; Zhang, X.; Liang, F.; Zhao, Z.; Liu, S. Enhancement of metal-metal interactions inside a large-cavity synthetic host in water. Chem. Commun. 2018, 54, 2169–2172. [Google Scholar] [CrossRef]
- Chan, A.K.-W.; Ng, M.; Wong, Y.-C.; Chan, M.-Y.; Wong, W.-T.; Yam, V.W.-W. Synthesis and characterization of luminescent cyclometalated platinum(II) complexes with tunable emissive colors and studies of their application in organic memories and organic light-emitting devices. J. Am. Chem. Soc. 2017, 139, 10750–10761. [Google Scholar] [CrossRef] [Green Version]
- Huo, S.; Carroll, J.; Vezzu, D.A.K. Design, synthesis, and applications of highly phosphorescent cyclometalated platinum complexes. Asian J. Org. Chem. 2015, 4, 1210–1245. [Google Scholar] [CrossRef]
- Chan, A.K.-W.; Yam, V.W.-W. Precise modulation of molecular building blocks from tweezers to rectangles for recognition and stimuli-responsive processes. Acc. Chem. Res. 2018, 51, 3041–3051. [Google Scholar] [CrossRef]
- Law, A.S.-Y.; Yeung, M.C.-L.; Yam, V.W.-W. Arginine-rich peptide induced supramolecular self-assembly of water-soluble anionic alkynylplatinum(II) complexes: A continuous and label-free luminescence assay for trypsin and inhibitor screening. ACS Appl. Mater. Interfaces 2017, 9, 41143–41150. [Google Scholar] [CrossRef]
- Li, K.; Tong, G.S.M.; Wan, Q.; Cheng, G.; Tong, W.-Y.; Ang, W.-H.; Kwong, W.-L.; Che, C.-M. Highly phosphorescent platinum(II) emitters: Photophysics, materials and biological applications. Chem. Sci. 2016, 7, 1653–1673. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aliprandi, A.; Genovese, D.; Mauro, M.; De Cola, L. Recent advances in phosphorescent Pt(II) complexes featuring metallophilic interactions: Properties and applications. Chem. Lett. 2015, 44, 1152–1169. [Google Scholar] [CrossRef]
- Chung, C.Y.-S.; Yam, V.W.-W. Induced self-assembly and Förster resonance energy transfer studies of alkynylplatinum(II) terpyridine complex through interaction with water-soluble poly(phenylene ethynylene sulfonate) and the proof-of-principle demonstration of this two-component ensemble for selective label-free detection of human serum albumin. J. Am. Chem. Soc. 2011, 133, 18775–18784. [Google Scholar] [PubMed]
- Yeung, M.C.-L.; Wong, K.M.-C.; Tsang, Y.K.T.; Yam, V.W.-W. Aptamer-induced self-assembly of a NIR-emissive platinum(II) terpyridyl complex for label- and immobilization-free detection of lysozyme and thrombin. Chem. Commun. 2010, 46, 7709–7711. [Google Scholar] [CrossRef]
- Law, A.S.-Y.; Yeung, M.C.-L.; Yam, V.W.-W. A luminescence turn-on assay for acetylcholinesterase activity and inhibitor screening based on supramolecular self-assembly of alkynylplatinum(II) complexes on coordination polymer. ACS Appl. Mater. Interfaces 2019, 11, 4799–4808. [Google Scholar] [CrossRef]
- Gabr, M.T.; Pigge, F.C. Rhenium tricarbonyl complexes of AIE active tetraarylethylene ligands: Tuning luminescence properties and HSA-specific binding. Dalton Trans. 2017, 46, 15040–15047. [Google Scholar] [CrossRef]
- Gabr, M.T.; Pigge, F.C. Rhenium complexes of bis(benzothiazole)-based tetraarylethylenes as selective luminescent probes for amyloid fibrils. Chem. Eur. J. 2018, 24, 11729–11737. [Google Scholar] [CrossRef]
- Gabr, M.T.; Pigge, F.C. Platinum(II) complexes with sterically expansive tetraarylethylene ligands as probes for mismatched DNA. Inorg. Chem. 2018, 57, 12641–12649. [Google Scholar] [CrossRef]
- Zhu, L.; Zhou, J.; Xu, G.; Li, C.; Ling, P.; Liu, B.; Ju, H.; Lei, J. DNA quadruplexes as molecular scaffolds for controlled assembly of fluorogens with aggregation-induced emission. Chem. Sci. 2018, 9, 2559–2566. [Google Scholar] [CrossRef] [Green Version]
- Georgiades, S.N.; Karim, N.H.A.; Suntharalingam, K.; Vilar, R. Interaction of metal complexes with G-quadruplex DNA. Angew. Chem. Int. Ed. 2010, 49, 4020–4034. [Google Scholar] [CrossRef]
- Wang, P.; Leung, C.-H.; Ma, D.-L.; Yan, S.-C.; Che, C.-M. Structure-based design of platinum(II) complexes as c-myc oncogene down regulators and luminescent probes for G-quadruplex DNA. Chem. Eur. J. 2010, 16, 6900–6911. [Google Scholar] [CrossRef] [PubMed]
- Cao, Q.; Li, Y.; Freisinger, E.; Qin, P.Z.; Sigel, R.K.O.; Mao, Z.-W. G-quadruplex DNA targeted metal complexes acting as potential anticancer drugs. Inorg. Chem. Front. 2017, 4, 10–32. [Google Scholar] [CrossRef] [Green Version]
- Tullius, T.D. DNA footprinting with the hydroxyl radical. Free Radical Res. Commun. 1991, 13, 521–529. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.-H.; Chung, T.D.Y.; Oldenburg, K.R. A simple statistical parameter for use in evaluation and validation of high-throughput screening assays. J. Biomol. Screen. 1999, 4, 67–73. [Google Scholar] [CrossRef]
- Kolarevic, A.; Yancheva, D.; Kocic, G.; Smelcerovic, A. Deoxyribonuclease inhibitors. Eur. J. Med. Chem. 2014, 88, 101–111. [Google Scholar] [CrossRef]
- Apostolov, E.O.; Soultanova, I.; Savenka, A.; Bagandov, O.O.; Yin, X.; Stewart, A.G.; Walker, R.B.; Basnakian, A.G. Deoxyribonuclease I is essential for DNA fragmentation induced by gamma radiation in mice. Radiat. Res. 2009, 172, 481–492. [Google Scholar] [CrossRef] [Green Version]
- Illic, B.S.; Kolarevic, A.; Kocic, G.; Smelcerovic, A. Ascorbic acid as DNAse I inhibitor in prevention of male infertility. Biochem. Biophys. Res. Commun. 2018, 498, 1073–1077. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds 1–4 are available from the authors. |







© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gabr, M.T.; Pigge, F.C. Expanding the Toolbox for Label-Free Enzyme Assays: A Dinuclear Platinum(II) Complex/DNA Ensemble with Switchable Near-IR Emission. Molecules 2019, 24, 4390. https://doi.org/10.3390/molecules24234390
Gabr MT, Pigge FC. Expanding the Toolbox for Label-Free Enzyme Assays: A Dinuclear Platinum(II) Complex/DNA Ensemble with Switchable Near-IR Emission. Molecules. 2019; 24(23):4390. https://doi.org/10.3390/molecules24234390
Chicago/Turabian StyleGabr, Moustafa T., and F. Christopher Pigge. 2019. "Expanding the Toolbox for Label-Free Enzyme Assays: A Dinuclear Platinum(II) Complex/DNA Ensemble with Switchable Near-IR Emission" Molecules 24, no. 23: 4390. https://doi.org/10.3390/molecules24234390